Abstract
Introduction
Few drugs are available for migraine prophylaxis in children. Levetiracetam is a broad-spectrum anti-seizure drug that has been suggested to be effective in reducing adult migraine episodes. We assessed the safety and efficacy of levetiracetam in the prevention of pediatric migraine.
Methods
A randomized double-blind placebo-controlled trial was performed. Eligible participants were aged 4–17 years old with at least four migrainous episodes monthly or had severe disabling or intolerable episodes. Primary endpoints were the mean changes in monthly frequency and intensity of headaches from the baseline phase to the last month of the double-blind phase. Safety endpoint was the adverse effects reported.
Results
Sixty-one participants (31 taking levetiracetam and 30 taking placebo) completed the study. All had a significant reduction in frequency and intensity of episodes that was significantly greater in the levetiracetam arm. Sixty eight percent of individuals in the treatment group reported more than 50% reduction of episodes at the end of the trial compared with 30% in the placebo group (p-value: 0.007). Irritability, day-time sedation, and mild tic were reported.
Conclusion
Levetiracetam may be useful in migraine prevention and may decrease migraine episodes and severity.
Trial Registration
The study is prospectively registered with Iranian Registry of Clinical Trials; IRCT.ir, number IRCT2017021632603N1.
Introduction
Up to 8% of children and adolescents may have migraine (1). About half of children and adolescents with migraine will continue to experience episodes into adulthood (2,3), leading to disability with substantial social and financial burdens to the individual as well as society.
Migraine prophylaxis may reduce the burden of the condition (4). To prevent analgesic overuse and to help children have normal daily activity, migraine preventive medications should be used when the number of headache episodes exceeds four per month or when quality of life, school attendance or daily activities are restricted (5,6). Few high-quality studies to identify medications with reasonable prophylactic effect have been conducted in pediatric migraine. Topiramate is the only prophylactic agent of migraine in children older than 12 years approved by the Food and Drug Administration (FDA). A recent phase 3, multicenter, double-blind, placebo-controlled trial, however, suggested that topiramate was not different from placebo in reducing events in children and adolescents (7). Further work is needed to identify the best evidence based pharmaceutical interventions.
Anti-seizure medications (ASMs) are commonly used in migraine prevention. Randomized controlled trials evaluated the efficacy of 15 ASMs as migraine prophylaxis in adults (8). Topiramate, valproic acid, levetiracetam, zonisamide and gabapentin have also been used in a few studies to prevent childhood and adolescent migraine (9). Our aim was to assess the safety and efficacy of levetiracetam in prevention of pediatric migraine.
Methods
Study design and participants
A randomized, double-blind placebo-controlled trial in the Children's Medical Center, a major referral hospital affiliated to Tehran University of Medical Sciences, was performed. The trial was divided into three phases: An initial screening phase (2 weeks), a baseline phase (4 weeks), and a double-blind treatment phase (12 weeks). Other preventive pharmacologic treatment was not allowed during the trial or in the 2 months prior to baseline (Appendix, p.1). Use of abortive migraine medications including simple analgesics (e.g. acetaminophen, ibuprofen, ketorolac), triptan-based (e.g. sumatriptan, zolmitriptan), and ergot-derivative-based (e.g. ergotamine, dihydroergotamine) migraine medications were permitted but participants were not to change the rescue drugs during the study period.
Children aged 4–17 years who met the diagnostic criteria for pediatric migraine (with or without aura) as defined by the International Headache Society (10) were enrolled. They had to have at least four migrainous episodes per month or to have severe disabling or intolerable headache. They were excluded if they had any of the following: History of cluster headache, hemiplegic migraine, or chronic daily headaches; headaches related to structural brain lesions; focal neurologic deficit; no therapeutic response to at least three adequate trials of medication for headache prophylaxis; history of levetiracetam sensitivity; pregnancy; other neurological conditions (e.g. epilepsy).
The ethics committee of Tehran University of Medical Sciences approved the final protocol (Number: IR.TUMS.VCR.REC.1395-811). The method was explained to all involved. Parents provided written informed consent before initiation of any study procedures. The study is prospectively registered with Iranian Registry of Clinical Trials; IRCT.ir, number IRCT2017021632603N1.
