Abstract
Approximately one third of depressed patients fail to respond to multiple trials of standard treatments. These patients continue to experience highly distressing and disabling symptoms [1], and their remaining treatment options are limited. Currently, electroconvulsive therapy (ECT) is the only established therapy for this large percentage of patients with treatment resistant depression (TRD). While ECT is both effective and commonly used, it has substantial problems including the occurrence of highly distressing cognitive side effects [2–3,4]. Despite considerable and ongoing research into ECT modification, the forms of ECT which have superior efficacy still possess unwanted cognitive consequences. The development of a new therapy for TRD which has the superior efficacy of ECT but without the cognitive side effects would have great clinical value. One highly promising possibility which is currently under investigation is magnetic seizure therapy (MST). The aim of this paper is to review the development of MST to date, present the first Australian case history and describe the context of its development in relationship to the development of ECT.
Electroconvulsive therapy: current status
ECT is widely used in most psychiatric treatment settings in Australia. In the 2006 reporting period, 17 414 ECT treatments were reported to the chief psychiatrist's office in Victoria alone, involving 1706 individual patients. These treatments were provided in both the public (69.6%) and private sectors. Despite its common use, there remain major difficulties with ECT. Primarily, its use is complicated by the production of substantial side-effects, especially the development of cognitive impairment and in particular, memory impairment. The cognitive effects of ECT have been shown to be dependent upon numerous ECT parameters, namely electrode placement, dose and pulse width [2,5–6]. Memory difficulties following ECT are clearly highly distressing for patients [4]. Patients may refuse initial treatment with ECT because of these side effects. In addition, and perhaps even more telling, are anecdotal reports of many patients who have had successful ECT and will not return for follow-up treatment upon relapse of their depression because of the cognitive consequences of treatment.
Such consequences include variable degrees of post-ictal disorientation, anterograde and retrograde amnesia; again the degree and duration of which are dependent upon various stimulation parameters including electrode placement, with bilateral ECT (BL-ECT) having the most profound effect on cognition (for detailed review see Ingram et al. [2]). Disorientation following ECT is common and has been shown to last from approximately 11 to 40 min depending on stimulation parameters. Duration of disorientation has also been related to longer term cognitive difficulties, in particular the extent of retrograde amnesia at 2 months post-ECT [7]. Where anterograde amnesia is present, research has found that improvements in memory function can continue for up to six months, while recovery from retrograde amnesia is considerably slower and may not always be absolute [2]. The amnestic effects of ECT are reported to occur independently of treatment response [3]. While memory impairment is the most commonly reported cognitive side-effect following ECT, there has been less research into other cognitive domains which may also be affected. As reviewed by Ingram et al. [2], the research that has been conducted into other cognitive domains (i.e. attention, information processing, visuospatial ability, executive functioning and language) has, as a result of methodological limitations, produced highly variable findings.
The development of a modified ECT treatment procedure, which is able to reduce or minimize memory-related side effects whilst maintaining efficacy, has been the subject of a considerable amount of investigation [8–10]. Studies of ECT modifications were occurring as early as the 1960s. Initially, d'Elia reported that non-dominant temporal lobe unilateral ECT (RUL ECT) was potentially as effective as BL ECT but with less cognitive impairment [11]. Subsequent research has indicated that both efficacy and cognitive side effects of RUL ECT are dose-dependent [6,10,12–13]. Low dose RUL ECT (generally defined as up to 1.5 times seizure threshold) results in less cognitive impairment but without the efficacy of standard BL ECT [6,9,12,14]; while RUL ECT delivered at dosages of at least 5 times the seizure threshold (considered high dose) appears to result in greater efficacy than low dose RUL ECT with reports of corresponding increases in cognitive side effects, however, findings of increased cognitive side effects are not universal [10,14]. Another alternative electrode placement which has been investigated is bifrontal ECT (BF ECT); with some studies suggesting that bifrontal ECT may be as efficacious as temporal BL ECT, but with less severe cognitive impairment [15–18]. More recent randomized trials of BF ECT have produced somewhat inconsistent findings with respect to the efficacy-side effect profile of BF-ECT [19] and additional research is clearly required to ascertain the relative clinical and cognitive outcomes of BF-ECT. Most recently, the Consortium for Research in ECT published the first randomized multisite study of BL-ECT versus BF-ECT versus High Dose RUL-ECT (i.e. 6 times seizure threshold) [20]. The study found that while all forms of ECT were highly efficacious, BL-ECT resulted in the most rapid response. With respect to cognitive outcomes there was little difference between the groups, with the only significant finding being significantly poorer performance on immediate and delayed recall on word lists following BF-ECT. It was concluded that BL-ECT was the most efficacious electrode placement, with respect to degree and speed of response; with the alternate placements also failing to result in better cognitive outcomes [20].
