Abstract
Mood stabilizers are effective treatments for bipolar affective disorder for both acute episodes and for relapse prevention. They are often part of long-term treatment plans for those suffering from bipolar disorder [1]. These mood stabilizer treatments include lithium carbonate and the anti-epileptic drugs (AEDs) sodium valproate, carbamazepine, and lamotrigine. Before the availability of these effective treatments of bipolar disorder this illness often resulted in long admissions to psychiatric institutions, significant functional impairment and even death from ‘mental exhaustion’ [2]. Research has suggested that, over the long term, bipolar disorder may cause impairment of psychosocial function approaching that of schizophrenia [3]. Thus the recent improvement in the range of effective treatments both for acute episode and relapse prevention has significantly improved the lives of those suffering from bipolar disorder. For women with bipolar disorder this has meant the capacity to form stable partnerships and the consideration of having children.
Evidence is accumulating concerning the interplay between bipolar disorder and pregnancy. A recent prospective study found that for bipolar disorder the risk of relapse in pregnancy was 85.5% for those who ceased their medication compared to 37% in those who continued their medication in pregnancy. In addition those who ceased their mood stabilizers in pregnancy spent a greater proportion of their pregnancy unwell [4]. An earlier retrospective study showed that in the postpartum period risk of bipolar relapse may increase sharply [5]. Both studies called for particular care concerning rapid discontinuation of mood stabilizers. In a separate study, women with a previous history of puerperal psychosis following childbirth were shown to reduce their risk of relapse to approximately 10% if they used prophylactic lithium in late pregnancy [6].
Studies which have examined the effect of untreated mental illness in pregnancy have consistently found associations with poorer child biological, psychological and developmental outcomes for a range of illnesses from depression, anxiety through to schizophrenia [7–9]. Maternal mental illness also impacts on maternal sensitivity which is one of the predictors of infant attachment [7,10]. As this evidence accumulates and is disseminated, the trend towards more assertive mental health interventions in pregnancy is likely to continue.
Clinical decision making regarding mood stabilizers in pregnancy can be conceptualized as balancing risks. There are risks for the mother including that of deterioration or relapse without the medication. These are balanced with the risks to the foetus and infant from exposure to the medication, but also the risks identified with having an unwell or untreated mother.
There are recent studies into treatments for bipolar disorder which have suggested a role for atypical antipsychotic medication, previously used as adjunctive treatments, as monotherapy [1]. Around pregnancy caution is required as recent reviews yield significantly less information on malformation, pregnancy and longer term developmental risks for this class of medication than for the mood stabilizers [11–13]. In contrast there is a significant amount of research into the anti-epileptic mood stabilizers (AEDs), most of which has come from large overseas pregnancy and birth defect registries; there has also been one international registry and a number of small studies into lithium. Some of the risks shown by these registries include both structural and neurodevelopmental teratogenicity, pregnancy complications and neonatal complications. It should be noted that most AED studies are of women with epilepsy, and include agents not used in the treatment of bipolar disorder. Epilepsy itself may affect outcome in these studies via genetics and the effects of seizure activity on foetal growth and development.
This review was one of three separate systematic reviews which respectively examined use of anti-depressants, antipsychotics and mood stabilizers in pregnancy. The reviews were undertaken as a joint initiative across the perinatal mental health services in Melbourne under the auspices of the Melbourne Perinatal Psychotropic Review Group.
Material and methods
Strategies used to identify eligible studies included a systematic search of electronic databases, a search of the grey literature (bibliographies of relevant reference books and review articles) and a hand search of the references of all included articles.