Randomization and blinding
Eligible participants were randomly assigned to receive either levetiracetam or placebo in a 1:1 ratio by permuted block randomization (block sizes of four) via an interactive web response system. The responsible statistician was not involved in study conduct or monitoring. Some hospital personnel involved in the preparation of the trial material were not blinded but had no contact with participants, their parents, or investigators. The study medications were coded and administered by a nurse who was not informed about the clinical characteristics of cases. The study personnel, investigators, participants, and their parents were blinded during the course of the study until the code was broken at trial end. Packaging, size (18 mm × 8 mm), shape (oval), and color (yellow) of placebo tablets were similar to the 500 mg levetiracetam tablets used.
Procedures
Phase 1 and 2
All children had complete physical examinations. Neuro-imaging was performed if clinically indicated. Laboratory screening tests including blood count (hemoglobin, white blood cells, and platelets), serum chemistry, and urine pregnancy testing were performed if appropriate (phase 1). Headache diaries were provided for each child, and the parent who had more contact with the child was responsible for recording events in the diary. A simple form of migraine diagnostic criteria (including criteria B–D) (10) translated into the Farsi language was also provided and explained to the responsible parent so that only migrainous episodes were reported. To avoid information bias, parents were asked to collect the data in real time, every day and not retrospectively. The number of migraine episodes and the intensity of the attacks were recorded using a questionnaire for a period of 4 weeks (phase 2) before initiation of the double-blind phase.
Phase 3
In the 12-week double-blind phase, oral levetiracetam was administered to the treatment group. The initial dose was usually about 20 mg/kg/day divided into two doses but varied at the discretion of the study team (up to 40 mg/kg/day). Parents recorded migraine frequency and intensity daily. The first follow-up visit was one week after the initiation of treatment to check whether the medication had been taken at the correct times and doses and whether any adverse effects had occurred. Subsequent follow-up visits were at 4, 8, and 12 weeks. Participation was discontinued if i) any adverse event occurred that, in the opinion of the investigator, required termination of medication; ii) there was progression of migraine attacks; iii) the participant became pregnant; or iv) consent was withdrawn.
Outcomes
The primary endpoints were the mean changes in monthly attack frequency and severity of migraine headaches from the baseline phase to the last four weeks of treatment. Migraine frequency was defined as the number of attacks that fulfilled the IHS criteria for migraine with or without aura (10). In each arm, we obtained the mean number of these episodes every four weeks after initiation of treatment.
Migraine intensity was reported using a visual analogue scale (VAS). This scale consists of a 10 cm line divided into 10 parts, numbered 0 (no pain) to 10 (most extreme pain). In each group, we obtained the mean of migraine intensity every four weeks after initiation of treatment.
The secondary endpoint was the >50% responder rate. The drug was defined as effective if it decreased headache frequency by more than 50% in the double-blind phase compared with the baseline frequency.
For the safety endpoint, all participants and their parents were asked to report any side effects of the medications during their monthly visits. A phone number was also provided so adverse events could be reported. Participants and their parents were asked to go to the emergency department if serious complications occurred.
Statistical analysis
Based on previous placebo-controlled trials in migraine, we assumed a 40% reduction in migraine attack frequency with standard deviation (SD) 50% for sample size calculation. Power analysis mandated at least 25 cases per group (11) with α (the probability of type I error) as 0.05, and β (the probability of type II error [1 − power of the test]) as 0.20. Considering a 20% dropout rate, a final sample size of 30 subjects in each treatment arm was estimated. The formula used for sample size calculation was:
Statistical analysis was performed by the statistician who was masked to the study arms. Continuous variables were reported as mean with SD, standard error mean (SEM), or 95% confidence interval (CI). Categorical variables were reported using percentages. Analyses of between-group differences were performed using the intention to treat (ITT) approach. All randomized individuals who received at least one dose of the study drug were included. Missing data were handled by multiple imputation. Secondary analyses were also performed for participants who completed the trial.