Ultra–brief ECT is the most recently investigated modification and involves shortening the width of electrical pulse delivered to around 0.3 ms (compared to 0.5 and 1.5 ms) [21]. Ultra-brief ECT may more efficiently stimulate neurons (reducing the overall charge required) and may result in less spread of electrical current and thus more focal stimulation. Sackiem et al. reported equivalent efficacy of ultra brief RUL-ECT to high dose standard pulse RUL-ECT with a significant reduction in cognitive side effects with ultra brief ECT [5]. Loo et al. also reported equivalent efficacy of ultra brief RUL-ECT unilateral when compared to high dose RUL-ECT again with a reduction in cognitive side effects [21]. Finally, Sienaert et al. compared two forms of ultra brief ECT, high dose unilateral and bifrontal, concluding that both were efficacious with minimal cognitive side effects [22]. While the research to date indicates that ultra-brief ECT does have antidepressant efficacy and seems to reduce cognitive side effects, it appears that a greater number of treatments are required to produce an equivalent therapeutic effect when compared to standard ECT [5,21–23]. Ultra brief ECT is a promising and emerging area in ECT research with more research needed to ascertain both its relative effectiveness and cognitive outcomes.
Despite the considerable research to date into ECT modifications, there are only a limited number of factors that can be varied in ECT. This is particularly true of the way in which the seizure is initiated [8]; the shunting of the electrical current produced by the resistance of the skull ensures the stimulus cannot be focused to any substantial degree regardless of electrode placement, stimulus intensity and pulse width. In addition, currently the form of ECT with the most proven efficacy still carries a significant cognitive side effect burden.
Magnetic seizure therapy
Introduction to magnetic seizure therapy
Magnetic seizure therapy (MST) is an alternative form of convulsive therapy which is able to produce a highly focal seizure, and thus potentially overcome one of the current limitations of ECT. Like ECT, MST involves the intentional induction of a seizure for therapeutic purposes [8]. However, the induction of a seizure occurs through the use of high frequency repetitive transcranial magnetic stimulation (rTMS) rather than a direct electrical current as occurs with ECT. rTMS is a procedure that involves the use of a rapidly timed variable high intensity magnetic field which is able to pass into the brain without resistance to induce changes in focal brain activity. It has been investigated in its own right as an antidepressant technique, and while rTMS appears to have value as a treatment, to date it has not shown equivalent efficacy in treating depression when compared to ECT [24–25]. During MST, stimulation is applied at frequencies and intensities much greater than that utilized in standard rTMS treatment. Therefore, in MST stimulation is delivered in such a way that the increase in brain activity it produces results in spontaneous electrical discharges, producing a transient seizure.
Seizure induction in MST: contrast with ECT
The seizure induced using MST is quite different from that produced with ECT, in that its origin is in superficial regions of the cortex. Also, unlike ECT, no electrical current passes deep through the brain. Therefore the main difference between MST and ECT relates to the capacity to focus stimulation [26]. As the electromagnetic pulse generated by MST passes into the brain without resistance, it is not subjected to the same diffusion that the electrical current applied in ECT is. Although a number of approaches attempt to apply ECT in focused ways (e.g. frontal, temporal), the substantial impedance of the scalp and skull means that the bulk of the electrical stimulus is shunted away from the brain, resulting in widespread stimulation of cortical and subcortical regions [8]. However, as there is no resistance to the passage into the brain of the magnetic stimulus, MST can be focused more precisely. Neurophysiological investigations into the seizure induced via MST in non-human primates, discussed below, have shown that the electrical field and the resultant seizure are indeed more focal than seen with ECT. Therefore, it is possible that MST may produce similar therapeutic benefits to ECT without inducing memory-related side effects, as there is no direct electrical stimulation of medial temporal lobe structures, such as the hippocampus, which are implicated in ECT-related memory impairment [8].