The electronic databases searched were MEDLINE, PsycINFO and Embase. These were initially searched from 1950 until June 2009. Additional electronic resources such as the Cochrane library and DARE were also searched. From the former we identified a Cochrane review of developmental outcomes for AEDs from 1966 until December 2003; while we searched this literature it was not included in our final results given this has been comprehensively covered within the Cochrane review [14]. Therefore this paper covers all outcomes for lithium, AED malformation, pregnancy complications and neonatal complications for the complete time period and AED developmental outcomes from 2004 onwards only. The search terms included: abnormalities (drug induced), anticonvulsants, antimanic agents, female, humans, lithium, lithium compounds, pregnancy, pregnancy complications, pregnancy outcome, triazines and valproic acid. Boolean operators were used with care and the searches run many times over 12–18 months (early 2008–mid 2009). All citations identified were initially reviewed as abstracts and if found to meet inclusion criteria for this review the article obtained and read by the authors of this review.
Studies met inclusion criteria if they were original research, English-language, human studies of malformation rates, perinatal outcomes (pregnancy and neonatal) and child neurodevelopmental outcomes following the use of sodium valproate, carbamazepine, lamotrigine or lithium in pregnancy. Review articles were excluded. As randomization in pregnancy presents ethical problems, the best available NHMRC level of evidence was III-2 (controlled cohort studies). We noted whether such studies were prospective, although as the window for true prospective recruitment in pregnancy is narrow, many studies combined different degrees of ‘prospectiveness’. Levels III-3 (uncontrolled cohorts) and Level IV (case series) were also examined, although for the AED malformation studies we did not include case studies in the final review and tabulation process due to the large number of prospective case controlled studies available.
Results
Malformations for AEDs
Case reports
Case reports were excluded from this review (see Methods).
Cohort studies
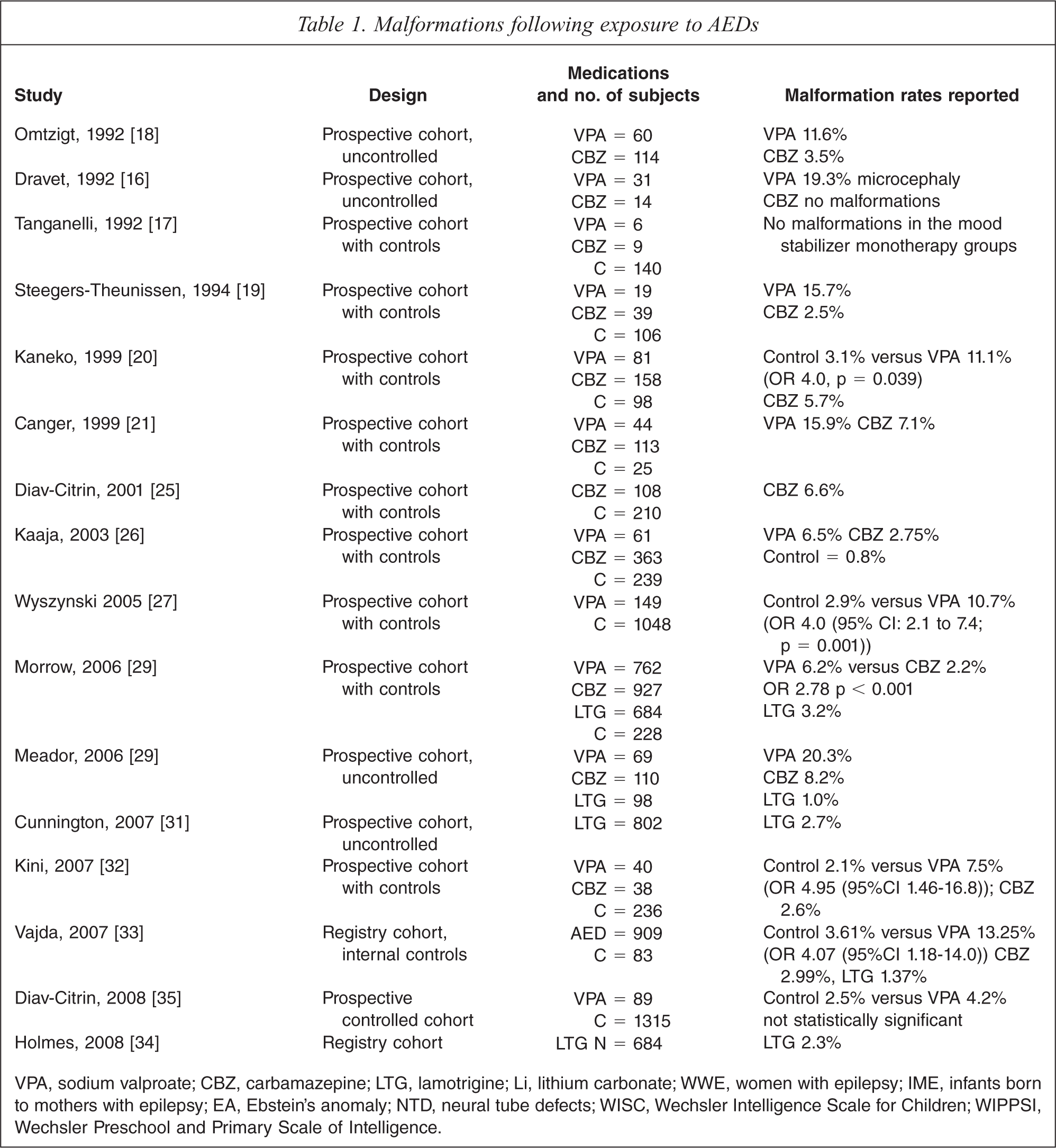
Prospective cohort with controls: 10 studies were identified and are included in Table 1.
Malformations following exposure to AEDs
