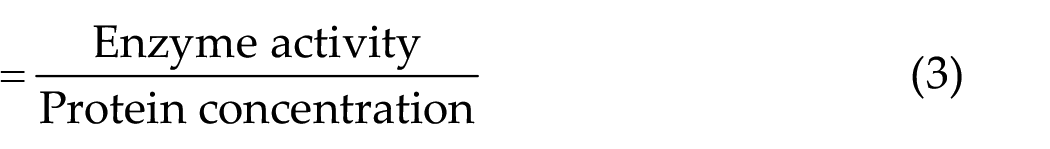Abstract
Background:
Aluminium-induced neurotoxicity has become a subject of substantial concern because of its consequential effects, including its potential role in neurodegenerative disease pathogenesis. It is thought to occur through multiple mechanisms including the generation of reactive oxygen species and the disruption of cellular homeostasis leading to neuronal damage and dysfunction. However, despite growing evidence of aluminium-induced neurotoxicity, the precise mechanisms underlying its effects and potential therapeutic strategies remain incompletely understood. Its manifestations therefore need to be explored so that its adverse effects can be attenuated.
Methods:
In the present study, we investigated the neurotoxic effects of aluminium chloride (AlCl3) in relation to neurobehavioural changes and oxidative damage. We also assessed the therapeutic role and protective effects of aqueous extract of Phoenix dactylifera L. fruit (AEPDLF) on antioxidant levels (catalase and superoxide dismutase) and neurobehavioural patterns in adult female Wistar rats.
Results:
Thirty adult female Wistar rats were divided into five groups (N=30, each group n=6). Control group (A) rats received 2 mL/kg body weight/day (bw/d) of distilled water, and rats in the other four groups (B, C, D, and E) received 200 mg/kg bw/d of AlCl3 for 28 days. In addition, the treated groups (C, D, and E) received 250, 500, and 1000 mg/kg bw/d, respectively, of AEPDLF for 28 days. On the last day of administration, neurobehavioural assessments were performed. Rats were then sacrificed and brains were harvested and homogenised for biochemical assays.
With AlCl3 treatment, body weight decreased more than brain weight. There was also significantly reduced locomotion with AlCl3 treatment; however, dose-dependent amelioration was observed with AEPDLF. Although group B had significantly fewer entries into baited arms, and more entries into non-baited arms than group A in the radial arm maze, these changes were ameliorated in groups C, D, and E in an AEPDLF dose-dependent manner. Furthermore, AlCl3 treatment lowered catalase and superoxide dismutase levels, and these changes were mitigated by AEPDLF in a dose-dependent fashion.
Conclusion:
The protective capacity of AEPDLF in female rats most efficiently combatted neurotoxicity at 1000 mg/kg bw/d in response to chronic (28 d) AlCl3 exposure.
1 Introduction
Neurodegenerative diseases such as Alzheimer's disease, Parkinson's disease and multiple sclerosis are common in the health sector. Unfortunately, many patients present late to the hospital and their insults are often caused by seemingly unavoidable bioavailable substances. Aluminium is a toxic non-biodegradable, and biologically unnecessary metal that is found in the biosphere component of the Earth; it has been linked to various diseases. The increased use of aluminium in industrial processes, domestic appliances and pharmaceuticals has led to increased human and animal exposure [1]. This, in turn, has led to its increased accumulation in the body, where it causes toxicity in various organs including the kidney, liver and brain [2], thereby requiring urgent intervention. Aluminium neurotoxicity begins with its entrance into the brain through the compromised blood-brain barrier, the choroid plexus, and/or the nasal cavity. It can also access the brain via the iron-binding protein, -transferrin or by binding to citrate to form aluminium-citrate [3]. Because its redistribution is slow, it is retained in the brain for a relatively long time.
Neurotoxicity can be defined as the exposure of the brain to insults beyond its metabolic capacity, thus affecting diverse neuronal processes. Aluminium is reportedly a neurotoxic agent capable of causing ionic imbalance, molecular disruptions, and cellular disturbances [4], which then translate to neuromuscular discoordination, neurobehavioural abnormalities and other illnesses.