Another substantial difference in regard to the mechanism of seizure induction between MST and standard, or brief pulse, ECT related to pulse width. Basic physiological studies have suggested that the best pulse width for stimulating cortical neurons may be briefer than that used in standard ECT; hence the impetus for investigating ultra brief ECT. Briefer pulses more efficiently excite neurons at lower charge densities and have a larger safety margin because of their lower charge per phase [27]. The pulse width of a MST stimulator (typically 0.2 ms) is in the ultra brief range. This may therefore be expected to enhance its efficacy of seizure induction, although the pulse waveform itself differs between magnetic stimulation and ECT.
Finally, the mechanism through which the seizure is propagated may also differ between ECT and MST. Although the location of the induced current remains focal with MST, the distribution of physiologic effects resulting from neuronal depolarization and trans-synaptic activity is expected to extend beyond the superficial cortex [8]. Stimulation with MST will result in the spread of seizure activity from the stimulated to other regions but there is also the possibility that the firing of depolarized neurons at the site of activation will produce effects in distant cortical regions independent of the seizure-related activity.
Early MST investigations
Non-human primate studies
The initial research with MST was conducted in non-human primates with the first MST-induced seizure reported in 1998 [28]. Seizures were induced across eight trials in two adolescent male Macaca mulatta using 40 Hz stimulation at 90% of maximal stimulator output for a period of 4–5 seconds. The monkeys were anaesthetized with ketamine hydrochloride (15 mg/kg) for the majority of the trials, and muscle relaxation was achieved with succinylcholine chloride (2 mg/kg). This initial study was able to demonstrate the feasibility of inducing a seizure using rTMS under general anaesthesia. Following this, neuropathological investigations were undertaken, in which 12 Macaca mulatta received 6 weeks of ECS (electroconvulsive shock, the animal equivalent of ECT), 6 weeks of MST or 6 weeks of anaesthesia as a control. There were no histological lesions found following the intensive courses of ECS and MST, thus providing strong evidence for the contention that neither therapy produces structural brain damage [29]. Further research then looked at the differential neurophysiological characteristic of the ECS and MST seizure [28,30]. Studies used both intracerebral and scalp EEG electrodes in non-human primates receiving ECS, MST or sham stimulation to provide seizure characterization. These studies found differential patterns of EEG activity following ECS and MST. Namely, following MST the electric field and resultant seizure is more focused than with ECS, the electric fields induced by MST within the cortex were less intense and more confined to the superficial cortex. MST-induced seizures were less robust, less generalized to deeper brain structures (i.e. hippocampus), and had less post-ictal suppression [28,30].
The effects of MST on cognition were also studied within the non-human primate using the validated Columbia University Primate Cognitive Profile [31]. The CUPCP is a computerized cognitive battery which measures post-intervention orientation time and long-term memory; working memory, immediate memory and delayed retention; and serial memory. Using this battery, investigators found that long-term (5 weeks) daily exposure to MST resulted in fewer cognitive side effects than ECS [32]. Specifically, monkeys were slower to complete tasks following ECS and exhibited more errors for long- and short-term memory and recall tasks when compared to MST.
There have been several important outcomes of the early non-human primate research. First, MST was shown to not produce identifiable histological lesions in the brain of primates [29]. Second, information in regard to optimal stimulation parameters and seizure characterization was gathered [30]. Third, there appeared to be fewer cognitive side effects with MST as opposed to ECS in this animal model [32].
