Abstract
Research findings from epidemiology, neurobiology, cognitive neuroscience and genetics have provided important insights into the potential causes of schizophrenia, and the advent of new technologies promises further advances in aetiological research. However, certain barriers hamper progress. The relatively low prevalence of schizophrenia, combined with difficulties in obtaining sufficient numbers of affected individuals who are willing to participate in research, tend to compromise the size and representativeness of samples. This problem is of particular significance in light of the very large samples that are required to achieve sufficient statistical power for modern genetics studies. In addition, the need for accurate phenotyping and thorough, multidimensional assessment of cases in order to define subgroups along key dimensions of potential aetiological importance adds substantially to the costs associated with the collection, processing, storage and analysis of large datasets. These barriers mean that large-scale genetic research is likely to fall beyond the resources of most research centres working in isolation.
To help overcome these barriers, a consortium of scientists has established a national research facility, the Australian Schizophrenia Research Bank (ASRB). This is a multi-site collaborative project involving centres in Brisbane, Melbourne, Newcastle, Perth and Sydney. The resource contains comprehensive, cross-referenced data to support schizophrenia research across the clinical, neurocognitive, genetic and brain imaging domains, and builds on four pre-existing schizophrenia research facilities, namely the Schizophrenia Research Register [1], the Hunter DNA Bank for Schizophrenia and Allied Disorders, the Newcastle Virtual Brain Bank, and the Western Australia Family Study of Schizophrenia (WAFSS) [2,3].
The purpose of the ASRB is to build up a repository of comprehensive, cross-referenced data and biological samples from a large number of people with schizophrenia and healthy controls, from which case and control subsamples can be selected that are matched for age, gender and handedness, and to make the contents of this repository available for scientific study to Australian and international researchers. While the research database allows the exploration of many schizophrenia related research questions, particularly the examination of relationships between brain structure and genetic factors, it is necessarily constrained with respect to patient descriptors in the psychological, neurocognitive and clinical domains, and consequently the scope of research questions that can be addressed in relation to the latter. The investigators had to limit the range of such data on the basis of feasibility and tolerability to patients, and make strategic choices as to which measures to include in constructing the test battery. This means that there are inherent but unavoidable limitations in the nature and scope of hypothesis driven research using existing ASRB data. However, the ASRB has developed a research register function that enables participants to join other research studies testing hypotheses that are beyond the capacity of the data collected at entry into the ASRB.
The management of the ASRB is based on the structures developed by the Schizophrenia Research Institute, a virtual institute with over twelve years experience in overseeing multi-site, collaborative research in Australia [4]. The following describes the procedures and protocols for sample recruitment, assessment and resource management used by the ASRB.
Method
Sampling
The ASRB is based on a case–control design. Recruitment is ongoing in five Australian states and territories (i.e. Australian Capital Territory, New South Wales, Queensland, Victoria and Western Australia) through a variety of new and existing networks associated with the ASRB collaborators.
People with schizophrenia are being recruited through treatment settings, such as hospital inpatient units, community mental health services, outpatient clinics and rehabilitation services, non-government mental illness support organizations, and, in the initial stages, through a large-scale, national, multi-media advertising campaign conducted via television and radio community service advertisements, web pages and media interviews. Initial recruitment strategies also included brochures and posters distributed to hospital waiting rooms, community mental health centres and other public locations. Psychiatrists, general practitioners, mental health nurses, allied health professionals, and community support agencies were given information about the ASRB through educational and scientific presentations and asked to facilitate recruitment of patients in their care. A web site (www.schizophreniaresearch.org.au) contains detailed information and an enrolment form for those wishing to join online. A national toll-free telephone number (1800 639 295) was also established so that potential participants could make enquiries about participation.
