Abstract
Background:
Use of hypnotics or anxiolytic drugs is common and various studies have reported increased mortality with hypnotics or anxiolytic use.
Objective:
To consolidate the evidence on mortality risk associated with hypnotics or anxiolytic use
Methods:
Major databases were searched through April 2014 for studies reporting mortality risk associated with hypnotics or anxiolytics use. A pooled hazard ratio with 95% confidence interval was estimated using random-effects model.
Results:
After screening 2188 articles, 25 studies (24 cohort, 1 case-control) enrolling 2,350,093 patients with 59% females (age 18–102 years) were included in the meta-analysis. Hypnotics or anxiolytic users had 43% higher risk of mortality than non-users (hazard ratio, 1.43; 95% confidence interval, [1.12, 1.84]). Eight studies reported risk estimates for each gender category and pooled results from these studies showed increased risk of mortality among men (hazard ratio = 1.60, 95% confidence interval = [1.29,1.99]) and women (hazard ratio = 1.68, 95% confidence interval = [1.38, 2.04]). Pooled results from 10 studies showed higher mortality among benzodiazepine users compared to non-users (hazard ratio = 1.60, 95% confidence interval = [1.03, 2.49]), while pooled results from five studies showed an increased risk of mortality with Z-drugs use although the effect could not reach statistical significance (hazard ratio = 1.73, 95% confidence interval = [0.95, 3.16]). Significant heterogeneity was observed in the analyses and the quality of included studies was good.
Conclusion:
This meta-analysis suggests that hypnotics or anxiolytics drugs use is associated with increased mortality and hence should be used with caution. Future studies focused on underlying mechanism of increased mortality with hypnotics or anxiolytics use are required.
Introduction
Benzodiazepines and hypnotics use is common in the general population and is estimated to range between 3.5% and 11.7% (Chong et al., 2013; Mellinger et al., 1985; Morgan et al., 1988; Ohayon and Caulet, 1996; Ohayon et al., 1998; Quera-Salva et al., 1991). Benzodiazepines are mainly used to treat anxiety, insomnia, alcohol withdrawal, seizure, panic disorder, and also used for conscious sedation and as a muscle relaxant. Long-term use of benzodiazepines and hypnotics increases with age, highest among the elderly, ranging from 7% to 43% (Cheng et al., 2008; Donoghue and Lader, 2010; Olfson et al., 2015; Paulose-Ram et al., 2007). Benzodiazepines and hypnotics use may be associated with daytime sleepiness, depressive symptoms, poor physical functioning, car accidents, increased falls and fractures (Cumming and Le Couteur, 2003; Pariente et al., 2008; van Vliet et al., 2009; Vermeeren, 2004), and raises the concern for abuse and dependency. In addition, benzodiazepines may have deleterious effects on memory and cognition (Curran, 1986), and may be associated with an increased risk of Alzheimer type dementia (Billioti de Gage et al., 2014).
In addition to the above negative effects, numerous studies have evaluated the association of hypnotics and anxiolytics (HA) use with mortality risk. However, inconsistent results have been reported (Belleville, 2010; Hausken et al., 2007; Hays et al., 1996; Kojima et al., 2000; Kripke et al., 1998, 2002, 2012; Mallon et al., 2009; Phillips and Mannino, 2005; Rumble and Morgan, 1992). Whereas few studies found no association (Hays et al., 1996; Jaussent et al., 2013; Kojima et al., 2000; Phillips and Mannino, 2005; Rumble and Morgan, 1992), others have reported an increased mortality risk associated with HA use (Belleville, 2010; Hausken et al., 2007; Kripke et al., 1998, 2002, 2012; Mallon et al., 2009). Thus, the association of HA use with mortality risk has remained inconclusive. Therefore, we performed a systematic review and meta-analysis of all available studies evaluating the mortality risk associated with HA use, to quantify the magnitude of this association and appraise the quality of the supporting evidence for this association.
Methods
Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement guidelines were used to report this systematic review and meta-analysis (Moher et al., 2009).
Study eligibility
We searched for randomized control trials (RCT), cohort, case control and cross-sectional studies that provide documentation of benzodiazepines and/or anxiolytics and/or hypnotics use, measured all-cause mortality as a predefined outcome of interest and reported risk estimates or frequency data from which we could calculate the mortality risk associated with HA use. Studies were included irrespective of language or publication status. Data were included from most recent comprehensive report in case of overlapping study samples. Studies reporting selective serotonin reuptake inhibitors, serotonin and norepinephrine reuptake inhibitors and tricyclic antidepressants as anxiolytics were excluded.
Data sources and search/study selection
Major databases including Ovid MEDLINE In-Process & Other Non-Indexed Citations, Ovid MEDLINE, EMBASE, PsycINFO, Cochrane Central Register of Controlled Trials, Cochrane Database of Systematic Reviews and Scopus were searched through 10 April 2014 by an expert librarian with the input from study team. The complete search strategy is available as online supplement. To further increase the yield of relevant articles, references cited in the potentially eligible articles and conference proceedings of major psychiatry, internal medicine and pharmacology organizations were hand-searched by the study team. This was followed by full article searches of relevant abstracts. All titles and abstracts were screened by two reviewers (A.K.P. and S.S.M.) independently in parallel using a standardized form. The same two reviewers did the full text review of selected articles. Inter-observer agreement on study selection was assessed by Cohen’s κ (Cohen, 1960), and any disagreements were resolved by consensus in the presence of a third reviewer.
Data extraction
Data were extracted independently by two reviewers from selected studies. A predesigned form was used to extract data regarding study characteristics (author, country, study design, study population, number of participants and conclusion), HA exposure and mortality. We collected the most adjusted risk estimates in studies where multiple models for risk estimates were reported. When cohort studies reported both cross-sectional and longitudinal models, we used risk estimates from the longitudinal models.
