Abstract
U.S. war planners knew lewisite was not suited for battlefield use, yet vast quantities of the agent were produced, saddling the government with a large, ongoing cleanup.
For years now, the Army Corps of Engineers has been digging up and removing contaminated soil and buried World War I-era poison gas munitions in Spring Valley, a well-to-do Washington, D.C. neighborhood. [See Jonathan Tucker's “Chemical Weapons: Buried in the Backyard,” September/October 2001 Bulletin.]
This massive $91 million cleanup project continues, and one of the thorns plaguing the effort is the residue of what is believed to be the most toxic agent of the era, an arsenic-based compound called lewisite–the “dew of death.” To date, 140 of 1,600 residences and businesses near Washington's American University, where chemical weapons research was conducted, have been found to have arsenic levels higher than 20 parts per million, requiring the removal and replacement of the upper two feet of soil from their properties. On September 3, 2003, the Corps announced that a sealed vial containing about a tablespoon of lewisite had just been excavated. Small as this find was, it heightened residents' fears because it was discovered in what was believed to be a “low-priority” area. Residents wonder how much more lewisite is still buried in their yards.
Julius Aloysius Nieuwland.

It is ironic, given the enormous commitment of resources devoted to the manufacture of lewisite in this country and others–and the equally enormous price tag for cleanup–that the compound's history is riddled with questions about why it was produced in the first place. Its usefulness as a weapon of mass destruction was rightfully questioned from the beginning.
Early days
Another irony of lewisite's history is its initial discovery in 1903 by a priest pursuing doctoral work at Catholic University of America, in Washington, D.C. Julius Aloysius Nieuwland was studying the reactions of acetylene when he combined it with arsenic trichloride in the presence of aluminum chloride, causing a toxic compound to form (dichloro-2-chlorovinyl arsine). Nieuwland described the reaction in his thesis and then went on to an illustrious career at the University of Notre Dame.
Nieuwland's discovery was ignored until 1917, when a Chemical Warfare Service research unit was established at Catholic University. The unit's director, Winford Lee Lewis, whose assignment was to develop an arsenic-based poison gas to counter the German success with mustard gas, was drawn to Nieuwland's work. Lewis's unit purified Nieuwland's compound and sent it for testing to the Chemical Warfare Service's main research campus at American University.
Animal and human testing at American convinced chemical weapons researchers that lewisite was the Allied answer to German successes in the development of more and more toxic agents. With lewisite, either inhalation or skin contact caused death–and less than a teaspoon was thought sufficient to kill a man.
Eventually, the equivalent of $60 million in today's dollars was spent to build a secret plant in Willoughby, Ohio, where lewisite could be manufactured by the ton. By the time the war ended in November 1918, the plant was producing about 10 tons a day.
What made the Chemical Warfare Service's commitment to lewisite curious, however, is that its analysis appeared to ignore some very basic chemistry. Lewisite hydrolyzes–breaks down–when it comes into contact with water, even water vapor. And although the Chemical Warfare Service believed in 1918 that it had scooped the Germans by developing lewisite, in actuality, German chemists had considered lewisite the year before and rejected it–because of hydrolysis.
A 1941 gas mask drill at Fort Dix, New Jersey, included sniffs of lewisite, mustard, phosgene, and chloropicrin; A World War II–era poster (right) stresses lewisite's strong, tell-tale odor.

Were the Germans wrong and the Americans right, or vice versa? There is no doubt that American chemists knew how rapidly lewisite hydrolyzes. Maybe they didn't think it was an issue–the compound appeared very effective when tested on animals (although not under battlefield conditions). Or perhaps the development, production, and hype about lewisite was partly a matter of prestige. Did the Americans need to show the other Allies that they could compete in the chemical warfare game? The United States did tell its allies that lewisite had great potential as a chemical warfare agent, and Britain, at least, started planning its own manufacturing facility.
James Bryant Conant was the chemist in charge of lewisite production during World War I. During World War II, Conant chaired the National Defense Research Council and in that role oversaw U.S. military research in both chemical and atomic weapons. About his work during World War I, Conant writes in his autobiography that he later learned there were “many doubts as to whether lewisite was, in fact, effective.”