VPA, sodium valproate; CBZ, carbamazepine; LTG, lamotrigine; Li, lithium carbonate; WWE, women with epilepsy; IME, infants born to mothers with epilepsy; EA, Ebstein's anomaly; NTD, neural tube defects; WISC, Wechsler Intelligence Scale for Children; WIPPSI, Wechsler Preschool and Primary Scale of Intelligence.
Prospective cohort without controls: 4 studies were identified and are included in Table 1.
Registry Cohort: 2 studies were identified and are included in Table 1.
More than 20 studies were identified for the commonly used mood stabilizer AEDs sodium valproate, carbamazepine and lamotrigine [15–36]. There is a degree of consistency of findings, in spite of variations in methodology and in the definition of malformation used across the studies. The malformations studied included neural tube defects for both sodium valproate and carbamazepine [18,22,26]. Craniofacial, cardiac and limb defects were also identified, as was a range of minor malformations [15,23,24,30,36].
Table 1 presents the 16 studies which yielded malformation rates for comparison [16–21, 25–29, 31–35]. There was a consistent finding of higher malformation rate with sodium valproate than with either carbamazepine or lamotrigine. Four of these studies identified doses greater than 1000 mg of sodium valproate as increasing further the risk of malformation [18,20,33,35]. Only one study of lamotrigine [29] noted a dose effect for malformation although another study[31] found no such effect up to 400 mg. The preponderance of orofacial clefts among malformations with lamotrigine was reported in one study [34] but refuted in another [37].
All studies were in women with epilepsy, although some included women treated for other indications. The role that genetic factors and seizure activity that are associated with epilepsy and the implication for malformation rate is unclear. However, studies which have used women with epilepsy not on AEDs in pregnancy have lower malformation rates compared to those with epilepsy and AED exposure in pregnancy [38].
A recent review of the results of the North American, UK and Australian AED registries has found the overall major malformation rate was 2.9% for carbamazepine, 8.7% for sodium valproate and 2.7% for lamotrigine these are similar findings to an earlier meta-analysis [39,40].
Perinatal outcomes (pregnancy and neonatal) for AEDs
Cohort studies
Prospective cohort with controls: 10 studies were identified and are included in Table 2.