Quality assessment
Two investigators (A.K.P., S.S.M.) performed the methodological quality assessment of included studies in parallel. Disagreements were resolved by consensus in the presence of a third reviewer. We used the Newcastle-Ottawa Quality Assessment Scale to assess the methodological quality of included studies (Wells et al., n.d.). Each study was assessed for risk of bias in selection, comparability of study groups and ascertainment of the outcome of interest.
Outcome assessment
Our primary outcome was all-cause mortality associated with HA use. The secondary outcomes were sex-stratified mortality risk and mortality associated with benzodiazepines or Z-drugs use only. A priori subgroup analysis based on study location, study population, age, duration of study drug exposure, follow-up, studies adjusted for minimal covariates versus those not adjusted and study quality were planned to explain the potential heterogeneity in the direction and magnitude of the effect estimate among different studies. Studies adjusted for age and medical co-morbidities were considered to be adjusted for minimal covariates.
Data synthesis and analysis
Continuous variables were reported as means with standard deviation (SD) or medians with interquartile range (IQR), and categorical variables were reported as frequency and proportions. Most studies reported risk estimate as an adjusted hazard ratio (HR), while a few reported an odds ratio (OR) and risk ratio (RR) which was converted into HR using a standard formula. The pooled HR for mortality associated with HA use was calculated using DerSimonian and Laird random-effects model (DerSimonian and Laird, 1986). I2 statistic and Cochran’s Q test was used to assess the heterogeneity across the included studies (Cochran, 1954). Heterogeneity due to real differences in the study population, methodology and outcome was indicated by an I2 value >50% and a small p (<0.10) of the Cochran’s Q test (Higgins et al., 2003). All statistical analyses except for publication bias were performed using Review Manager (RevMan) Version 5.1 (Copenhagen: The Nordic Cochrane Center, The Cochrane Collaboration, 2011). A probability level <0.05 was considered statistically significant (except for publication bias and heterogeneity assessment) and all p were 2-tailed. Publication bias was assessed quantitatively using Egger’s precision test (weighted linear regression) (Egger et al., 1997) and was considered present if p < 0.10. To assess publication bias, we used Comprehensive Meta-Analysis (CMA) Version 2 (Biostat, Englewood, NJ). Quality of evidence was assessed based on Grading of Recommendations Assessment, Development and Evaluation (GRADE) working guidelines (Atkins et al., 2004).
Results
Study selection
A database search identified a total of 2182 articles and six additional articles were found by a references search of relevant articles. An initial screening of titles and abstracts was done by two reviewers in parallel. During the initial screening, 2144 articles were excluded and 44 articles were included for second screening (Figure 1). A full text review of 44 articles was done by the same two reviewers in parallel and 25 studies were included for final data synthesis. Agreement between the two reviewers was high during both steps (kappa > 0.90).
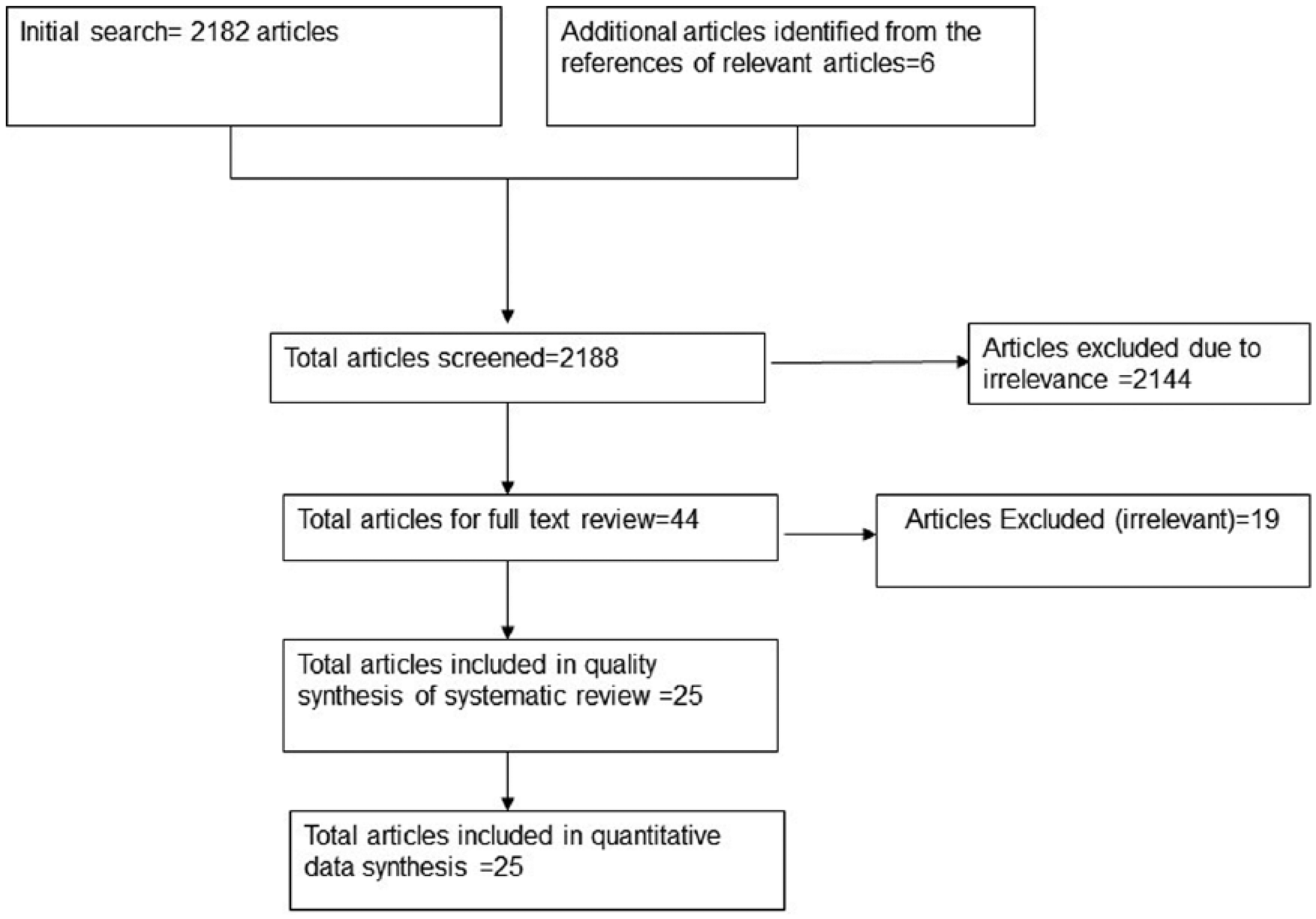
Flow diagram for the selection of references.
