Abstract
Background:
To create and validate nomograms for the personalized prediction of survival in octogenarians with newly diagnosed nonsmall‐cell lung cancer (NSCLC) with sole brain metastases (BMs).
Methods:
Random forests (RF) were applied to identify independent prognostic factors for building nomogram models. The predictive accuracy of the model was evaluated based on the receiver operating characteristic (ROC) curve, C‐index, and calibration plots.
Results:
The area under the curve (AUC) values for overall survival at 6, 12, and 18 months in the validation cohort were 0.837, 0.867, and 0.849, respectively; the AUC values for cancer‐specific survival prediction were 0.819, 0.835, and 0.818, respectively. The calibration curves visualized the accuracy of the model.
Conclusion:
The new nomograms have good predictive power for survival among octogenarians with sole BMs related to NSCLC.
1. Introduction
Lung cancer poses a serious threat to human health, with nonsmall‐cell lung cancer (NSCLC) accounting for approximately 80%–85% of lung cancers [1]. In recent years, there has been a high incidence of lung cancer among aging adults, and it is estimated that in the United States, approximately two‐thirds of lung cancer patients are over 65 years old, with a rapidly growing group of octogenarians in this cohort. Approximately four‐fifths of lung cancer deaths occur after the age of 65, and one‐fifth of lung cancer‐related deaths occur in octogenarians [2–4]. Octogenarians are less suitable for aggressive treatment than young patients, with a 7.4% survival rate and a worse prognosis after 5 years [5].
Brain metastases (BMs) are the most complicated situation for patients with NSCLC. BMs have an estimated incidence of 30%–50% and are considered to be the most common metastases in single sites [6,7]. BM is a vital element leading to death and can cause neurological symptoms as well as functional and emotional impairment. The natural average survival period for patients who suffer from NSCLC with BM is approximately 1–2 months [8,9], and a population‐based study found a median survival of 16 months in adult patients with NSCLC‐related BM [10]. Aging issues in patients with NSCLC and BM disease heterogeneity were discussed in another population‐based study that included 2,465 research subjects monitored between 2010 and 2013. Octogenarians with sole BM had a median OS of only 6.7 months and had worse cancer specificity than other age groups [11]. Accordingly, octogenarians with brain metastases should be given special consideration.
Underrepresentation of elderly patients in clinical trials is a recognized problem in current practice [12], and clinicians are grossly misinformed about the relative efficacy of changing system therapies in this population. Some scholars have indicated that relatively novel individualized treatment strategies should be designed for the elderly population. Assessing the remaining life expectancy of patients assists clinicians in making treatment decisions.
Expected survival is an important consideration when making treatment decisions. The diagnosis‐ specific graded prognostic assessment (DS‐GPA) ratings system for NSCLC BMs is generally understood. The DS‐GPA is currently the approved prognostic tool for NSCLC‐related BM. The DS‐GPA was developed in one study to predict the survival rate of patients with lung cancer BMs; nevertheless, the median survival time predicted by this assessment model still differed from the actual survival time [13]. Furthermore, in this approach, the age was defined as 70 years old, with no further subdivision for patients aged ≥80 years old. There is a scarcity of research into prognostic models conducted with this population.
This study aimed to determine the overall survival (OS) and disease‐specific survival (DSS) outcomes as well as independent prognostic factors in octogenarians with sole BM from NSCLC. We developed a nomogram survival prediction model to predict the survival of this group of patients.
2. Materials and methods
2.1 Data source
The SEER database is a large cancer database that represents 34.6% of the US population [14], and information on BM during diagnosis could be obtained on SEER after 2010. This study included octogenarians with NSCLC diagnosed between 2010 and 2015. SEER‐Stat version 8.3.5 (National Cancer Institute, Bethesda, Maryland, USA) was used to collect data on typical patients (http://seer.cancer.gov/). Because these data are public and do not involve patient privacy, no ethics committee review or consent was required.
