Abstract
Neurocognitive deficits in schizophrenia have been consistently reported [1,2], with impairment relatively resistant to antipsychotic medication [3], apparent early in the illness [4] and remaining stable over the course of the illness [5]. Schizophrenia patients who demonstrate lower ratings on assessments of global functioning exhibit greater neurocognitive impairment [6]. They show impaired neurocognitive performance across multiple cognitive domains including memory, attention, executive function, motor skill and language [2,7]. Memory impairment, however, is one of the most replicable and reliable findings; with schizophrenia patients showing deficits on a range of memory tasks including short- and long-term memory, verbal and non-verbal memory, episodic, semantic and working memory [8–14]. Problems with attentional capacity and processing speed are also significant in schizophrenia, as measured by tasks such as the continuous performance test [15,16]. Extensive research also indicates deficient executive functioning; performance deficits are observable on tasks such as the Wisconsin Card-Sorting Test [17], Hayling Sentence Completion Test [18] and verbal fluency [19].
The Repeatable Battery for the Assessment of Neuropsychological Status (RBANS) is a brief, easily administrable and standardized tool, which can provide a general, overall evaluation of several cognitive domains [20,21]. The RBANS measures five domains: immediate and delayed memory, attention, visuospatial/constructional ability, and language. These domains combine to provide a total score of global cognitive ability. The RBANS has become increasingly useful in schizophrenia research, with a number of studies reporting cognitive deficits [6,22–26]. Further, the RBANS has been used as an early detection measure in psychotic adolescents [27] and as an evaluative tool of cognitive change after drug treatment in schizophrenia [28–30]. This tool demonstrates good sensitivity (Cronbach's α = 0.88), convergent validity (high correlations with Wechsler Adult Intelligence Scale-III and Wechsler Memory Scale-III) and test–retest reliability in a schizophrenia population [22,24,25,31].
Previous investigations into neurocognitive functioning in schizophrenia suggest that performance can be affected by demographic variables, that is, age, education and gender [26,32], and also clinical characteristics such as negative symptom severity [23]. The effects of gender are of particular significance because gender differences exist in the psychopathology of schizophrenia [33]. Epidemiological evidence indicates that men have an earlier illness onset, compared with women, of approximately 3–4 years [34,35]. Furthermore, only in women does the distribution peak for age at onset of schizophrenia show a second peak around 50 years of age, argued to correspond with menopause [36,37]. Compared to men, women demonstrate a less severe course of illness, showing milder symptoms, superior treatment response to antipsychotic medication and improved social, intellectual and occupational outcome [38–40].
In studies examining gender differences in cognition in large healthy populations, women performed better on language and memory tasks, while men performed better on visuospatial tasks [41,42]. In schizophrenia, studies have shown that male schizophrenia patients have lower performance than female patients on tests of verbal ability, executive function and attention; and female schizophrenia patients performed worse on visuospatial tasks [32,43,44]. In contrast, female schizophrenia patients performed worse than male patients on tests of verbal and spatial memory and visual processing [45]. One methodological problem with these previous investigations is the use of different tasks to examine the same cognition. Use of a standardized cognitive battery can assist with such cross-study comparisons. Gender differences have been found using the RBANS, where male schizophrenia patients outperformed female patients on the attention and visuospatial/constructional index, and female patients outperformed male patients on the delayed memory index [22,26]. Previous studies using the RBANS in schizophrenia, however, have additional methodological limitations in that they did not match the number of male and female participants across groups. Given the importance of gender in schizophrenia and the discrepant findings in neurocognition, further investigation is warranted. Based on the previous literature, we predict that gender differences in cognition will be exacerbated in schizophrenia, with female patients demonstrating enhanced verbal abilities and male patients, enhanced visual processing skills.
A further issue is the diagnostic specificity of cognitive impairment in schizophrenia. Are the performance deficits secondary to the illness and a result of psychopathological characteristics common to all mental illness, or are the deficits specific to the underlying aetiology of schizophrenia? This issue has been addressed in the current study by examining the performance of a bipolar disorder (BD) sample. Both BD and schizophrenia patients experience psychotic symptoms and as a result share several psychopathological characteristics [46]. Although distinctly separate disorders, both groups can experience long periods of time in psychiatric institutions and both are often prescribed with antipsychotic medication for extended periods of time. BD patients also show extensive neurocognitive deficits including memory, attention and executive function [47]. We have identified only one recent study to investigate gender and cognition in BD compared with controls: Barrett et al. found that a spatial working memory deficit was more apparent in male-only comparisons than in female-only comparisons [48]. Three studies have investigated cognitive functioning in both schizophrenia and BD using the RBANS. These studies showed that patients with schizophrenia performed worse on the RBANS relative to BD patients [23,24,49]. To our knowledge, no study has specifically investigated gender differences in schizophrenia and BD in terms of neurocognition.