Characteristics of included studies and qualitative summary
A detailed description of included 25 studies is reported in Table 1. Of the 25 included studies, 24 were cohort studies (22 prospective cohorts and two retrospective cohorts), while one study was case control, wherein for mortality analysis, only cases with community-acquired pneumonia were included (Obiora et al., 2013). Studies were conducted in different countries across the world. Total sample size included in this meta-analysis was 2,350,093 (range 500–1,099,830) participants with 59% being females. Age of participants ranged from 18–102 years. Study populations differed across the included studies with detailed description given in Table 1. Duration of follow-up ranged from 1–22 years. A detailed quality assessment of the included studies is reported in eTable 1 (online supplement). All studies scored reasonably highly on the quality assessment tool. Agreement between the reviewers was high on each item of the Newcastle-Ottawa scale (kappa > 0.90).
Characteristics of included studies.
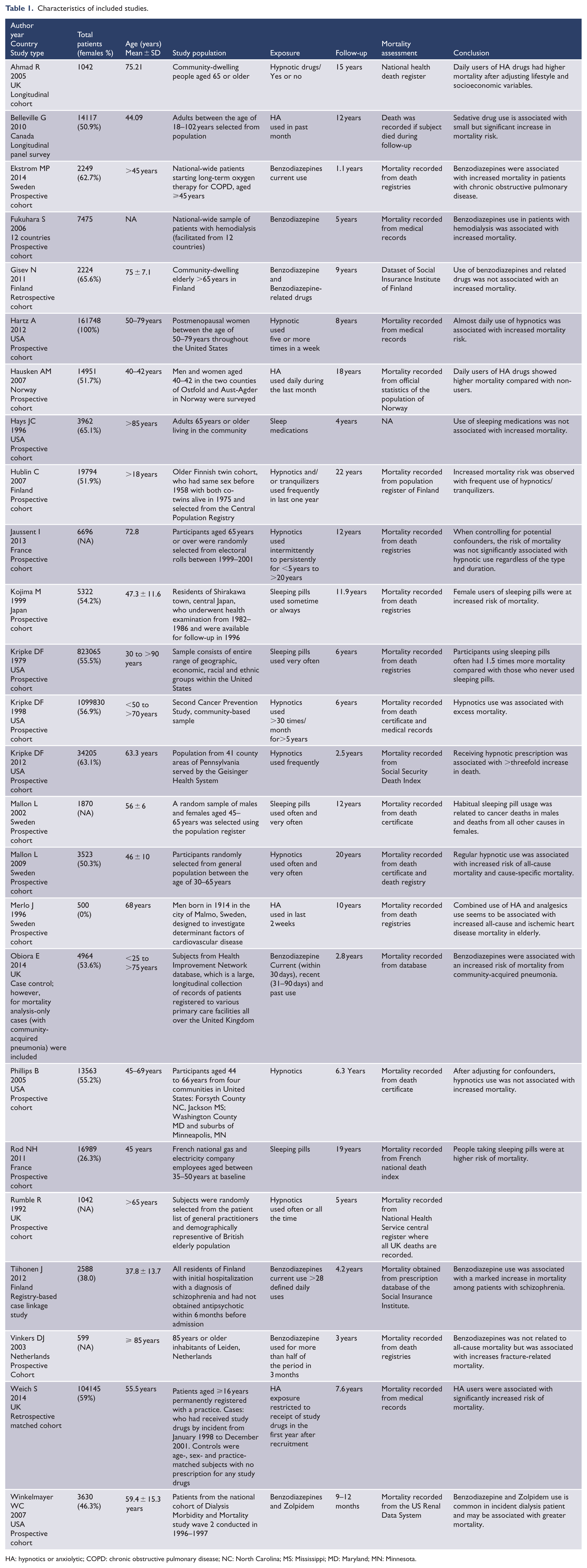
HA: hypnotics or anxiolytic; COPD: chronic obstructive pulmonary disease; NC: North Carolina; MS: Mississippi; MD: Maryland; MN: Minnesota.
Meta-analysis
The pooled adjusted HR for mortality among HA users was significantly higher as compared to non-users (HR = 1.43; 95% confidence interval [CI] = [1.12,1.84]). Overall, HA users had 43% higher risk of mortality compared with non-users (low quality). However, significant heterogeneity was observed between the studies (Cochran’s Q test p < 0.001, I2 = 99%) (Figure 2). After removing six studies causing heterogeneity, HA users had 21% higher risk of mortality compared with non-users and results were homogenous (Figure 3) (Ahmad and Bath, 2005; Kripke et al., 2012; Mallon et al., 2002, 2009; Vinkers et al., 2003; Weich et al., 2014). Eight studies had reported a risk estimate for each sex category and pooled results from subset analysis of these studies showed similar increase in the risk of mortality among men (HR = 1.60, 95% CI = [1.29,1.99]) (Figure 4) and women (HR = 1.68, 95% CI = [1.38, 2.04]) (Figure 5). On stratification, 10 studies reported mortality risk associated with only benzodiazepines use and pooled analysis showed 60% higher mortality among benzodiazepines users as compared to non-users (HR = 1.60, 95% CI = [1.03, 2.49]) (Figure 6). Only five studies reported association of mortality with Z-drugs use and pooled effect showed a 73% increased risk of mortality with Z-drugs use, although the effect did not reach statistical significance (HR = 1.73, 95% CI = [0.95, 3.16]) (Figure 7).