2.2 Identified patients
In the US SEER 2010–2015 population, 49,511 cases of brain metastases were diagnosed. We excluded patients under the age of 80 (n = 44,005), nonprimary lung cancer (n = 1,207), non‐initial diagnoses (n = 1,174), cases with different histopathology types (small‐cell lung cancer, n = 1,382), American Joint Committee on Cancer (AJCC) stage T = 0 cases (n = 276), combined metastases from other sites (n = 650), surgical treatments (n = 26), and AJCC stage M1a/M1 NOS cases (n = 28). A total of 540 patients met the criteria and were allocated randomly in a 7:3 ratio to the training cohort (n = 378) and the test cohort (n = 162).
2.3 Variables of interest
The following information was obtained from SEER for each case: age (80–85 years or ≥86 years), sex (male or female), race (white, black, or other), year of diagnosis (2010, 2011, 2012, 2013, 2014, or 2015), marital status (single, married, divorced/ separated/widowed, etc.), primary site (upper lobe, lower lobe, middle lobe, main bronchus, or other), tumor laterality (left, right, or other), histopathology, AJCC stage, treatment, cause of death, and survival. Disease‐specific survival (DSS) and overall survival (OS) were the outcomes of interest. OS was recorded from the day of diagnosis to the day of death of any cause (including lung cancer) or the last follow‐up visit, whereas DSS was recorded from the day of diagnosis to the day of lung cancer death or the last follow‐up visit. The cut‐off point for the study was December 31, 2017, which is the most recent date for follow‐up.
2.4 Statistical analysis
To compare the clinical characteristics of patients with different outcome events, Fisher’s exact or chi‐square tests were used. The Kaplan–Meier method was used to form the survival curve, and the log rank test was applied for further comparisons. Multivariate and univariate Cox proportional risk models were used to determine the correlations between the clinical variables and the outcomes, and each variable included in the statistical analysis was checked for violations of proportional hazard assumptions. Important variables were screened using random forest (RF) machine learning methods. The factors in the RF model were used to create a nomogram for OS and DSS. The calibration curve was used to determine whether the prediction model was correctly calibrated. In a fully calibrated model, the predicted values should fall along the diagonal line of the calibration plot. The area under the receiver operating characteristic (ROC) curve can be used to assess the model’s discrimination and is plotted based on the model’s predicted probability. Harrell’s C‐index was used to evaluate model discrimination. The Briser score is an evaluation of the model’s discriminative power and accuracy. The P values were bilateral, and statistical significance was considered at P values less than 0.05.
R version 4.1.1 (http://www.r‐project.org/) was used in almost all statistical analyses. The randomForest package in R was used to run RF, and the survival and replot packages in R were used to create the nomograms.
3. Results
3.1 Baseline patient characteristics
Our study cohort included 540 eligible senior patients diagnosed between 2010 and 2015, the training cohort comprised 378 patients, and the validation cohort comprised 162 patients. Of these patients, 519 had an outcome event with OS as the endpoint, and 472 had an outcome event with DSS as the endpoint. The overall cohort median age was 83 years, with 434 patients (80%) in the 80–85 age group and 106 patients (20%) in the 86 and older age group. Male participants accounted for 52% of all patients, whereas females accounted for 48%. White people made up 79.5% of the population. Adenocarcinoma of the lung was the most prevalent pathology, accounting for 70% of all cases. Thoracic radiotherapy was administered to 74% of patients, whereas systemic chemotherapy was administered to only 29% of patients. Table 1 and Table 2 displays the demographic and clinical features of the study subjects with various outcomes.
Sociodemographic characteristics of the cohort.

0: No OS endpoint; 1: Appearance of OS endpoint; NOS: Not otherwise specified
clinicopathological characteristics of the cohort.