Consequently, the aims of the current study were twofold: (i) to investigate deficits in neurocognition using the RBANS, comparing schizophrenia and BD to a healthy non-psychiatric control group; and (ii) to investigate the effects of gender on neurocognition in schizophrenia, BD and control participants specifically matched for gender. We expected the patient groups to show a different pattern of cognitive performance, with schizophrenia patients demonstrating the greatest deficits. We also hypothesized differential gender effects for the different cognitive domains; men would perform better on the visuospatial domain and women would perform better on the language domain. These effects would be particularly apparent for schizophrenia patients given the gender differences that exist in the psychopathology of schizophrenia.
Methods
Participants
Thirty-eight schizophrenia patients and 40 BD patients were recruited via community support groups and care units and were all outpatients (Table 1). Diagnosis was confirmed using the Structured Clinical Interview for DSM-IV (SCID) [50]. Current symptomatology was assessed using the Positive and Negative Syndrome Scale (PANSS) [51]. Only patients with no other comorbid Axis 1 diagnoses were included in the study. All patients were in a chronic phase of illness and were not experiencing an acute psychotic or mood episode at the time of testing. BD patients were screened for current mood episodes during recruitment and then a euthymic state was confirmed during the SCID.
Subject data (mean ± SD)
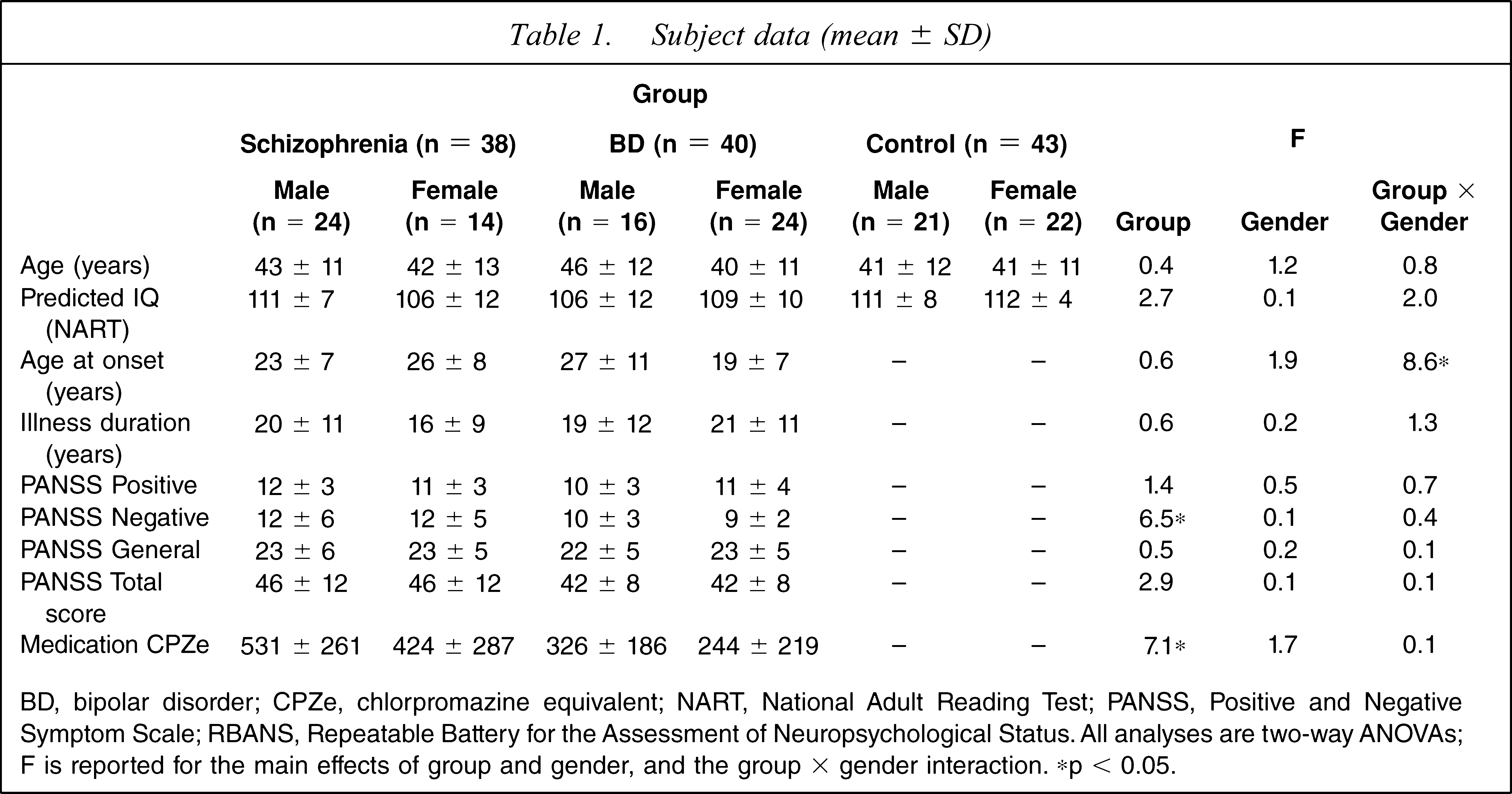
BD, bipolar disorder; CPZe, chlorpromazine equivalent; NART, National Adult Reading Test; PANSS, Positive and Negative Symptom Scale; RBANS, Repeatable Battery for the Assessment of Neuropsychological Status. All analyses are two-way ANOVAs; F is reported for the main effects of group and gender, and the group × gender interaction. ∗p < 0.05.
