Abstract
Obsessive–compulsive disorder (OCD) is characterized by unwanted and persistent thoughts, images, or impulses (i.e. obsessions) and/or complex behaviours that include, among others, repetitive hand washing, checking, and ordering (i.e. compulsions). In order to meet current DSM-IV-TR diagnostic criteria for OCD, a patient has to exhibit either obsessions and/or compulsions that result in marked anxiety or distress, consume time (take more than 1 h a day), or significantly interfere with the person's normal routine, occupational (or academic) functioning, or usual social activities or relationships [1]. In a recent review of studies that employed the Composite International Diagnostic Instrument as a diagnostic instrument [2], the one month prevalence of obsessive–compulsive disorder in the general population ranged from 0.3 in Brazil to 3.1% in US samples [3].
A number of correlates and potential risk factors associated with the development of OCD have been identified through epidemiological studies, including demographic characteristics (such as age, gender, employment, marital condition, education, race, and socioeconomic status), genetic features (such as familial background), and environmental factors (such as pregnancy and birth complications, substance abuse, and life events, including trauma) [4]. Indeed, the role of environmental factors in the pathogenesis of OCD, particularly life events and traumatic experiences, is still a largely under studied [4,5]. In a two-stage epidemiological study, Valleni-Basile et al. [6] found that adolescents who had experienced more undesirable and less desirable life events, as measured by the Coddington Life Events Scale for Adolescents, were at higher risk for developing OCD later in life. Likewise, at least three studies found a significant link between victimization and the development of OCD in the general population [7–9]. For instance, experiencing a situation during which the person feared serious injury or death was associated with increased rates of several psychiatric disorders, including PTSD, major depressive episode, agoraphobia, social anxiety disorder, and OCD [7–9]. More strikingly, while PTSD acted as a strong mediator between victimization and most Axis I disorders, in one study rape remained a significant and independent predictor of OCD [8].
Although it had been suggested that traumatic events could shape the expression of OCD, only lately has evidence concerning the validity of a specific post-traumatic subtype of OCD emerged [10–13]. In a recent study, patients who developed OCD after traumatic events were compared to patients with non-trauma-related OCD and found to be characterized by a later age at onset of obsessions, higher prevalence of obsessions with aggressive content; increased levels of ‘suicidality’, greater severity of depression, anxiety, and aggression, symmetry, and miscellaneous obsessive–compulsive symptoms; increased rates of mood, anxiety, impulse control and tic disorders, and a positive family history for PTSD, major depressive disorder and generalized anxiety disorder, but not for OCD [14].
Based on these findings, we have proposed that OCD developing after a traumatic event may constitute an etiologically distinct subtype of OCD. To validate this incipient psychiatric phenotype, more studies investigating whether ‘post-traumatic’ OCD is also associated with particular genetic and neurocognitive characteristics or different treatment outcomes are badly needed [15]. With this in mind, the present study was designed to investigate whether OCD occurring simultaneously or after PTSD (PsT-OCD) is associated with distinctive executive, memory, and intellectual patterns of dysfunction, therefore adding greater validity to its existence. Further, since traumatic events have shown to be particularly detrimental to memory abilities [16,17], we predicted that individuals for whom traumatic events/PTSD were related to OCD (PrT-OCD and PsT-OCD) would be characterized by decreased learning abilities when compared individuals with OCD without a history of similar traumatic events//PTSD, and healthy controls.
Methods
Subjects
The data set was composed of 101 consecutive outpatients with OCD diagnosis according to the DSM-IV criteria. Patients were recruited from four different university outpatient clinics: Universidade Federal do Rio Grande do Sul (UFRGS) (n = 35), Universidade Federal da Bahia (UFBA) (n = 25), Universidade Federal do Rio de Janeiro (UFRJ) (n = 23), and Universidade de Pernambuco (UPE) (n = 18), each located in a different Brazilian city (including Porto Alegre, Salvador, Rio de Janeiro and Recife, respectively) and interviewed between August 2003 and August 2008. Healthy controls were selected among the health and administrative staff of University of São Paulo (n = 17). All institutions belonged to the neuropsychology core of the Brazilian Research Consortium on Obsessive–Compulsive Spectrum Disorders (CTOC), a collaborative enterprise of seven Brazilian centres aiming to investigate characteristics of OCD patients and their families, spanning all areas of OCD research [18].
To be included in the study, patients were required to have a DSM-IV diagnosis of OCD confirmed by the Structured Clinical Interview for DSM-IV Axis I disorders (SCID-I) [19]. Individuals suffering from schizophrenia, dementia, or any other condition that could limit their understanding of the procedures involved in the research protocol were excluded. The institutional review board of each institution involved in the CTOC approved this project. Accordingly, all participants signed written informed consent after a receiving a full description of the study and the assurance that the decision to participate in the project would not interfere with their access to treatment. A complete account of the methodology of the CTOC can be found elsewhere [18].
Psychiatric assessment
Research subjects were interviewed by clinical psychologists or psychiatrists with experience in the diagnosis and treatment of individuals with OCD. An array of standardized instruments was applied: Structured Clinical Interview for DSM Disorders (SCID), Yale–Brown Obsessive–Compulsive Scale (Y-BOCS) [20,21], and Beck Depression (BDI) and Anxiety Inventories (BAI) [22]. Examiners from different CTOC sites were intensively trained in two-day meetings twice a year (2004-2006) in order to guarantee satisfactory reliability among participants, which reached 96% [18].