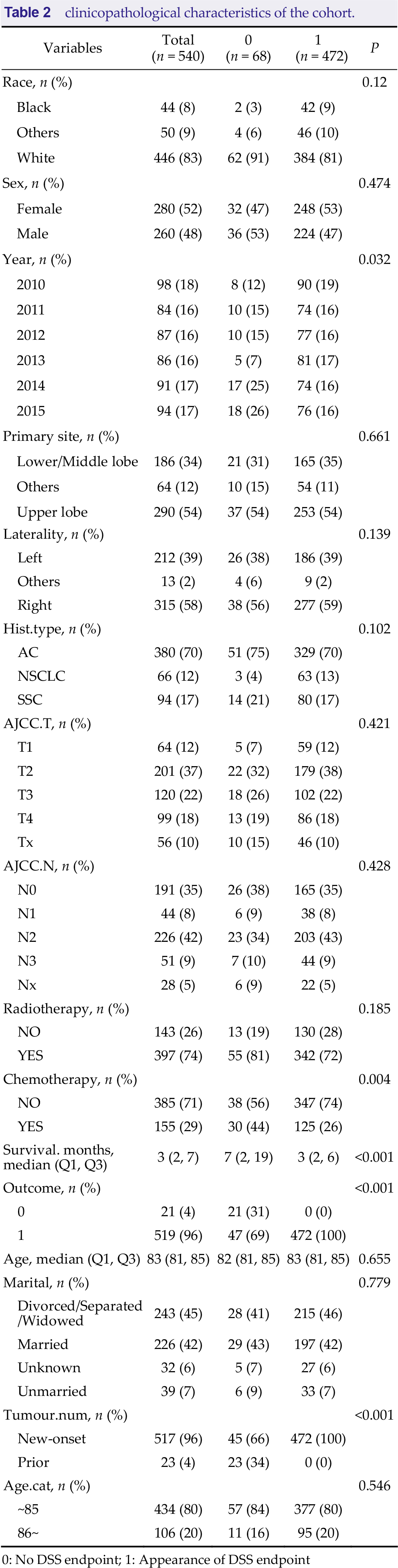
0: No DSS endpoint; 1: Appearance of DSS endpoint
3.2 Survival analysis
According to the survival analysis, the median OS and DSS were both 3 months. There was no significant difference between genders (P = 0.399). Neither OS nor DSS was statistically significant in the comparison of the two age groups (P = 0.097). Regarding OS, patients who received chemotherapy had a relatively good prognosis, with a median OS of 6.5 months (95% CI 5–8 months) and survival rates of 50.0%, 20.9%, and 16.2% at 6, 12, and 18 months, respectively, whereas those who had not received chemotherapy had a median OS of only 2 months (95% CI 2–3 months), corresponding to a statistically significant difference (P < 0.001). In terms of DSS, patients receiving chemotherapy had a median survival time of 7 months (95% CI 6–8 months), with survival rates of 52.6%, 22.9% and 17.9% at 6, 12 and 18 months, respectively. The 6‐, 12‐, and 18‐month OS rates were 29.7%, 13.3%, and 10.3%, respectively, for patients with adenocarcinoma (AC) and decreased to 15%, 3.3%, and 1.7%, respectively, for patients with squamous carcinoma (SCC) (P = 0.031). The 6‐, 12‐, and 18‐month DSS rates were 32.3%, 15.4%, and 12.3%, respectively, for patients with AC, and 25.7%, 7.0%, and 1.7%, respectively, for patients with SCC (P = 0.060). Patients with tumors originating from the upper lobe of the lung had higher survival rates than those with tumors originating from the lower/ middle lobe, with OS rates at 6, 12, and 18 months of 30.9%, 14.9%, and 11.8%, respectively, for the upper lobe and 20.0%, 7.7%, and 4.7% for the lower/middle lobe, with a statistically significant difference (P = 0.008). The 6‐, 12‐, and 18‐month DSS rates were 34.4%, 15.4%, and 13.1% for the upper lobe and 22.4%, 9.6%, and 5.7% for the lower/middle lobe, with statistically significant differences (P = 0.014). Interestingly, we found that patients diagnosed in 2013 had lower OS (P = 0.023) and DSS (P = 0.011) than patients in other years. Figure 1 shows the Kaplan–Meier curves in the training group with OS and DSS as outcomes.

Overall survival (OS) and disease‐specific survival (DSS) in the training cohort. The patients were stratified by tumor site (A, B), pathological type (C, D), whether they had received chemotherapy (E, F), and year of diagnosis (G, H).
3.3 Univariate and multivariate Cox hazard regression analysis
Fitting a Cox proportional hazards model, the hazard ratio was determined. (Schoenfeld residuals tests proved the feasibility of the proportional hazards assumption). Multivariate analysis implied that men [hazard ratio (HR), 1.271; 95% CI, 1.001–1.616] and tumors in the lower lobe of the lung (HR 0.734. 95% CI, 0.578–0.933), SCC (HR, 1.36. 95% CI, 1.005–1.84), AJCC stage N2 (HR, 1.296. 95% CI, 1.003–1.674), not having received chemotherapy (HR, 0.449; 95% CI, 0.348–0.58) and diagnosis made in 2013 (HR, 1.471. 95% CI, 1.025–2.112) were all linked to a lower chance of survival and considered independent risk factors for predicting the OS.