Forty-three healthy control participants were recruited via local advertisements. Control participants were excluded if they had any history of psychiatric disorder or a first-degree relative with either schizophrenia or BD. All participants met the following criteria: no history of neurological disorder or head trauma, no current substance abuse, English as first language, between the ages of 18 and 65 years and predicted IQ >80 as scored on the National Adult Reading Test [52]. The procedures described in this study were approved by the Mental Health Research and Ethics Committee (Melbourne Health, Vic., Australia) and the Health Sciences Human Ethics Subcommittee of the University of Melbourne; participants gave their written, informed consent prior to testing.
All participants were tested once, with most female patients tested during low levels of circulating oestrogen and progesterone to eliminate the confounding variable of cycling hormone levels. Therefore, women with a natural menstrual cycle were tested during the early follicular phase (days 1–8 of cycle; control, n = 10; schizophrenia, n = 7; BD, n = 9), women prescribed the oral contraceptive pill were tested during administration of the inactive pills (control, n = 2; schizophrenia, n = 3; BD, n = 2), and postmenopausal women (lack of period for > 2 years) were tested at any time; control, n = 4; schizophrenia, n = 2; BD, n = 4). A small number of women had very irregular cycles and thus we were unable to determine their menstrual cycle status (control, n = 6; schizophrenia, n = 2; BD, n = 9). No women were pregnant. Although most women were tested during low levels of circulating hormones, it is unclear whether the varying hormonal conditions (natural cycle vs contraceptive pill vs postmenopause) may be associated with endocrine effects on cognition. There was, however, a similar distribution of control, schizophrenia and BD women in each of the three hormonal conditions.
Medication
Schizophrenia patients were taking a higher dose of antipsychotic medication than the BD patients, as represented in equivalent mg of chlorpromazine (CPZe; Table 1). Of the schizophrenia patients, 24 were taking antipsychotic medication alone, 11 were taking a combination of antipsychotic and antidepressant and/or mood stabilizer, one was taking an antidepressant alone and two were medication free. Of the BD patients, nine were taking mood stabilizer medication alone, one was taking antipsychotic medication alone, one was taking antidepressant medication alone, 15 were taking a combination of antipsychotic and mood stabilizer and/or antidepressant, eight were taking a combination of antidepressant and mood stabilizer and six were medication free.
Procedure
The RBANS took approximately 30 minutes to complete, and each participant was individually tested using Form A. Participants were given the battery in accordance with the manual guidelines [20], with stimuli presented from a booklet. The RBANS results were scored by two independent raters using criteria in line with the manual guidelines. The RBANS consists of 12 subtests that were used to calculate five index scores; each subtest is described in the order of presentation. Index scores were age adjusted and standardized such that the normal mean was equal to 100 with an SD of 15, based on a normative sample [20]. The index scores were combined to form the Total score, a summary measure of RBANS performance.
Immediate memory/learning
The score range for this is 40–152 and the subtests are as follows: (i) List Learning, the immediate recall of a list of 10 words over four learning trials (1 point for each word of each trial recalled); and (ii) Story Memory, the recall of a story composed of 12 key elements over two trials (1 point for each item of each trial recalled).
Visuospatial/Constructional
The score range = 50–136 and the subtests are as follows: (i) Figure Copy, copying a 10-part geometric drawing (1 point for each part accurately copied and 1 point for correct placement of each part); and (ii) Line Orientation, the identification of two target lines out of an array of 13 lines spanning 180°, over 10 trials (1 point for each correctly matched line of each trial).
Language
Score range = 40–137 and the subtests are: (i) Picture Naming, providing the name of each item in 10 line-drawings (1 point for each correctly named picture); and (ii) Semantic Fluency, the generation of as many words as possible for a given semantic category (i.e. fruits and vegetables) within 60 s (1 point for each word generated).