Perinatal outcomes following exposure to AEDs

VPA, sodium valproate; CBZ, carbamazepine; LTG, lamotrigine; Li, lithium carbonate; WWE, women with epilepsy; IME, infants born to mothers with epilepsy; EA, Ebstein's anomaly; NTD, neural tube defects; WISC, Wechsler Intelligence Scale for Children; WIPPSI, Wechsler Preschool and Primary Scale of Intelligence.
Table 2 presents the findings of the 10 studies identified for perinatal outcomes, including pregnancy and neonatal complications, associated with the AEDs [22,41–51]. Compared with the malformation data these studies were smaller, with findings for mood stabilizer monotherapy failing to reach statistical significance, and with the equivocation of several negative studies [44–47,51]. However, a significant and replicated association was found between carbamazepine and reduced head circumference [42,48], with findings also for lower birth weight and length [48,49]. A significant association was found between sodium valproate and neonatal hypoglycaemia, with weaker findings for hepatic dysfunction, withdrawal symptoms and reduced birth dimensions [41,43,50]. One cohort study challenged the association, drawn mostly from case reports, between carbamazepine and neonatal bleeding [51].
Neurodevelopmental outcomes for AEDs
Case Reports
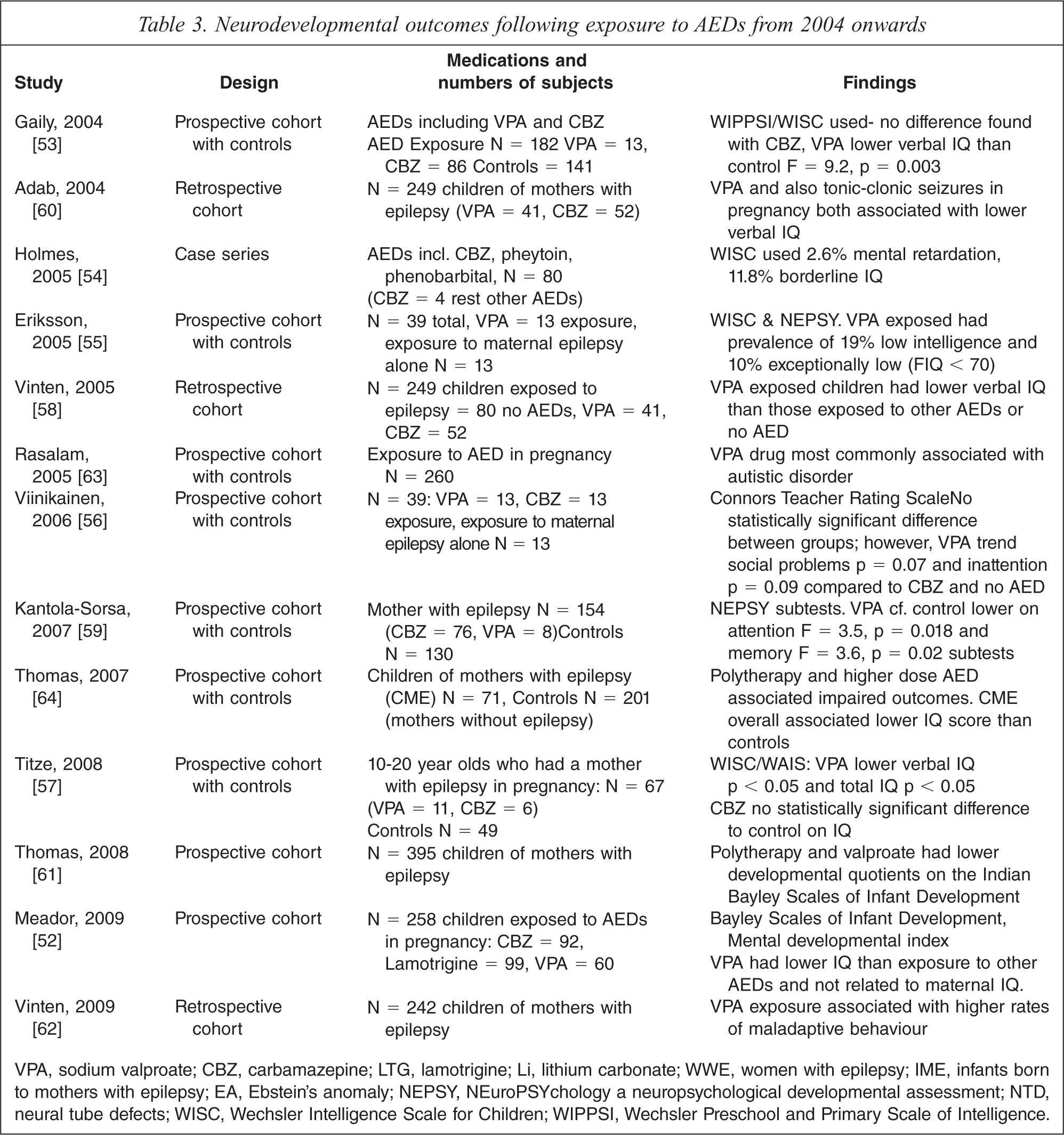
One case series from 2004 onwards was identified and is included in Table 3.
Neurodevelopmental outcomes following exposure to AEDs from 2004 onwards