Following the diagnostic algorithm provided by the SCID, which is based on DSM-IV criteria, a simultaneous diagnosis of OCD and PTSD was made only if the patient's obsessions and compulsions were not better explained by comorbid PTSD. For example, while a PTSD patient who develops compulsions to check the stove after a fire accident would not receive an additional diagnosis, someone with PTSD who exhibits clinically relevant washing compulsions after the same accident would be diagnosed with OCD.
Neuropsychological assessment
The C-TOC neuropsychological core selected a battery of neuropsychological tests aimed at evaluating different aspects of cognitive performance, including executive function, memory, and intellectual abilities. The evaluation of executive functioning included the administration of the Wisconsin Card Sorting Test (WCST), an instrument designed to assess, among other cognitive functions, set-shifting skills, an ability presumably related to dorsal aspects of the prefrontal cortex, and the Iowa Gambling Task (IGT), a test that is thought to tap decision-making biases and, as a consequence, to probe the more ventral and medial aspects of the prefrontal cortex. The battery for the assessment of memory comprised the Wechsler Memory Scale – R Logical Memory (LM), a test that investigates verbal memory, and the Brief Visual Memory Test Revised (BVMT-R), to evaluate visuospatial memory abilities. Intellectual skills were evaluated by means of the Wechsler Abbreviated Scale of Intelligence (WASI).
The methods of administration and correction of the neuropsychological tasks were standardized across the five CTOC sites. Examiners were re-trained and reassessed three times a year. A PhD-level neuropsychologist supervised the application of the neuropsychological battery on each site. A detailed description of each of these tasks is given below.
Executive function tests
Wisconsin Card Sorting Test
We employed the computerized version of the Wisconsin Card Sorting Test (WCST) [23]. In this test, subjects sit in front of a monitor exhibiting four stimulus cards with symbols that differ in colour, form, and number and are instructed to match each card of a series of 128 cards to one of the stimulus cards according to a specific criterion (colour, form, or number). Subjects are not informed of the matching criterion, but are told after each trial whether the match was correct or not. The criterion changes automatically after 10 consecutive correct matches. Perseveration consists in continuing to sort the cards according to a previous rule, even after the computer's negative feedback shows that this rule has been changed. The number of categories completed is the number of times an individual gave 10 consecutive correct responses.
Iowa Gambling Task
We employed the computerized version of the Iowa Gambling Task (IGT) [24]. In this test, subjects sit in front of a monitor exhibiting four decks of cards (termed A, B, C and D), each containing 40 cards. They are asked to choose a card from any of the four decks. The ‘disadvantageous’ decks (A and B) result in large short-term monetary gains but even larger long-term losses, whereas ‘advantageous’ decks (C and D) lead to small short-term monetary gains but even smaller long-term losses. Thus, the disadvantageous decks appear, at first, to be the best choice, at least with respect to the amount to be won. Individuals with orbitofrontal lesions tend to choose an excessive number of cards from decks A and B.
Memory tests
Wechsler Memory Scale – R Logical Memory
Immediate and 30-min delayed trials of the WMS-R LM were administered to test verbal memory [25]. Briefly, story A is read once to the subject, who then orally provides any information recalled. Next, story B is read twice to the examinee, with any recalled information provided after each reading. The examiner records the number of free recall units and thematic units. After 30 min of other testing, the subject is asked to provide any piece of information recalled from Story A and then Story B. A standard cue is provided if the subject has no recollection of a story. The recall scores are again recorded.
Brief Visual Memory Test – Revised
The BVMT-R is a test of visuospatial memory [26]. It consists of three learning trials in which six abstract designs are presented for 10 s. A learning score is calculated by summing all designs recalled across the three encoding trials (maximum score = 36). Recall is scored by the accuracy and correct placement of each figure (maximum score = 12). Delayed recall of the designs takes place after a 25-min delay. A recognition trial then followed, where subjects had to recognize six core stimuli from 12 designs (maximum score = 6).
Intellectual assessment
Wechsler Abbreviated Scale for Intelligence
The Wechsler Abbreviated Scale for Intelligence is a short test of adult intelligence composed of four subtests: similarities (evaluating abstract verbal reasoning), vocabulary (measuring the extent to which one has learned, been able to comprehend, and give definitions of some words), block design (assessing spatial perception, visual abstract processing and problem solving) and matrix reasoning (a subtest that evaluates nonverbal abstract problem-solving skills) [27].
Statistical analysis
The OCD sample was divided into three groups based on the presence of comorbid PTSD and its temporal relationship with OCD, i.e. ‘post-traumatic OCD’ (PsT-OCD) (n = 16), ‘pre-traumatic OCD’ (PrT-OCD) (n = 18), non-traumatic OCD (NT-OCD) (n = 67). Demographic, clinical, and neuropsychological features of these three OCD groups were compared to those of healthy controls (n = 17) using (χ2 test or Fisher's exact test for categorical variables and multivariate analysis of variance (MANOVA) for continuous variables. When the overall MANOVAs were significant (p < 0.05), post hoc pairwise comparisons were done using partitioned (χ2 test for categorical variables and least significant difference (LSD) or Dunnett's tests (according to the homogeneity of variance) for continuous variables.
