Abstract
Rapid advances in genetic research over the past decade have led to identification of a substantial number of candidate genes associated with susceptibility to common complex disorders of public health significance including coronary artery disease, breast cancer, type 2 diabetes and major depression [1].
Identification of groups with an increased genetic risk for such disorders presents an opportunity to target interventions that modify specific environmental risk factors at a pre-symptomatic stage, with the potential to signifi-cantly reduce burden of disease. Clinical utility, acceptability and potential health impact of pre-symptomatic genetic testing as a preventive intervention is currently the subject of contentious debate, but few data exist to guide policy and ethical decision-making [2]. Given current concerns about the rapid expansion of commercial predictive genetic tests for multifactorial diseases marketed direct to consumer (DTC), research about how the public might use genetic risk information to change health behaviour is needed.
Around 50 predictive genetic tests purported to indicate risk for a wide range of common multifactorial diseases are currently marketed DTC via the Internet. Although some of these tests are available in clinical practice, many are based on a small number of unrepli-cated studies with uncertain clinical validity [3].
In psychiatry, a psychiatric gene–disease association that has been widely replicated by a large number of studies thus far is an interaction between a functional polymorphism in the promoter region (5-HTTLPR) of the serotonin transporter gene (SCL6A4) and exposure to stressful life events in increasing the likelihood of depression in non-clinical populations of adults [4–7], adolescents [8] and children [9]. The evidence suggests that homozygosity for the short allele (s/s) of the serotonin transporter gene-promoter region is associated with depression on exposure to multiple stressful life events [4–9]. The s/s variant is thought to play a role in mediating response to stress, with s/s individuals demonstrating hyperreactivity to stressors and/or deficient problem-solving coping, which may convey increased risk to future depression [10].
Since the completion of the present study a recent meta-analysis found no evidence for a 5-HTTLPR by environment interaction in association with an increased risk of depression [11]. However, 5-HTTLPR genotyping remains a good hypothetical example by which to evaluate attitudes to preventive interventions based on genetic testing for a risk of a common complex disorder.
Effective mental health intervention, based on genetic susceptibility, will depend upon community attitudes towards behavioural change to reduce risk, and understanding of uncertain penetrance, relationship between genes and environment, and potential to modify environmental risk factors.
Recent debate highlights popular attitudes about the right to know one's own genetic information [12], and that predictive genetic tests, especially those available DTC, offer autonomy and empowerment for the individual [13]. Critics question whether it is responsible to offer genetic tests if their predictive value is low, and if there is no associated treatment available [14]. Implications cited include a potential for a low-risk result to provide false reassurance, or a high-risk result to cause fatalistic thinking based on a belief that a genetic component for a disorder makes the disorder less preventable [15,16]. Both circumstances could demotivate an individual to engage in mental health interventions [14].
Previous studies evaluating potential to change health behaviours in association with genetic risk information have focused on breast cancer [17], heart disease [15], smoking [18], familial hypercholesterolemia [15,19], and Alzheimer's disease [20], but not psychiatric disease. It is generally thought that intention to change behaviour is a poor indicator of uptake of an intervention [21]. Fatalistic attitudes towards genetic risk for common complex disorders have been more commonly observed in general populations rather than among individuals informed of a genetic predisposition [19]. Empirical evidence suggests that provision of genetic risk information to the individual may prompt uptake of new health behaviours [17–19].
Only anecdotal evidence is available about how genetic risk information involving psychiatric disorders might be interpreted and used by patients [22]. Because serotonin transporter genotype-major depression associations are replicated and 5-HTTLPR genotyping has been commercially available DTC in the USA, we use this genetic test as an example to qualitatively evaluate among the general population, preparedness to modify risk at a pre-symptomatic stage through preventative behaviour based on a hypothetical genetic susceptibility to major depression.
Methods
The present results were obtained as part of a broader qualitative study, which also explored interest in genetic testing [23] and perceived impact of media portrayal of genetics and mental illness. The results regarding the latter topics will be reported separately.
Because this is a relatively unexplored area of enquiry, a qualitative methodology was used. There has been an upsurge in interest in studies that examine attitudes, beliefs and experiences of people in connection to health-care issues, and qualitative methodology has been increasingly recognised in evidence-based clinical research [24].
Participants
A market research company was engaged to randomly recruit 10 participants each to four or more focus groups from their database of 10 000 members of the public, ensuring an even mix of gender, age and sociodemo-graphic backgrounds. Eligibility criteria included being ≥18 years, fluent in English, resident in the Sydney metropolitan area and not having participated in any research in the previous 6 months. Ethical approval for the study was provided by the relevant Institutional Review Board (Human Research Ethics Committee, University of New South Wales, Australia).