VPA, sodium valproate; CBZ, carbamazepine; LTG, lamotrigine; Li, lithium carbonate; WWE, women with epilepsy; IME, infants born to mothers with epilepsy; EA, Ebstein's anomaly; NEPSY, NEuroPSYchology a neuropsychological developmental assessment; NTD, neural tube defects; WISC, Wechsler Intelligence Scale for Children; WIPPSI, Wechsler Preschool and Primary Scale of Intelligence.
Cohort studies (from 2004 onwards)
Prospective cohort with controls: 7 studies were identified and are included in Table 3.
Prospective cohort without controls: 2 studies were identified and are included in Table 3.
Retrospective cohort: 3 studies were identified and are included in Table 3.
Therefore 13 studies were identified in total from 2004 onwards examining developmental outcomes for children exposed to AEDs in pregnancy [52–64]. These are described in Table 3. Although methodologically diverse, all of the 11 studies which examined the effect of sodium valproate on child development found an association with poorer developmental outcomes. These ranged from a global reduction in IQ through to specific developmental concerns such as lower verbal IQ, autism, memory and attention [53,55,56]. One study found an increase in myopia among children exposed to AED in utero [65]. 11 of the studies identified included carbamazepine and of these none found an association with carbamazepine and poorer developmental outcomes. Only one study identified included lamotrigine and no effect on development was found [52].
Studies which examined a dose effect and polytherapy found both doses of sodium valproate over 1000 g and polytherapy associated with later poorer neurodevelopmental outcomes [64].