Because we found, as expected, some significant demographic and psychopathological differences between the groups, we employed a multiple analysis of covariance (MANCOVA) with diagnostic group as the fixed factor, neuropsychological tests as dependent variables, and the above-mentioned MANOVA findings as covariates. Statistical tests were two-tailed, and p values < 0.05 were considered significant. Corrections for multiple comparisons were not made to avoid missing potential effects.
Results
General description of the sample
We recruited a total of 118 participants who fulfilled the inclusion criteria: 101 patients with OCD and 17 healthy controls. Patients with OCD were predominantly female (n = 59; 58.4%) with a mean age of 35.4 (±13.2) years. Of the participants 59 (59.6%) were single, 29 (28.7%) were married, 7 (6.9%) lived together with a partner, and 4 (4%) were divorced or separated. The volunteers with OCD had 13.0 ± 3.7 years of study and 83% of them belonged to classes B or C (A being the highest and E, the lowest), according to Brazilian norms. They reported the onset of obsessions and/or compulsions at age 13.8 ± 9.1 years. Their mean scores on the Y-BOCS, BDI, and BAI were, respectively, 26.9 ± 7.3 (severe obsessive–compulsive symptoms), 16.5 ± 11.2 (mild-moderate depression), and 17.0 ± 12.7 (moderate anxiety). In terms of comorbid disorders, 58.2% of the patients with OCD exhibited a current major depressive episode, 26.5% had specific phobia, 24.5% had social phobia, 17.8% had generalized anxiety disorder, and 11.2% presented panic disorder with or without agoraphobia. Most patients were already under psychological and/or pharmacological treatment. A description of the socio-demographic features of the healthy controls can be found in Table 1.
Comparison of socio-demographic features between the groups
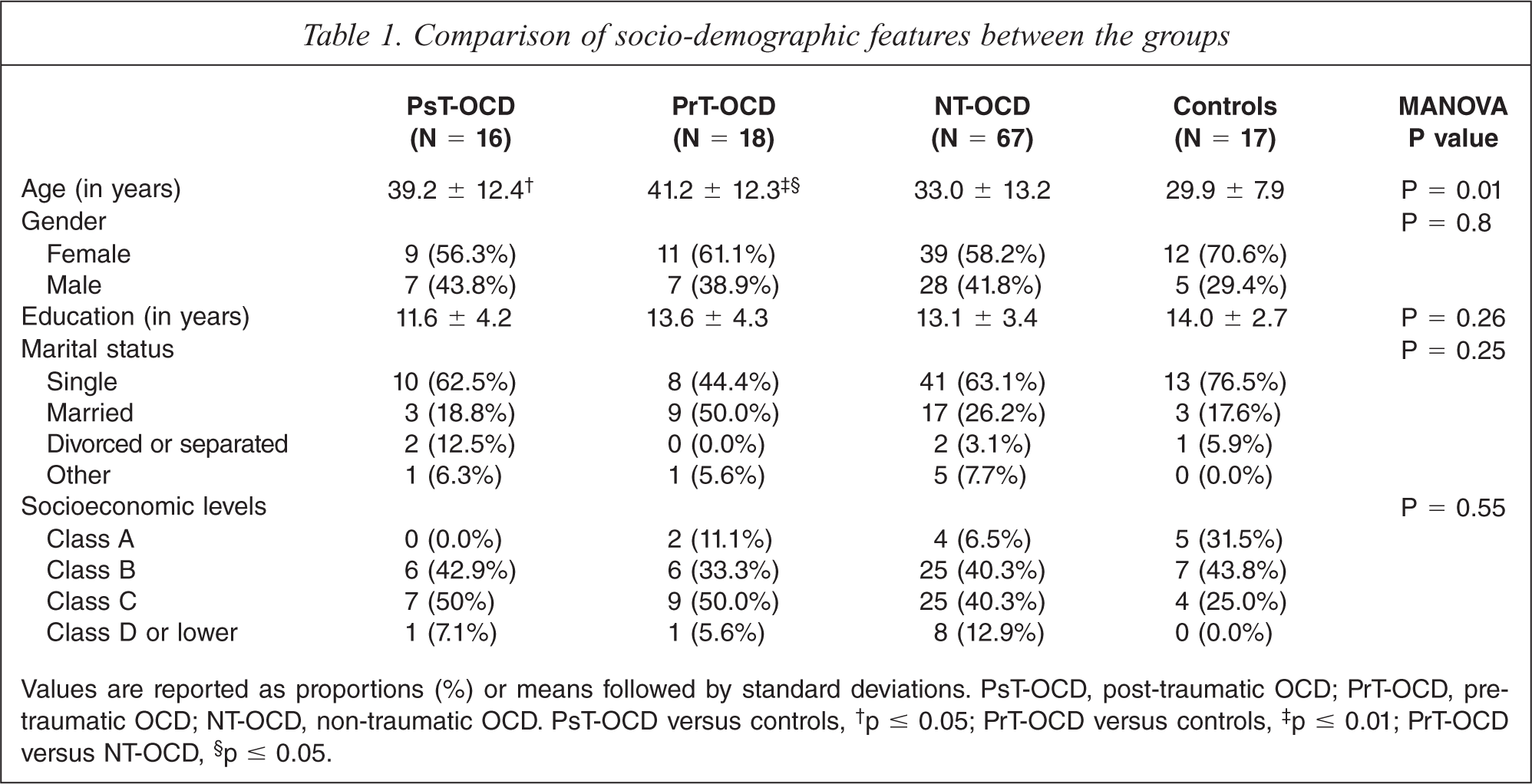
Values are reported as proportions (%) or means followed by standard deviations. PsT-OCD, post-traumatic OCD; PrT-OCD, pre-traumatic OCD; NT-OCD, non-traumatic OCD. PsT-OCD versus controls,† p ≤ 0.05; PrT-OCD versus controls,‡ p ≤ 0.01; PrT-OCD versus NT-OCD,§ p ≤ 0.05.
