Abstract
Objective:
Recent evidence suggests that adjuvant anti-inflammatory agents could improve the symptoms of patients with schizophrenia. However, the effects of the adjuvant anti-inflammatory agents on cognitive function, general functioning and side effects have not yet been systematically investigated. The present meta-analysis aimed to explore the effects of anti-inflammatory agents in patients with schizophrenia comprehensively.
Method:
We performed a literature search in online databases, including PubMed, EMBASE and the Cochrane Database of Systematic Reviews. Randomized, placebo-controlled double-blind studies that investigated clinical outcomes including psychopathology, neurocognition, general functioning and extrapyramidal side effects were included. The examined anti-inflammatory agents included aspirin, celecoxib, omega-3 fatty acids, estrogen, selective estrogen receptor modulator, pregnenolone, N-acetylcysteine, minocycline, davunetide and erythropoietin.
Results:
Sixty-two double-blind randomized clinical trials studying 2914 patients with schizophrenia met the inclusion criteria for quantitative analysis. Significant overall effects were found for anti-inflammatory agents for reducing total, positive and negative symptom scores in the Positive and Negative Syndrome Scale. Cognitive improvements were significant with minocycline and pregnenolone augmentation therapy. General functioning was significantly enhanced by overall anti-inflammatory agents. There were no significant differences in side effects compared with placebo. Baseline total Positive and Negative Syndrome Scale score and illness duration were identified as moderating factors in the effects of anti-inflammatory augmentation on psychiatric symptom improvements.
Conclusion:
The comparative evaluation of efficacy and safety supported the use of anti-inflammatory adjuvant therapy over the use of antipsychotics alone. However, future studies could focus on patients with homogeneous clinical profile to figure out more detailed effects of anti-inflammatory therapy.
Introduction
Schizophrenia is a psychiatric illness characterized by psychotic symptoms and accompanied by impairments in cognitive and social functioning (Janicak et al., 2014). The chronic course of this illness, along with its early age of onset, imposes tremendous direct and indirect costs that accumulate to rank schizophrenia as one of the most burdensome disorders worldwide (Rössler et al., 2005). The use of antipsychotic medication is efficacious for positive symptoms; however, reports of treatment resistance and limited response, especially in the realms of negative and cognitive symptoms, are abundant (Dixon et al., 1995; Erhart et al., 2006; Kucharska-Pietura and Mortimer, 2013; Miyamoto et al., 2012; Wiersma et al., 1998). Consequently, the importance of developing alternative or adjuvant treatment options has been advocated.
With evidence accumulating for the adverse role of an excessive inflammatory response in the pathophysiology of schizophrenia, anti-inflammatory agents comprise one of the promising options for adjunctive therapies in schizophrenia (Hanson and Gottesman, 2005; Mitra et al., 2017; Müller et al., 2013). Inflammation in the central nervous system causes neurons to emit inflammatory mediators (e.g. cytokines) that trigger cell death (Ransohoff et al., 2015), and the process of neurodegeneration resulting from such excessive inflammatory responses can lead to abnormal brain morphology (Najjar and Pearlman, 2015). Recent reports on the increased level of pro-inflammatory markers in first-episode psychosis (FEP) and acutely relapsed inpatients have suggested the influence of immune dysfunction in the pathogenesis and progression of schizophrenia (Miller et al., 2011; Upthegrove et al., 2014), and findings from preclinical studies have further corroborated the important role of the inflammatory response in the pathogenesis of schizophrenia (Monji et al., 2009; Torrey and Peterson, 1973). The association between inflammation and risk of schizophrenia has also been demonstrated by longitudinal studies reporting the effects of maternal exposure to infection during pregnancy (Brown and Derkits, 2009; Khandaker et al., 2013; Miller et al., 2013), as well as by a recent genome-wide association study that demonstrated the link between single nucleotide polymorphisms on chromosome 6p22.1, which includes immunity-related genes, and schizophrenia susceptibility (Ripke et al., 2014).
Increasing interest in the use of anti-inflammatory agents as adjunctive therapies for schizophrenia accompanies the need to assess treatment efficacy comprehensively. However, current studies report inconsistent findings. For instance, celecoxib, one of the most widely used nonsteroidal anti-inflammatory drugs (NSAIDs), selectively inhibits cyclooxygenase-2 (COX-2), which is upregulated by pro-inflammatory mediators and induces oxidative stress in brain tissues (Meyer et al., 2011). Significant improvements in the cognitive factor of the Positive and Negative Syndrome Scale (PANSS) have been demonstrated following 5 weeks of celecoxib augmentation (Müller et al., 2005), but no significant advantage of the 8-week celecoxib add-on therapy over antipsychotic monotherapy has been reported (Rapaport et al., 2005). Furthermore, a 26-week intervention with adjunctive omega-3 fatty acids, which regulate inflammatory processes in terms of eicosanoid metabolism and cytokine production (Seibert et al., 1994; Simopoulos, 2002), remarkably improved general psychopathology (Pawelczyk et al., 2016). In addition, 12 weeks of ethyl-eicosapentaenoic acid (E-EPA) supplementation significantly reduced symptoms in patients on clozapine (Peet and Horrobin, 2002). However, no significant effect on symptoms was also reported for 16-week treatment with daily E-EPA augmentation (Fenton et al., 2001).
Current meta-analytical studies examining the clinical effects of anti-inflammatory strategies in schizophrenia also have limitations. These studies have primarily focused on outcome measures indexed by changes in total PANSS or Brief Psychiatric Rating Scale (BPRS) scores (Müller, 2013; Nitta et al., 2013; Sommer et al., 2012, 2013). The effects of anti-inflammatory strategies on other significantly relevant clinical factors, such as cognition, real-life functioning and side effects, remain largely unexamined. The largest meta-analysis conducted to date examined seven anti-inflammatory agents and reported the therapeutic potential of N-acetylcysteine (NAC), estrogens and aspirin for schizophrenia (Sommer et al., 2013). However, the potential moderator variables of effects were not examined by meta-regression. Furthermore, the amelioration was only assessed as to PANSS total scores, and the specific changes in symptom subscales were not discussed. Recently published trials which evaluating the cognitive benefits of adjuvant anti-inflammatory therapy also remain unexamined. With these current limitations in mind, the present study aimed to provide an updated meta-analysis of the treatment efficacy of anti-inflammatory drugs in schizophrenia. We comprehensively examined the therapeutic profile of 10 anti-inflammatory agents, previously reviewed as effective therapeutic options for schizophrenia (Keller et al., 2013; Müller et al., 2013; Sommer et al., 2013), and assessed the efficacy of these treatment options in terms of psychopathology, cognitive functioning, general functioning and side effects. We also implemented a meta-regression framework to examine the possible effects of moderators for the efficacy of anti-inflammatory agents.