Human studies
Human studies of MST began in 2000. In Bern, Switzerland, a 20-year-old female with treatment resistant major depression received four MST treatments before beginning a course of ECT [33]. Following this the same group provided 12 MST treatments to a patient with treatment-resistant depression. In these initial case reports, MST was applied at 40–50 Hz at 100% of machine output. The MST was found to be well-tolerated and effective [33–34]. Tonic-clonic seizures were elicited after each MST session, both patients tolerated the treatments well (the first patient reported a mild headache after one treatment) with no subjective memory complaints (in the first patient mini mental status exams were performed throughout the treatments and remained at 30/30), time to reorientation was more rapid than that seen in ECT and in the first patient the Hamilton Rating Scale for Depression decreased from 20 to 13 after the fourth treatment [33–34]. In a subsequent study of 10 inpatients with a major depression episode, ECT sessions were replaced with MST within a treatment course of ECT [26,35]. These patients were administered MST at threshold in two of their first four convulsive therapy sessions, and suprathreshold MST in one of the last two sessions of their treatment course. MST sessions were better tolerated and resulted in fewer acute side effects than the ECT [26,35]. Specifically, following the MST sessions, patients had a significantly reduced time to reorientation and performed better on tests of attention, memory and verbal fluency when compared to post ECT. Subjectively, patients reported fewer side effects following the MST sessions. Of note, both raters and patients were blind to treatment type, i.e. ECT or MST, for each session.
A controlled efficacy investigation was undertaken subsequent to the initial safety and feasibility investigations. Twenty patients with major depressive disorder were treated with a full course of MST using the same 50 Hz device across two sites [35–36]. Mood improvement was seen following the MST treatment course, along with fewer side effects and dramatically more rapid reorientation post stimulation than an ECT comparison group. However, the magnitude of improvement did not seem as great as that which is generally seen with ECT, with only 53% of patients achieving a 50% or greater improvement following MST. The stimulation dose was only on average 1.3 times above magnetic seizure threshold, and as such the authors suggested that substantially greater response rates may have been achieved with higher stimulation intensity. Especially as ECT response rates have been shown to be highly sensitive to dose relative to seizure threshold [14]. However, this was not possible with the MST stimulators used at that time, which could only produce short trains (8 s) of 50 Hz stimulation at high intensity. Therefore, the advancement of MST relied on the development of stimulators capable of producing longer trains at higher stimulation intensities.
100 Hz MST
In the late 2000s the technology used to produce MST advanced considerably, with two companies having developed MST devices capable of stimulating continuously at 100 Hz for up to 10 s at full machine power. 100 Hz stimulation has been shown in primate experiments to much more reliably induce seizures while still demonstrating fewer cognitive side effects than conventional ECS [37]. 100 Hz MST has been tested in human subjects, with the first patient receiving treatment in 2006. The first 11 patients to receive 100 Hz MST did so in a single session during a regular course of ECT [38]. In 10 of 11 patients seizures were elicited. All patients had a more rapid recovery of orientation (on average 15 minutes shorter) and reported less confusion than following ECT. Initial clinical outcome data with this type of high dose 100 Hz MST is also promising. In 10 patients treated twice a week with 100 Hz MST at 100% for between one and six seconds in Bonn, Germany, 7 patients (70%) met criteria for clinical response without any side effects such as headache, nausea, dizziness or cognitive/memory impairment [39]. To date, MST has only been compared with the more clinically utilized forms of ECT, i.e. standard pulse BL-ECT and RUL-ECT, but not with ultra-brief ECT.
Further clinical trials are currently underway to investigate the comparable efficacy and safety of 100 Hz MST to ECT. In the following case study we describe the outcomes of the first patient treated in Australia with MST.