Attention
The score range is 40–154 and the subtests are as follows: (i) Digit Span, the immediate repetition of a string of numbers that progressively increases in length from two to nine digits, that is, eight trials (2 points for each correctly repeated string or 1 point for a second attempt); and (ii) Coding, filling in as many boxes as possible within a 90 s time window, by matching a symbol with a number (1–9; the symbols are indicated in a key; 1 point for each box filled in with the correct number corresponding to the presented symbol).
Delayed Memory
Score range = 40–137 and the subtests are: (i) List Recall, the free recall of the words from the List Learning subtest (1 point for each word recalled); (ii) List Recognition, the yes/no recognition of the List Learning words from a total of 20 words (1 point for each word recognized); (iii) Story Recall, the free recall of the story from the Story Memory subtest (1 point for each item recalled); and (iv) Figure Recall, the free recall of the figure from the Figure Copy subtest (1 point for each part accurately copied and 1 point for correct placement of each part).
Statistical analysis
The statistical software package SPSS (version 15; SPSS, Chicago, IL, USA) was used for all data analysis. Demographic group differences were assessed via the Pearson's χ2 test and two-way between-groups ANOVA for group (controls, schizophrenia, BD) and gender (male, female). Two-way ANOVAs were conducted to compare groups and gender on the RBANS Total score, the five index scores (immediate memory/learning, visuospatial/constructional ability, language, attention, delayed memory) and 12 subtests. Significant main effects of group were followed up with Bonferroni post-hoc tests. Partial η2 was used to determine effect sizes (small = 0.01, medium = 0.06, large = 0.14).
Relationships between the RBANS Total score and clinical variables identified in Table 1 were investigated for the two patient groups using Pearson's r correlations with Bonferroni adjustment, to assess confounding factors. Further correlations between the index scores or subtests were not performed due to the problems associated with multiple testing, such as increased Type I error. Clinical characteristics included age at onset (i.e. first appearance of symptoms), illness duration, medication (CPZe), and positive, negative, general and total PANSS; correlations were considered significant at the corrected level of p < 0.007.
Results
Demographic and clinical variables
The schizophrenia, BD and control groups were matched for gender (Pearson's χ2(2,n = 121) = 4.3, p = 0.1), further, the male and female participants for each group were matched for age and predicted IQ (Table 1). Overall, the two patient groups were matched for age of illness onset, with no effect of group or gender, but there was a group × gender interaction indicating that BD female patients had an earlier age at onset compared to BD male patients (t(23) = 2.7, p = 0.01). When comparing the two patient groups, there was no difference in illness duration, positive, general or total PANSS scores, nor were there any differences between male and female patients on these variables. Schizophrenia patients scored significantly higher on the negative factor of the PANSS and had a higher CPZe compared to BD patients (main group effect, Table 1), but there were no differences between the genders. Thus, the patient groups showed similar levels of psychopathology and the genders were matched on all aspects except age at onset.
RBANS analyses
Group differences