But if he did believe those doubts, why did he permit lewisite research to continue during World War II? And Conant did more than permit the research to continue–he was instrumental in the U.S. adoption of an improved manufacturing technique developed by the British.
Consistent with Conant's autobiography, however, a 1918 Chemical Warfare Service report on lewisite concluded that “little effect should be expected” from lewisite vapor. The report indicated that most troops were by then equipped with gas masks, and unlike its toxicity as a liquid, lewisite vapor had low skin toxicity and did not penetrate clothing easily. At the same time, the report suggested that hydrolysis was not an issue because the compounds that formed after its chemical reaction with water were still toxic and effective in contaminating the surrounding environment.
Efforts by Winford Lee Lewis, lewisite's namesake (left), and James Conant Bryant (right) led to the agent's mass production.

The report concluded that the pain and blisters resulting from direct contact with contaminated material, and perhaps the respiratory and ocular effects on unmasked troops, would give lewisite an advantage over mustard. In the end, the report was contradictory in its assessment of lewisite's military value.
Between the wars
After World War I, the public was inundated with popular press articles touting American prowess in the development of the dew of death. Both the New York Times and the Washington Post reported on May 25, 1919 that lewisite was the “deadliest poison ever known.” The Times described how two airplanes carrying lewisite could have wiped out “every vestige of life–animal and vegetable–in Berlin. A single day's output would snuff out the millions of lives on Manhattan Island.”
On June 15, 1919, the Cleveland Plain Dealer ran an article describing how methyl (a code name for lewisite) would have killed half the German army because it was 72 times as toxic as mustard gas. Harper's Magazine declared in 1919 that lewisite belonged at the top of all the American inventions in the war effort; it was “the most powerful weapon of war ever wielded.” In 1922, lewisite even appeared in a serialized short story, “The Slayer of Souls,” published in the Chicago Herald & Examiner. According to a 1925 New York Times story, lewisite was purportedly put into bank vaults to prevent robberies.
During the interwar period, lewisite's value as a chemical warfare agent continued to be debated and evaluated, based both on limited testing and theory. A 1923 Chemical Warfare Service report described the difficulty of developing sufficiently toxic concentrations of vapor. Thirteen years later, the service indicated in a classified publication, A Handbook of Chemical Warfare Agents, that lewisite “is useful in cold frozen countries and in hot very dry countries; i.e., where hydrolysis is of little importance,” perhaps suggesting that the service did not believe it was valuable in temperate climates. The document also indicated that one of the products of the hydrolysis reaction, lewisite oxide, would contaminate an area for a long time because it was still highly toxic and vesicant (caused severe blisters), did not evaporate, and was not washed away by rain.
The debate pertaining to the effectiveness of lewisite also appeared in publicly available books written by military personnel who had access to classified data. In 1925, The Medical Aspects of Chemical Warfare by Lt. Col. Edward B. Veddar indicated that because of lewisite's tendency to hydrolyze, sufficient vapor levels to produce casualties would be unattainable. Maj. Gen. M. W. Ireland, then the U.S. surgeon general, wrote in his 1926 book, Medical Aspects of Gas Warfare (volume 14 of The Medical Department of the United States Army in the World War), that although hydrolysis might impede lewisite's effectiveness, after some had been hydrolyzed by moisture on the skin, successive droplets would penetrate and cause injury. Lt. Col. Augustin M. Prentiss, in his 1937 book Chemicals in War, said that lewisite was effective even after hydrolysis because the products of hydrolysis were toxic. Prentiss also claimed that most battlefields would be dry enough.
Some information on lewisite's potential also appeared in literature outside the United States. Heinz Liepman's Death From the Skies was published in Germany in 1937. The book described an “eyewitness” account of a man entering a room containing an imperceptible amount of lewisite vapor. The man is quickly seized with violent pains and uncontrollable vomiting; four days later, suffers from chronic arsenical dermatitis and nervous breakdown; and dies from lewisite poisoning three months after exposure.
On the other hand, in 1931, in his book, Green and Yellow Cross, German physician-scientist Hermann Büscher described his experiments testing mustard gas and lewisite on human skin. Büscher, who used an impure form of lewisite, found its effects to be less serious and less painful than those of mustard. And another German author, Ulrich Müller, wrote in 1932 in Die chemische Waffe (The Chemical Weapon) that if the Allies had used lewisite in World War I, they would have been greatly disappointed.