Case recruitment has gradually shifted from respondents to the national multi-media campaign to clinical services. Healthy controls have also been recruited as a result of the multi-media campaign and in response to local initiatives, but recruitment of controls shifted in 2010 to the use of epidemiological methods, namely cold telephone calling of people identified through the Australian White Pages telephone directory. This recruitment strategy was used to help achieve a national control sample that was representative of the Australian population and thereby limit the potential confounds of sampling bias arising from reliance on non-systematically recruited community volunteers alone. Further, this alternative provides the potential to stratify sampling in relation to the schizophrenia cohort recruited at each site on the basis of age, sex and handedness matches to cases.
Screening and consent
Potential participants are provided with an information sheet and consent form, either by mail or online. Participants are screened by a trained clinical assessment officer (CAO) using a detailed telephone checklist prior to enrolment to assess the likely eligibility of participants and their suitability for the project.
Participants are English speaking (required for neuropsychological assessments) and aged 18–65 years. Exclusion criteria are organic brain disorder, brain injury with greater than 24 hours post-traumatic amnesia, mental retardation (IQ <70), movement disorders, current diagnosis of substance dependence, electroconvulsive therapy received in the last 6 months and, for controls, a personal or family history of psychosis or bipolar 1 disorder.
Data collection
All eligible participants are allocated the next available unique ID code generated automatically from the ASRB database and their details are entered into the ASRB system in preparation for assessment. Clinical interviews and neuropsychological testing are conducted by CAOs. They are registered psychologists or intern psychologists, trained in the administration of the diagnostic interview and neuropsychological test instruments, and work under supervision. Clinical assessment takes approximately three hours and consists of three one-hour sections (i.e socio-demographic information, including medical and family history; neuropsychological evaluation; and diagnostic interview) and three self-report questionnaires. Sections are independent and can be completed in any order.
Clinical assessment data are recorded in most centres direct to laptop computers at the time of interview and then uploaded directly onto the ASRB database, thereby avoiding double handling and minimizing the likelihood of data transcription errors.
Clinical assessment, blood collection and structural neuroimaging
Assessments consist of the Diagnostic Interview for Psychosis (DIP) [5,6], which is used to establish a lifetime diagnosis of a psychotic disorder, as well as present and lifetime substance use disorder diagnoses, according to DSM-IV and ICD-10 criteria. The DIP diagnostic algorithm also enables classification according to other systems, including DSM-IIIR and Research Diagnostic Criteria. In addition, the DIP provides for the collection of socio-demographic and medical history data. Premorbid and current IQ are assessed using the Wechsler Test of Adult Reading (WTAR) [7] and the Wechsler Abbreviated Scale of Intelligence (WASI) [8], respectively. The Repeatable Battery for Assessment of Neuropsychological Status (RBANS) [9] is used to assess neuropsychological performance on measures of immediate and delayed memory, attention, learning and language. Additional tests of working memory (Letter Number Sequencing) [10] and executive functioning (Controlled Oral Word Association Test) [11] are also administered. Handedness is measured using the Edinburgh Handedness Inventory [12]. A modified version of the Neurological Evaluation Scale [13] is used to assess neurological ‘soft signs’. General functioning is assessed using the Global Assessment of Functioning scale (GAF) [14]. Negative symptoms are rated using the Scale for Assessment of Negative Symptoms (SANS) [15]. Self-report questionnaires administered include the Childhood Adversity Questionnaire [16], Schizotypal Personality Questionnaire (SPQ) [17] and the International Personality Disorders Examination (IPDE) [18].
A 40 mL blood sample is collected at the time of clinical interview. Half the sample is collected in the presence of ethylene-diamine-tetra-acetic acid (EDTA) for the extraction of DNA, while the rest is collected using a lithium heparin tube for storing lymphocytes. Blood samples are collected by trained pathology staff, transported using existing blood collection and transport networks, and delivered by these services to the ASRB facilities in New South Wales, Queensland and Western Australia within 24 hours of collection. The Newcastle site in New South Wales holds blood samples collected in Victoria and acts as the central repository for the ASRB, holding a duplicate of every blood sample in two separate locations in Newcastle.