Lithium carbonate
Case reports
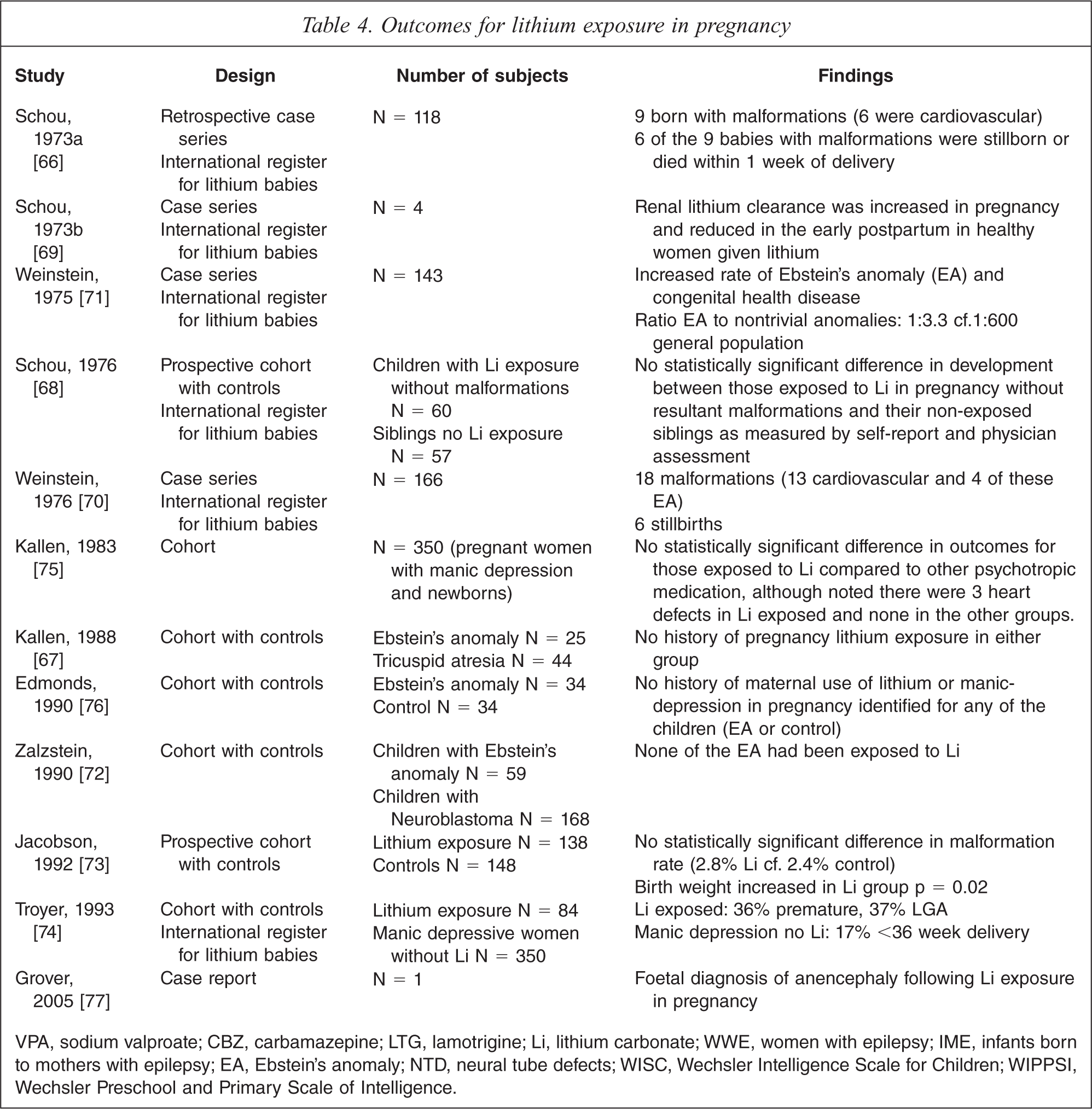
One case report and four case series were identified and are included in Table 4.
Outcomes for lithium exposure in pregnancy
VPA, sodium valproate; CBZ, carbamazepine; LTG, lamotrigine; Li, lithium carbonate; WWE, women with epilepsy; IME, infants born to mothers with epilepsy; EA, Ebstein's anomaly; NTD, neural tube defects; WISC, Wechsler Intelligence Scale for Children; WIPPSI, Wechsler Preschool and Primary Scale of Intelligence.
Cohort studies
Cohort with controls: 5 studies were identified and are included in Table 4.
Cohort without controls: 1 study was identified and is included in Table 4.