Aluminium fuels the production of reactive oxygen species which at high levels cause oxidative stress. Worldwide, oxidative stress is currently a leading cause of aberrations within the metabolic homeostatic environment [5]. It has become a mainstay of illnesses and diseases, causing irreparable and irreversible damage to organs of the body. It also cause imbalances at ionic, cellular, and tissue levels, thereby leading to illnesses such as autoimmune dysfunction, cardiovascular disease, neurological disorders, pulmonary diseases, cancer, rheumatoid arthritis, nephropathy, ocular diseases, accelerated ageing, and reproductive system diseases [6]. Furthermore, it is a causative agent for brain illnesses such as neurodegenerative diseases (e.g., Parkinson's disease and Alzheimer's disease) [7, 8].
To combat the effects of oxidation, we need to explore how to mitigate this worldwide problem by providing protective and/or ameliorative measures.
Declining health systems in developing countries have given way to advances in plant-based medicine. This has led to increased research, discoveries, and usage of natural plants and plant parts to treat various diseases, which may be attributed to the bioavailability, cost-effectiveness and low-to-no side effects of plant parts.
Treatments to counteract metal toxicity have been performed using medicinal plants with potential therapeutic effects [9]. Furthermore, neurotoxicity can be ameliorated using medicinal plants and plant parts, including date palm (or Phoenix dactylifera L.). Date palms are one of the oldest trees in the world; the earliest evidence for their cultivation is in the 3rd-millennium BCE (before Common Era) and has progressively shown unequivocal tendencies for centuries. It is an ancient fruit that has been consumed and used for various purposes for centuries. Aside from the usefulness of various other parts of the date palm, date fruit is a relatively cheap source of micronutrients such as carbohydrates, proteins, amino acids, and dietary minerals (selenium, potassium, calcium, and magnesium) [10]. It is also rich in flavonoids and phenols, which have antioxidant properties, and contain polyphenols, anthocyanins, carotenoids, tannins, and sterols [11]. These micronutrients confer anti-mutagenic, anti-cancer and antimicrobial properties on its consumer [12]. Date fruit have also been revealed to have hepatoprotective, nephroprotective, and neuroprotective properties [13]. Moreover, this fruit mediates neuroprotective effects against oxidative stress, toxicity, and illnesses, and its beneficial effects against neurodegenerative diseases have been established. The antioxidant activity of date palm has been attributed to the presence of selenoproteins, anthocyanins, and both phenolic and flavonoid compounds [14]. Its potency, enhanced by its bioactive phytochemical constituents, aids in the detoxification of excess reactive oxygen species (ROS) and reverses oxidative damage.
However, sex discrepancies may occur in terms of how oxidative stress affects the body; as some studies have reported ROS increases in specific organs in males but not in females, whereas other studies have not shown these effects [15, 16, 17, 18]. In this study, we therefore investigated the changes in antioxidation and neurobehavioural expression with aqueous extract of Phoenix dactylifera L. fruit (AEPDLF) treatment following aluminium chloride-induced neurotoxicity in female rats.
2 Materials and methods
2.1 Animal Grouping, Care and Ethics
Thirty healthy adult female Wistar rats (aged 9-10 weeks, weighing 150 - 180 g) were obtained from breeding stock from the Animal House of College of Health Sciences, Obafemi Awolowo University, Ile-Ife, Nigeria. They were housed in the Animal Holding facility of the Department of Anatomy and Cell Biology, Obafemi Awolowo University. The rats were randomised by weight into five groups (A - E, n=6), kept in clean plastic cages and given food daily. They were also given water ad libitum. Ethical approval for the study was obtained from the Health Research Ethics Committee of the Institute of Public Health, Obafemi Awolowo University (approval number IPH/OAU/12/1957).