Comparison between OCD groups and healthy controls
The clinical (PsT-OCD, PrT-OCD, and NT-OCD) and the healthy control groups had their socio-demographic, clinical and neuropsychological characteristics compared to one another as seen in Tables 1, 2 and 3, respectively.
Comparison of psychiatric features between the groups
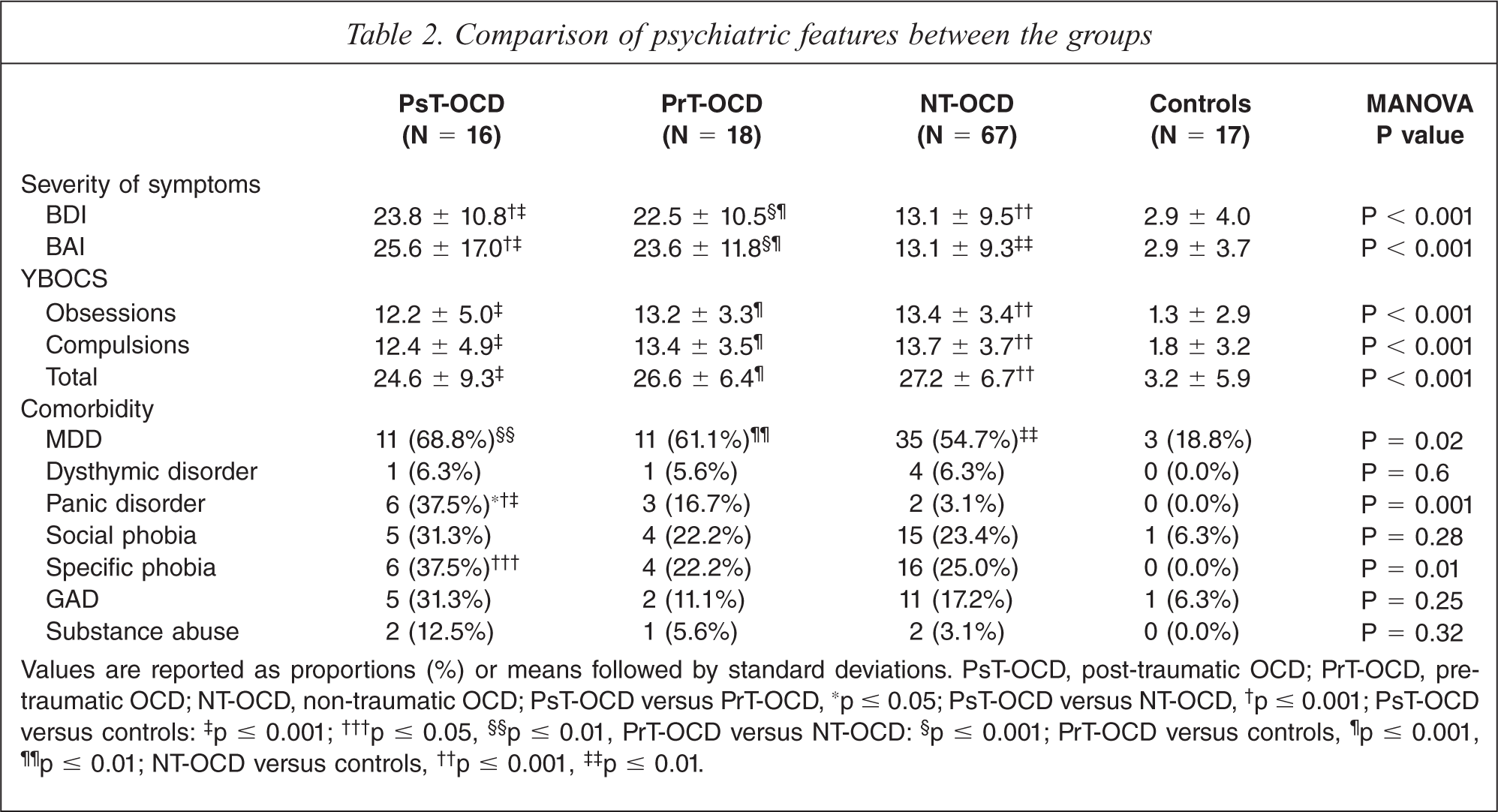
Values are reported as proportions (%) or means followed by standard deviations. PsT-OCD, post-traumatic OCD; PrT-OCD, pre-traumatic OCD; NT-OCD, non-traumatic OCD; PsT-OCD versus PrT-OCD,∗ p ≤ 0.05; PsT-OCD versus NT-OCD,† p ≤ 0.001; PsT-OCD versus controls:‡ p ≤ 0.001;††† p ≤ 0.05,§§ p ≤ 0.01, PrT-OCD versus NT-OCD:§ p ≤ 0.001; PrT-OCD versus controls,¶ p ≤ 0.001,¶¶ p ≤ 0.01; NT-OCD versus controls,†† p ≤ 0.001,‡‡ p ≤ 0.01.