Structural magnetic resonance imaging (sMRI)) brain scans are performed in as many cases as possible and a sub-sample of healthy controls at designated facilities. The sMRI brain scans also incorporate DTI sequences and are acquired using a standard data acquisition protocol on Siemens Avanto 1.5 Tesla scanners located in the four participating states. All data are forwarded to the Newcastle site for review and quality control before inclusion in the database.
Data storage and processing
A centralized database of commercial standard has been developed by Intersect (www.intersect.org.au), a major e-research organization in New South Wales, in conjunction with the University of Newcastle, to manage securely the large volume of data acquired. New purpose-built software loaded on laptop computers for the collection of clinical data has also been developed allowing seamless integration of the collected clinical data into the ASRB database for secure aggregation and storage [19].
Several levels of access control are available to the ASRB database allowing authorized, internet-connected individuals to read and download data. Users are grouped according to access privileges (i.e. management, worksite, research project/group, public). Access for a particular research project is limited to the subset of data authorized for that project by the ASRB Access Committee (see below); all researchers associated with a particular project are then afforded access to the unique dataset compiled for that project.
Quality control
Procedures have been developed to ensure the quality and integrity of the ASRB data. These include regular reviews of clinical assessments, quality assurance questionnaires for participants and researchers, clinical assessment software for automated data entry that also contains fixed data input parameters to reduce data entry errors, and inter-rater and inter-site reliability assessments. With regard to blood samples, optical density readings are undertaken on each genomic DNA sample using a nano-photometer to obtain A260:A280 ratios >1.8 as an indication of the DNA quality. Prior to sending samples to researchers, DNA quantitation may be checked using PicoGreen (Invitrogen) assay for double stranded DNA. For brain scans, a Siemens head coil phantom is regularly used at each participating site to establish and monitor scanning quality across sites. After scanning acquisition, data is transferred to a central processing office where it is checked for artefacts and general image quality.
To help monitor the quality of the assessment experience, all participants receive a quality assurance questionnaire at the completion of their assessment. Problems raised by participants about their assessment experience are dealt with initially by the ASRB manager in consultation with the CAO concerned; adverse events are reported to the Scientific Committee (see below) and, where appropriate, to the relevant Ethics Committee(s).
Organizational structure
The operations of the ASRB are overseen by a governance committee. Two committees report directly to the governance committee; the scientific committee and the access committee. The scientific committee comprises the chief investigators on the NHMRC enabling grant, the core funding source of ASRB, and this committee oversees all operational details of establishing and maintaining the ASRB. The access committee reviews and has discretion to grant or deny approval of requests for access to the data and samples.
Accessing the ASRB
Access to the ASRB is available to Australian scientists from July 2010 and to international scientists from July 2011. The application form and guidelines for researchers are available electronically (www.schizophreniaresearch.org.au). Researchers must have their relevant health research ethics committee approval for their project before applying. Applications are assessed by the access committee. Should a project application be judged by this committee to be of insufficient scientific merit or if insufficient information is provided, feedback will be given, researchers will be notified that the application has been unsuccessful, and the applicant may be invited to reapply. In the event that two applications are received for data to conduct identical studies, the first application received will be given priority.
Results
The first set of cases with a confirmed diagnosis of schizophrenia (ie schizophrenia or schizoaffective disorder) have been assessed according to the protocol described above. Approximately equal numbers of volunteers have been recruited across states, with most volunteers recruited, as expected, from the larger metropolitan centres of Sydney (21%), Melbourne (21%), Brisbane (26%), and Perth (27%), and smaller proportions from the Hunter region and ACT (5%).