Method
Literature search
A systematic search strategy was employed to identify relevant studies. Two independent researchers conducted a two-step literature search. In the first step, a literature search was conducted in PubMed, EMBASE and the Cochrane Database of Systematic Reviews databases. The search spanned from database inception to August 2017, and the following keywords, including their synonyms and combinations, were used as search terms: ‘schizophrenia’, ‘anti-inflammatory agents’, ‘aspirin’, ‘celecoxib’, ‘Omega 3 fatty acids’, ‘EPA’, ‘docosahexaenoic acid (DHA)’, ‘estrogen’, ‘minocycline’, ‘pregnenolone’, ‘N-acetylcysteine’, ‘davunetide’ and ‘erythropoietin’. In the second step, the reference lists of the retrieved studies and reviews were manually searched for additional relevant publications. Eligible articles were selected according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) Statement (Figure 1).
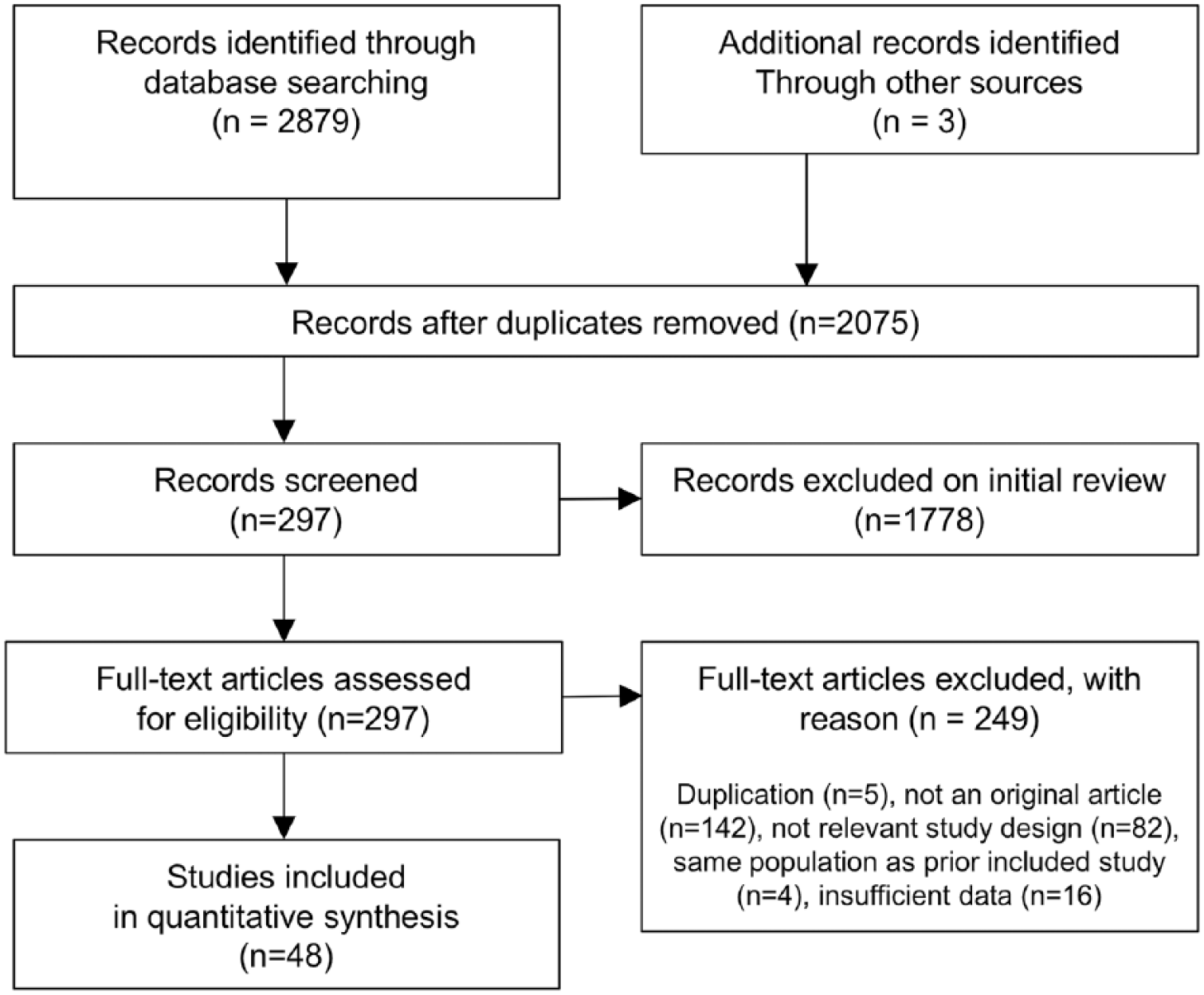
Preferred Reporting Items for Systematic Reviews and Meta-Analyses flow diagram.
Selection criteria
Studies included in the present meta-analysis met the following criteria: (1) studies published as an original article; (2) randomized, double-blind, placebo-controlled studies regarding antipsychotic augmentation with celecoxib, aspirin, davunetide, minocycline, estrogens (or raloxifene), omega-3 fatty acids, NAC or erythropoietin; (3) studies including patients with a diagnosis of a schizophrenia spectrum disorders; (4) studies providing sufficient information to calculate effect size (ES) statistics of changes in at least one outcome measure of interest and (5) literature written in English. We also included crossover studies in our analysis to obtain as much information as possible. Studies that used additional treatments, cognitive therapy for instance, along with anti-inflammatory agents were excluded. The eligibility of each randomized trial was assessed according to Cochrane’s tool for assessment of the risk of bias.