DSS multivariate analysis revealed that divorce/ widowhood [HR, 0.561; 95% CI, 0.325–0.969] and tumors in the lower lobe of the lung (HR, 0.751. 95% CI, 0.587–0.962), AJCC stage N2 (HR, 1.407. 95% CI, 1.072–1.846), not having received chemotherapy (HR. 0.449; 95% CI, 0.343–0.588) and diagnosis made in 2013 (HR, 1.634. 95% CI, 1.128–2.368) were all independent predictors of DSS. Detailed information is shown in Figure 2.
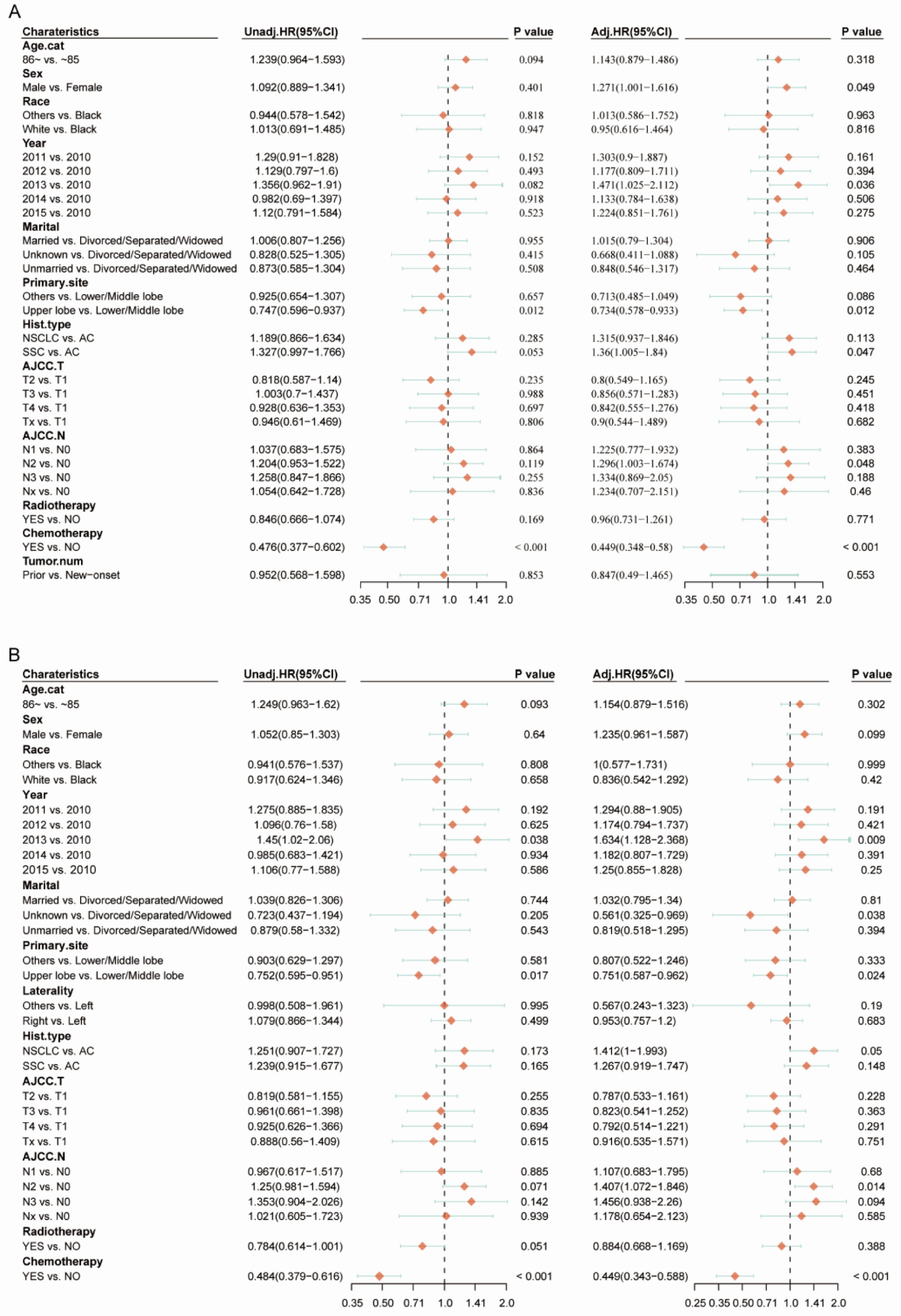
A) Cox proportional hazard model for predicting OS in the training cohort. B) Cox proportional hazard model for predicting DSS in the training cohort.