The mean change in frequency and intensity of migraine attacks during weeks 9–12 were compared between the two treatment arms using t-tests for two independent samples. To more rigorously evaluate the robustness of the treatment effect, analysis of covariance (ANCOVA) models with adjustment for covariates including age, gender, and baseline migraine frequency or intensity were performed as a supplemental analysis. Fisher's exact test was used for the secondary endpoint (>50% responder rate) analysis. Participants who failed to complete the study were considered non-responders. IBM SPSS Statistics version 25 was used for statistical analyses. Figures were created using GraphPad Prism version 7.04.
Results
Participants
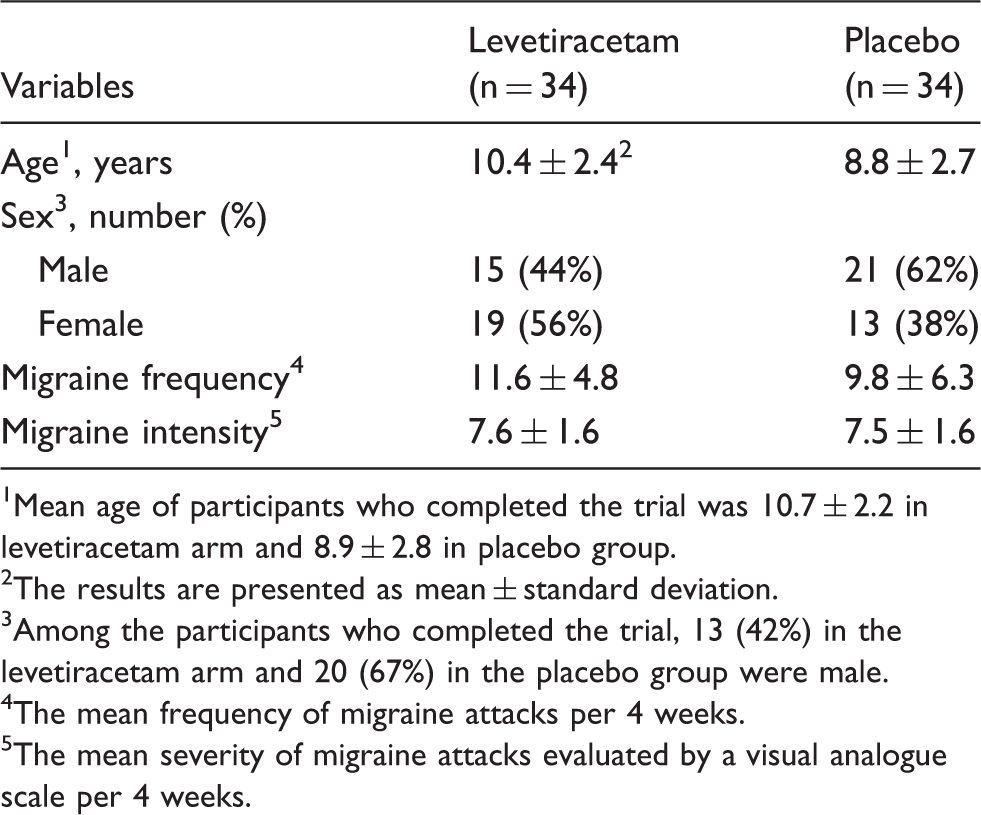
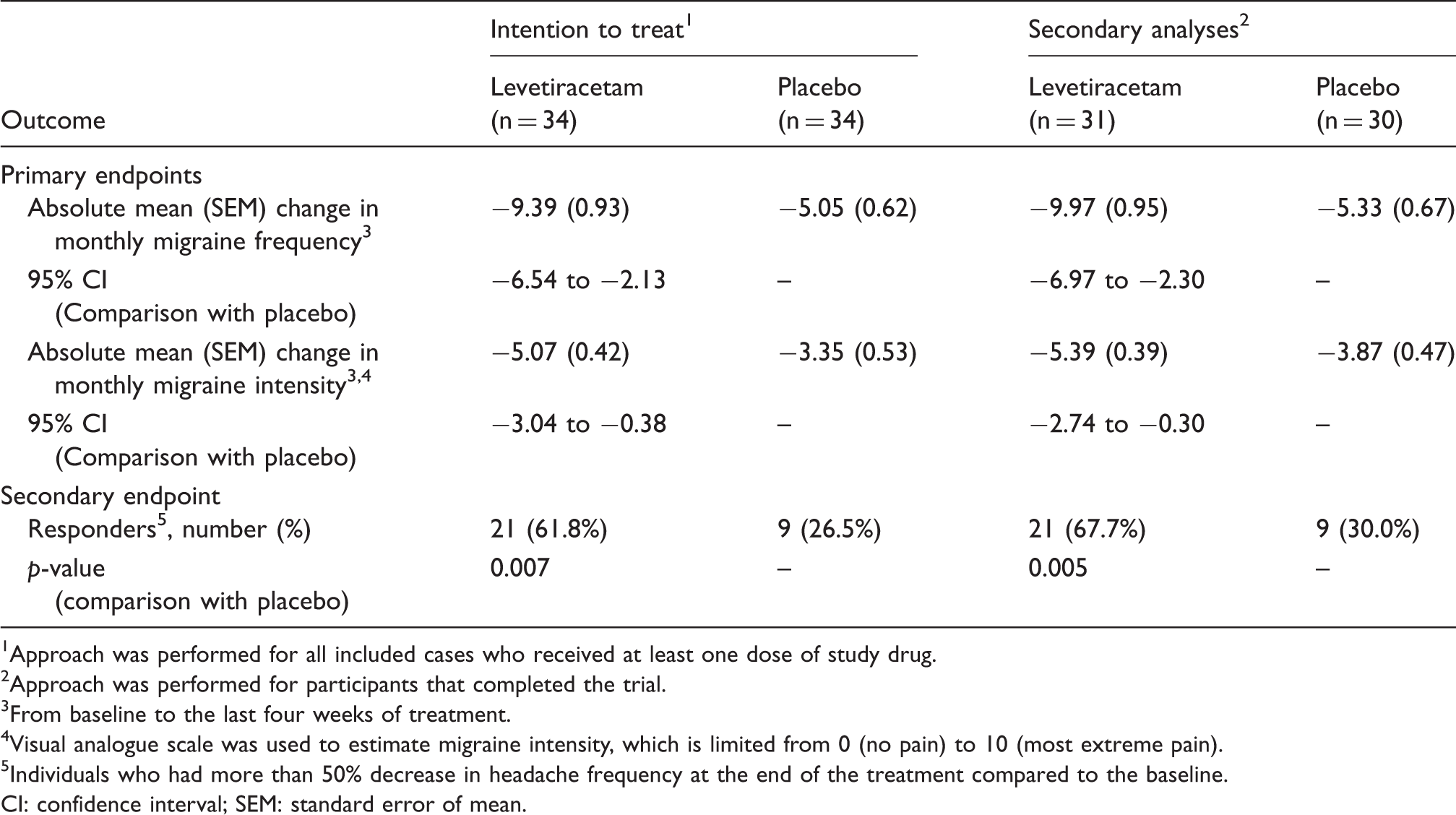
The initial phase started in July 2017. Double-blind treatment was given from August 2017, when the first participant was assigned, to August 2018. Eighty-six individuals were screened for eligibility, of whom 68 were eligible and were randomly assigned to a treatment group (34 in each group). Each took at least one dose of the allocated drug and was therefore eligible for the ITT analysis. Of these 68 included subjects, three receiving levetiracetam and four receiving placebo did not complete the study. Reasons for drop-out were: Lost to follow-up (n = 3), poor compliance (n = 3), and withdrew consent without providing any specific reason (n = 1). No adverse event was reported by these participants. Overall, 61 individuals (31 on levetiracetam) completed the study (Figure 1). The baseline characteristics of the participants are summarized in Table 1 and Figure 2.