Of the first 500 cases presenting to the ASRB reporting a clinical diagnosis of schizophrenia, 459 (91.8%) met ICD-10 criteria for schizophrenia, 34 (6.8%) for schizoaffective disorder and 7 (1.4%) met criteria for other mental disorders. Comparisons between the first 493 cases with schizophrenia or schizoaffective disorder and 293 control participants are presented in Table 1. These show that schizophrenia/schizoaffective participants had a higher proportion of males, were marginally older than the control sample, had fewer years of education, and had lower scores on measures of current and premorbid IQ, and RBANS total score compared to controls. The schizophrenia/schizoaffective sample was also less likely to be currently living in a married or de facto relationship, more likely to be unemployed, and displayed lower levels of general functioning. The groups did not differ on handedness.
Comparisons between ASRB schizophrenia and control samples
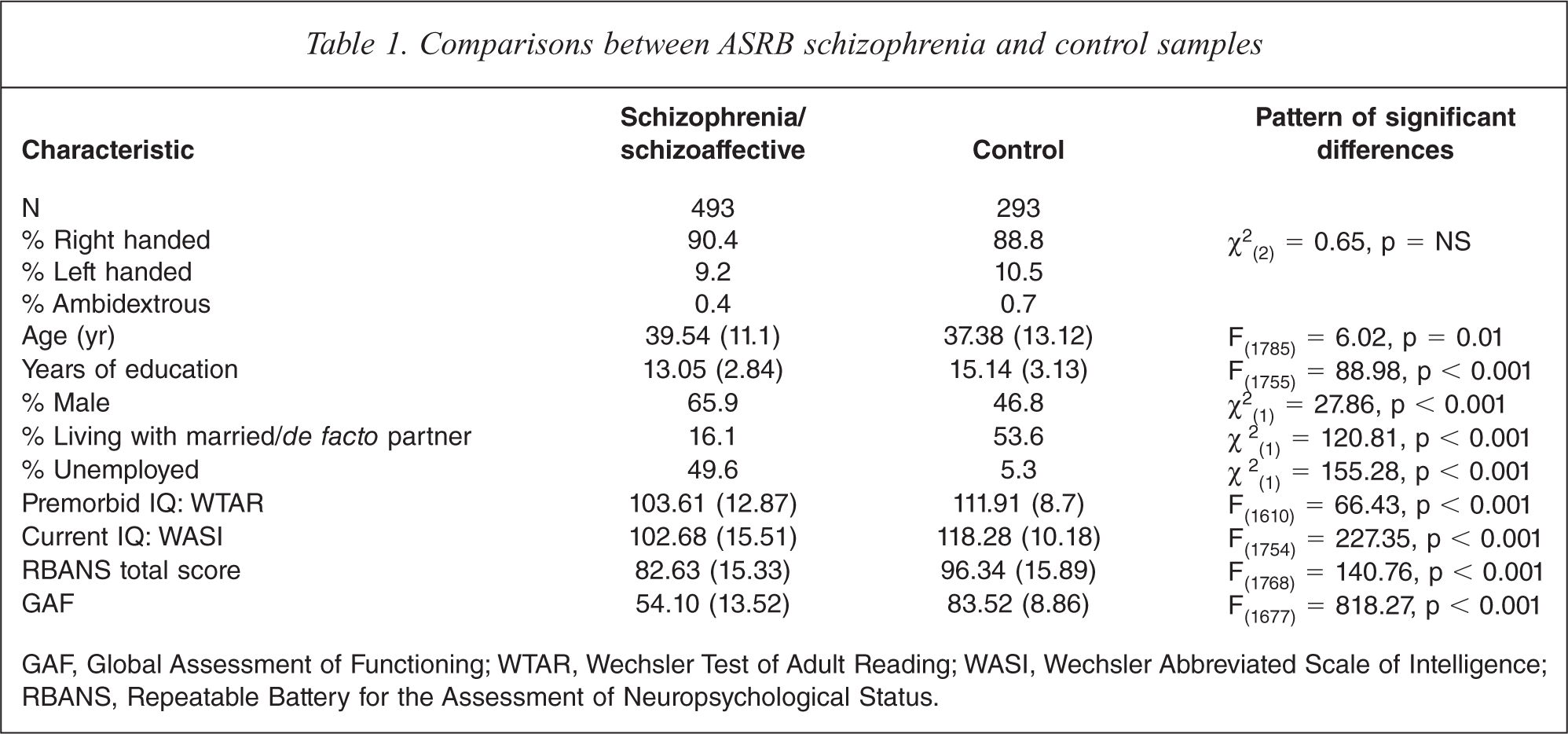
GAF, Global Assessment of Functioning; WTAR, Wechsler Test of Adult Reading; WASI, Wechsler Abbreviated Scale of Intelligence; RBANS, Repeatable Battery for the Assessment of Neuropsychological Status.