Focus group interviews
Participants completed a short questionnaire that included items about age, sex, and highest education level. Participants were asked to indicate whether they had prior knowledge or experience of the subject of mental illness. They were not obliged to disclose personal or family history of mental illness.
The focus groups were conducted in accordance with widely accepted standards of focus group methodology [25]. In particular, at the beginning of each focus group the participants were assured of confidentiality, the discussions were videotaped and the facilitator periodically summarised discussions to check correct understanding.
The focus groups were facilitated by the first author (a health research scientist and medical journalist) and observed by the second author (a research psychologist). The observer also took written notes of the main themes discussed. An interview guide was developed on the basis of a review of the relevant literature with input from all authors. The analysis of the focus group interviews involved the use of verbatim transcripts.
Genotyping for the 5-HTTLPR polymorphism was framed to participants as a ‘genetic test to determine whether an individual has an increased risk for developing depression in the event of experiencing significant adversity’. A genetic test result that was positive for the short-short (s/s) variant of the 5-HTTLPR polymorphism was framed to participants as indicating an ‘increased risk for depression’.
Analysis
The conceptual approaches of Patton, and Miles and Huberman were used to guide the analysis [26,27]. A detailed coding scheme was developed and transcripts were coded by the first author. This involved coding each unit of meaning and comparing these with recurring patterns and discrete categories. A conceptually clustered coding tree was prepared to facilitate analysis both within and across themes.
Ten per cent of the transcripts were recoded by the second author, to identify any discrepancies in interpretation of codes and refine the coding scheme. Discrepancies were resolved by discussion and consensus. Coded transcripts were subsequently analysed for emergent themes arising from the transcripts [26,27]. Data analyses were iterative and the results from each focus group were used to suggest additional lines of questioning in subsequent focus groups to ensure that divergent points of view could be expressed.
The qualitative data analysis computer program QSR N6 (NUD∗IST6; QSR International, Melbourne, Vic, Australia) was used to facilitate comparisons between affected and unaffected participants as well as other aspects of the analysis.
Corresponding to the qualitative nature of the data, focus group discussions were designed to identify the range of beliefs rather than extent to which participants held particular beliefs. Immediately prior to discussing attitudes towards mental health interventions based on a hypothetical personal genetic risk for depression, participants had discussed perceived implications of predictive genetic testing and beliefs about the causes of mental illness.
Results
Participation and demographics
Thirty-six (18 female, 18 male) of 40 invitees were recruited to a total of four focus groups held in four locations across Sydney. Recruitment was discontinued after the fourth focus group, when informational redundancy was achieved, in accordance with widely accepted standards of qualitative methodology [27].
During focus group discussions, 14 participants spontaneously disclosed a personal or family history of depression, bipolar disorder or schizophrenia. Hereafter, participants reporting a personal or family history of depression are referred to as ‘affected’, while those who did not are referred to as ‘unaffected’. Citations referring to affected participants are denoted with [A] and those referring to unaffected participants with [U]. Demographic characteristics of unaffected and affected participants are shown in Table 1. The mean age was 41 years (range=20–65 years).