The RBANS Total score differed significantly between the three groups (Figure 1; Table 2); Bonferroni post-hoc tests showed that schizophrenia patients performed worse than controls (p < 0.001) and BD patients (p < 0.01). Further, there was a significant cognitive deficit in BD patients compared to controls (p = 0.03).
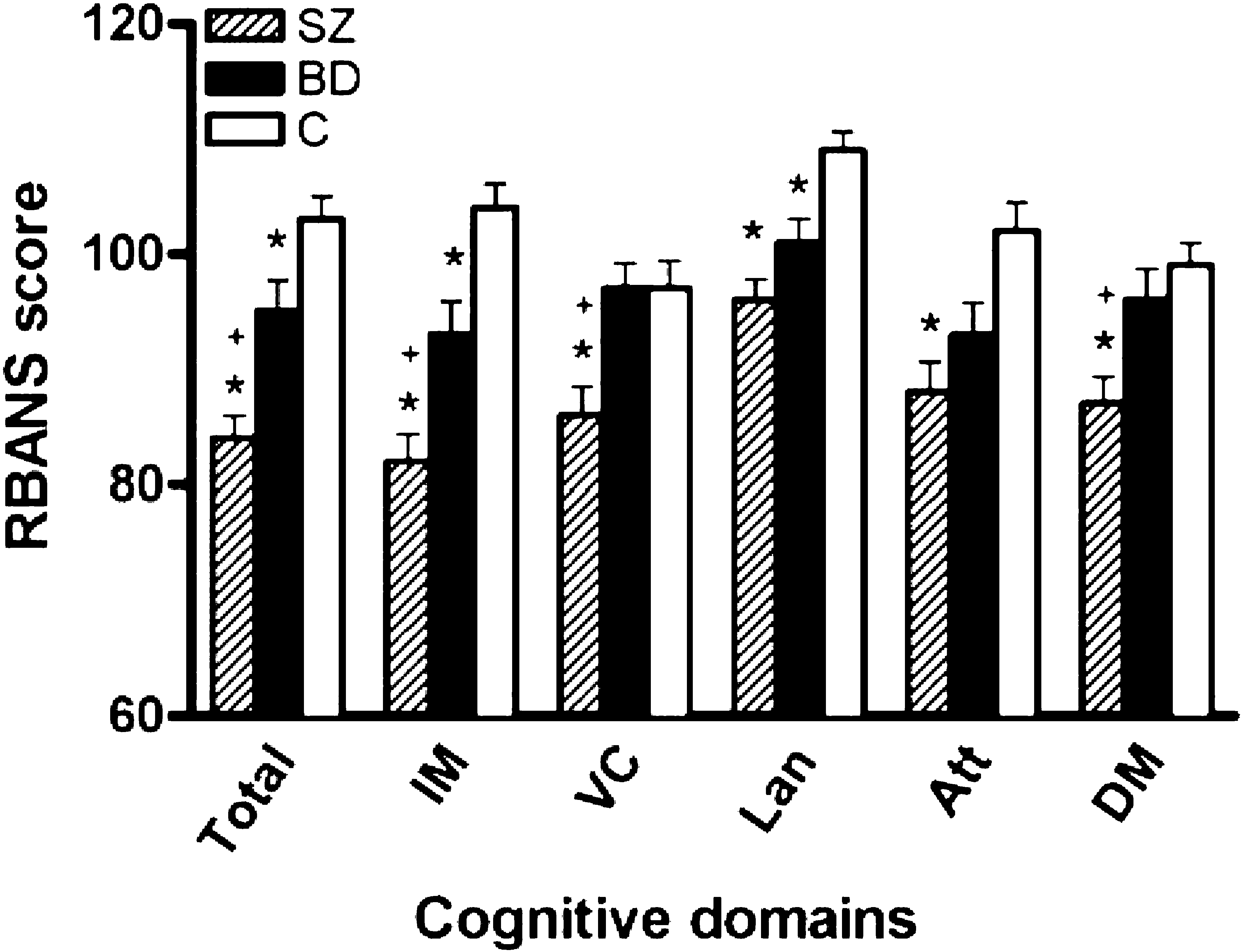
Mean (SEM) Repeatable Battery for the Assessment of Neuropsychological Status (RBANS) scores for schizophrenia patients (SZ, n = 38), bipolar disorder patients (BD; n = 40) and non-psychiatric controls (C; n = 43), for the overall Total score and the five index scores: immediate memory/learning/(IM), visuospatial/constructional ability (VC), language (Lan), attention (Att) and delayed memory (DM). The standard score is 100. ∗p < 0.05 compared to controls, + p < 0.05 compared to BD.
RBANS Total and index scores vs gender (mean ± SD)

BD, bipolar disorder; RBANS, Repeatable Battery for the Assessment of Neuropsychological Status. All analyses are two-way ANOVAs; F is reported for the main effects of group and gender, and the group × gender interaction. # p ≤ 0.07; ∗∗p < 0.01.
There was a main effect of group for each of the five index scores (Figure 1; Table 2). Overall, control participants performed at the highest level, while schizophrenia patients performed at the worst. Post-hoc tests showed that controls significantly outperformed schizophrenia patients on each domain (all p < 0.01). The effect sizes were medium–large for all domains when comparing schizophrenia patients and controls: immediate memory/learning = 0.42, visuospatial ability = 0.12, language = 0.27, attention = 0.15, delayed memory = 0.18. BD patients showed intermediate performance, whereby they performed similar to schizophrenia patients on two domains (language, attention) and significantly better on three domains (immediate memory/learning, visuospatial ability, delayed memory; all p < 0.05). Post-hoc tests showed that controls outperformed BD patients on two domains (immediate memory/ learning, language; both p < 0.01), but these groups performed similarly on the visuospatial and delayed memory domains. There was a trend for the controls to perform better than BD patients on the attention domain (p = 0.05). For the three domains showing a difference between BD and control participants, the effect sizes were medium: immediate memory/learning = 0.11, language = 0.12, attention = 0.08.
The 12 subtest scores were consistent with the results of the index scores. There was a significant group difference in nine of the 12 sub-tests (Table 3). Two subtests that did not show a group difference, that is, picture naming and list recognition, were at ceiling performance levels, which may have caused the lack of a group effect. For the picture naming subtest, 96% of participants (100% controls, 92% schizophrenia, 95% BD) performed at ceiling (i.e. scoring 9–10). For the list recognition subtest, 89% of participants (95% controls, 82% schizophrenia, 88% BD) performed at ceiling (i.e. scoring 19–20). The other subtest that failed to show a group difference was the digit span subtest of the attention domain. Of the nine subtests showing a group difference, post-hoc tests indicated that controls outperformed schizophrenia patients in all nine subtests (all p < 0.05) and outperformed BD patients in five out of nine subtests (all p ≤ 0.05; list learning, Story Memory, semantic fluency, coding, list recall). Schizophrenia and BD patients performed similarly on five out of nine subtests (Figure Copy, Semantic Fluency, Coding, Story Recall, Figure Recall), while schizophrenia patients performed worse on four out of nine subtests (all p < 0.05; List Learning, Story Memory, Line Orientation, List Recall; Table 3).