Case study
Background
Mrs S is a 63-year-old woman with a history of severe recurrent depression since the age of 27. She has had numerous in-patient hospital admissions and has been treated with variable success with sertraline, fluoxetine, venlafaxine, amitriptyline, escitalopram, reboxetine, fluvoxamine, duloxetine, paroxetine, olanzapine, valproate, lithium and ECT. She has previously responded to some of these antidepressant medications, however, the benefit she achieves has mostly been quite short term. She has also responded to ECT in the past. In 1976 she underwent a course of 16 bilateral ECT treatments, in 2001 she was treated with a course of 18 bilateral ECT and in 2003 she had five bilateral treatments. Despite a favourable mood response, Mrs S experienced considerable disorientation and anterograde amnesia following these treatments. Both she and her family found these side effects distressing, and as such Mrs S was not prepared to undergo repeat ECT. In addition, Mrs S underwent and failed to respond to a course of rTMS immediately prior to her MST treatment. She received 20 active bilateral rTMS treatments (low frequency right stimulation followed by high frequency left stimulation) to the dorsolateral prefrontal cortex over a four-week period. As Mrs S did not benefit from the course of rTMS she was referred to the MST programme.
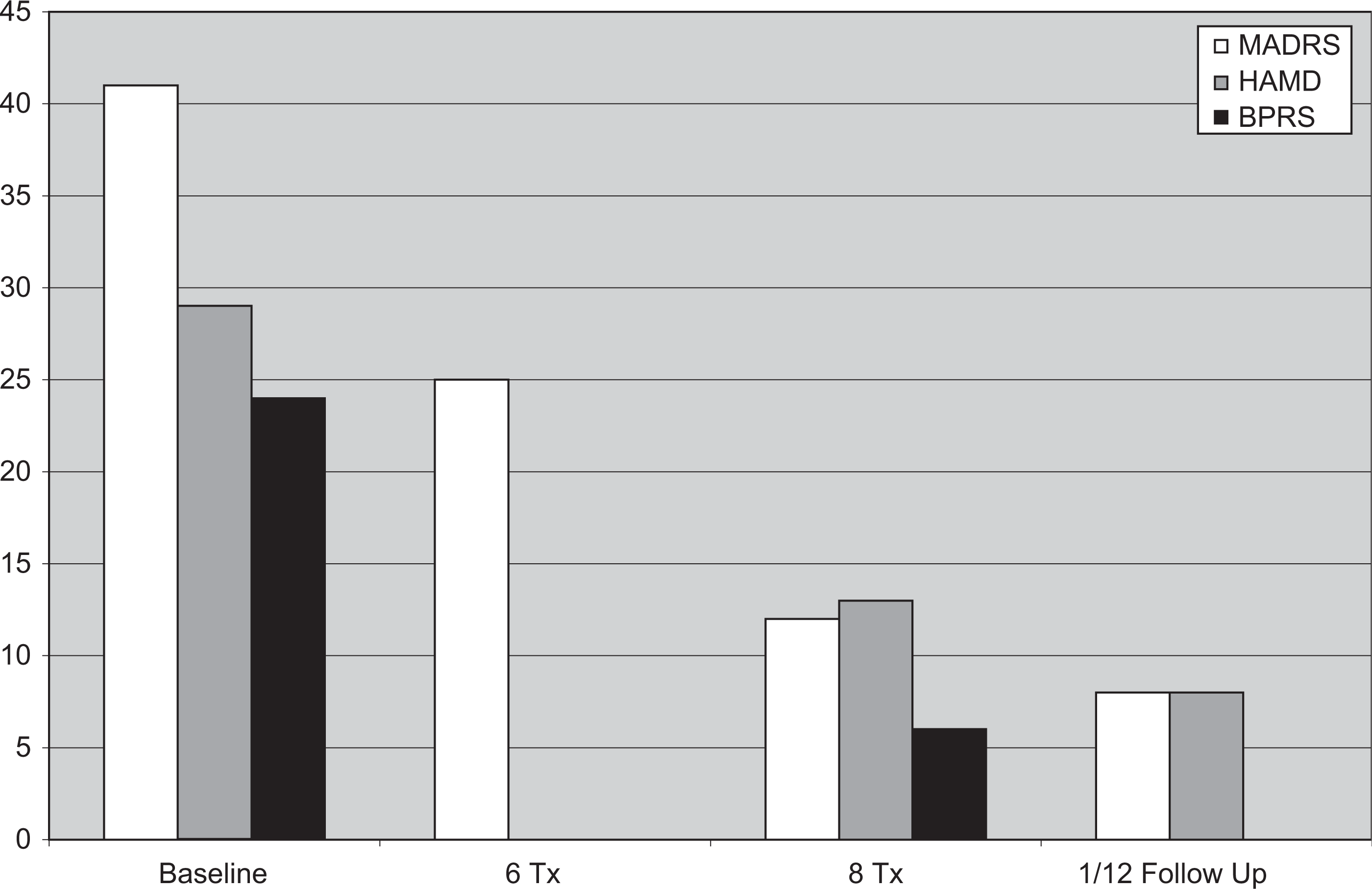
Clinical Scale Scores on the MADRS, HAMD and BPRS from baseline to one month post treatment.