Subject characteristics
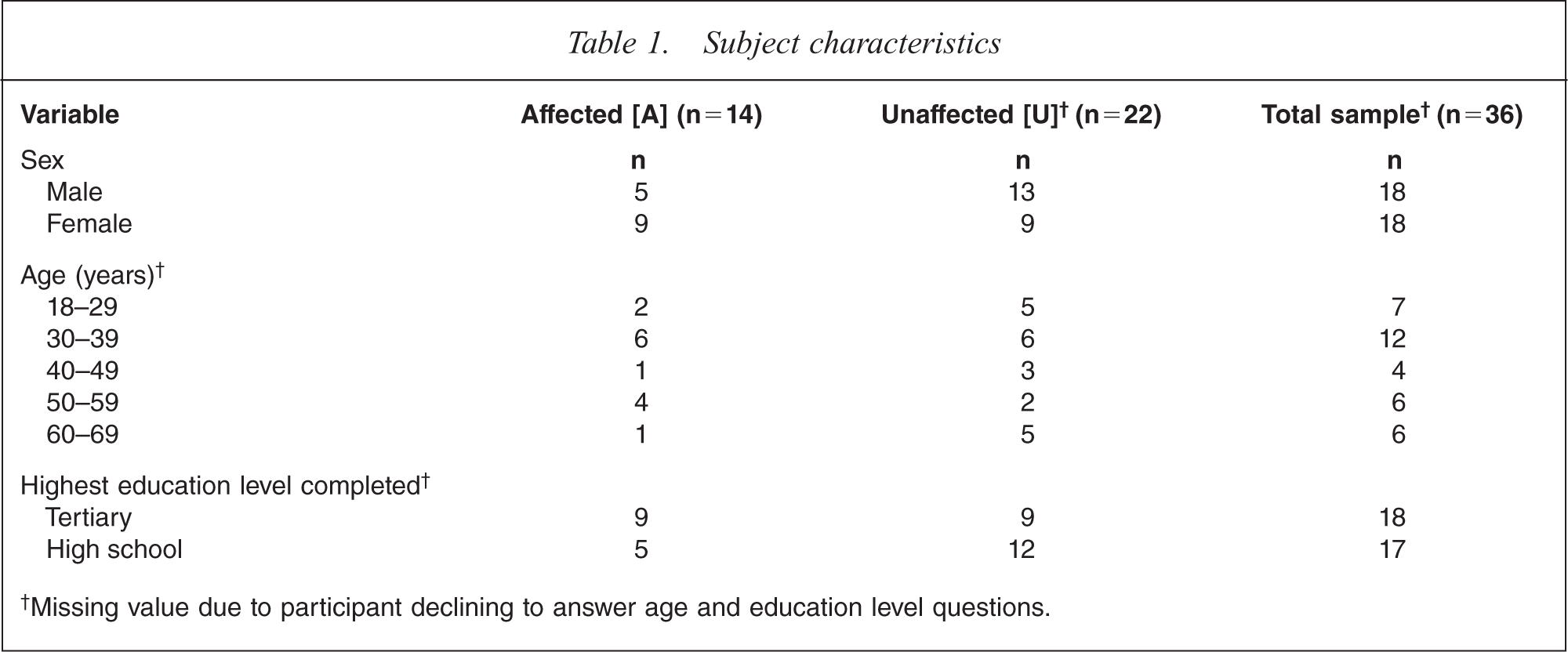
†Missing value due to participant declining to answer age and education level questions.
Anticipated health behaviours on receiving s/s genotype result
The majority of participants (11 affected, 20 unaffected) thought that being identified with an increased genetic risk of major depression would have a personal impact. Participants anticipated that they would increase vigilance for symptoms, seek information about depression, make lifestyle changes, undertake preventative strategies or do nothing.
Increased vigilance
The majority of affected participants and approximately half of the unaffected participants agreed that receiving a genetic test result indicating an increased risk for depression would encourage them to be vigilant for signs and symptoms of the disorder. Several affected participants thought vigilance would make them more likely to act on warning signs for depression and seek medical help as appropriate: ‘So if the symptoms and signs are showing up… you're aware so you're more likely to notice them.’ [A].
One participant observed that public education about the familial aspect of depression would be an important intervention to enable family members to be vigilant for symptoms in each other [U]; while another remarked that this strategy could be life-saving [U].
Prompt information seeking
Many participants said that an increased risk result would prompt them to seek information about depression, its symptoms and the meaning of being at increased genetic risk. One said: ‘I'd want to get a better educated person … just understand what the implications may or may not be ….’ [U].
Many participants showed trust in being advised by their doctors: ‘But if I did have that sort of thing I would go and see the doctor and do something because I wouldn't like to be caught out.’ [U].
One unaffected participant said she would ‘go down the natural path’ rather than see a general practitioner (GP) [U]. Another pointed out that people with an increased genetic risk should also be made aware of treatment options for depression and be advised on how to access medical services [U].
One participant, despite suggesting he would seek further information if he received an increased risk result, had a fatalistic view that could negatively impact on effectiveness of genetic counselling and behavioural intervention: ‘It's a done deal isn't it? You've got your DNA, you've got your genetics and you're in no position to alter them’ [U].
Prompt lifestyle changes
Participants who said they would make lifestyle changes if genotyping identified an increased risk for depression considered the potential to modify environmental risk factors including stress, diet, exercise and drug and alcohol intake. Several participants were in favour of minimising stress as an intervention: ‘You'd have to try and get as many stresses out of your life as possible…if you've got a stressful job, get rid of the job’ [U].
Other participants, while agreeing that drugs and alcohol intake were modifiable risk factors, were cautious about whether stress could be modified or avoided: ‘… you can cut down … the drugs and alcohol and stress you can try but you're not going to erase that from your lifestyle’ [U]. ‘Yeah, … marijuana and drugs and alcohol…definitely something to be avoided if you've got a disposition but you can't avoid stress throughout life, you just can't’ [A].
One participant said a genetic test result indicating an increased risk for depression would encourage him to maintain a healthy lifestyle [U]; while another remarked that she would adjust her diet and take more exercise as well as ‘seek some sort of help so as you can be steered in the right direction’ [A]. Two participants observed that individual differences in response to stress would impact on attempts to implement preventive strategies [A] [U].