Comparison of neuropsychological performance between the groups
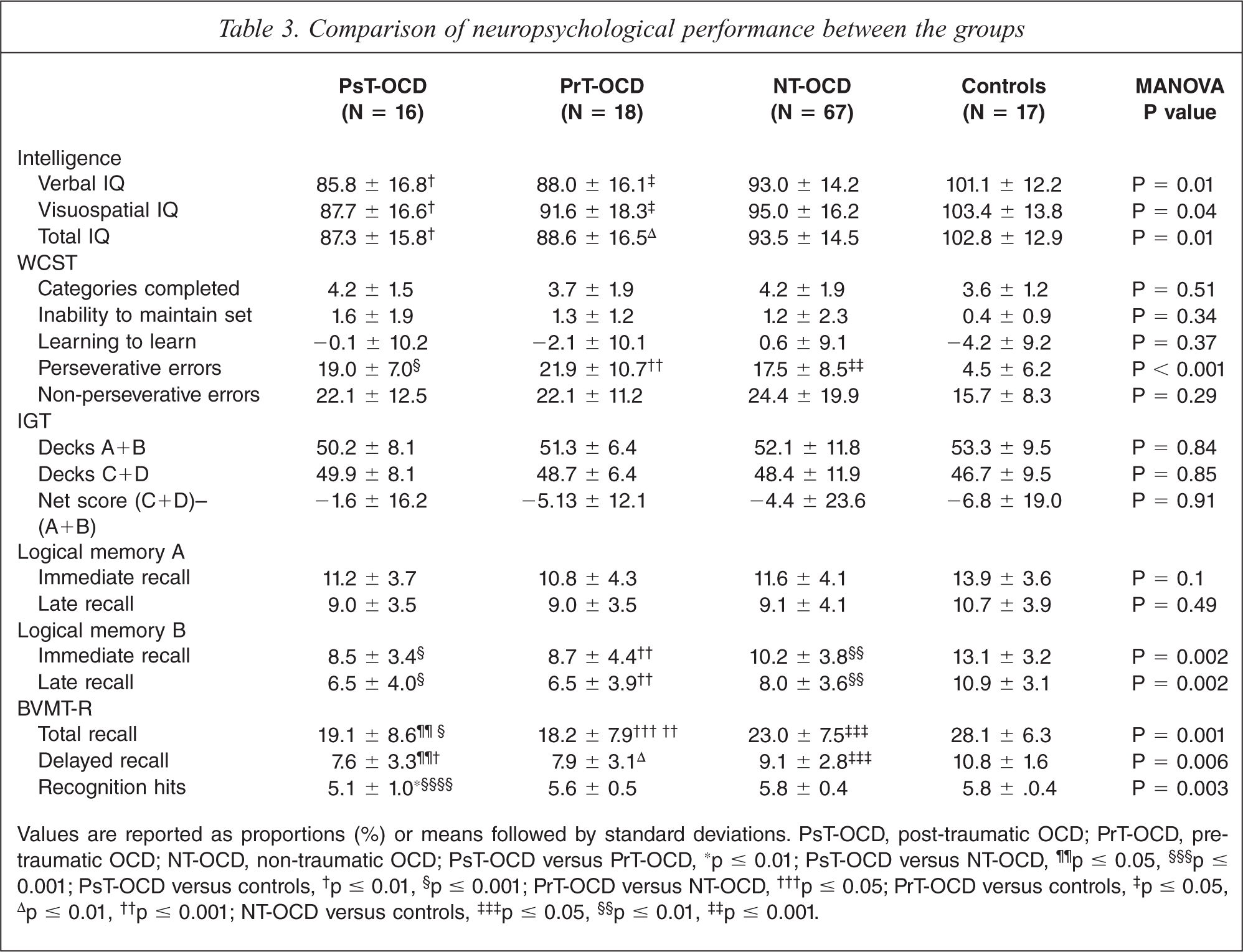
Values are reported as proportions (%) or means followed by standard deviations. PsT-OCD, post-traumatic OCD; PrT-OCD, pre-traumatic OCD; NT-OCD, non-traumatic OCD; PsT-OCD versus PrT-OCD,∗ p ≤ 0.01; PsT-OCD versus NT-OCD,¶¶ p ≤ 0.05,§§§ p ≤ 0.001; PsT-OCD versus controls,† p ≤ 0.01,§ p ≤ 0.001; PrT-OCD versus NT-OCD,††† p ≤ 0.05; PrT-OCD versus controls,‡ p ≤ 0.05,Δ p ≤ 0.01,†† p ≤ 0.001; NT-OCD versus controls,‡‡‡ p ≤ 0.05,§§ p ≤ 0.01,‡‡ p ≤ 0.001.
Because there were some significant differences between the groups, spanning current age, severity of symptoms, patterns of comorbidity, and intelligence levels (Tables 1, 2 and 3), comparisons were made using multiple analysis of covariance (MANCOVA) with the diagnostic group as the fixed factor, neuropsychological tests as dependent variables, and age at presentation, Beck's anxiety and depression scores, and IQ scores as covariates. As seen in Table 4, only age of presentation reached a level of significance that allowed it to be kept in the MANCOVA model.