RBANS subtest scores vs gender (mean × SD)
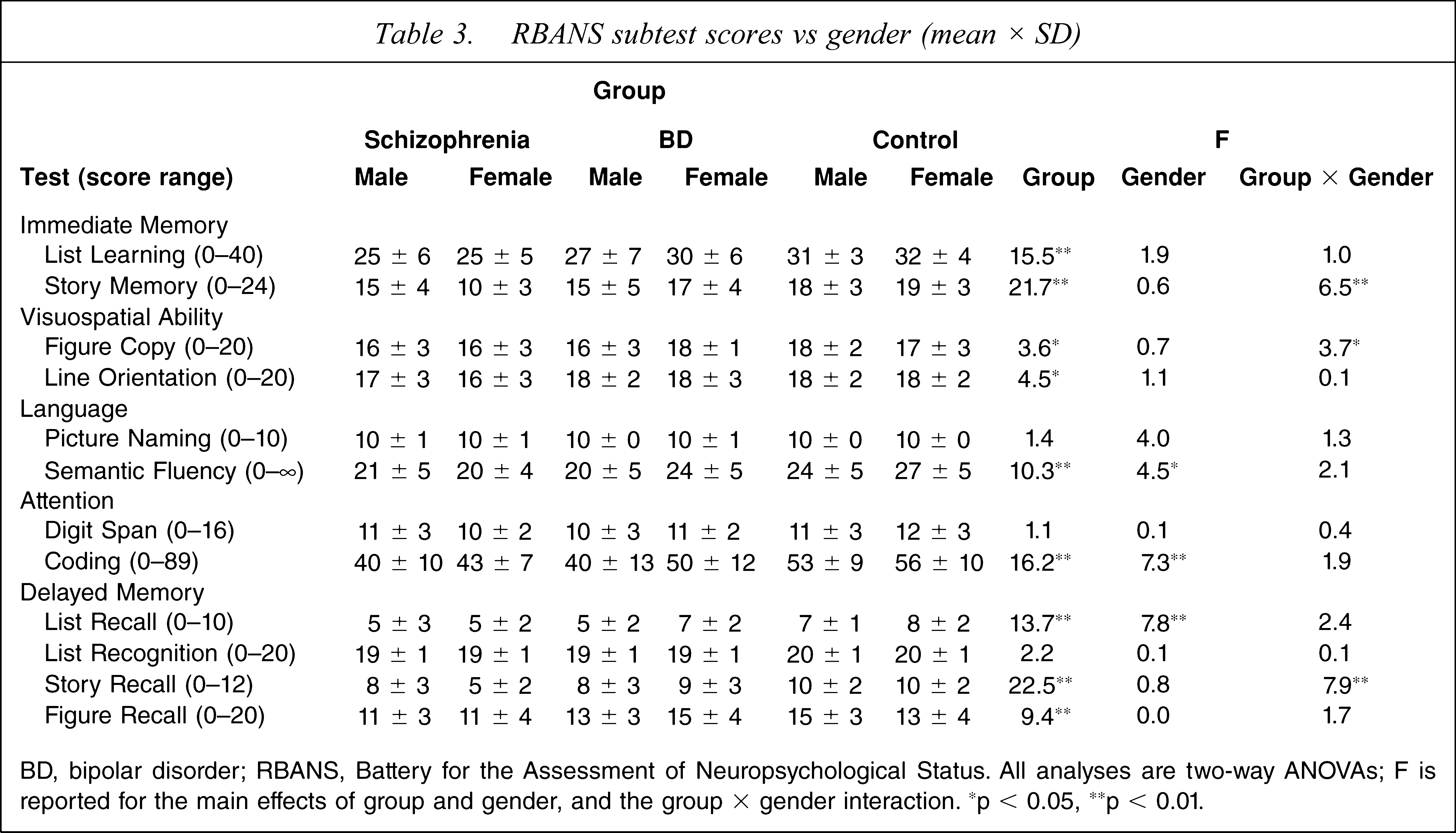
BD, bipolar disorder; RBANS, Battery for the Assessment of Neuropsychological Status. All analyses are two-way ANOVAs; F is reported for the main effects of group and gender, and the group × gender interaction. ∗p < 0.05, ∗∗p < 0.01.