3.4 Random Forests
We incorporated all extracted variables using the RF machine learning algorithm, and the five most important parameters for predicting OS were chemotherapy and AJCC stage T, AJCC stage N, histology type, and primary site, whereas the five most important parameters for predicting DSS were chemotherapy, AJCC stage N, marital status, AJCC stage T, and primary site. The constructed RF obtained the lowest out‐of‐bag error rates (40.2% and 39.5%). In addition, we observed that four of the five variables corresponded to the most important predictors identified in the multivariate analysis. These results support our combination of the most predictive features to improve the prediction of survival in elderly patients. The variables screened on the basis of the RF model were used for subsequent column line graph model development.
3.5 Calibration and validation of nomograms
According to the OS and DSS, nomograms were drawn from the independent factors in the RF model for 6, 12, and 18 months (Figure 4). As shown in the figure, each factor was assigned a score by calculating all scores and placing them on the total score table. Then, a straight line was generated to predict the prognostic possibilities at each time point.

A) Random Forest model for predicting OS in the training cohort. B) Random Forest model for predicting DSS in the training cohort.
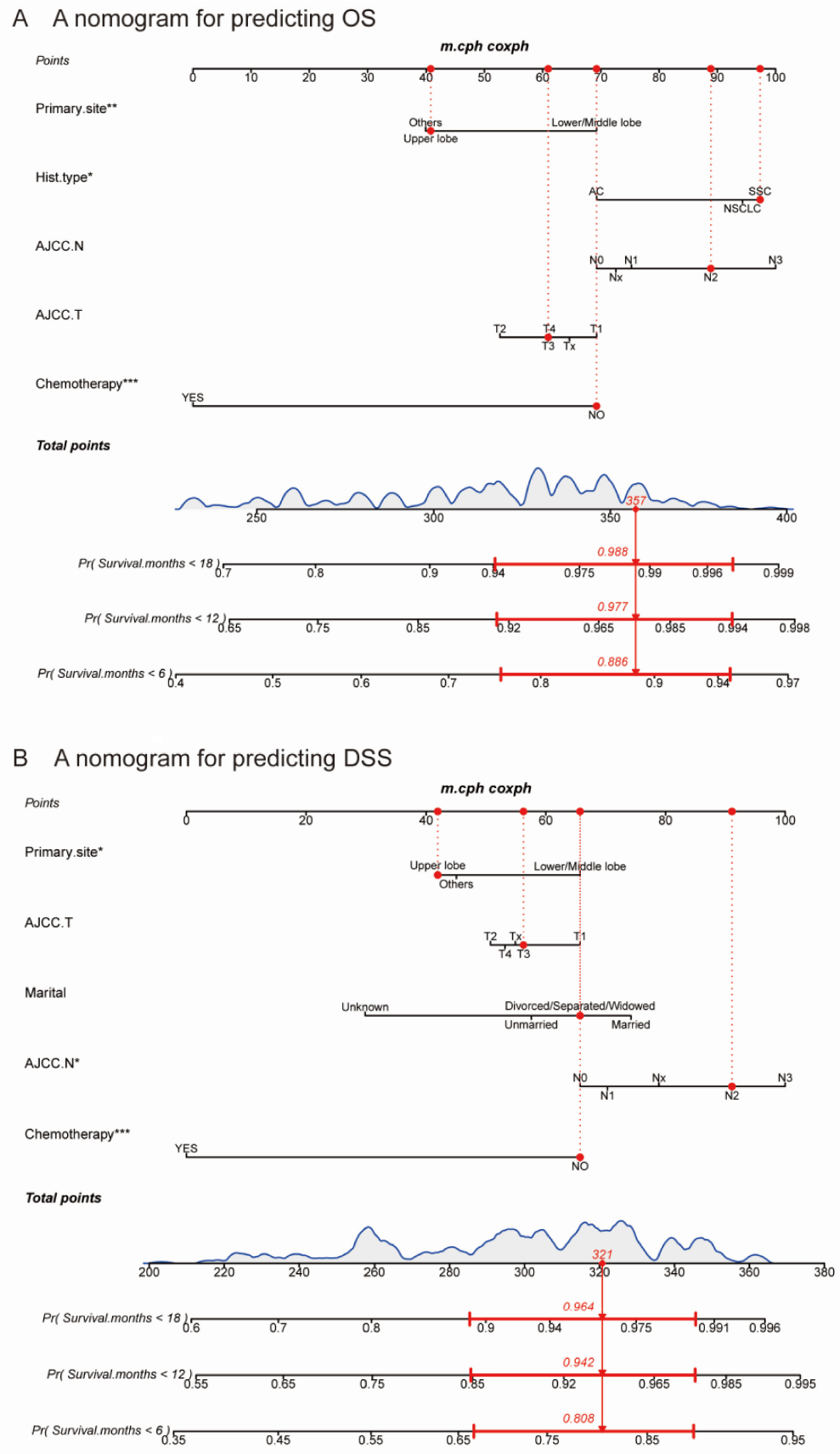
Nomograms for predicting patient OS. A) and DSS B) at 6, 12, and 18 months.
These calibration curves visualized the accuracy of the model, with high correlations between the estimated and real survival possibilities shown by the histogram in the training cohort and satisfactory agreement in the validation cohort (Figure 5). In addition, the area under the curve (AUC) was counted for discrimination in the novel bar graph model. The AUC values for OS at 6, 12, and 18 months in the training cohort were 0.749, 0.719, and 0.730, respectively; those for cancer‐specific survival (CSS) at 6, 12, and 18 months were 0.744, 0.708, and 0. 694, respectively (Figure 6), and those for OS at 6, 12, and 18 months in the validation cohort were 0.837, 0.867, and 0.849, respectively. The AUC values for CSS prediction were 0.819, 0.835, and 0.818, respectively. The C‐index values of the constructed nomograms predicting OS and DSS in the training cohort were 0.641 and 0.654, respectively. The C‐indices of the nomograms predicting OS and DSS in the validation cohort were 0.743 and 0.703, respectively.