Data extraction
The following details were collected from the included studies: study location, year of publication, group characteristics, subject characteristics, experimental design, psychometric data, effect estimates and information required to complete the CONSORT checklist (Schulz et al., 2010). Pre- and post-treatment scores and F or p values for treatment effect were extracted when the mean and standard deviation (SD) value of the change scores were not reported (Lipsey and Wilson, 2001). When more than one study shared a study population, we included the one with a larger sample. The authors of the articles were contacted for unpublished information.
Outcome measures
Our primary outcome measures were the mean change (initial minus end-point ratings) in PANSS total and subscale scores. Our second outcome measures included the mean change in the scores of cognitive scales—e.g. MATRICS Consensus Cognitive Battery (MCCB; Nuechterlein et al., 2008), Brief Assessment of Cognition in Schizophrenia (BACS; Keefe et al., 2008), global assessment of functioning (GAF) and side effects measured on the Abnormal Involuntary Movement Scale (AIMS; Gharabawi et al., 2005) or Extrapyramidal Symptom Rating Scale (ESRS; Munetz and Benjamin, 1988). Results for cognitive functioning were grouped by the domains of attention, processing speed, language, visuospatial and constructional skills, visual learning/memory, verbal learning/memory, executive function or social cognition (Supplemental Table 1).
Statistical analysis
For outcome measures, we calculated the ES of the mean differences between the two treatment groups. Significant differences were determined by Hedges’ g test. A random-effects model was employed to take into account the variation between the retrieved studies. Primary analyses including the ES of all trials were conducted, and we then repeated the meta-analysis per drug. The composite of ES of each drug was also presented as a more accurate indicator of the overall effects of anti-inflammatory agents. When a study of cognitive functioning reported data on multiple cognitive tasks classified in the same domain, a combined ES from those tasks was used. The individual study with standardized residual >1.96 for each analysis was considered as an outlier and was excluded from the meta-analysis. Study heterogeneity was measured using I2 statistics. In the case of high heterogeneity, meta-regression or subgroup analysis was performed to identify the possible source of the heterogeneity. Potential moderator variables included treatment duration, treatment dose, age, sex, illness duration, antipsychotic dose (olanzapine equivalent) and PANSS total scores at baseline. The presence of publication bias was assessed by visually inspecting funnel plots. If the number of studies was 10 or larger, Egger’s test was conducted to test for asymmetry. To account for significant publication bias, we used the fixed-model trim-and-fill method to estimate the influence of small study effects. All statistical tests were two-sided and used a significance level of p < 0.05. The analyses were performed with Comprehensive Meta-Analysis V2 (Biostat, Inc., Englewood, NJ, USA) or Stata V14 (StataCorp, College Station, TX, USA).
Results
Our literature search yielded 62 eligible trials from 48 articles (PRISMA flowchart, see Figure 1). This search led to a dataset comprising 2914 patients with schizophrenia in total. All included studies compared the efficacy of using anti-inflammatory agents adjunctively with antipsychotics with that of antipsychotic monotherapy and provided information necessary to calculate changes in PANSS scores (k = 57), cognitive functioning (k = 14), GAF scores (k = 9) or extrapyramidal symptoms (k = 15). All included trials were double-blind randomized controlled trials (RCTs), and two of the included trials adopted a crossover design (Ji et al., 2016; Weickert et al., 2015). Study characteristics and patient demographics are listed in Table 1. The methodological quality of each study was evaluated in accordance with the Cochrane Handbook for Systematic Review of Interventions (Supplemental Table 2). Examined drugs included aspirin, celecoxib, omega-3 fatty acids, estrogens, pregnenolone, minocycline, NAC, erythropoietin and davunetide. Publication bias was detected in two of the analyses and was corrected with the trim-and-fill method.
Summary of study characteristics and demographics.
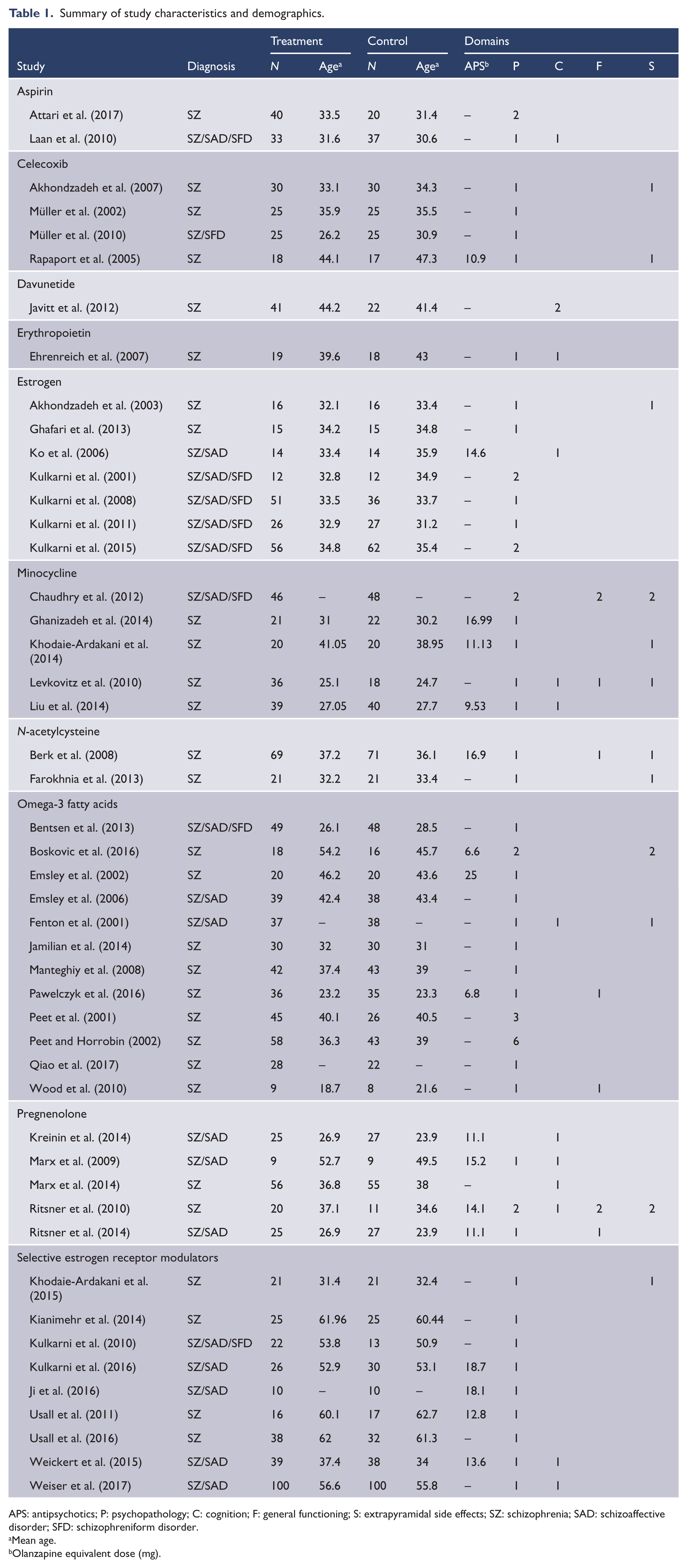
APS: antipsychotics; P: psychopathology; C: cognition; F: general functioning; S: extrapyramidal side effects; SZ: schizophrenia; SAD: schizoaffective disorder; SFD: schizophreniform disorder.