MST treatment procedure
MST was provided three times a week on Monday, Wednesday and Friday. Propofol (100 mg) and succinylcholine (50 mg) was used for anaesthetic induction and muscle relaxation. Her ictal EEG was monitored throughout using rTMS/MST compatible frontal and mastoid electrodes. Single simulation trains of 1000 pulses were applied at 100 Hz and 100% of total machine output at each treatment session. At this stage as insufficient knowledge is available as to the relative benefits of dosing MST in relation to individual seizure thresholds, maximal dosing was utilized to ensure the best possible chance of seizure induction. The MST (Mag Venture A/S, Farum, Denmark) coil used was a bilateral dual cone coil, and stimulation was applied to the vertex.
Mrs S had a total of eight MST treatments and experienced a grand mal seizure during each. The seizures lasted on average 22.65 seconds (16 seconds visual seizure). Her orientation was also assessed after each treatment. She was fully oriented upon initial assessment after waking from anaesthetic following each seizure. This equated to on average 5 min 55 seconds from end of visual seizure, and 68 seconds on average from initial waking from anaesthetic, which was essentially the time taken to conduct the orientation assessment.
Outcomes
Mrs S underwent comprehensive clinical and neuropsychological assessments at baseline and at the end of her treatment course; she also underwent brief clinical review following six treatments. Mrs S was beginning to show improvement at the brief clinical review, and had meet response criteria after eight treatments. A follow up review was undertaken at one month post treatment course, and Mrs S showed continued improvement. Her clinical scale scores are shown in Table 1.
Clinical scale scores on the MADRS, HAMD and BPRS from baseline to one month post treatment

Mrs S did not experience any disorientation following the MST treatments.
The neuropsychological assessment covered the following domains; attention and concentration, speed of information processing, anterograde and retrograde memory, perceptual ability, and executive functioning; the results of which are provided in Table Two. Mrs S experienced mild improvements across a number of cognitive tasks, including verbal memory for stories and visual memory, verbal fluency and speed of information processing. These improvements are most likely to be related to the improvements in her mood. She did exhibit greater attentional fluctuation during the post MST assessment, with some subsequent mild memory inefficiencies for unstructured verbal information (i.e. word lists), which is consistent with a degree of ongoing depression symptomology and the fatigue as noted at the time of assessment. Assessments revealed no apparent MST-related cognitive side effects.