The recruitment of participants using various methods (treatment settings, community outpatient services and multimedia advertising) raises the question as to how representative the ASRB sample is relative to the overall population of people with schizophrenia. A preliminary examination of this question is described below. As a partial control for the number of statistical tests, the threshold for significance was set at p < 0.01.
Comparisons with the Low Prevalence Disorders Study sample
Data from the first 493 ASRB participants with schizophrenia/schizoaffective disorder were compared with a subsample drawn from the 1999 Low Prevalence (psychotic) Disorders Study (LPDS) [6]. The LPDS recruited primarily through mental health services across four Australian states: Queensland, Victoria, Western Australia and the ACT [6]. The LPDS subsample comprised 612 individuals with an ICD-10 diagnosis of schizophrenia (N = 510) or schizoaffective disorder (N = 102) on the basis of the DIP, and excluded any individuals with inpatient psychiatric admissions of 6 months or more during the last year. Table 2 shows comparisons between samples of individuals with an ICD-10 diagnosis of schizophrenia or schizoaffective disorder from the ASRB and the LPDS. These groups did not differ significantly in age, gender distribution, level of premorbid social or work adjustment, age at illness onset, duration of illness or proportion with a psychosocial stressor at onset. However, ASRB participants comprised a significantly larger proportion of individuals who were born in Australia, a higher rate of involvement in relationships (married or de facto), a lower rate of unemployment and a higher rate of high school completion or advanced education than the LPDS sample. The ASRB sample also had a higher proportion with a positive family history of schizophrenia. Groups also differed on the proportions of schizophrenia versus schizoaffective disorder diagnoses, with the ASRB subsample having a higher proportion of participants with schizophrenia. Finally, the groups differed in their reported lifetime course of illness, with the ASRB sample displaying a lower proportion with a chronic course of illness compared to the LPDS sample.
Comparisons between ASRB sand LPDS schizophrenia/schizaffective disorder sample