Mean age.
Olanzapine equivalent dose (mg).
Psychopathology
Overall effects of all anti-inflammatory agents
In total, 57 trials investigated the effects of nine anti-inflammatory agents in terms of psychopathology (Table 2). Davunetide was excluded from the present meta-analysis due to the lack of eligible studies. Our meta-analysis revealed the superior efficacy of anti-inflammatory agents in reducing PANSS total (g = 0.41, 95% confidence interval (CI) = [0.26, 0.56]), positive (g = 0.31, 95% CI = [0.14, 0.48]) and negative (g = 0.38, 95% CI = [0.23, 0.52]) scores. Heterogeneity was moderate to high (I2 = 74.2%, 75.5%, 66.0%, each), and subsequent meta-regression analysis revealed that baseline total PANSS scores significantly modulated the effects of anti-inflammatory agents for PANSS positive scores (n = 43, p < 0.01). Publication bias was present for PANSS total and positive symptoms (p < 0.05). For total symptoms, trim-and-fill correction identified three missing studies, but the ES was still significant (g = 0.32, 95% CI = [0.16, 0.49]). The ES for positive symptoms also remained the same after correction.
Treatment efficacy of anti-inflammatory agents.
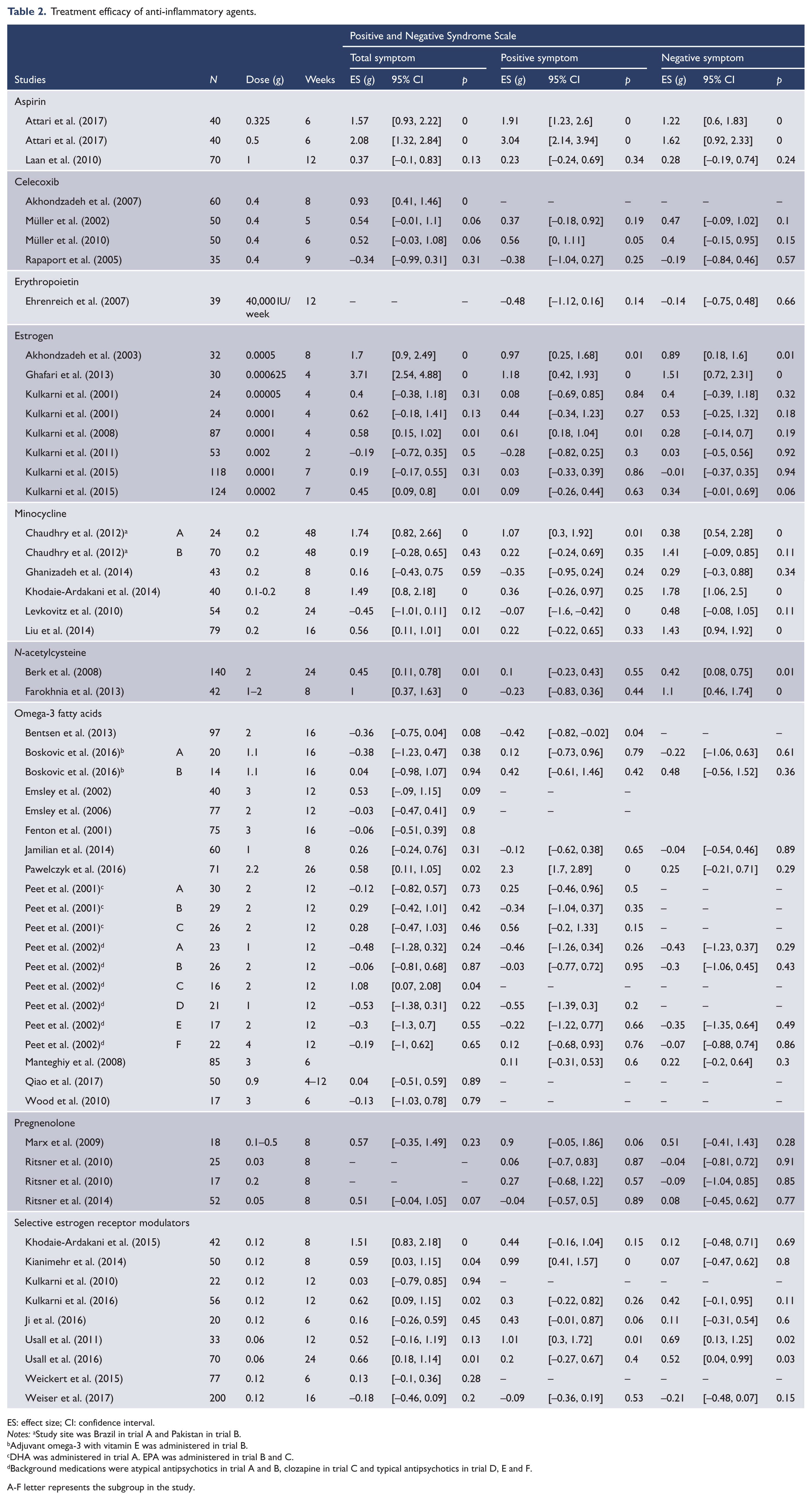
ES: effect size; CI: confidence interval.