World War II
When World War II began, the Chemical Warfare Service had, surprisingly, not developed any new chemical warfare agents. The old standards from the first war, including lewisite, were resurrected, and 23,000 tons of lewisite were manufactured at three U.S. military factories. Lewisite was tested both under restricted and battlefield conditions. Tens of thousands of soldiers “volunteered” for these tests and were exposed to lewisite. The long-term consequences of these tests were compiled by the Institute of Medicine in 1993 in Veterans at Risk: The Health Effects of Mustard Gas and Lewisite. The report definitively linked lewisite exposure to respiratory illnesses and concluded it was possibly associated with ocular diseases, cancer, and immune system dysfunction. [See also, “The VA's Sorry, the Army's Silent,” March 1993 Bulletin.]
During and after World War I, newspapers touted lewisite's deadly effectiveness.

Considering the doubts raised about the battlefield effectiveness of lewisite during the interwar years, the decision to renew its production and testing at the beginning of the war seems open to question.
Curt Wachtel, a German who had migrated to the United States, had evaluated lewisite for the German military in 1918. Wachtel published Chemical Warfare in 1941, in which he described lewisite as having little to offer on the battlefield. He wrote in the foreword that he hoped his knowledge would be “helpful to the military of his adopted country.”
Unfortunately, no one in the Chemical Warfare Service appears to have paid any attention to Wachtel's book or to their own reports.
Lewisite testing and production may have been initiated because it was feared that the Axis powers might use it. If that was what the leaders of the Chemical Warfare Service were thinking, they were right–in part.
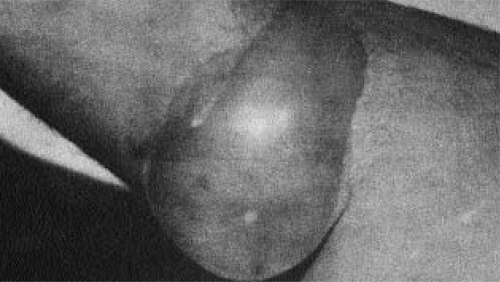
Picture of a lewisite blister 24 hours after a drop was placed on the skin. From Hermann Büscher's book, Green and Yellow Cross (1931).
Despite numerous allegations that Adolf Hitler's Germany would produce lewisite, there is no credible evidence that it was ever in that country's chemical arsenal. Apparently, German chemists trusted the evaluations done earlier by their World War I counterparts.
In contrast, though, Japan did make and use lewisite. In 1929, Japan secretly built a plant on Okunojima, where it manufactured lewisite and other chemical agents. In 1931, Japan invaded Manchuria and began using biological and chemical weapons, including lewisite, on thousands of soldiers and defenseless Chinese civilians. Approximately 10,000 people were killed in gas attacks.
Between 700,000 (a Japanese estimate) and 2 million (a Chinese estimate) abandoned Japanese chemical munitions remain in China today. Numerous casualties and deaths (estimated at about 2,000) have resulted from accidental discoveries of these munitions. In 2000, the Chinese and Japanese governments agreed to a 10-year cleanup project to destroy the remaining munitions.
In most cases, Japanese chemical shells were filled with a lewisite/mustard mixture. Why combine the two? One reason is that the two compounds have somewhat different effects–lewisite causes immediate pain and blister formation and has much greater systemic toxicity than mustard, but mustard achieves higher vapor concentrations. The mixture potentially combines the most toxic effects of each agent. However, after the war, Japanese officers testified that the reason lewisite was added to mustard was that it made the mustard usable at lower temperatures (as in Manchuria). Mustard normally freezes at 55-59 degrees Fahrenheit; lewisite at 0.
U.S. testing of lewisite during World War II revealed that although vaporized lewisite may reach high concentrations immediately after an explosion, its spatial range is small, perhaps no larger than that of an explosive shell. Similarly, vapor sprayed from an airplane was found to be transient. World War II-era reports on lewisite also indicated that soil contaminated with lewisite releases vapor that is odorous, but not injurious. The results of 11 tests indicated that lewisite would only be effective as a direct, liquid contaminant.