2.2 Drugs, Plant Materials and Preparation of the Plant Extract
AlCl3 (anhydrous) was purchased from Molychem, (Mumbai, India) and distilled water was obtained from the Histochemistry Laboratory of Obafemi Awolowo University. Dried P. dactylilfera L. fruit was procured from new Ile-Ife township market and, authenticated by a taxonomist in the Botany Department of Obafemi Awolowo University. A voucher specimen (reference number IFE-18030) was deposited at the department's herbarium. The procured date fruit was pitted, cut into pieces, washed under clean runny water, and air-dried at room temperature in a well-ventilated room for approximately 2 months. Next, 8 kg of the dried date fruit was pulverized into a fine powder using a grinding machine. Subsequently 40 g of this powder was mixed with 120 mL of distilled water and placed on a rotary shaker at 190 - 220 rpm with continuous agitation for 48 hours. The mixture was then filtered through muslin cloth [19], and the filtrate (i.e., AEPDLF) was freeze-dried, kept in a desiccator and preserved in the refrigerator (4oC) until further use.
2.3 Administration Protocols
The oral median lethal dose (LD50) of AEPDLF in rats is 5000 mg/kg body weight (bw) [20] and this was used for the present study. AEPDLF was reconstituted in accordance with Al-Sayyed et al., [21] (200 mg to 1 mL). The AlCL stock solution was compounded by adding 2 g of anhydrous AlCl3 to 20 mL of distilled water, to make 200 mg/mL of AlCL3.H2O solution [22]. The oral LD50 of AlCl3 in rats is 3.45 g/kg bw [23]. The AlCl3 was prepared twice weekly, using distilled water, stored in an air-tight bottle, refrigerated after use and administered to rats according to the stated doses while the AEPDLF was prepared daily. Group A (control) rats received 2 mL/kg bw/day (d) of distilled water, and groups B, C, D and E received 200 mg/kg bw/d, respectively, of AlCl3 for 28 days. In addition, groups C, D and E (the treated groups) received 250, 500 and 1000 mg/kg bw/d, respectively, of AEPDLF.
2.4 Neurobehavioural Assessments
Neurobehavioural assessments were performed on each rat on the last day of administration. The radial arm maze (RAM) was used to assess memory, spatial learning, and navigation, and the open field test (OFT) was used to evaluate locomotion and anxiety-related behaviour.
2.4.1 RAM Procedure
The rats were fasted for approximately 24 hours before starting this assessment to create an appetite for food which was the bait that was used. The experimental animals first underwent a random trial individually before the test trials were initiated. Four alternate arms of the eight-arm maze were baited with standard rat pellet, which was hidden from view in a recess at the end of each arm. The rats were placed in the centre compartment, all facing the same arm, and they were then allowed to explore the arms in search of the bait. The identification of the pellets by the rats in any of the four baited arms served as the commencement of the test trial. Each rat was removed from the maze after 2 minutes of exploration and the sequence of entries into the baited and non-baited arms of the maze was recorded. The number of visits to repeated arms was also recorded. Three test trials (T1, T2 and T3) were conducted and analysed and the standard error of mean (SEM) was calculated.
2.4.2 OFT Procedure
A single trial was performed in an open field maze (OFM) which comprised a square wooden box (72 cm χ 72 cm χ 36 cm) that was painted white; the floor was divided by visible lines into 16 smaller squares, each measuring 17 cm χ 17 cm. The open arena of the open field box was divided into two chambers (central and outer chambers). The rats were placed, individually, in the centre of the box, facing the same direction and allowed to freely explore the chamber for 2 minutes. The locomotion frequency (number of line crossings with all four paws, from one square to another) was noted and recorded. After each rat completed its test session, faecal boli and urine were removed. The apparatus was then cleaned with 70% ethanol and, allowed to dry completely before testing a new animal, to eliminate any possible bias from odour cues. Upon completing the test, rats were returned to their home cages. The results were then analysed in terms of the locomotion frequencies (i.e., lines crossed).
2.5 Determination of Body and Brain Weights
Rats were weighed twice weekly using a top-loading digital balance. Total weight gain was expressed as the difference between the final and initial body weights. Percentage weight change was expressed as the percentage difference between the final and initial body weights divided by the initial weight. Brain weights were measured using a sensitive balance (electronic digital scale MH-03B, Shenzhen LapSun store, China) at sacrifice. Relative brain weight was expressed as percentage ratio of the brain weight to the final body weight.