CONSORT diagram of trial flow. Age distribution of study participants. Baseline demographic data and headache characteristics of participants. Mean age of participants who completed the trial was 10.7 ± 2.2 in levetiracetam arm and 8.9 ± 2.8 in placebo group. The results are presented as mean ± standard deviation. Among the participants who completed the trial, 13 (42%) in the levetiracetam arm and 20 (67%) in the placebo group were male. The mean frequency of migraine attacks per 4 weeks. The mean severity of migraine attacks evaluated by a visual analogue scale per 4 weeks.

Seven subjects were included due to severely disabling migrainous attacks (two taking levetiracetam and five taking placebo) and the rest had at least four migraine episodes per month. At least one other prophylactic medication had been unsuccessful in eight participants taking levetiracetam and nine taking placebo, while 51 individuals had no history of previous preventive treatment. Simple analgesics were the most commonly used acute migraine treatment in both arms (Supplementary Table 1). For individuals who received levetiracetam, doses ranging from 500 mg to 3000 mg per day were administered (Mean ± SD: 1120.9 ± 487 mg).
Primary endpoints
During the last four weeks of the double-blind phase, the mean migraine frequency reduced significantly in the levetiracetam and placebo arms compared to baseline (Figure 3). The mean reduction of migraine frequency was statistically greater in levetiracetam than in the placebo arm (95% CI: −6.54 to −2.13) (Table 2). The mean migraine intensity also reduced significantly in both treatment groups during the last four weeks of the study period compared to baseline (Figure 4), but the difference was significantly greater in those taking levetiracetam (95% CI: −3.04 to −0.38) (Table 2).
Mean of migraine frequency per 4 weeks. Mean of migraine intensity per 4 weeks. Primary and secondary endpoints. Approach was performed for all included cases who received at least one dose of study drug. Approach was performed for participants that completed the trial. From baseline to the last four weeks of treatment. Visual analogue scale was used to estimate migraine intensity, which is limited from 0 (no pain) to 10 (most extreme pain). Individuals who had more than 50% decrease in headache frequency at the end of the treatment compared to the baseline. CI: confidence interval; SEM: standard error of mean.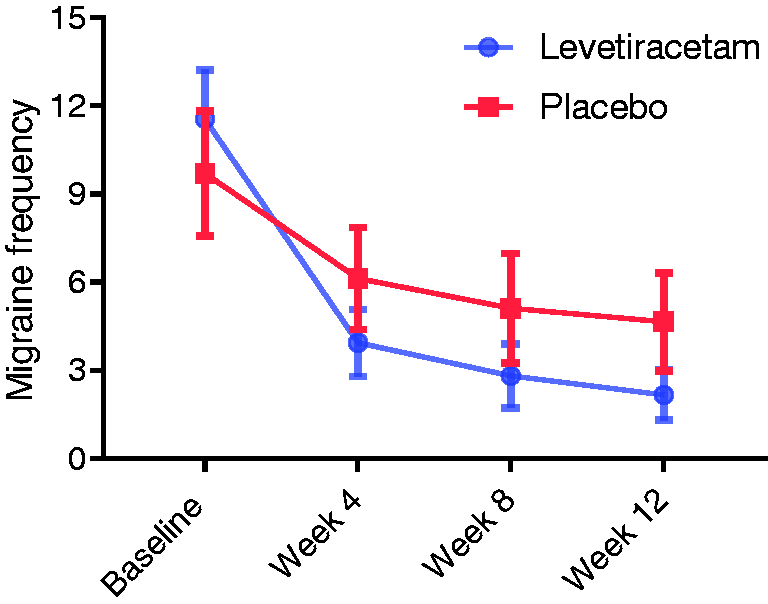
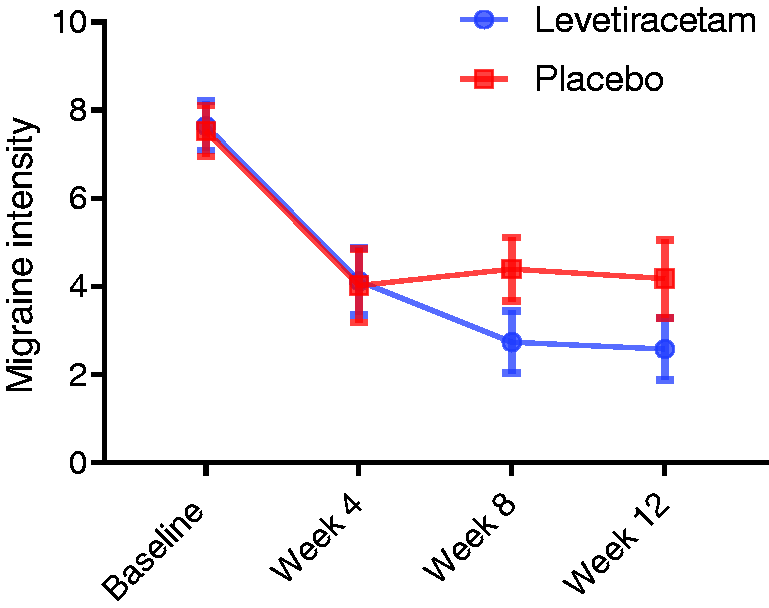