Cleanup in Russia
Russia, which inherited the former Soviet Union's entire chemical weapons stockpile, signed the Chemical Weapons Convention in 1993 and ratified it in 1997, renouncing chemical weapons development, manufacture, stockpiling, and future use. Although Russia pledged to destroy its arsenal by 2007, financial constraints mean that the deadline will not be met.
Today, the world's largest stockpile of lewisite (approximately 7,500 tons) is stored mainly at Kambarka in the Urdmurtia region, with remaining stores at Gorny in the Saratov region. The Kambarka facility, founded in 1941, has no alarm system for the detection of dangerous vapor levels and is situated about 3 kilometers from the city of Kambarka, which has a population of 17,100. More than 7,000 tons of lewisite are stored in 80 huge steel containers.
The storage site at Gorny was founded in 1943 as NKO Warehouse No. 276. Currently, 248 tons of lewisite and 231 tons of a lewisite/mustard mixture are stored there. More than 8,000 people live within 10 kilometers of the site.
Because of the environmental dangers associated with lewisite and its degradation products, European countries and the European Union are assisting Russia with the destruction of lewisite at these two sites. Germany committed $60 million in 1992 to build a plant at Gorny to destroy the lewisite and mustard stored there, and an additional $6 million was contributed by the European Union. At the opening ceremony in 2002, German Amb. Hans-Joachim Daerr said this was the first “small step” in the destruction of the Russian stockpile of chemical weapons. Before the plant opened, the governor of the Saratov region, Dmitri Ayatskov, waged an intense public relations campaign to stop the opening of the plant, arguing that no decision had been made regarding the disposal of the toxic byproducts resulting from the destruction of the agents. (Some believe that the governor was simply pressing the plant's financial backers for more aid.) The facility began test runs in May 2003.
In 1996 Joris Voorhoeve, the Netherlands' minister of defense, pledged that his country would provide $16 million to Russia, primarily for the elimination of the lewisite storage facility at Kambarka. The program includes soil remediation around the site, purchasing and equipping a mobile environmental laboratory to monitor the site, medical treatment for workers affected by the agent, and installation of a facility to pump the lewisite from bulk containers to smaller ones to facilitate future transport to a destruction facility.
Sweden has also allocated funds for the Kambarka site. It pledged approximately $125,000 for risk analysis associated with the storage and destruction of lewisite and potential transportation of agents from the site. In a second phase of the project, $350,000 will be committed specifically to accident-reduction measures.
Human field tests conducted by the United States showed that unless a soldier was defenseless or unconscious, the rapid pain and eye irritation and the geranium-like odor associated with lewisite compelled him to move out of the area immediately. Because of hydrolysis, only ineffective vapor concentrations could be achieved.
Once the Chemical Warfare Service concluded that lewisite would not work under battlefield conditions, production was suspended on November 8, 1943. However, stockpiles of lewisite were maintained as a supplement to mustard to “confuse and harass the enemy.” After the war ended, the National Defense Research Council's summary report pertaining to lewisite declared that because the British had developed an effective antidote in 1941, and because it was likely that the antidote would become available to other countries in the future, “there seems little likelihood that there will ever be any incentive for the use of lewisite as a chemical warfare agent.”
After 1943, the U.S. military seems to have finally accepted its own evaluations and not produced lewisite, except in small quantities for testing and identification purposes. In 1948 the vast majority of the 20,000 tons produced during World War II were dumped into the Atlantic and Pacific Oceans in “Operation Geranium.” An unknown quantity is stored in one-ton containers at the Deseret Chemical Depot in Utah (since 9/11, data on the exact amount is no longer available to the public).
The story elsewhere
Britain produced about 156 tons of lewisite during World War II. The United States and Britain discontinued production before the war ended, but the Soviet Union, which apparently considered lewisite to be one of its counters to the U.S. development of atomic weapons, continued to produce it after the war. The Soviet Union produced about 22,000 tons between 1940 and 1945.