Prompt preventative behaviour
One participant, who disclosed a history of depression, commented that predictive genetic testing, had it been available to her prior to her diagnosis, would have enabled her to learn coping strategies in advance so that her depression ‘could possibly have been minimalised or prevented’ [A]: ‘I would have liked to have known [in advance] because the things I've learnt how to cope with it over time like panic attacks…how to breathe prop-erly…I think maybe I could have implemented some of those tools earlier. It might have stopped me from getting really sick when I did’ [A].
Another participant said that if she received an increased risk result she would start a course of antide-pressants as a preventive strategy [A]. Two participants agreed that preventative medication could be used as a preventative measure while observing that there could be potential for harm [U] [U]; while two were against such a strategy [A] [A].
Doing nothing
Two of the four unaffected participants who said that they would do nothing if they received a predictive genetic test result that showed an increased risk for depression expressed the views: ‘…why treat something if you don't have it?’ and ‘… why educate yourself on something and worry yourself when it's probably not going to happen.’
Causal attributions for mental illness
The study found support for a genetic model for major depression with genetic factors viewed as predisposing rather than causal. Some participants perceived depression as less severe, less enduring and more likely to be attributed to stress rather than genetic factors than other psychiatric disorders including bipolar disorder and schizophrenia. Two participants observed that individual differences in response to stress would impact on attempts at preventive strategies.
Both affected and unaffected participants suggested that possible environmental factors that could trigger a mental illness were ‘alcohol, drugs, stress, chemical imbalance, poverty, general trauma, emotional disturbance, relationship breakdown, family environment, isolation, trauma in childhood, social environment, disadvantage’ and ‘arguments all the time’.
Discussion
The present study supports previous findings that positive attitudes towards a range of mental health intervention strategies at a pre-symptomatic stage exist [28]. These include interest in information and genetic counselling from GPs about the implications of having an s/s genotype, about depression and its risk factors and symptoms, and about future options for treatment and management. There was minor support for preventive medication among affected individuals as a pre-symptomatic intervention.
Although some participants were ambivalent about whether stress could be modified, positive attitudes were reported towards willingness to engage in lifestyle interventions such as reducing stress, drugs and alcohol intake and increasing exercise. The results suggest that mental health interventions that facilitate learning of effective coping skills are likely to be well-received as preventive strategies for target groups at a pre-symptomatic stage.
A number of findings have the potential to moderate uptake of future preventative mental health strategies among individuals identified as having the s/s variant of the 5-HTT promoter polymorphism. These include fatalistic attitudes that one's DNA is immutable, thus rendering environmental modification useless, perceptions of pointlessness of interventions in the absence of symptoms, and mixed or confused views on causal attributions for major depression.
Finding community endorsement of a contribution of both genetic and environmental factors in the development of mental illness and perceptions that genetic predispositions can be modified by adjusting environmental risk factors supports previous studies [29–31]. These endorsements suggest that target groups might be receptive to preventive programmes that involve predictive genetic testing associated with preventive cognitive and behavioural interventions that modify environmental risk factors. This is especially true in the light of greater endorsement of environmental risk factors as a cause for major depression than other psychiatric disorders.
Because provision of information about individual genetic risk alone may not be sufficient to change health-related behaviour [12,13,32], it will be necessary to evaluate the synergistic effects of individual genotype with personal and family history of psychiatric disorders, and lifestyle and environmental factors that regulate gene expression [5,22].
Ethical issues surrounding the use of predictive genetic testing in psychiatry, such as risk of discrimination and loss of privacy, require further investigation. Effective mental health interventions and appropriate genetic counselling should be established before 5-HTTLPR genotyping is offered in clinical practice.
Limitations of the study should be mentioned. Some participants may have interpreted the term ‘significant adversity’, or stressful life events, to mean everyday life stress, which could have affected anticipated health behaviour based on perception of modifiable nature of risk factors. Intention to change health behaviours in response to genetic risk information shown in the present study may not reflect actual change. Although every effort was made to include all participants throughout the focus group discussion, there may be a bias towards the views of a dominant minority. Reporting of a personal or family history of mental illness was voluntary, which may have resulted in the affected group being represented only by those willing to disclose such information.
This qualitative study has identified only the range of attitudes towards anticipated health behaviours based on genetic risk information, and not the extent to which they are held. These qualitative findings now require quantitative replication using a survey design in large representative non-clinical general population samples before recommendations about mental health interventions based on genetic risk can be made on a broader scale.
Footnotes
Acknowledgements
We thank Dr Nadine Kasparian for training in the use of QSR N6 software, Dr Catriona Bonfiglioli for advice on methodology, and study participants who gave their time. This study was supported by a 2006 Australian National Health and Medical Research Council (NHMRC) Public Health PhD scholarship (455414), an NHMRC Career Development Award (350989) and 2008 NHMRC Program Grant (510135).