Notes: aStudy site was Brazil in trial A and Pakistan in trial B.
Adjuvant omega-3 with vitamin E was administered in trial B.
DHA was administered in trial A. EPA was administered in trial B and C.
Background medications were atypical antipsychotics in trial A and B, clozapine in trial C and typical antipsychotics in trial D, E and F.
A-F letter represents the subgroup in the study.
When the pooled ESs for each anti-inflammatory agent were combined, the estimated efficacy increased for PANSS total scores (k = 8, g = 0.48, 95% CI = [0.22, 0.73]; see bar plot in Figure 2), while it decreased for positive (k = 9, g = 0.19, 95% CI = [0.03, 0.35]) and negative (k = 9, g = 0.30, 95% CI = [0.10, 0.50]) scores. Heterogeneity was low to moderate (I2 = 65.9%, 30.9%, 59.0%, each). The ES was still significant when we narrowed the criteria for the anti-inflammatory agent to celecoxib and aspirin with a direct anti-inflammatory mechanism (g = 0.59, 95% CI = [0.14, 1.04]).
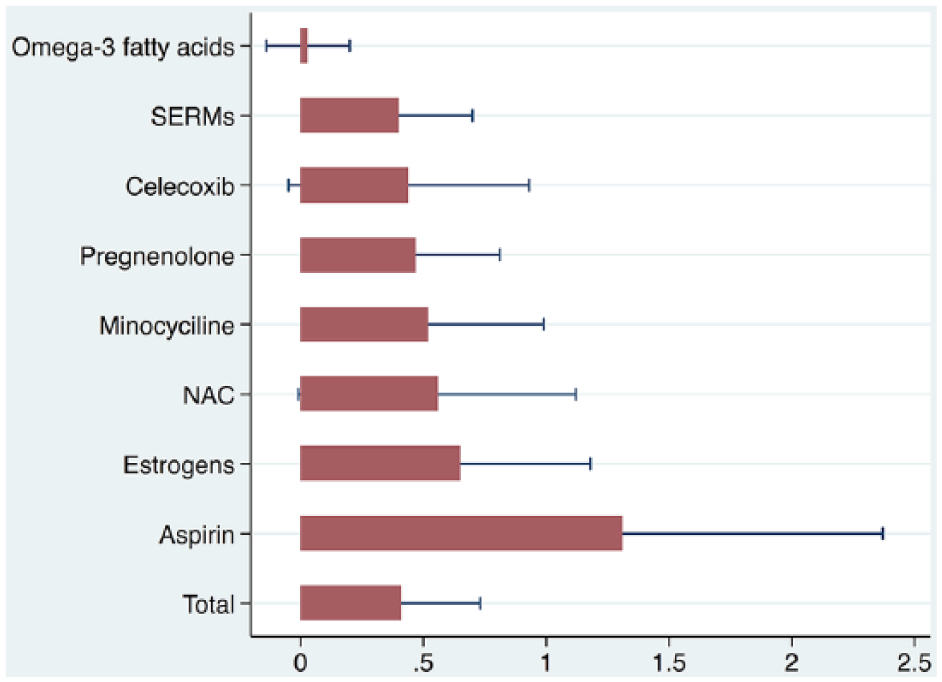
Effects of anti-inflammatory agents on total PANSS scores.
Aspirin
Three trials evaluated the effects of aspirin in a total of 130 patients (Attari et al., 2017; Laan et al., 2010). Adjunctive use of aspirin was more effective than antipsychotic monotherapy in improving PANSS total (g = 1.31, 95% CI = [0.25, 2.37]), positive (g = 1.69, 95% CI = [0.05, 3.33]) and negative (g = 1.01, 95% CI = [0.18, 1.83]) scores. Heterogeneity was high (I2 = 88.8%, 94.5%, 83.0%, each). Subgroup analysis showed that aspirin treatment was more effective for clinical trials with higher baseline PANSS total score, lower treatment dose and shorter treatment duration (Supplemental Table 3).
Celecoxib
Four RCTs investigated the effects of celecoxib in 195 patients (Akhondzadeh et al., 2007; Müller et al., 2002, 2010; Rapaport et al., 2005). There was no significant difference between add-on celecoxib and standard antipsychotic treatments in terms of PANSS total (g = 0.44, 95% CI = [–0.05, 0.93]), positive (g = 0.24, 95% CI = [–0.31, 0.79]) and negative (g = 0.26, 95% CI = [–0.12, 0.64]) scores. Heterogeneity was moderate to low (I2 = 66.6%, 62.4%, 11.8%, each).
Omega-3 fatty acids
Twenty trials examined the effects of omega-3 in a total of 778 patients (Boskovic et al., 2016; Emsley et al., 2002, 2006; Fenton et al., 2001; Jamilian et al., 2014; Pawelczyk et al., 2016; Peet et al., 2001; Peet and Horrobin, 2002; Qiao et al., 2017; Wood et al., 2010). Eleven trials administered eicosapentaenoic acid (EPA), one trial administered DHA and six trials administered a mixture of EPA, DHA or oleic acid. Six trials with patients taking different antipsychotics were retrieved from a dose-ranging study (Peet and Horrobin, 2002). No significant treatment efficacy was found for omega-3 in terms of PANSS total (g = 0.03, 95% CI = [–0.14, 0.20]), positive (g = 0.13, 95% CI = [–0.28, 0.55]) and negative (g = 0.03, 95% CI = [–0.18, 0.24]) scores. Heterogeneity was low (I2 = 21.4% and 5.4%) for total and negative symptoms but high (I2 = 81.2%) for positive symptoms. Meta-regression analysis revealed a trend-level effect of illness duration on changes in positive PANSS scores (n = 6, p = 0.074). Within the range of 0 to 16 years, there was a trend for shorter illness duration yielding larger ESs.
Estrogens
Eight trials assessed the effects of estrogens in 342 female patients with schizophrenia (Akhondzadeh et al., 2003; Ghafari et al., 2013; Kulkarni et al., 2001, 2008, 2011, 2015).
Among studies included for total symptom analysis, one study (Ghafari et al., 2013) with ES of 3.7 was excluded for high standardized residuals (p < 0.05). The results demonstrated the superior efficacy of estrogens in reducing PANSS total (g = 0.47, 95% CI = [0.13, 0.81]), positive (g = 0.33, 95% CI = [0.02, 0.64]) and negative (g = 0.40, 95% CI = [0.11, 0.69]) scores. Heterogeneity was moderate (66.2%, 62.6%, 56.7%, each).