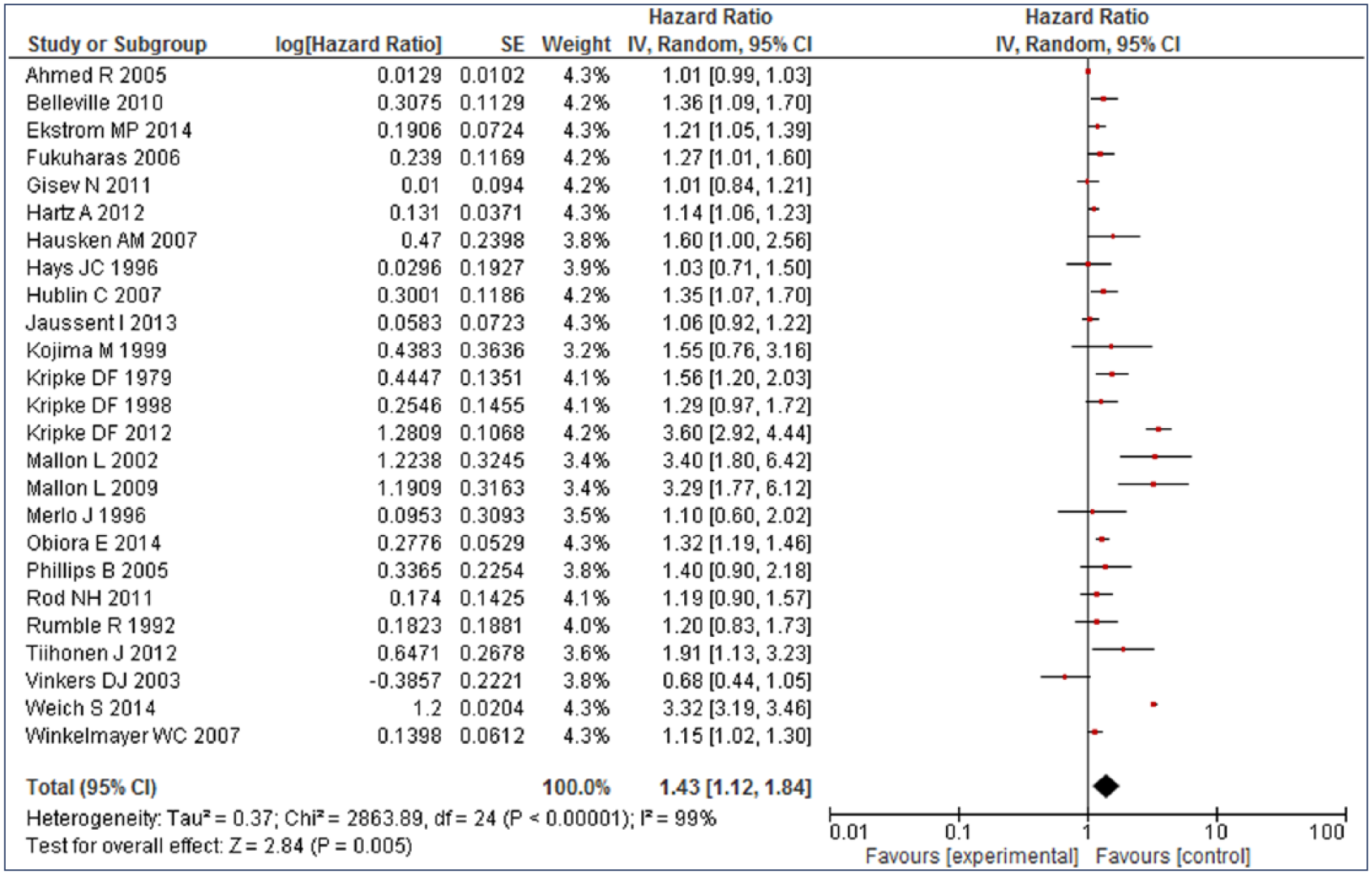
Mortality risk associated with hypnotics and anxiolytics use.
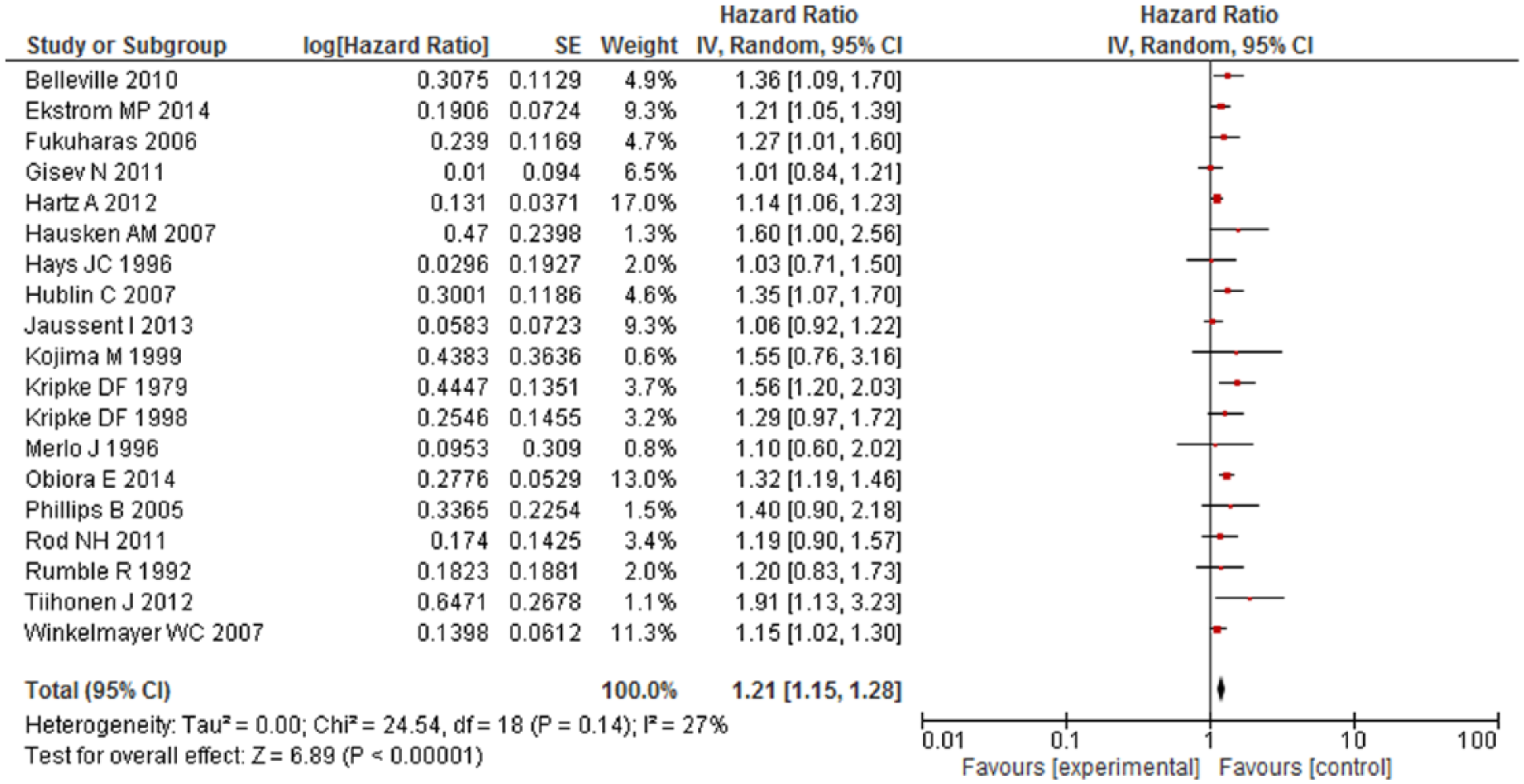
Mortality risk associated with hypnotics and anxiolytics use after excluding six studies causing heterogeneity.