Cognitive outcomes following successful course of MST
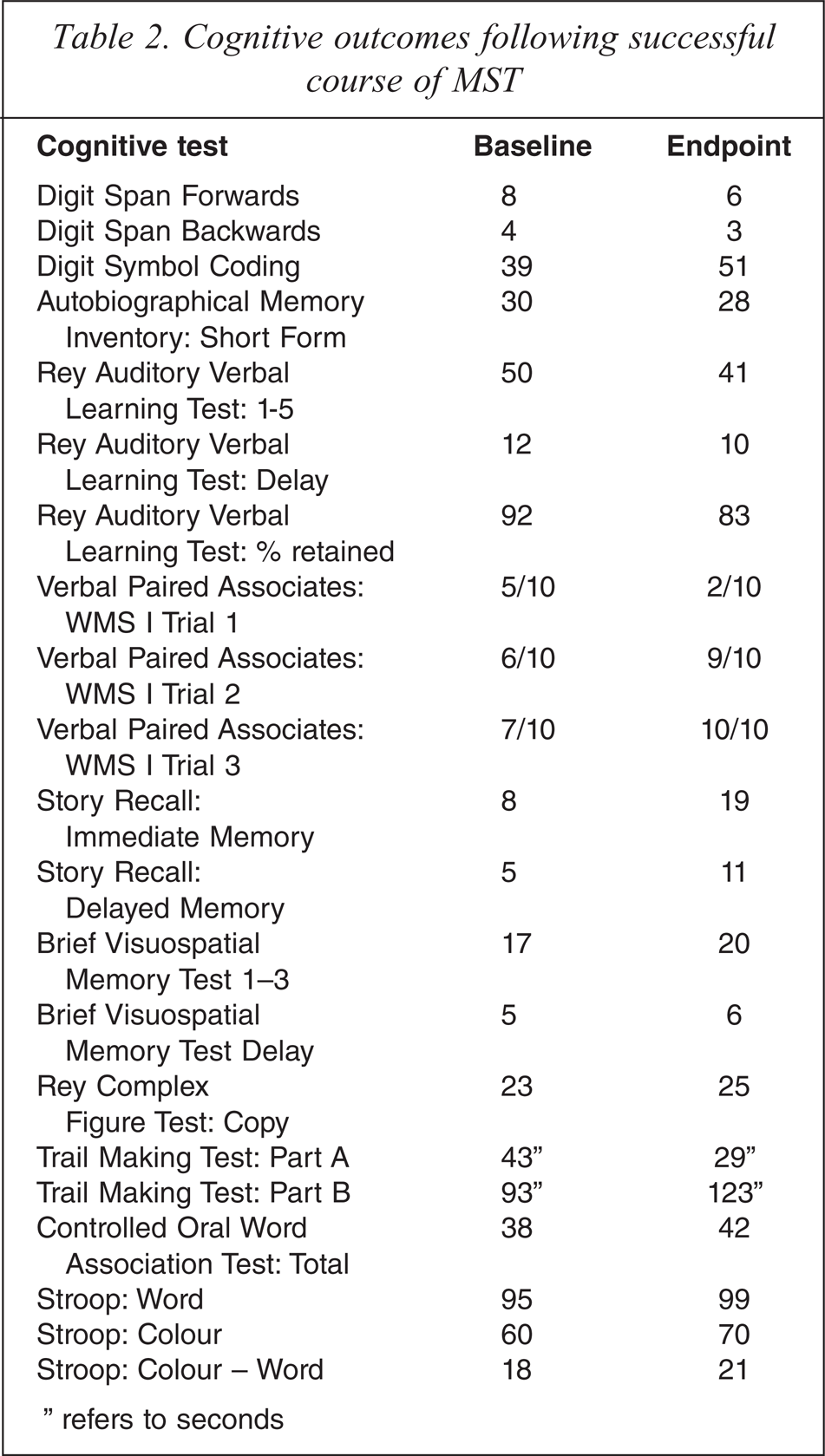
″ refers to seconds
Conclusion
The development of MST has been necessitated by the presence of cognitive side effects following ECT. Initial research with MST suggests that it may be able to produce similar clinical effects to ECT but without these disabling cognitive side effects. However, experience with the technique to date is limited and further research is required. MST has the potential to provide substantial ongoing benefits to patients, such as greater tolerability of the procedure and potential acceptability to patients; and extremely rapid rate of recovery of post procedure orientation compared to ECT as seen in the initial pilot studies. The faster rate of recovery also suggests that MST would be a more realistic outpatient treatment. The period of observation following ECT is determined by both the anaesthetic and the time taken for orientation, as MST patients are more rapidly orientated they would not require such lengthy periods of observation post procedure. Such benefits would potentially make MST much more available to patients, reduce currently overburdened psychiatric hospital bed use and decrease health care costs.
Footnotes
Acknowledgements
The authors would like to acknowledge the assistance of Ms Anne Maree Clinton and Dr Revi Nair in the provision of the MST.