Gender differences
There was no significant gender difference for the RBANS Total score, but there was a trend for a group × gender interaction (F(2,115) = 2.8, p = 0.07; Table 2). The BD patients’ performance tended to be better in female patients compared to male (F(1,38)= 3.9, p = 0.06), while male and female control or schizophrenia participants showed similar performance. Similar to the Total score, there was no main effect of gender for any of the five index scores (Table 2). There was a trend for a group × gender interaction for the immediate memory/learning domain (F(2,115) = 2.9, p = 0.06), reflecting the tendency for reduced performance in female compared to male schizophrenia patients (F(1,36)= 3.4, p = 0.07). There were no other significant interactions.
Three of the 12 RBANS subtests showed a significant main effect of gender: semantic fluency, coding and list recall (Table 3). In these tasks, female participants outperformed male participants.
There was a group × gender interaction for three subtests: Story Memory, Figure Copy and Story Recall (Table 3). For these three subtests, male and female controls had similar performance, but female schizophrenia patients had significantly reduced performance for Story Memory and Story Recall (p < 0.01), and female BD patients had enhanced performance for Figure Copy (p = 0.01), compared to male patients in the same group.
RBANS performance correlations with clinical characteristics
Given the lack of a main effect of gender for the Total RBANS score, correlations were conducted only for the patient groups. For schizophrenia patients (n = 38), there was a significant correlation with Total RBANS and general PANSS score (r = −0.5, p = 0.003). (Entering the PANSS general score as a covariate into our multivariate statistics with only the two patient groups as between-group comparisons, did not change the main effects or interactions reported in the main text.) In schizophrenia patients there were no significant correlations with Total RBANS and other PANSS measures or any other clinical characteristics. For BD patients (n = 40), there were no significant correlations with RBANS Total and any clinical characteristics.
Discussion
The present study used the RBANS to assess neurocognitive performance in schizophrenia, compared to both BD patients and a non-psychiatric control group. Schizophrenia patients had marked cognitive deficits in their Total RBANS score compared to both other groups. The BD patients performed at an intermediate level, between schizophrenia patients and controls. This study also investigated the effects of gender on cognitive performance in these groups, in which the groups were specifically matched for gender. On the overall measure of cognition, there were no major gender differences across or within groups; there were some specific gender effects on individual cognitive tasks.
The present findings support the hypothesis that schizophrenia patients have the greatest cognitive deficit. Compared to the control group, schizophrenia patients had deficits across all five cognitive domains (immediate and delayed memory, language, attention, and visuospatial/ constructional ability), which was further reflected on most of the subtests. This is consistent with meta-analyses indicating broad cognitive impairment in schizophrenia [2]. Although the results indicated relatively generalized impairment for the schizophrenia patients, it appeared that they performed worst on measures of immediate memory/ learning and had the best performance on measures of language and visuospatial ability, which is consistent with previous work [22,23,26].
The RBANS Total and index scores are standardized such that the average level of performance is 100 ± 15. The current results indicate that performance of the schizophrenia patients was approximately 1 SD below the normal mean, similar to the Australian sample assessed by Loughland et al. [6]. This highlights that the current patient sample had levels of cognitive functioning within the range of other larger groups of schizophrenia patients and can thus be considered a relatively cognitively representative patient group.
Schizophrenia patients showed an overall cognitive deficit compared to BD patients, as indicated by a significant difference in the Total RBANS score. Specifically, compared to BD patients, schizophrenia patients showed deficits in immediate and delayed memory and visuospatial ability, but performed similarly in terms of language and attention. Three studies have investigated cognitive functioning in both schizophrenia and BD using the RBANS [23,24,49]. Two of these studies found similar cognitive performance in schizophrenia and BD patients on all measures except the immediate memory/learning domain [24,49], but in a later study using a larger sample, these authors found significant differences across all cognitive domains [23]. Further, a review of 31 studies examining cognitive performance in patients with schizophrenia and BD, found that patients with BD performed better than patients with schizophrenia [53].
We found a significant overall cognitive deficit in BD patients compared to control participants, which remained significant for two cognitive domains (immediate memory/ learning and language). This suggests that schizophrenia and BD patient groups have qualitatively similar cognitive deficits on some domains, but with differences in severity. This is consistent with Dickerson et al., who found that BD patients had cognitive deficits in most domains of the RBANS compared to controls, but these were milder than those exhibited by schizophrenia patients [23]. In the current study, BD patients did not demonstrate a deficit in visuospatial ability or delayed memory compared to controls, while schizophrenia patients did. Therefore, cognitive deficits are confirmed to be prominent in schizophrenia but, given the substantial overlap of deficits in BD, one cannot conclude that these are a unique feature of the illness.