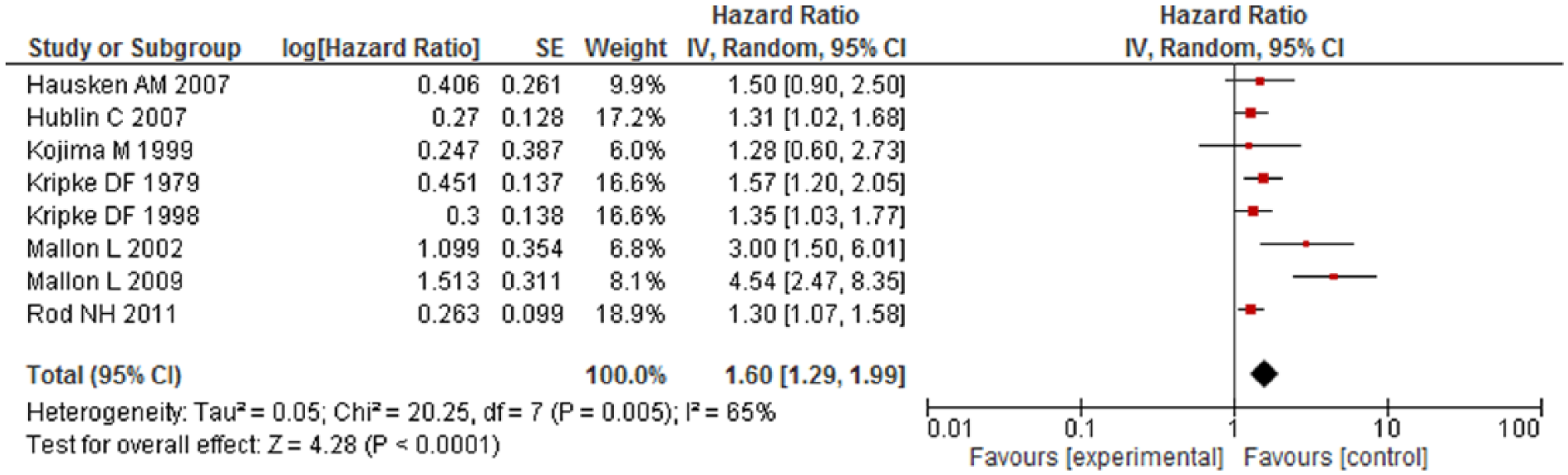
Mortality risk associated with hypnotics and anxiolytics use among males.
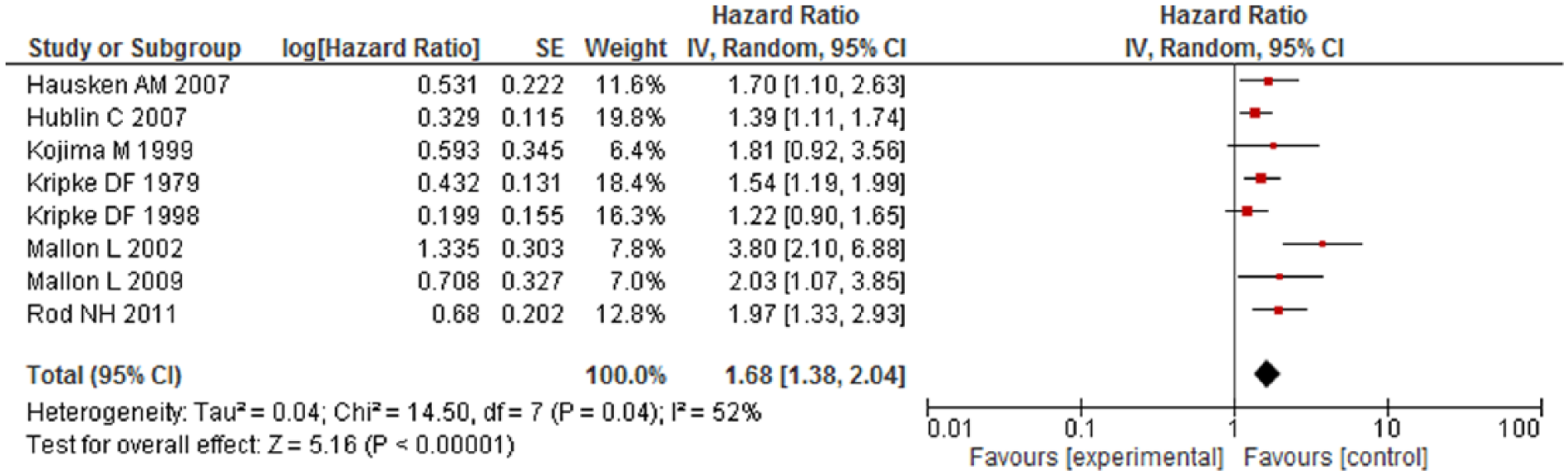
Mortality risk associated with hypnotics and anxiolytics use among females.