Selective estrogen receptor modulators
Nine trials investigated the effects of selective estrogen receptor modulators (SERMs) in a total of 583 patients (Ji et al., 2016; Khodaie-Ardakani et al., 2015; Kianimehr et al., 2014; Kulkarni et al., 2010, 2016; Usall et al., 2011, 2016; Weickert et al., 2015; Weiser et al., 2017). The included trials all administered raloxifene. Adjuvant raloxifene therapy significantly reduced PANSS total (g = 0.40, 95% CI = [0.10, 0.70]) and positive (g = 0.41, 95% CI = [0.10, 0.72]) but not negative (g = 0.19, 95% CI = [–0.06, 0.45]) scores. While heterogeneity was moderate (I2 = 74.0%, 65.6%, 49.8%, each), publication bias was detected from the funnel plot. The trim-and-fill procedure significantly reduced the ESs for the total (g = 0.20, 95% CI = [–0.08, 0.50]), positive (g = 0.12, 95% CI = [–0.11, 0.36]) and negative (g = 0.09, 95% CI = [–0.12, 0.30]) symptom scores.
Pregnenolone
Four trials assessed the effect of add-on pregnenolone therapy for schizophrenia in 101 patients (Marx et al., 2009, 2014; Ritsner et al., 2010). Pregnenolone was more effective than antipsychotic monotherapy in reducing PANSS total (g = 0.52, 95% CI = [0.05, 0.99]) scores, while its effect on positive (g = 0.17, 95% CI = [–0.19, 0.54]) and negative (g = 0.09, 95% CI = [–0.27, 0.46]) symptom scores was not significant. Heterogeneity was low (I2 = 0%) for all analyses.
Minocycline
Six trials evaluated the effects of minocycline in a total of 310 patients (Chaudhry et al., 2012; Ghanizadeh et al., 2014; Khodaie-Ardakani et al., 2014; Levkovitz et al., 2010; Liu et al., 2014). The results demonstrated that adjunctive minocycline was more efficacious for reducing PANSS negative (g = 0.93, 95% CI = [0.42, 1.44]) scores than antipsychotic monotherapy. However, there was no significant difference between the two therapies in terms of total (g = 0.56, 95% CI = [–0.01, 1.12]) and positive (g = 0.19, 95% CI = [–0.11, 0.49]) symptom scores. Heterogeneity was high for total and negative symptoms (I2 = 82.4%, 42.0%, 77.6%, each). No influential moderator variable was suggested from the subgroup analysis since the heterogeneity remained high in each subgroup (Supplemental Tables 4 and 5).
NAC
Two trials, including 182 patients, assessed the effects of NAC (Berk et al., 2008; Farokhnia et al., 2013). NAC was superior to placebo in PANSS total (g = 0.65, 95% CI = [0.13, 1.18]) and negative (g = 0.70, 95% CI = [–0.04, 1.36]) scores, while the two treatments were not significantly different in terms of positive (g = 0.02, 95% CI = [–0.27, 0.31]) symptoms. Heterogeneity was moderate to low (I2 = 56.3%, 0%, 71.03%, respectively).
Erythropoietin
Only one trial using add-on erythropoietin was retrieved; it included 37 patients (Ehrenreich et al., 2007). There were no significant differences between the treatment efficacy of erythropoietin and placebo in terms of PANSS positive (g = –0.48, 95% CI = [–1.12, 0.16]) or negative (g = –0.14, 95% CI = [–0.75, 0.48]) scores.
General functioning (GAF)
GAF scores were reported as an outcome measure in nine trials that administered omega-3 fatty acids, NAC, minocycline or pregnenolone (Berk et al., 2008; Chaudhry et al., 2012; Levkovitz et al., 2010; Pawelczyk et al., 2016; Ritsner et al., 2010, 2014; Wood et al., 2010). Overall, GAF scores were significantly higher in the anti-inflammatory augmentation group than in the control group (g = 0.22, 95% CI = [0.04, 0.40]). However, the ES measured for each agent was not significant. The majority of the included trials reported no significant improvement in functioning except for one 26-week intervention study providing a 2.2-g combination dose of EPA and DHA (g = 0.61, CI = [0.14, 1.08]; Pawelczyk et al., 2016). Heterogeneity was low (I2 = 0–20%) for all analyses.
Neurocognition
Fourteen studies on aspirin, omega-3, estrogen, SERM, davunetide, minocycline, pregnenolone or erythropoietin were included in our analysis of cognitive improvement (Table 3). Significant cognitive benefits were found for minocycline (k = 10, g = 0.21, 95% CI = [0.04, 0.38]) and pregnenolone (k = 20, g = 0.19, 95% CI = [0.08, 0.29]).
Outcomes of cognitive tasks in each intervention trial.