Using the ANCOVA sensitivity analysis, the treatment effect defined by the primary endpoints remained significant and migraine frequency and intensity of participants taking levetiracetam had greater reduction than cases taking placebo (migraine frequency [mean difference (95% CI)]: −2.84 (−4.52 to −1.17); migraine intensity [mean difference (95% CI)]: −1.68 (−2.95 to −0.41)).
Exploratory analyses of between-group differences in mean migraine frequency and intensity during the first and second four-week periods of the double-blind phase also showed greater reductions in the levetiracetam arm than in the placebo arm (Supplementary Table 2).
Secondary endpoint
Levetiracetam was associated with a greater than 50% reduction from baseline migraine episodes in 17 (50%) participants at four weeks after initiation of medication; 19 (56%) after eight weeks, and 21 (62%) after 12 weeks. Six of the participants in the levetiracetam arm reported complete elimination of headaches. Conversely, six (18%) of those in the placebo arm had a greater than 50% reduction from their baseline frequency at four weeks after the beginning of double-blind treatment; eight (24%) after eight weeks, and nine (26%) after 12 weeks. One had a complete cessation of headache attacks. Overall, the > 50% responder rate was significantly greater in the levetiracetam arm than the placebo (p = 0.007) (Table 2).
Safety endpoint
Adverse events occurred in eight individuals taking levetiracetam. None was significant, and these did not lead to discontinuation of therapy. Overall, irritability (four cases), day-time sedation (two cases), and mild tic (two cases) were reported, which all resolved during treatment. No side effect was reported in participants receiving placebo.
Discussion
Summary of results
Levetiracetam and placebo significantly reduced migraine frequency and intensity compared to baseline. The reduction in the levetiracetam arm was, however, significantly greater than with placebo. Over two thirds of participants in the treatment arm reported a reduction of more than 50% from baseline headaches with levetiracetam, which was significantly greater than with placebo. More participants reported adverse events on levetiracetam, but safety was similar to placebo.
The annual health-care costs of children diagnosed with migraine are threefold greater than controls (12). These individuals are also prone to developing different neurological and mental co-morbidities in the future (13). About 18% of participants (with baseline mean migraine frequency of 12 per month) became headache-free after 12 weeks of treatment with levetiracetam. Migraine episodes only stopped in one individual on placebo. This difference can be significant from a clinical standpoint.
Previous studies
Studies evaluating the efficacy of levetiracetam in prevention of pediatric migraine.