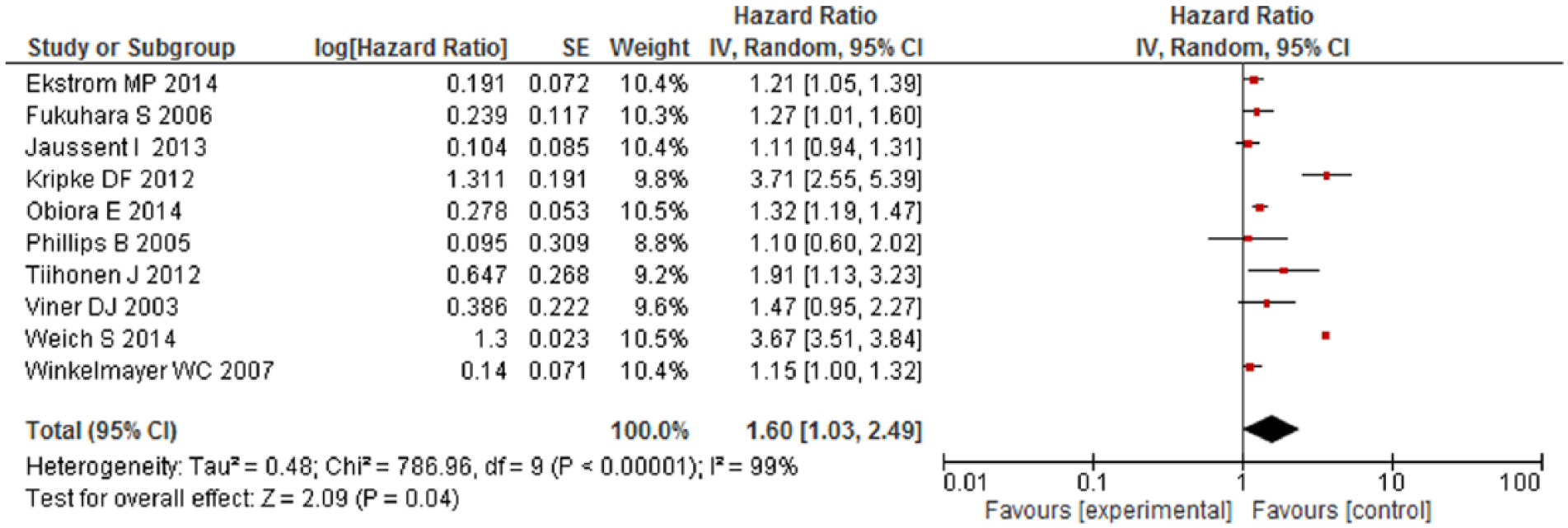
Mortality risk associated with benzodiazepines (only) use.
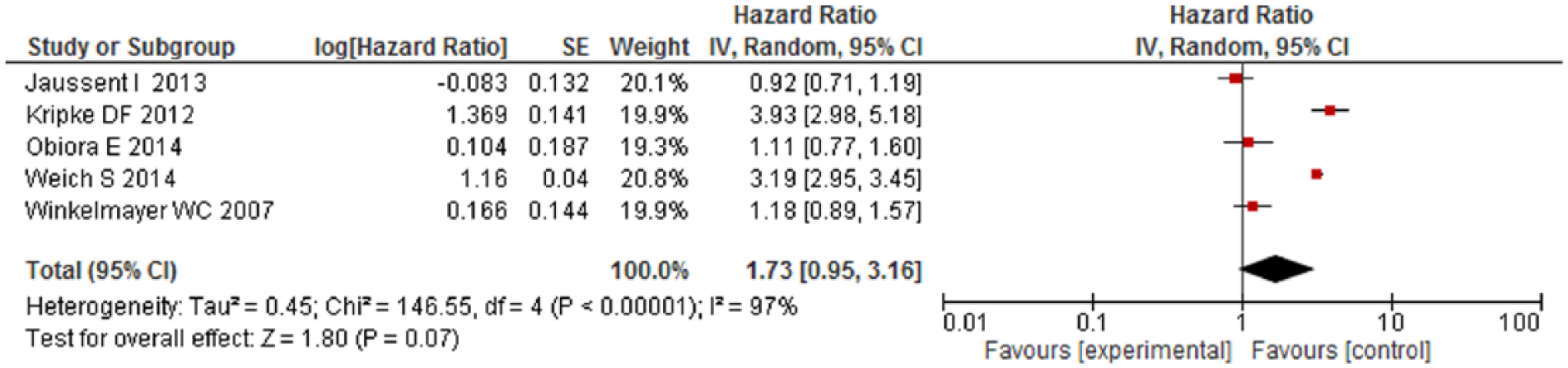
Mortality risk associated with Z-drugs (only) use.