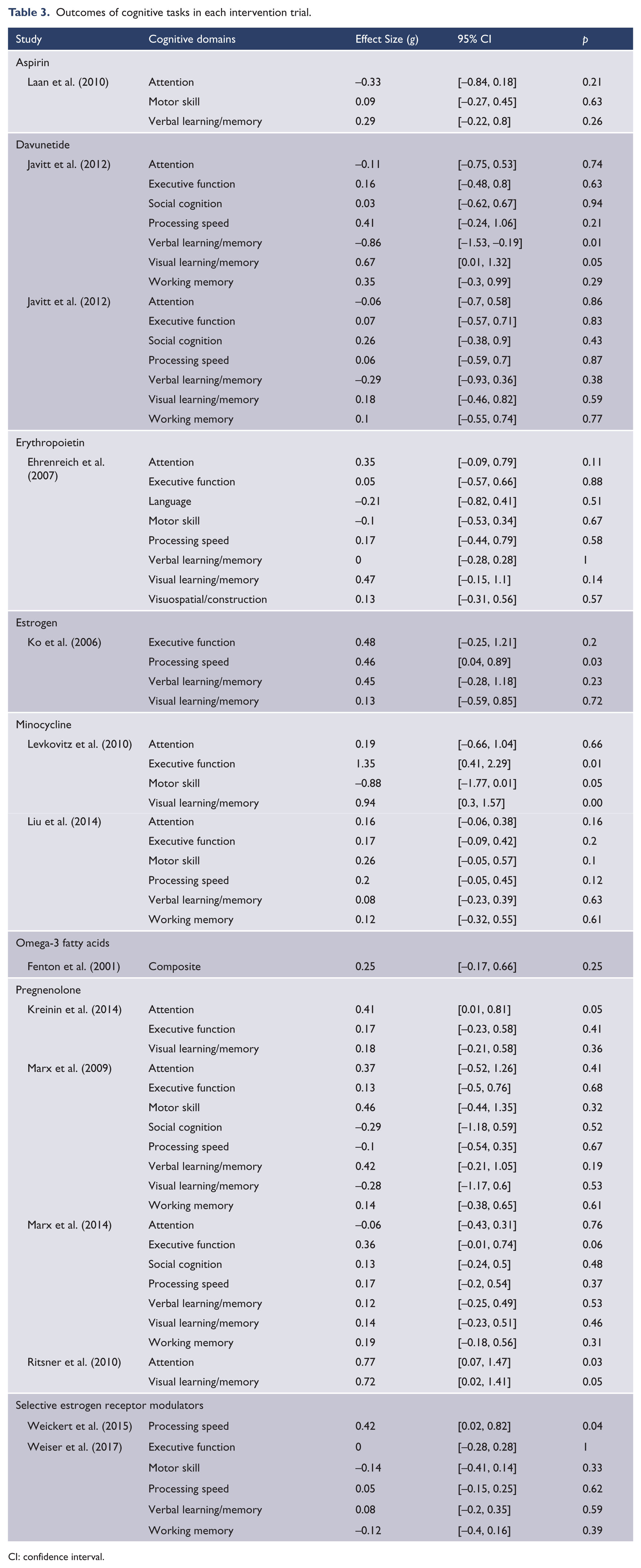
CI: confidence interval.
In minocycline augmentation trials, significant improvements were found in the cognitive domains of visual learning (Levkovitz et al., 2010), executive function (Levkovitz et al., 2010) and attention (Liu et al., 2014). For pregnenolone, one large study reported significant improvement in visual learning (Ritsner et al., 2014) and attention (Kreinin et al., 2014; Ritsner et al., 2014). There was no significant cognitive improvement when the effects of omega-3, estrogens, SERM, aspirin, davunetide and erythropoietin were pooled. Heterogeneity was low to moderate (I2 = 0–63.4%) for each augmentation strategy.
Side effects (ESRS/AIMS)
ESRS or AIMS scores were reported as outcome measures in 15 trials that administered omega-3 fatty acids (Boskovic et al., 2016; Emsley et al., 2002; Fenton et al., 2001), estrogens (Akhondzadeh et al., 2003; Khodaie-Ardakani et al., 2015), NAC (Berk et al., 2008; Farokhnia et al., 2013), pregnenolone (Ritsner et al., 2010), minocycline (Chaudhry et al., 2012; Khodaie-Ardakani et al., 2014; Levkovitz et al., 2010) or celecoxib (Akhondzadeh et al., 2007; Rapaport et al., 2005). No significant difference in ESRS or AIMS scores between groups (g = 0.07, 95% CI = [–0.08, 0.21]) was revealed by quantitative analysis. Moreover, subgroup analyses for omega-3 fatty acids, estrogens, SERM, NAC, pregnenolone, minocycline and celecoxib did not reveal any significant effects for side effects.
Discussion
We have comprehensively reviewed the efficacy of anti-inflammatory agents with various outcome measures, including psychopathology, neurocognition, general functioning and extrapyramidal side effects. The meta-analytic results of nine drugs showed that adjuvant anti-inflammatory agents could effectively reduce psychiatric symptoms measured on PANSS, with a small to moderate ES. Significant cognitive benefits were observed in minocycline and pregnenolone augmentation trials. The safety of five included anti-inflammatory drugs was also confirmed with the absence of additional side effects.
Psychopathology
The two NSAIDs included in our analysis greatly differed in their effects in PANSS score changes. The efficacy of aspirin was evident with a substantial ES (g = 1.31, 1.69, 1.01, each), but celecoxib augmentation trials did not show significant therapeutic potential. This finding is consistent with a previous review of NSAID augmentation (Nitta et al., 2013). This finding was largely driven by a patient subgroup of high baseline PANSS scores. Both adjuvant estrogen and raloxifene were superior to antipsychotics alone in ameliorating the psychopathology of schizophrenia, which is in line with a recent meta-analysis of sex hormones (Heringa et al., 2015). Nonetheless, the estimated ESs of raloxifene did not remain significant after the trim-and-fill procedure. Pregnenolone, an estrogen precursor, was also significantly beneficial in reducing total PANSS symptom scores, with a moderate ES. A previous meta-analysis including a sample of a 10-week intervention trial with patients having minimal baseline symptoms (Marx et al., 2014) did not yield significant effects for pregnenolone (Heringa et al., 2015). Considering the high baseline PANSS total scores (⩾61) of samples included in our analysis, this discrepancy might have resulted from the selective benefits of pregnenolone dependent on the clinical status of patients.
Omega-3 exerts anti-inflammatory properties by modulating eicosanoid metabolism and cytokine production (Calder, 2015; Mori and Beilin, 2004; Simopoulos, 2002). Nonetheless, the treatment efficacy of omega-3 fatty acids was not significant in the present review. However, the results of meta-regression suggested the therapeutic potential of omega-3 for participants with a short duration of illness. This result is mirrored by the subgroup analysis of a previous review, which revealed that the therapeutic action of omega-3 might be specifically beneficial for patients in the prodromal phase or FEP (Chen et al., 2015). Minocycline effectively reduced negative symptoms, but the high heterogeneity limited the reliability of the results. A recent meta-analysis of minocycline intervention, including trials with an open-label design or BPRS assessment, found beneficial effects of minocycline in reducing both total and negative symptoms (Xiang et al., 2017). Their subgroup analysis suggested several modulating factors to explain the high heterogeneity, e.g. the types of baseline antipsychotics, age and ethnicity of study participants. However, our analysis of potential moderator variables did not account for the between-study heterogeneity. NAC had significant efficacy in treating total and negative symptoms (g = 0.65 and 0.70, each), which confirms the results of the previous meta-analysis (Sommer et al., 2013), with larger patient samples. Notably, the effect of NAC on total symptom improvement was mainly driven by negative symptom improvement. The single study with erythropoietin did not reveal any significant effects in terms of psychopathology.