Previous investigations have implicated gender as a potential mediating factor in cognitive performance for schizophrenia patients, but with inconsistent findings [33]. We expected, independent of group, that men would perform better on the visuospatial domain and women would perform better on the language domain. Overall, there was no significant gender difference on any of the cognitive domains measured. There was a gender effect, however, for the semantic fluency subtest of the language index, indicating a female superiority. Problematically, the performance of the other subtest of the language index, the picture naming task, was at ceiling level, thus we suggest that its influence within the language domain should be ignored. Therefore, when we consider only the fluency data, there was an overall gender effect for language. In the current study, coding (within the attention domain) and list recall (within the delayed memory domain) showed better performance by women overall. One study using the RBANS reported greater performance in healthy, mature women on the language and delayed memory domains [41].
Of particular interest for the present study was whether the three participant groups would show differential gender effects. Data analysis indicated no significant interactions between gender and group for any of the RBANS domains, although there was a trend on the RBANS Total score for women with BD to perform better than men, and a trend on the immediate memory/learning index for male schizophrenia patients to perform better than female patients. Further, three subtests had a differential gender effect across the groups: Story Memory, Figure Copy and Story Recall. For these tasks, male and female controls performed similarly, female schizophrenia patients showed deficits in Story Memory and Story Recall, and male BD patients showed deficits in Figure Copy. Interestingly, we recently reported that female BD patients have superior performance on a sensorimotor gating or information processing paradigm [54]. Such data may indeed suggest that the psychopathology of male and female BD patients is in fact different, with male patients having greater cognitive disruption.
The lack of a significant gender difference on the index scores in schizophrenia is in contrast to other studies using the RBANS, in which male schizophrenia patients outperformed female patients on the attention and visuospatial index; and female patients outperformed male patients on the immediate and delayed memory index [6,22,26]. This inconsistency may be due to the fact that the Gold et al. and Wilk et al. studies included a sample that was predominantly male, and only made within-patient-group gender comparisons [22,26]. They used t-tests to compare men and women for each index score. The present study examined gender differences in schizophrenia and BD compared to a non-psychiatric control group, thus we used gender as a between-subjects factor in a two-way ANOVA. If we compare only the present schizophrenia group, the means presented in Table 2 show that the data do not reflect the trends previously reported. Overall, there seems to be a preconceived assumption that clear gender differences in cognition exist, with little empirical data to support these statements. In the present study, small gender differences were found between the clinical groups, but only on specific subtests within a particular cognitive domain, and not globally. Further investigations examining whether these subtle cognitive differences affect the functional outcome of patients will be important.
One limitation of the present study may be the sample size. Although the present sample produced significant group differences, it may not have been large enough to identify gender differences in cognition. For example, Beatty et al. included 631 participants and the significant gender effects were only a difference of 3–6 points on the index score [41]. Larger samples would also allow for a more complete investigation of the relationship between cognition and other clinical variables, especially considering that the current study recruited patients with low levels of psychopathology. In the present study we demonstrated a relationship between general PANSS symptoms and cognition in schizophrenia only. The patient groups were not matched on the negative PANSS factor or for CPZe, but these variables were not significantly correlated with RBANS performance.
There is evidence that drug treatments can improve cognitive functioning [29,30]. In the present study, schizophrenia patients were prescribed a higher dose of antipsychotic medication than the BD patients. Of the schizophrenia patients, most were taking antipsychotic medication alone, whereas the BD patients were mostly taking a combination of antipsychotic and mood stabilizer and/or antidepressant medications. Given these differences in medication dose and type, it is possible that medication may have influenced the group differences that were observed. Because most patients were on multiple and various medications, it is very difficult to extract the effect of medication on cognition. Future research in drug-naïve patients should investigate the role of medication on cognitive deficits.
Importantly, in the present study, the groups were carefully matched and the results were not confounded by differences in demographic or clinical variables. The RBANS is a brief cognitive test and not a comprehensive neuropsychological battery. Nevertheless, it is a good screening test that correlates highly with more extensive test batteries. In the present study the RBANS was sensitive enough to differentiate performance between all three groups and between patient groups. A review of the schizophrenia literature shows that neurocognitive function is clearly linked to functional outcome and is a better indicator of outcome than negative or positive symptom severity [55,56]. This has direct implications for the treatment of schizophrenia, whereby understanding of which neurocognitive deficits compromise functional outcome may determine which areas of neurocognition are best targeted for remediation [55,57].
Footnotes
Acknowledgements
Funding for this study was provided by the J & P Clemenger Trust; the National Health and Medical Research Council of Australia (Peter Doherty Fellowship, ID 435690 to A.G.); and the Menzies Foundation (Research Scholarship to N.J.). This research was also supported by Operational Infrastructure Support (OIS) from the Victorian State Government. We would like to acknowledge the assistance of Ms Alison O’Regan with some of the clinical interviewing and Professor David Castle for assistance in patient recruitment.