Twelve studies provided duration of HA use, while remaining 13 studies reported HA use as ‘yes or no’ without giving details about the duration of use. Subgroups analysis showed 1.25 times higher risk of mortality among HA users reporting use as ‘yes’ compared with non-users reporting use as ‘no’. Subgroup analysis as per study location, study population, age, duration of HA use, duration of follow-up and quality of included studies, has been reported in Table 2. Of the different variables, only population characteristics—age was able to explain the study heterogeneity (p = 0.01, Table 2). Studies with mixed-age subjects had the highest risk of mortality (OR 1.74, 95% CI = [1.25, 2.44]). To assess whether any individual study or group of studies had a dominant effect on the meta-analytic HR, each study was excluded and its effect on the main summary estimate and Cochran’s Q test p for heterogeneity was evaluated. No individual study markedly affected the summary estimate or p value for heterogeneity but after removing six studies from the pool(Ahmad and Bath, 2005; Kripke et al., 2012; Mallon et al., 2002, 2009; Vinkers et al., 2003; Weich et al., 2014), risk estimate decreased but heterogeneity in the pooled HR was resolved.
Subgroup analysis.
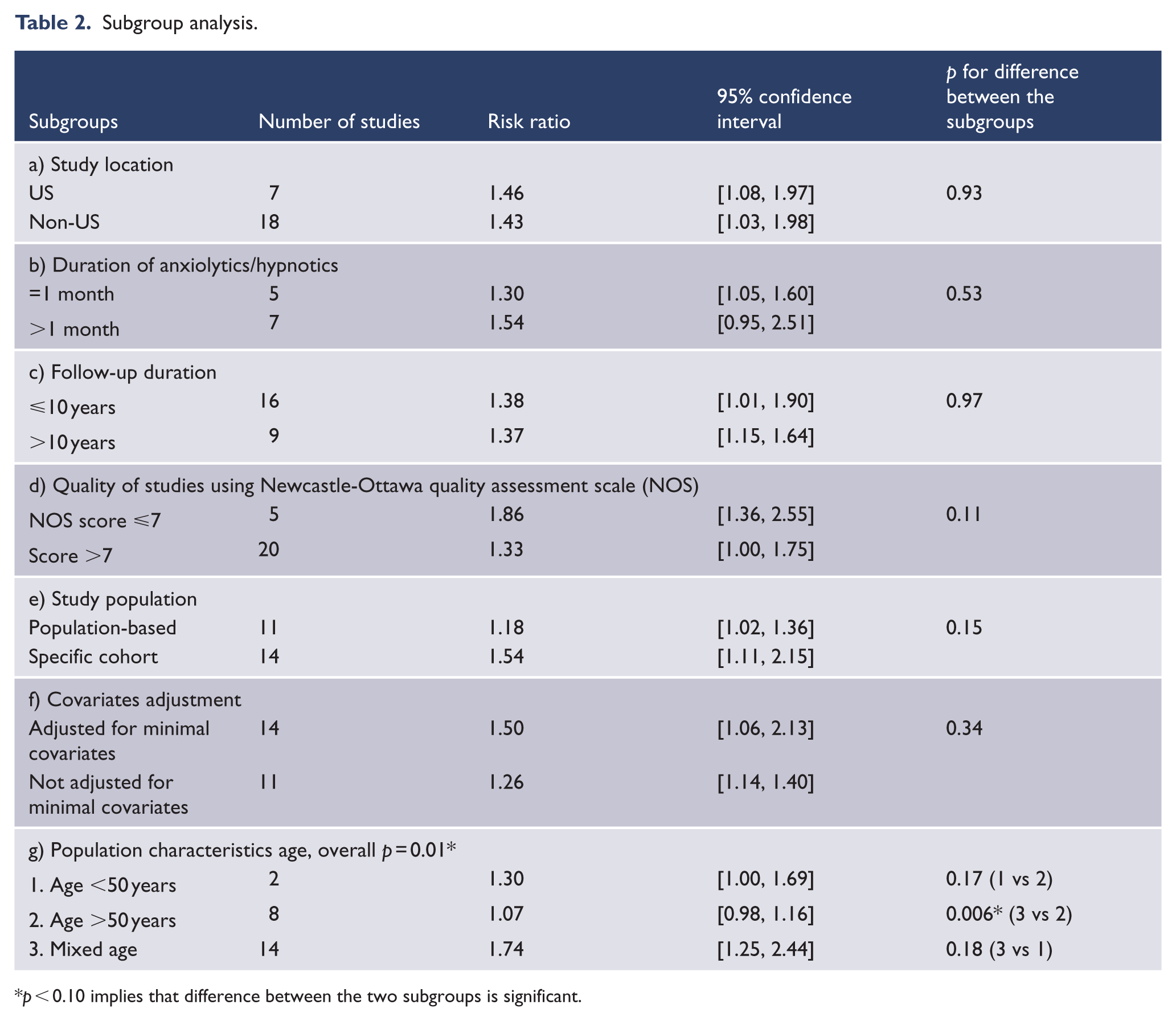
p < 0.10 implies that difference between the two subgroups is significant.
Publication bias
Quantitatively, there was no evidence of publication bias (Egger’s regression test: t = 0.66, df = 23, p = 0.51).
Mortality causes
Most studies reported all-cause mortality, but 12 studies reported cause-specific mortality with suicides, cardiovascular and cancer-related deaths among the common causes (Table 2).
Discussion
To the best of our knowledge, this is the first meta-analysis that examines the cumulative effects of HA on mortality risk. Our pooled results from 25 observational studies including 2,350,093 subjects showed that HA users had 43% higher risk of mortality during a follow-up period of 1–22 years. Similarly, higher mortality was found among population using benzodiazepine and Z-drugs, even though the effect did not reach statistical significance for Z-drugs likely due to limited number of studies. Mortality risk was significantly higher among men and women with no significant interaction of sex with the mortality. Based on limited evidence, higher deaths due to suicides, cancers and cardiovascular-related causes were observed among HA users; however, no casual association was reported. Increased suicides may be secondary to increased depression. Patients with depression have higher sleep disturbances, which may lead to higher sedatives and hypnotics use, but only limited studies controlled for depression (Jaussent et al., 2013; Mallon et al., 2002, 2009; Phillips and Mannino, 2005; Weich et al., 2014). Furthermore, HA may impair judgment and promote risky behavior including suicides (Drummer, 2002; Neutel and Patten, 1997). Depression may increase the mortality risk in subjects with underlying cardiovascular disease (Shah et al., 2014). Therefore, it is possible that subjects using HA had higher depression leading to increased cardiovascular deaths given that limited studies controlled for depression. HA may increase the risk of cancer by suppressing immune function or promoting viral infection (Kripke, 2008). These findings are supported by carcinogenic effects of hypnotics in rodents, by causing chromosomal damage (Kripke, 2008). However, it is possible that patients receiving hypnotics get more medical care and thus higher cancer detection than non-users.