The total amount of lewisite produced by the Soviet Union during the Cold War is unknown, but approximately 132,000 tons were dumped into the Arctic Sea in the 1940s and 1950s. Like the lewisite produced by Japan, most Soviet-produced lewisite was combined with mustard in munition shells. Considering the severely cold weather in much of the former Soviet Union, it seems likely that combining the two was an effort to extend the temperature range for mustard. However, some lewisite was put in tanks for spraying from airplanes–suggesting that the Soviets had some faith that it would be effective on its own, despite the fact that U.S. and British testing had found spraying to be ineffective.
2001: Cleanup workers at the lewisite plant at Dzerzhinsk, Russia, continued to wear gas masks.

Perhaps the Soviets viewed lewisite as part of a “scorched earth” policy. They may have believed that by spraying the land with lewisite, they could make it virtually unusable by an invading army. The huge quantities they produced would be consistent with such an approach.
In 1982, the Reagan administration asserted that the Soviets had used poison gas to kill 3,000 people in Afghanistan. Lewisite was among the suspected agents. However, the use of chemical weapons in Afghanistan was never confirmed.
Soviet production methods were primitive and dangerous. One reason the Soviets may not have conducted formal testing with lewisite was the high toll among workers in its chemical weapons plants.
During World War II, the Soviet Union produced lewisite at a chemical munitions plant at Chapaevsk, in the Samara region. It was produced in Shop 7, which had a capacity of 4,400 tons a year. Shells were filled with lewisite in the “women's shops,” where it was poured from a kettle; safer technology was not introduced until the 1950s. In 1992, an article in Izvestia described conditions at the plant: “During the war young men unfit for the front line and girls arrived by the trainload–and just as quickly became ill and died.” Local residents were also adversely affected. Exhaust from the shops went directly into city air, and waste water often spilled into the Chapaevka River and from there into the Volga.
Conditions were no different at a mustard/lewisite plant at Dzerzhinsk in the Nizhni Novgorod region, where large quantities of toxic wastes were buried, dumped into the Oka River, or burned in the open air.
A threat today?
On April 7, 2003, Maj. Michael Hamlet of the U.S. 101st Airborne Division reported finding 14 barrels of chemical agents, described as nerve agents tabun and sarin, as well as lewisite, in Iraq. But these findings were later found to be erroneous. To date, chemical weapon stockpiles have not been found in Iraq. However, Major Hamlet's initial report, which was released to the media, clearly indicates that U.S. forces fully expected to find lewisite; its manufacture would be consistent with Iraq's reliance on Soviet military technology. During the Iraq-Iran war in the 1980s, some Iranian injuries were considered consistent with those caused by lewisite. And it is also possible that lewisite might be more effective on a Middle Eastern battlefield than in a more temperate climate.
Since the Al Qaeda attacks on 9/11, there seems to be renewed interest in lewisite as a potential terrorist weapon. In early 2002, mock terrorist attacks were conducted with lewisite as the purported agent in three U.S. cities: Lemon Grove (a suburb of San Diego), St. Petersburg, and Washington, D.C.
Is a lewisite attack realistic? How dangerous would it be? Lewisite (or at least an impure version of it) is not difficult to manufacture, and raw materials can be easily acquired without attracting too much attention. Once produced, lewisite is relatively stable and easy to transport. If terrorists could find a direct way to disperse the compound, it could be effective–it would certainly induce panic.
In 1997, Ch'oe Ju Hwai, a former member of the North Korean military, testified before Congress that Lizut (lewisite) was only one of many chemical warfare agents North Korea possesses. No hard data exists on North Korea's estimated stockpiles, although it is believed that the country has a total of between 2,500 and 5,000 tons of assorted chemical weapons. A 2002 Commander of U.S. Forces in Korea report asserted that North Korea was self-sufficient in the production of World War I-era chemical agents, including lewisite. If the North is producing lewisite for battlefield use, it is likely that it is the only country in the world currently doing so.
Lewisite is an enigmatic chemical warfare agent. Despite basic chemical data suggesting that its battlefield effectiveness would be minimal, some of the world's greatest military powers produced massive quantities that have gone on to negatively impact the environment. Not enough is known about lewisite degradation or optimum methods for its destruction, compounding the environmental problems that have been created. At a minimum, arsenic-containing residues will continue to haunt Spring Valley and areas in Russia and China for many years to come.