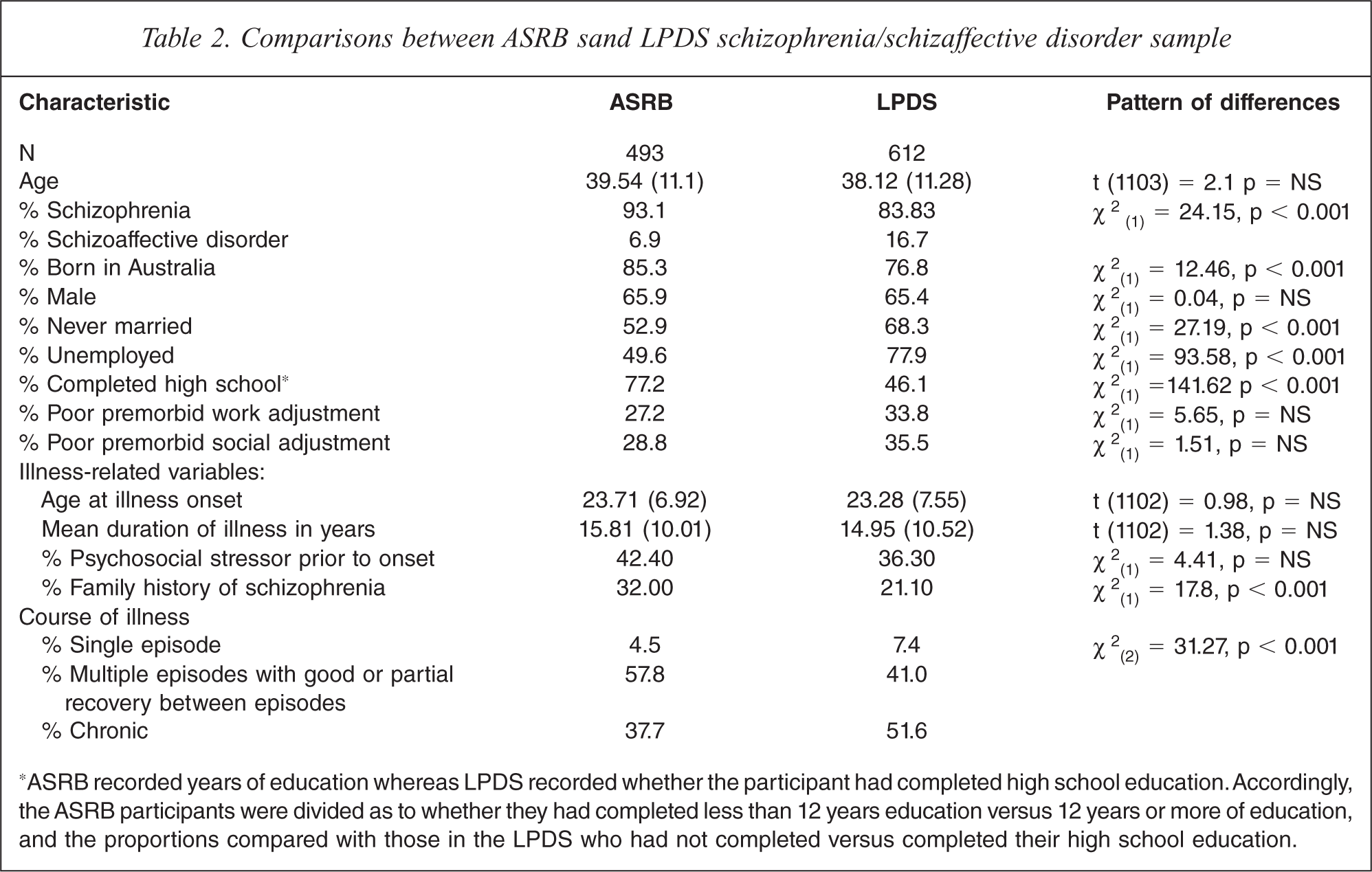
∗ASRB recorded years of education whereas LPDS recorded whether the participant had completed high school education. Accordingly, the ASRB participants were divided as to whether they had completed less than 12 years education versus 12 years or more of education, and the proportions compared with those in the LPDS who had not completed versus completed their high school education.
Discussion
The early stages of recruitment to the ASRB relied heavily on a national multi-media campaign and the strategy has since shifted to potentially more representative methods of recruitment through clinical services. Previous experience with recruitment through non-clinical services and general practitioners, rather than mental health inpatient units or community mental health facilities, has revealed that these recruitment methods deliver a sample of people with schizophrenia who are more highly functioning than those recruited through the latter more conventional sources [20]. In particular, volunteers to a schizophrenia research register and those recruited in general practices tend to have reported a pre-onset psychosocial stressor, display lower levels of disability post-onset, are more likely to be living in a married or de facto relationship, have higher levels of family support, are more likely to have completed school, report fewer reality distortion symptoms, are more likely to be employed and have fewer negative symptoms: they also report a less chronic course of illness [20]. Similarly, the initial method of recruitment to the ASRB has captured a sample with some bias apparent when compared to the LPDS sample where recruitment was predominantly through mental health services, supplemented by such sources as facilities for the homeless, and with relatively few participants recruited via general practitioners. In particular, the ASRB sample had a significantly lower proportion of individuals who had never been married, a lower rate of unemployment and higher educational achievement than the LPDS sample: the proportion reporting a chronic course of illness was also lower (and the proportion with multiple episodes and good or partial recovery was higher) in the ASRB sample.