Calibration plots of OS (A, C) and DSS (B, D) in the training and validation cohorts. OS, overall survival; DSS, disease‐specific survival.

ROC curve for nomograms. ROC curves of the OS nomogram (A, C) and DSS nomogram (B, D) in the training and validation cohorts. ROC, receiver operating characteristic; OS, overall survival; CSS, cancer‐specific survival; AUC, area under the curve.
The Briser score is able to interpret the model’s discrimination and precision in a comprehensive manner. Our model generated Briser scores of 0.17, 0.09, and 0.07 for OS at 6, 12, and 18 months in the training cohort; scores of 0.18, 0.11, and 0.08 for DSS at 6, 12, and 18 months in the validation cohort; and scores of 0.16, 0.11, and 0.08 for DSS prediction at 6, 12, and 18 months in the validation cohort. The combined evaluation showed that the nomogram model had a high predictive ability.
4. Discussion
In this study, we aimed to build the first clinical prediction model on the basis of an RF machine learning algorithm in an octogenarian NSCLC BM population. This study used a large sample with little selection bias to validate the model’s performance by partitioning the dataset. Nomograms were constructed to predict OS and DSS in this study. The results of the calibration curves, AUC values, C‐index, and Briser scores indicated that our nomograms possess good predictive power.
Older adults, particularly octogenarians, are largely excluded from clinical studies; therefore, little is known about the outcomes in this population (including survival rates, quality of life, etc). Even in guidelines, it is stated that “Really quite limited data are available for octogenarians, and, as a result, no specific recommendations can be made for this group” [15] and no evidence‐based treatment recommendations can be provided. Patients diagnosed with NSCLC with BM have a poor prognosis, and when facing patients aged 80 years, both clinicians and patients are susceptible to the influence of “fatalism” and “futility,” exacerbating the undertreatment of older patients [5,16]. Octogenarians should be considered as a separate group, and it is increasingly urgent to predict the viability of this population using simple and applicable predictive scores, which are essential for making individualized clinical decisions, especially follow‐up strategies, and designing future clinical trials [17].