Identifying moderator variables for the effectiveness of anti-inflammatory agents would be of value for the design of future clinical trials. According to our meta-analysis, adjuvant anti-inflammatory drugs are recommendable for patients with severe baseline total symptoms, as a strong positive relationship was observed between the efficacy of anti-inflammatory agents and the baseline severity of total symptoms in the meta-regression of overall anti-inflammatory augmentation trials. This association was also replicated at a trend level among omega-3 trials (p = 0.10, n = 11) and was further supported by a trend for a lower baseline antipsychotic dose yielding larger ESs for positive symptom improvement in overall trials using anti-inflammatory agents (p = 0.12, n = 17). Previous literature reviews have mainly suggested that patients in an early disease stage tend to benefit more from anti-inflammatory drugs (Mitra et al., 2017; Müller, 2017). Our results have also verified this finding by showing that the efficacy of omega-3 in positive symptoms was negatively impacted by the illness duration of participants. Taken together, these results suggest that acutely ill patients with schizophrenia would benefit more from anti-inflammatory augmentation.
General functioning
A combined ES revealed a significant improvement in GAF scores, which assess psychological, social and occupational functions (g = 0.22). However, no single agent could be recommended in particular for functional improvement, since subgroup analyses of individual agents did not yield significant effects. The lack of effects on functional improvement could have been influenced by uncontrolled variables such as social support and vocational opportunities (Green et al., 2004).
Cognition
An increased level of systematic inflammation in patients with schizophrenia has been linked to changes in cognition and behaviors (Khandaker et al., 2015). Excessive activation of the immune system causes cognitive dysfunction directly through the effects of peripheral cytokines (Harrison et al., 2014; Yirmiya and Goshen, 2011) and activation of microglia (Doorduin et al., 2009; Van Berckel et al., 2008) or indirectly through modulation of neurotransmitters (Meyer and Feldon, 2009) and neurodegenerative processes (Davis et al., 2012). However, whether anti-inflammatory therapies can mitigate cognitive deterioration in schizophrenia has not been examined with meta-analytic approaches due to a lack of data. In our meta-analysis, the cognitive improvement was significant with minocycline and pregnenolone. Visual learning/memory, attention and executive function were the cognitive domains with significant changes in minocycline or pregnenolone augmentation trials. Although these results are promising in that attention, memory and executive function are the most damaged cognitive domains in schizophrenia (Buoli and Altamura, 2015), some caution seems justified as the cognitive scales used in each trial are quite heterogeneous.
Side effects
We did not find any significant changes in ESRS or AIMS scores with celecoxib, omega-3, estrogen, raloxifene, pregnenolone, minocycline and NAC augmentation. Moreover, no significant adverse effects were observed in clinical trials with other agents. Davunetide 5 or 20 mg resulted in a mild degree of self-reported restlessness (Javitt et al., 2012). Chronic administration of NSAIDs could cause gastric side effects (Hirsh et al., 1992), but the safety of aspirin was increased by the concomitant use of omeprazole, a proton inhibitor, in a previous clinical trial (Laan et al., 2010). Erythropoietin has hematopoietic properties, but thromboembolic effects on blood pressure were not reported in the treatment group (Ehrenreich et al., 2007).
The overall meta-analytic results indicated that a wide range of substances with anti-inflammatory actions could help patients with schizophrenia, thereby supporting the involvement of heightened inflammatory responses in the pathology of schizophrenia. Since the use of anti-inflammatory agents for schizophrenia is still in early stages, further investigation is required for the potential efficacy of other promising anti-inflammatory agents. Sodium benzoate, for instance, has been reported to enhance the antipsychotic effect in patients with chronic schizophrenia (Lane et al., 2013; Lin et al., 2018). As suggested in previous reviews, anti-inflammatory agents might inhibit excessive microglial activation and N-methyl-
Limitations
This study was subject to several limitations. First, other than primary anti-inflammatory agents, we have also examined broad-acting agents with anti-inflammatory properties. The therapeutic efficacy of those drugs should not be entirely ascribed to anti-inflammatory action. However, the ES of PANSS score change was still significant when the analysis was limited to celecoxib and aspirin. Second, subjects in any phase of illness and baseline symptom were included in our analysis. Meta-regression and subgroup analysis suggested that the variability of baseline clinical characteristics increased the overall heterogeneity of our meta-analytic results. Future studies with more specific patient selection are warranted to control for confounding factors. Finally, we have focused on the most frequently used scales (AIMS, ESRS and GAF) for our secondary outcome measures. Including other scales, Glasgow Antipsychotic Side-effect Scale (GASS), Medication Adherence Rating Scale (MARS) or Personal and Social Performance (PSP), Social and Occupational Functioning Assessment Scale (SOFAS) for instance, could permit a more comprehensive understanding of the effects of anti-inflammatory drugs.
Conclusion
Overall, the quantitative evidence proved that anti-inflammatory adjuvant treatments were favorable to antipsychotic monotherapy in terms of psychopathology and real-life functioning. Three cognitive domains, attention, memory and executive function, were enhanced by adjuvant pregnenolone or minocycline. However, whether these agents derived their treatment efficacy from their anti-inflammatory actions remains unclear. Future clinical trials with anti-inflammatory agents could examine the association between changes in inflammatory markers and symptom improvements to clarify this important issue.
Supplemental Material
supplementary_materials – Supplemental material for Adjunctive use of anti-inflammatory drugs for schizophrenia: A meta-analytic investigation of randomized controlled trials
Supplemental material, supplementary_materials for Adjunctive use of anti-inflammatory drugs for schizophrenia: A meta-analytic investigation of randomized controlled trials by Myeongju Cho, Tae Young Lee, Yoo Bin Kwak, Youngwoo Brian Yoon, Minah Kim and Jun Soo Kwon in Australian & New Zealand Journal of Psychiatry
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research was supported by the Brain Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Science, ICT and Future Planning (Grant Nos. 2017M3C7A1029610 and 2016R1E1A1A02921618).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