Multivariate analysis of variance of features that differed between groups

Although controlling for age attenuated some of the findings described in the previous model, most differences remained significant (Table 5). In our final MANCOVA model, while patients with PsT-OCD, PrT-OCD and NT-OCD exhibited more perseverative errors and verbal and visuospatial memory problems than healthy controls, individuals with PsT-OCD and PrT-OCD seemed to be particularly impaired from the cognitive point of view. Of note, patients with PsT-OCD exhibited fewer recognition hits than patients with PrT-OCD.
Comparison of neuropsychological performance between the groups controlling for age at presentation
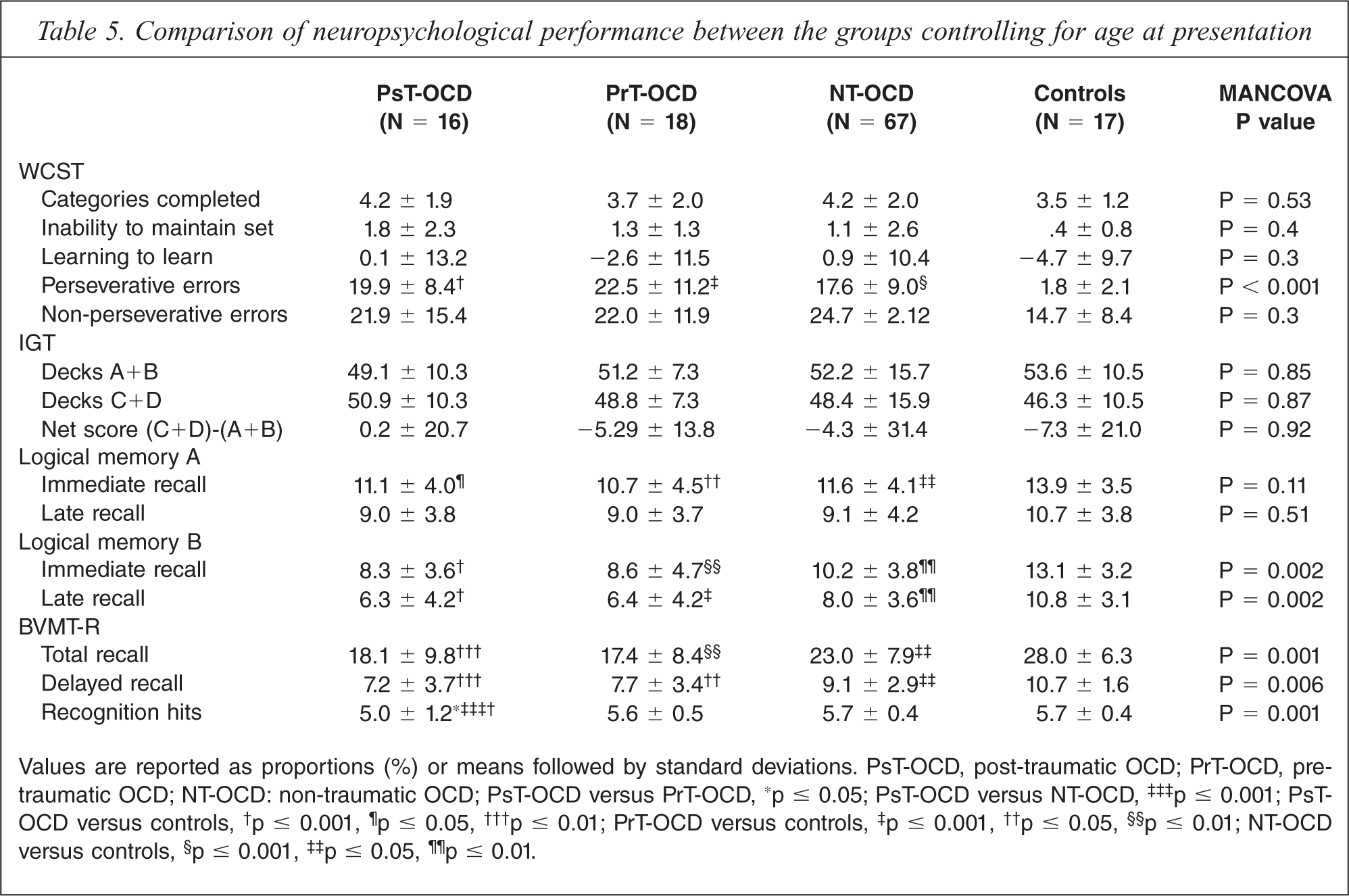
Values are reported as proportions (%) or means followed by standard deviations. PsT-OCD, post-traumatic OCD; PrT-OCD, pre-traumatic OCD; NT-OCD: non-traumatic OCD; PsT-OCD versus PrT-OCD,∗ p ≤ 0.05; PsT-OCD versus NT-OCD,‡‡‡ p ≤ 0.001; PsT-OCD versus controls,† p ≤ 0.001,¶ p ≤ 0.05,††† p ≤ 0.01; PrT-OCD versus controls,‡ p ≤ 0.001,†† p ≤ 0.05,§§ p ≤ 0.01; NT-OCD versus controls,§ p ≤ 0.001,‡‡ p ≤ 0.05,¶¶ p ≤ 0.01.