Several rating models have been employed to predict the survival rate in patients with BM [18], and these models consider four factors: age, primary tumor control, Karnofsky physical status (KPS), and extracranial metastases. The DS‐GPA [19] was developed in 2012 and considers age, KPS, extracranial metastases, and the number of BMs. Sperduto et al. [17] modified this model in 2017 to develop the lung‐molGPA, a new lung cancer survival prediction tool that includes patient age, KPS, extracranial metastases, number of BMs, and EGFR and ALK alterations. The overall cohort’s median OS reached 12 months, with patients with EGFR and ALK mutations and lung‐molGPA scores of 3.5–4.0 having a nearly 4‐year median survival. However, as described in the introduction, the age classification in these models may not be applicable to patients over 80 years of age. Recently, Japanese researchers [20] recommended the elderly‐specific BSBM (ES‐BSBM), which combines BM‐related factors, such as BM number and cumulative tumor volume, and concluded that the model is applicable to all primary tumors and patients over the age of 80. However, the cases in this study were subjected to a high degree of selection bias, and this was not a population‐based study. Neurological symptoms in BM diagnosis had an independent connection with survival prognosis, emphasizing the importance of including neurological symptoms in the prognostic evaluation of patients with BM in NSCLC [21].
Relying on the SEER database, we discovered that NSCLC patients over the age of 80 who were newly diagnosed with BMs had a poor prognosis in terms of both OS and DSS, with no notable difference in survival between the 80–85 age group and the 86+ age group, and a median OS and DSS of 3 months, which is lower than that reported by Gu et al. [11]. Divorce/ widowhood status was found to be an independent prognostic factor for DSS in our study, which is similar to a previous study [22] and may be associated with a lack of social and economic support. Marital status was also included in the DSS nomogram model.
Several studies have shown that octogenarians are reluctant to accept systemic chemotherapy [23, 24], and this phenomenon was indeed present in our analysis, where only a small proportion (29%) received systemic chemotherapy. However, the results showed that chemotherapy was the strongest predictor of patient survival, with receipt of chemotherapy significantly prolonging the OS (median OS 6.5 months vs. 2 months, HR 0.449, 95% CI 0.348–0.58, p < 0.001) and DSS (median DSS 7 months vs. 3 months, HR 0.449, 95% CI 0.343–0.588, p < 0.001). Thus, chemotherapy should be considered as part of the treatment of BM [25]. However, prospective studies have also suggested that most chemotherapy regimens recommended by the NCCN guidelines can achieve some degree of intracranial as well as extracranial response, with a median survival time of 4–12.7 months [26, 27]. Many people have questioned whether chemotherapeutic drugs can cross the blood‐brain barrier (BBB), claiming that chemotherapy has little or no effect on BM [28] and that the role of the BBB should not be overestimated. First, the BBB possesses some chemotherapeutic drug penetration for metastatic brain tumors, which is related to low P‐glycoprotein (P‐gp) expression [29]. Second, BBB disruption is a normal physiological aging phenomenon [30], suggesting that the BBB in the elderly may possess better chemotherapeutic drug permeability. As a result, carboplatin‐based doublets could be regarded as the first‐line therapy in advanced octogenarians; indeed, monotherapy with gemcitabine, vincristine, or paclitaxel has proven to be an eligible option for patients in poor physical condition [15].