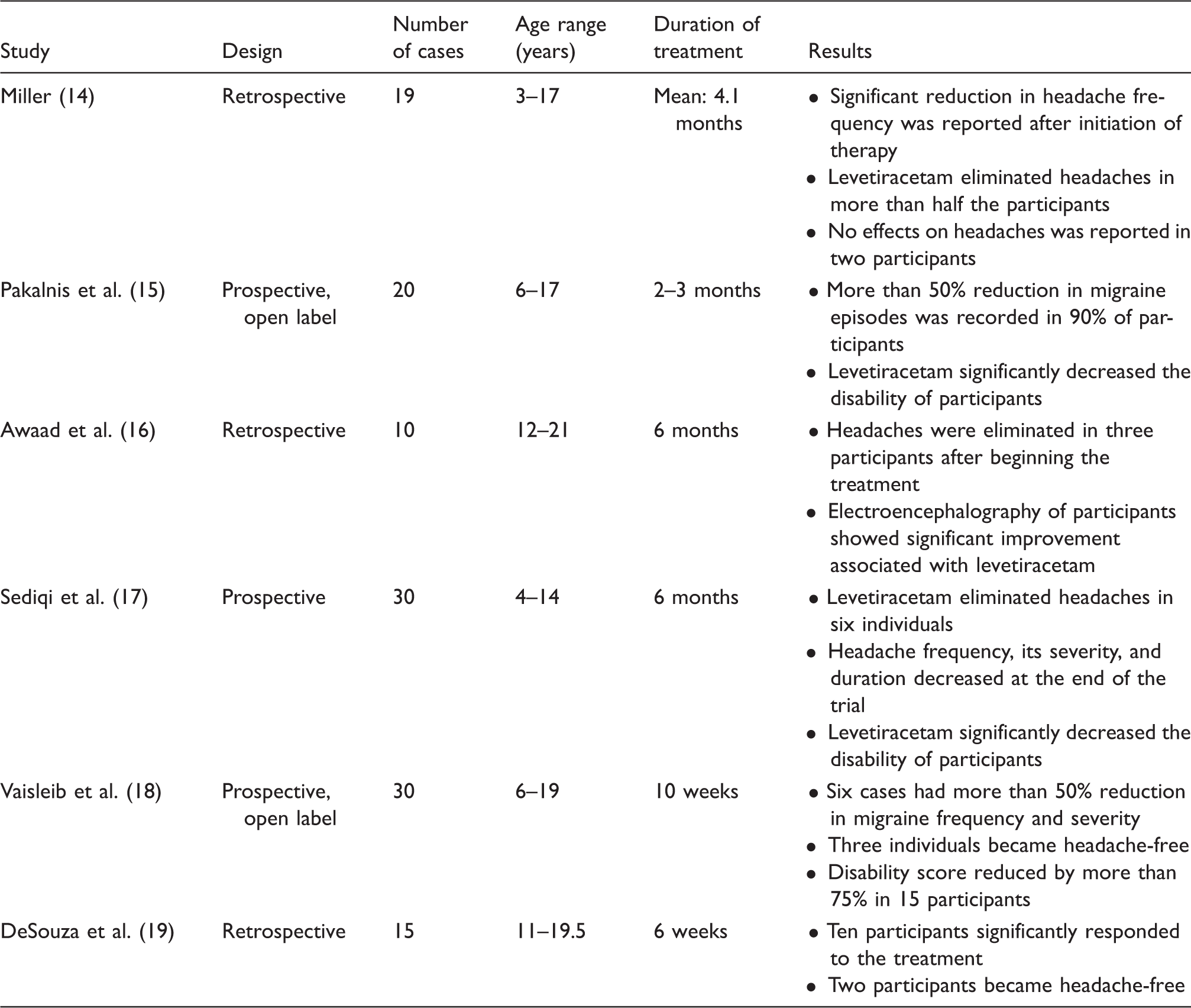
Levetiracetam was also used as a prophylactic agent for episodic and chronic migraine in adults. Prospective double-blind placebo-controlled trials showed that this medication produced significant reductions in migraine episodes, as well as attack severity, in adult episodic migraine (20,21). One trial showed that individuals in levetiracetam and valproate groups had more improvements in headache frequency than those in the placebo arm, but no significant difference was reported when they were compared to each other (21). Randomized double-blind studies conducted in adults with chronic migraine did not show significant efficacy of levetiracetam compared to placebo (22) or valproate (23). Side effects including irritability, somnolence, dizziness, hyperactive behavior, moodiness, and hostility were also reported in adults (21).
Levetiracetam mechanisms of action
The exact mechanism by which levetiracetam acts is unknown but it probably has unique mechanisms of action, binding to the synaptic vesicle protein (SV2A), leading to changes in the release of numerous neurotransmitters (24).