Further, we also found that BVMT recognition hits correlated significantly both with WCST categories completed (rho = 0.72; p = 0.001) and perseverative responses (rho = 0.61; p = 0.01) in patients with PsT-OCD, but not in healthy controls (rho = 0.15; p = 0.57 and rho = −0.21; p = 0.4, respectively), NT-OCD (rho = 0.16; p = 0.19 and rho = 0.01; p = 0.89, respectively), and PrT-OCD (rho = 2.9; p = 0.28 and rho = −0.16; p = 0.57, respectively).
The severity of obsessive–compulsive symptoms (Y-BOCS obsessions, compulsions, and total scores) correlated significantly with WCST perseverative responses in patients with NT-OCD (rho = 0.47; p = < 0.001, rho = 0.47; p < 0.001, and rho = 0.5; p < 0.001, respectively) and PrT-OCD (rho = 0.45; p = < 0.05, rho = 0.58; p = 0.01, and rho = 0.56; p = 0.01, respectively), but not in PsT-OCD (rho = 0.26; p = 0.35; rho = 0.11; p = 0.71, and rho = 0.2; p = 0.49, respectively).
Finally, we also investigated the relationship between OCD symptom severity with the recognition hits across different groups, but only one significant result emerged, i.e. Y-BOCS compulsions scores correlated negatively with BVMT-R recognition hits in patients with PrT-OCD (rho = −0.57; p = 0.02).
Discussion
We found that patients with OCD were characterized by poor set-shifting abilities and impaired verbal and visuospatial memory even after controlling for age, education, intellectual level and severity of emotional (i.e. depression and anxiety) symptoms. Further, impaired set-shifting abilities were found to correlate with the severity of obsessive–compulsive symptoms in all groups of patients with OCD, with the exception of PsT-OCD. Also, patients who developed OCD after PTSD were distinctively characterized by impaired visuospatial recognition, a dysfunction that was found to correlate with poor set-shifting abilities, but not to severity of OCD, in this particular group of patients.
In our study, all OCD samples exhibited higher average perserverative errors than the healthy control group, a result that could not be ascribed to differences in terms of age, education, intellectual level and severity of emotional symptoms. Also, the severity of obsessive–compulsive symptoms (i.e. obsessions, compulsions, and total scores) correlated significantly with perseverative responses in the WCST in the NonT-OCD and the PrT-OCD, but not in PsT-OCD. Accordingly, it seems that only subgroups of patients with OCD exhibit increased perseverative errors/responses in the WCST. For example, while some studies have suggested that associated depression [28,29], schizotypal symptoms [30,31], or intellectual impairment [32] are responsible for this association, other investigations, such as the current study, support the idea that patients with OCD display a primary deficit of set-shifting ability that is independent of other confounding factors. As it was previously proposed, although poor WCST performance may be worsened by a number of confounding factors, it may also be negatively influenced by certain clinical features such as symmetry/ordering symptoms [33] or genetic polymorphisms (DA-48G, DSer9Gly, 5-HTT102C and 5-HTT267C) [34] that may be present in individuals with OCD free from other different comorbidities.
We found that all groups of patients with OCD exhibited a reduction in immediate and delayed verbal episodic memory for short prose passages (LM from the WMS-R) when compared to healthy controls. These findings remained significant after controlling for the effects of age, education, intellectual levels, and severity of emotional symptoms and had been already reported in sub-clinical checkers [35] and in other samples of patients with OCD [36,37]. Of note, severity of obsessive–compulsive symptoms correlated with impairment in logical memory, particularly in patients with NonT-OCD. Recently, however, Andrés and coworkers argued that, at least in children with OCD, impaired delayed recall might be ascribable to comorbid depression [38] and resistant to treatment [39]. Therefore, if left untreated, it is possible that episodic memory impairment of children with OCD might consolidate later in life independently of depression.
Traditionally, verbal memory impairment in patients with OCD has been assessed on the basis of list-learning (California Verbal Learning Test, (CVLT) CVLT or others) or paired associates paradigms [40]. Nevertheless, the recall of orally presented stories, such as employed in the current study, has been shown to predict every day memory problems in a wide range of clinical conditions [41]. The inability of patients with OCD to memorize complex verbal information may be related to their reduced capacity for semantic processing [42,43]. As suggested by Exner et al. [37,44], patients with OCD might miss the overall semantic essence of the story while trying to keep track of its every single detail.
On the other hand, patients with OCD, either trauma- or non-trauma-related, displayed significantly reduced total and delayed recall of visuospatial information when compared to healthy controls. As reported for set-shifting abilities and verbal memory functioning, results for visuospatial memory could not be ascribed to differences in terms of age, education, emotional problems, or intellectual levels. Although not previously employed in OCD, the BVMT results replicated visuospatial memory deficits seen in previous studies that used the WAIS-R Visual Construction, the Benton Visual Retention, or the Rey Complex Figure Test [45]. Further, the fact that all OCD groups, with the exception of patients with PsT-OCD, exhibited preserved recognition hits suggests that patients with ‘classical’ OCD (PrT-OCD and NonT-OCD) display a typical fronto-striatal retrieval deficit syndrome, instead of a simple impairment in the ability to store visuospatial information [46].