The treatment of BM always necessitates a multidisciplinary team that consolidates brain treatment, systemic treatment, and optimal supportive care [31]. Surgical resection is an important local treatment [32], and surgical resection of limited BM in the NCCN may improve survival in selected stage IV patients. However, given the ultra‐advanced age of patients undergoing surgery, the likelihood of survival is small; therefore, we did not include patients who underwent surgery in this study. For BMs, aggressive local radiotherapy (RT), whole‐brain RT (WBRT), and stereotactic radiosurgery are defined as standard of care and have also been shown to be effective in patients ≥80 years of age [33–35]. Although radiotherapy alone does not achieve significantly better survival, the benefit may be in improved intracranial response rates and neurological improvement of symptoms [27]. Recently, novel therapies have achieved extraordinary results in patients with advanced NSCLC, and current studies confirm that ALK‐TKIs [36–40] are effective in treating BM and significantly prolong patient survival. Guidelines strongly recommend EGFR‐TKIs as first‐line therapy for elderly patients with EGFR mutations, and chemotherapy is preferred over TKIs as the first‐line therapy for patients with EGFR mutations[15]. Data on the impact of PD‐1 and PD‐L1 inhibitors [41,42] on BM in patients with NSCLC also seem to be restricted but promising, and one meta‐analysis found that, in patients with NSCLC‐related BM, PD‐1 or PD‐L1 inhibitors reduced disease progression and fatality [43]. Based on these findings, these drugs may be useful in the older population, particularly in patients with driver mutations. In an era of personalized medicine, an increasing number of older patients aged 80 and older are willing to receive aggressive anticancer therapy, and the perfect multimodal combination ofSRT(SRS), WBRT, and systemic agents [44–48] should continue to be explored in prospective studies to develop effective treatments in this population with high clinical needs.
Our research has certain limitations. First, because it is a population‐based study, the SEER database lacks some important data related to patient prognosis (e.g., chemotherapy strategy, radiotherapy dose, gene status, and neurologic symptoms). As new treatment regimens continue to be completed, the SEER registry may also include data on the use of targeted and immune drugs, as well as data on possible changes in patient prognosis. The nomogram information data also needs to be updated. Second, this study has the inherent limitation of retrospective studies (i.e, low level of evidence in evidence‐based medical theory). Finally, it is worth noting that the predictive value calculation nomogram is reference information only and not an absolute accurate prediction. In the future, we will collect more important variables to develop a more rigorous nomogram and further validate our model using external validation methods to make the nomogram a useful clinical prediction tool.
5. Conclusions
Our analysis revealed that chemotherapy, AJCC stage, histological type, marital status, and primary tumor site were independent parameters affecting OS and DSS. The SEER database nomogram model provides a reliable and practical tool for assessing OS and DSS in patients with NSCLC over the age of 80 years with sole BMs.
Footnotes
Future perspectives
Chemotherapy is the main factor influencing survival in octogenarians with newly diagnosed NSCLC with sole BMs. Traditional TNM staging will remain the main method to predict survival outcome. We believe that the nomograms can be used as a new visual prediction tool to assist future clinicians in selecting the most appropriate treatment for patients.
Summary points
Octogenarians with sole BMs from NSCLC should be considered as a separate group, and there is an increasing urgency to use simple and applicable predictive scores to predict the viability of this population, which is critical for making individualized clinical decisions and designing future clinical trials. Japanese scholars have built a predictive model for the elderly; however, it is not a population‐based study and still needs to be improved. The SEER database is a large available database of cancers, representing nearly 34.6% of the US population. Information on patients with BMs diagnosed between 2010 and 2015 was downloaded from the SEER database. The RF machine learning algorithm found that five variables were independent prognostic factors for OS and DS. New nomograms predicting 6‐month, 12‐month and 18‐month OS and DSS, respectively, were constructed. Internal and external validation of the nomograms revealed that the nomograms had good predictive power. Our research still has some limitations, and the prediction model needs to be continuously improved.
Funding information
This work is supported by the key specialty of traditional Chinese medicine promotion project.
Acknowledgments
The authors are grateful to the staff at the National Cancer Institute (USA) for their contribution to the SEER program.
Author contribution
Lj Song: literature search, study design, data collection, data analysis and interpretation, writing, figures
Y Wang: data analysis and interpretation, writing, figures, final approval of manuscript
X Li: data collection, data analysis, final approval of manuscript
Y Liu: data collection, data analysis, final approval of manuscript
By Yin: data collection, data analysis, final approval of manuscript
Hs Lin, Dr Li: Study design,
Yuqi Zhang: approval of final manuscript
Declaration of conflicting interests
The authors have no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter discussed in the manuscript.
Ethics approval
Because these data are public and do not involve patient privacy, no ethics committee review or consent was required.
Consent to participate
Not applicable.
Data availability statement
The data that support the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