This suggests that the ASRB sample is moderately biased in the direction of a more highly functioning group of patients than was obtained in the LPDS, in spite of the fact that the proportion with schizoaffective disorder, which is generally associated with less disability and a more favourable course of illness, was higher in the ASRB than the LPDS sample. However, in partial mitigation of this trend, it is noteworthy that the ASRB and LPDS samples did not differ significantly on other variables of interest, including premorbid work and social adjustment, age at onset, duration of illness or presence of a pre-onset psychosocial stressor.
It is not clear what to make of the findings that the ASRB sample contained a higher proportion of individuals born in Australia and a higher rate of reporting a family history of schizophrenia. This could reflect the initial recruitment strategy involving the national multi-media campaign, which may have been more effective in encouraging volunteers from Australian than other national backgrounds as well as prompting patients whose family history of schizophrenia may have sparked an interest in contributing to research on the genetic aspects of schizophrenia, given that the ASRB was promoted as a genetic epidemiology resource.
Selection bias is also apparent in the healthy controls. Although the statistically significant case-control differences shown in Table 1 were as expected, it is striking that the scores for premorbid and current IQ among the healthy controls were two thirds to one standard deviation above the norm, indicating that this is a relatively highly intelligent group, educated significantly beyond the high school level (mean 15.14 years of education). There is little doubt that this pattern is directly due to the multi-media recruitment methods to which individuals of higher intelligence and better education are presumably more likely to respond.
Now that recruitment of cases has moved to more conventional means via clinical services it will be important to keep track of the variables analysed above to determine whether recruitment methods are enabling acquisition of a sample whose clinical profile and other characteristics are similar to the LPDS sample while ensuring that we have a sample that represents the entire spectrum of possible outcomes of schizophrenia. Likewise, as the current sample of healthy control volunteers is supplemented by much larger numbers of individuals recruited by less biased epidemiological sampling methods, it is expected that the biases arising from the earlier recruitment methods will be counteracted.
Provided that the skewing towards higher functioning and higher intelligence in cases and controls, respectively, can be balanced by different recruitment methods as the ASRB progresses towards its target of 2,000 cases and 2,000 controls, it is likely that we will have a resource that is representative of a much broader spectrum of cases of schizophrenia in terms of functioning and level of disability than would be achieved by one recruitment method alone. If successful in this endeavour, the ASRB will be an especially valuable resource.
Conclusions
Schizophrenia is a complex and heterogeneous disorder. As such, there are likely to be multiple genetic and environmental factors that play causal roles in its pathogenesis, and there are likely to be multiple phenotypes distinguishable on the basis of combinations of clinical and cognitive variables, structural brain changes and other variables.
The ASRB aims to provide the foundation for advancing the understanding of schizophrenia by linking genetic, neuroanatomical, cognitive and clinical information in a large sample. The intended size of the sample will provide sufficient statistical power to allow researchers to address a range of important questions in relation to schizophrenia using the new technologies available to neuroscience. For example, it will be possible to specify certain sample characteristics (e.g. age range, gender, negative symptoms, cognitive impairments, structural brain changes, etc.) and determine whether these are associated, alone or in combination, with particular patterns of gene expression in cases. Such approaches have the potential to identify aetiologically significant subgroups with characteristic genetic, neuropsychological, and neuroimaging profiles.
The ASRB will also enable scientists who have promising original hypotheses, but no access to large clinical populations, to test their hypotheses on a sample that will contain representation of a wider than usual range of patients with schizophrenia, which they would not be able to do without such a facility. It will also be possible to combine ASRB data with other comparable datasets to help achieve the extremely large sample sizes needed to identify rare genetic variants and susceptibility genes of small effect.
Footnotes
Acknowledgements