The existence of a post-traumatic subtype of OCD has been speculated for decades [47], though no previous study has attempted to delineate its differential pathophysiology. In order to investigate the etiological role played by traumatic events in certain cases of OCD, we focused our study on the neuropsychological functioning of patients with PsT-OCD. As hypothesized, patients with PsT-OCD were found to be distinctively impaired, i.e. they were characterized by significantly lower visuospatial recognition hits than healthy controls, NonT-OCD, and more importantly PrT-OCD (Table 5). Indeed, the comparison between PsT-OCD and PrT-OCD allowed us to control for the impact of PTSD in brain functioning, i.e. lower visual recognition could not be ascribed to the presence of PTSD in general but to OCD occurring after PTSD. Whether lower visual recognition is a risk factor, a correlate, or a consequence of PsT-OCD, however, is not possible to answer with a cross-sectional study like this.
Further, we also found that BVMT recognition hits correlated significantly both with WCST categories completed and perseverative responses in patients with PsT-OCD, but not in healthy controls, non-trauma-related OCD and PrT-OCD. Taken together, these findings probably suggest that PsT-OCD may be related to greater right hippocampal dysfunction, and secondarily to the impaired set-shifting abilities that are thought to underlie obsessive–compulsive symptoms. These results are similar to those reported in individuals with PTSD, who exhibited impaired verbal memory [16,48] and left hippocampal dysfunction [49–51] in several studies. We can only speculate about the significance of these findings. For example, while left hippocampal dysfunction could confer greater susceptibility to PTSD, right hippocampal dysfunction may be a marker for the development of obsessive–compulsive symptoms in the aftermath of PTSD.
The current study has several limitations that need to be taken into account. First, the number of patients with pre- and post-traumatic OCD was relatively small. Clearly, a larger sample would contribute greatly to elucidate the relationship between neuropsychological deficits and different clinical groups. Nevertheless, one should consider that we are dealing with an atypical, probably rare, OCD phenotype. Indeed, we have made the best of our efforts to identify the greatest possible number of patients with PTSD-related OCD, a strategy that was only possible in the context of a multicentre study conducted by a research consortium such as ours.
Second, the fact that our study involved different research groups may raise questions regarding the reliability of the methods of evaluation of cognitive abilities employed by our neuropsychologists. In fact, although we did not test inter-rater reliability of neuropsychological assessments formally, we tried to circumvent this potential limitation by adopting strict instructions, routine calibration meetings, and regular supervision by an expert neuropsychologist who would visit each centre to check for data quality.
Third, since we have reported data on the neuropsychology of a clinical sample of patients with OCD, most of our research subjects were under pharmacotherapy, including serotonin re-uptake inhibitors (as the main therapeutic anchors), atypical antipsychotics and, in cases of treatment resistance or comorbid disorders, benzodiazepines (especially clonazepam). It must be underlined, though, that traditional anti-OCD treatment did not seem to affect neuropsychological performance in other OCD adult samples [52]. Nevertheless, despite these reassuring results, we acknowledge that future studies on the neuropsychology of PsT-OCD should ideally investigate non-clinical samples or drug naïve patients.
Fourth, we did not employ any instrument to assess for the severity of PTSD symptoms. It has already been demonstrated that severity of PTSD has a negative impact on measures of cognitive performance, mainly memory tasks [53]. Therefore, we cannot exclude the possibility that the lower number of recognition hits obtained by PsT-OCD patients might reflect, perhaps, more severe PTSD symptoms. Along the same line, it could be suggested that our PsT-OCD group is not OCD but rather a PTSD subtype. However, since we did not include a sample of patients with PTSD without comorbid OCD in our study, we were unable to look for potential cognitive differences and similarities between patients with PsT-OCD and PTSD and support the existence of a ‘pseudo-OCD’ subtype of PTSD. Although this type of comparison would be interesting, the final number of experimental groups (five) could be considered excessively high for a study that already has three comparison groups.
Finally, since PTSD and OCD (especially PsT-OCD) share a substantial number of features (including unpleasant, vivid, repetitive and intrusive cognitions [54] and behaviours performed in an attempt to increase the sense of safety [55]), it could be argued that we have mislabelled PTSD as PsT-OCD. Nevertheless, it should be mentioned that PsT-OCD and PTSD could be differentiated on the basis of a number of additional clinical features. For example, while PTSD patients ruminate about past traumatic events, OCD patients typically develop a range of behaviours aimed at preventing future harm. Further, whilst numbing or persistent symptoms of increased arousal are critical features for patients with PTSD, they are not needed for OCD diagnosis. Lastly, and most importantly, according to DSM-IV-TR, patients with PTSD should only receive an additional OCD diagnosis if obsessions and compulsions are unrelated to trauma (e.g. symmetry and ordering symptoms after a fire accident).
Despite these shortcomings, the current study revealed that OCD patients in general were characterized by impaired set-shifting abilities, verbal and visuospatial memory that were independent from age, education, depression and intellectual levels. Further, impaired set-shifting abilities were found to correlate with the severity of obsessive–compulsive symptoms in all groups of patients with OCD, with the exception of PsT-OCD. Patients who developed OCD after PTSD were distinctively characterized by impaired visuospatial recognition, a dysfunction that was found to correlate with poor set-shifting abilities in this particular group of patients.
Footnotes
Acknowledgements
The authors are indebted to Samantha N. Santos and Curt H. Menezes for helping with the neuropsychological assessment.