The published research available for outcomes for pregnancy, neonates and child longer term development following lithium exposure in pregnancy is limited compared with that available for AEDs. In total there were 12 studies identified and of this one was a prospective case control, one a cohort and two studies were case control [66–77]. The rest were case series. These studies are presented in Table 4. Case reports regarding cardiovascular malformations were not included in the table however one case report of anencephaly was included given this has not been examined in other studies. There was a trend in cardiovascular malformations with reports in four of the identified studies. These were predominantly an increase in Ebstein's anomaly. This is a rare cardiovascular malformation which occurs in 0.005% of the general population. While initially the studies indicated a much higher risk of Ebstein's anomaly a review of all the available studies identified a risk of between 0.05% and 0.01% following exposure to lithium in first trimester [78]. Interestingly a review of 59 children that had diagnosed Ebstein's anomaly none had been exposed to lithium carbonate [72].
The difficulty with the malformation data available for lithium is the small number of studies and participants and the lack of prospective case control studies. In a discussion of the available AED malformation data Meador et al. estimated that at least 500 cases are needed to determine differences in occurrence of major malformation and larger numbers to adequately control for other variables [79]. The largest number of women on lithium in the studies identified was 138 in the prospective case control study [73].
The perinatal outcomes identified with lithium exposure include prematurity and increased birth weight (large for gestation) [73,74]. There was only one study identified which examined longer term neurodevelopmental outcomes for children exposed in pregnancy [68]. Sixty children who had been enrolled in the lithium registry at birth were followed up to 7 years of age, along with 57 of their non-exposed siblings. None of the children was malformed. Information was collected regarding their development based on self report by the mother; no specific developmental assessment tool was used. Ten of the 60 exposed children and 6 of the non-exposed siblings had developmental anomalies, including goitre, being described as ‘nervous’ or ‘dull’, stuttering and bed wetting; some were confirmed via information from the treating doctor [68].
Discussion
This review examined the literature available on the risks and benefits of mood stabilizer medication in pregnancy. It has tried to address the risks and benefits of these agents when used to treat mental illness, although much of the AED literature targets women with epilepsy. While there are many existing reviews of psychotropic medications in pregnancy, this review was designed as one of three covering psychotropic use in pregnancy, allowing for a more intense focus on a narrowed area within the literature. The search conducted was thorough, combining electronic and hand searching of thousands of citations and abstracts, and the careful reading of hundreds of articles with critical analysis of the original research they contained.
The principal limitation concerns the target population. With the exception of lithium, the mood stabilizers under review here have mostly been studied in women with epilepsy (WWE); antipsychotic mood stabilizers have been reviewed by another arm of our group. The question underlying the review of these articles is whether the findings in the WWE population are generalizable to those using AEDs as psychotropics. While confounding factors diminish the applicability of findings both to the heterogeneous WWE population and to other populations, a recently published review of AEDs in pregnancy that detailed such methodological problems still found sufficient evidence for AED teratogenicity over the background effect of epilepsy [39]. This is relevant to all populations taking AEDs in pregnancy, whatever the indication. To further address the matter we selected data which was specific to the AEDs used as mood stabilizers (VPA, CBZ, LMT), excluding unstratified data that included other AEDs (e.g. phenobarbitone, phenytoin), or where the specific agents used were not made clear. Fortunately, due to prescription patterns and advances in the literature most of the studies thus excluded were older and less robust. Nonetheless, it remains that most of the women studied in our review, given the relatively small numbers in the lithium studies compared with the large multinational cohorts of WWE, were not taking the agents as mood stabilizers. This limitation applies as much to the literature available, therefore more robust studies of women taking these agents as mood stabilizers in pregnancy are clearly needed.
All mood stabilizers reviewed showed associations with increased malformation risk in pregnancy. These were predominantly structural malformations with the AEDs, in particular, neural tube defects but also cardiac and craniofacial defects. Our findings were consistent with a recent review which found respective major malformation rates of 2.9%, 8.7%, 2.7% in carbamazepine, sodium valproate and lamotrigine [39]. Sodium valproate, especially in doses above1000 mg/day, carried the highest malformation rate. Polytherapy also carried an increased risk, but in most studies this included agents not used in the treatment of bipolar disorder. There is limited evidence that some of the AEDs affect folic acid metabolism and that the risk of malformations may be reduced if folic acid 5 mg is taken for one month pre-conception and throughout first trimester [39,80,81]. Such use of folic acid is therefore recommended, although how much it reduces risk of malformation remains unclear. In addition, early pregnancy investigations such as high resolution morphological ultrasound with assessment of nuchal translucency, are indicated to assess for neural tube defects and other malformations [39].