A significant heterogeneity was observed in the outcome analysis. Heterogeneity could be due to several methodological issues including study design, differing study populations, follow-up duration, sample size, type and duration of HA use and differing adjusted confounders. Heterogeneity in our study could not be explained by the origin of study, study population, sex, duration of HA use, covariates adjustment, follow-up duration or quality of studies. On subgroup analysis by age, significant subgroup difference was seen in the outcome, suggesting that heterogeneity was possibly secondary to differing age of the study population. We also assessed whether any individual study or group of studies had a dominant effect on the meta-analytic HR. After removing six studies, mortality risk decreased to 21% and results were homogenous (Ahmad and Bath, 2005; Kripke et al., 2012; Mallon et al., 2002, 2009; Vinkers et al., 2003; Weich et al., 2014). Ahmad et al. analyzed data on community elderly (>65 years old) enrolled in longitudinal study of activity and aging, to identify risk factors for 15-years mortality (Ahmad and Bath, 2005). They used Cox Regression with genetic algorithms model for analysis in which they adjusted for age, stomach ache, chest pain, joint pain, handgrip strength, ability to raise GBP200 during emergency, self-rated activities, time since last visit to social worker and dentist, but did not adjust for actual medical illnesses. Kripke et al. (2012) reported mortality risk associated with currently popular hypnotics use only in a matched cohort study adjusted for age, gender, smoking, body mass index, ethnicity, marital status and alcohol use. They collected hypnotic information from electronic medical record which provided details about medication orders but not about dispensing or use by the subjects, which might have overestimated the risk. Mallon et al. (2002) studied association between sleep complaints and coronary artery disease-related mortality in a middle-age population. They reported increased mortality among sleeping pills users when adjusted for age; however, it was unknown if sleeping pills included sedatives and hypnotics only. It may be possible that sleeping pills included other sleeping promoting medications like antipsychotics or antidepressants. In another study on population-based cohort, authors reported increased mortality among sleeping pills users; however, the survey was done in 1983 without any descriptions of sleeping pills (Mallon et al., 2009). Mortality data were collected for the period of 1983–2003 with no follow-up on sleeping pills usage which might have changed significantly over the long duration of follow-up. Welch et al. (2014) found increased mortality risk among HA users over 7 years; however, they used primary care prescription database, which did not include psychiatrists’ prescription. Since they used prescription data, it was unknown if patient took the medications or not. Vinkers et al. (2003) found no association between benzodiazepine use and mortality risk; however, they enrolled only community-dwelling elderly individuals aged 85 years or older and used pharmacy registries for benzodiazepine use, which did not confirm if patient actually took the medications. Quality of included studies was reasonably good and ranged from 7–9 on Newcastle-Ottawa Quality Assessment Scale. However, due to significant heterogeneity and limited covariate adjustments among the studies, the overall quality of evidence according to the GRADE guidelines for association of HA is low to moderate. Therefore, the results need to be interpreted with caution.
The strengths of our meta-analysis include the detailed literature search, thorough screening of studies and quality assessment in duplicate, subgroup analysis and the good methodological quality of included studies. There are certain limitations of this meta-analysis which include analysis performed with study-level data rather than using an individual patient data, which would be the preferred methodology to account for patient-level characteristics. Different studies evaluated the mortality risk associated with different groups of HA, and usage varied in duration across the included studies. Another limitation is that adjustment for confounders varied across the studies. However, on subgroup analysis, there was no significant difference among studies who adjusted for major covariates versus who did not. Thus, it could not explain the heterogeneity.
Implications for clinical practice
Based on pooled results from 25 observational studies conducted across the world, there is low to moderate quality evidence (based on GRADE working group guidelines (Atkins et al., 2004)) suggesting that HA use is associated with increased mortality although the evidence supporting cause-specific mortality was limited. Therefore, physicians should carefully consider the increased mortality risk while prescribing HA drugs in addition to other risk factors and contraindications. Serious adverse effects and contraindications of sedatives and hypnotics include benzodiazepine abuse and dependence; concomitant alcohol, opioid and barbiturate abuse due to the risk of life-threatening interactions; pregnancy due to potential for harm in the unborn; myasthenia gravis, sleep apnea, bronchitis and chronic obstructive pulmonary disease (COPD) because of their muscle relaxant action, which may cause respiratory depression, and severe depression which may precipitate suicidal tendencies. Sedatives and hypnotics should be used with cautions in older age due to increased risk of dependence, and elderly are more sensitive to the adverse effects such as memory problems, daytime sedation, impaired motor coordination and falls. However, that does not mean that all patients should be treated identically or not be started on HA. Patients should be informed about the increased association of mortality with HA use. This further reiterates the importance of involving patients in decision making and explaining the merits of alternatives including non-pharmacological management for sleep and anxiety disorder so that patients can make well-informed decisions.
In conclusion, evidence warranting moderate confidence suggests an increased mortality with HA drugs use including benzodiazepine. Further well-designed studies adjusted for potential confounders are warranted to evaluate the temporal relationship between mortality risk and HA. Future studies should also focus on evaluating the underlying mechanism for increased mortality associated with HA use.
Footnotes
Acknowledgements
Ajay K. Parsaik has full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. Contents of this manuscript are solely the responsibility of the authors and do not represent the official view of any institutions. These data have been presented at the APA 168th Annual Meeting in Toronto, Canada, 16–20 May 2015.
Declaration of interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