To date, levetiracetam and many other ASMs (e.g. topiramate) have been used in human and animal studies for prevention of migraine, but the underlying mechanisms of efficacy are yet unclear. The pathophysiologic link between migraine and epilepsy may resolve some of our ambiguities. Increased prevalence of migraine among individuals with epilepsy as well as of epilepsy in people with migraine (25) suggests possible shared pathophysiology between these conditions. Similarities in clinical features of epilepsy and migraine such as presence of premonition, aura, and postictal phases may suggest elements of pathophysiologic overlay (26). Immune system dysfunctions including elevated levels of pro-inflammatory cytokines and autoantibodies are now considered in occurrence of epilepsy and migraine (27,28).
Placebo effects
Our results suggest that placebo could also be useful in improving pediatric migraine. A recent trial suggested that placebo was more efficacious than topiramate or amitriptyline in producing at least 50% reduction in headache days in children and adolescents (7). High placebo-response rates have also been reported in previous headache and pain trials (reviewed in 29,30). Placebo effects are neurobiologic changes that may occur due to expectations of people with migraine, their caregivers and clinicians (31).
Limitations and future directions
The small sample size, short follow-up period, and lack of active comparator are major limitations of this trial that should be resolved in future studies. As migraine cannot be objectively assessed, the outcomes of the study were reliant on reports from participants or their parents, which could lead to information bias. Multiple factors including diet, physical activity, and stressors affect migraine occurrence and further studies should estimate their impacts on results. Future trials are needed to test the most effective preventive medications in pediatric migraine. Trying to understand the pathophysiologic links between migraine and its co-morbidities may also identify new treatments for migraine. Future studies should also identify subgroups that may have the most benefits from levetiracetam and other ASMs.
Conclusion
Levetiracetam was more successful than placebo in decreasing migraine episodes and their intensity. It was also more effective than placebo in reducing headache frequency by 50% in participants in a 12 weeks period.
Clinical implications
Few drugs are available for migraine prophylaxis in the pediatric population. Levetiracetam may be effective in the prevention of pediatric migraine. Levetiracetam may reduce the number and intensity of migraine episodes. These findings need replication in larger groups.
Supplemental Material
Supplemental material for Levetiracetam for prophylactic treatment of pediatric migraine: A randomized double-blind placebo-controlled trial
Supplemental Material for Levetiracetam for prophylactic treatment of pediatric migraine: A randomized double-blind placebo-controlled trial by Hadi Montazerlotfelahi, Man Amanat, Ali Reza Tavasoli, Elmira Agah, Gholam Reza Zamani, Josemir W Sander, Reza Shervin Badv, Mahmoud Mohammadi, Mahdieh Dehghani, Morteza Heidari, Seyed Ahmad Hosseini, Mona Salehi and Mahmoud Reza Ashrafi in Cephalalgia
Footnotes
Ethics approval
The ethics committee of the Tehran University of Medical Sciences approved this study (Number: IR.TUMS.VCR.REC.1395-811).
Study protocol
The study is registered with Iranian Registry of Clinical Trials; IRCT.ir, number IRCT2017021632603N1. The study protocol can be observed in Appendix, p. 5-7.
Acknowledgements
We are grateful to Cobel Darou for providing levetiracetam and placebo. We are also very grateful to all parents and participants for partaking in this study. Our special thanks to Mrs Leila Forotan, who dispensed the trial medications to the participants. JWS is based at the NIHR University College London Hospitals Biomedical Research Centre, which receives a proportion of funding from the UK Department of Health’s Research Centers funding scheme. He receives research support from the Dr Marvin Weil Epilepsy Research Fund and from the UK Epilepsy Society.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JWS has received research funding from Eisai and UCB, personal fees from Eisai, UCB, GW and Zogenix Pharma, outside the submitted work. The other authors declare no competing interests.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The trial was funded by the Research Deputy of Tehran University of Medical Sciences (grant number: 95-01-30-31762), who had no role in study design, data collection, analysis, or interpretation of data, in the writing of the report, or in the decision to submit the article for publication.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