Exposure to lithium increased the risk of cardiovascular defects, in particular a risk of Ebstein's anomaly of 0.01–0.05% compared to a population risk of 0.005% [78]. Ebstein's anomaly is the displacement of the tricuspid valve into the right ventricle; the prognosis depends on the presence of other cardiac abnormalities as well as the severity of the lesion [82]. Cardiac malformations associated with lithium exposure should be assessed with high resolution ultrasound and foetal echocardiogram at 16 weeks in pregnancy [83].
All four mood stabilizers reviewed may affect foetal growth and length of gestation. Therefore foetal growth surveillance is recommended, especially in late pregnancy. Assessment of the neonate, in light of the risks identified, should include serum glucose and liver function tests in those exposed to sodium valproate [39,81]. It is recommended that all exposed infants be observed for sedation, withdrawal and toxicity. In those infants exposed to lithium, cord blood lithium levels, neonatal thyroid function tests and urea and electrolytes are recommended [81]. Maternal drug levels should be monitored throughout pregnancy and, in particular, in the third trimester as the pregnancy-related physiological changes alter metabolism and clearance of these medications [39,81,83,84].
The neurodevelopmental evidence is comparatively quite limited. There appears to be an increased risk of a range of poor developmental outcomes, such as cognitive impairment, with sodium valproate exposure compared to other AEDs, but again, these studies were predominantly in women with epilepsy and not bipolar disorder. The neurodevelopmental data for lithium is limited to one study which used no standardized assessment for development.
In light of the risks involved with exposure to these treatments in pregnancy, it would seem prudent to discuss treatment regimens collaboratively with women before pregnancy occurs, making modifications accordingly. However, over 50% of pregnancies are unplanned and many women are not aware until after first trimester when the risks around malformations have been conferred [85]. Therefore, risks associated with these medications in pregnancy should be discussed with women of child-bearing age as part of standard care rather than waiting until a specific pre-pregnancy assessment occurs. It is concerning that studies of mental health clinicians’ counselling around family planning show that most clinicians do not discuss these issues with their patients [86].
The specific risks for exposure to mood stabilizers in pregnancy have been identified in this review. These must be balanced with the risks of untreated or under-treated major mental illness in pregnancy. There is evidence that ceasing mood stabilizers in pregnant women with bipolar disorder will increase the risk of relapse and time spent unwell in pregnancy [4,5]. Moreover, the early postpartum period is a time of greater vulnerability to relapse which may be reduced if treatment is maintained [4,6]. The potentially severe impact of untreated maternal illness on foetal, infant and child well-being must also be considered, although some of the related evidence is not specific to bipolar disorder [9,87–92]. Identified risks from exposure to particular treatments should therefore be placed alongside the risks of maternal relapse and the risks for the infant/child of having an unwell mother. All this should be discussed carefully with women and their partners or other support people, to devise a management plan for pregnancy and the postpartum [84].
While this review has focused on pharmacological treatments and pregnancy it is important that psychosocial needs and adequate supports are also addressed. A comprehensive management plan should include clear communication with all involved clinicians in general practice, obstetrics, paediatrics, maternal and child health, and mental health.
Footnotes
Acknowledgements
Thanks are due to Martien Snellen, Sue Walker, Michael Permezel, and the Melbourne Perinatal Psychotropic Review Group members: Fiona Judd, Lia Laios, Justin Dwyer, Klara Szego, Adaobi Udechuku, Tram Nguyen, Rebecca Hill.