2.6 Biochemical Assay and Determination of Antioxidant Enzyme
2.6.1 Preparation of Brain Homogenates
Brains of the experimental animals were harvested and weighed before half of each cerebrum was homogenized with an electric homogeniser in 10 mL of phosphate buffer (0.1 M), to give a 10% (weight/volume) homogenate. The homogenate was then centrifuged at 704.34 relative centrifugal force ( χ g) for 10 minutes and the supernatant was decanted and used to estimate the levels of superoxide dismutase (SOD) and catalase (CAT). The brain homogenates were centrifuged in precooled ice tubes at 704.34 relative centrifugal force ( χ g) for 10 minutes. Supernatants were then obtained and stored in a freezer (below -18 °C). Aliquots of the supernatants were diluted at a 1:110 ratio and used as the enzyme source for the in vivo assay.
2.6.2 Estimation of Superoxide Dismutase Activity Levels in the Brains of Experimental Rats
SOD activity of the homogenised brain was determined using the Misra and Fridovich method [24]. First, 0.5 mL of brain homogenate was diluted in 4.5 mL of distilled water (1:10 dilution factor), and 0.2 mL of the diluted sample was then added to 2.5 mL of 0.05 M carbonate buffer (pH 10.2) to equilibrate the spectrophotometric cuvette. The reaction was initiated by adding 0.3 mL of substrate (0.3 mM Epinephrine) and 0.2 mL of distilled water and the absorbance was read at 480 nm. The increase in absorbance at 480nm was monitored every 30 s for a period of 150 s and enzyme activity was then calculated using the following formula:
Increase in absorbance per min
Where A0 = absorbance after 30 s and A1 absorbance after150 s
Percentage inhibition
Specific enzyme activity (μmol/minute)
2.6.3 Estimation of Catalase Activity Levels in the Brains of Experimental Rats
CAT activity was determined according to the direct ultraviolet assay method of Aebi [25] which is based on the disappearance of hydrogen peroxide (H2O2) in the presence of CAT. The reaction mixture consisted of 50 μL of the test sample, 950 μL of 50 mM phosphate buffer (pH 7.0), and the reaction was initiated by the addition of 500 μL of freshly prepared 30% (volume/volume) H2O2. The rate of peroxide decomposition was measured by reading the absorbance at 240 nm at 5 s intervals for 30 s. The enzyme activity was then calculated using the following formula:
CAT activity (units/minute) =
Where d = dilution of the original sample
V = sample volume in the CAT reaction (in mL)
0.0436 = extinction coefficient for CAT
K = total reaction volume (in mL)
Note: 1 unit of CAT will decompose 1.0 μΜ of
H2O2 to oxygen and water per minute at 25 °C
and a pH of 7.0.
2.7 Statistical Analysis
The obtained data were tested for normality and homogeneity and were analysed using GraphPad Prism software (v 8.3.0, GraphPad Software, Boston, MA, USA). Data were analysed using one-way analysis of variance followed by Tukeys multiple comparisons test. ImageJ software (National Institutes of Health, Bethesda, MA, USA) was used for image analysis. Results are expressed as the mean ± SEM, the significant difference was set at p < 0.05, and the confidence interval was set at 95%. The significance nomenclature used is as follows; α = p < 0.05 vs. group A, β = p < 0.05 vs. group B, γ = p < 0.05 vs. group C and δ = p < 0.05 vs. group D.
3 Results
3.1 Efficacy of AEPDLF on Brain and Body Weight Following AlCl3 Neurotoxicity
Compared with group A, significantly lower (p < 0.0001, F = 25.68) body weight change were
noted in groups B (p = 0.0070), C (p = 0.0006), D

Effect of AlCl3 & AEPDLF. (A)Effect of AlCl3 & AEPDLF on total body weight changes; (B)Effect of AlCl3 & AEPDLF on percentage body weight changes.
3.2 Neurobehavioural assessments
3.2.1 AEPDLF Confers Protective Effect on Locomotion Following AICI3 Neurotoxicity
Compared with group A significantly fewer (p < 0.0001, F = 100.1) lines were crossed by rats in groups B

Effect of AlCl3 & APDLF. (A)Effect of AlCl3 & APDLF on locomotion; (B)Effect of AlCl3 & APDLF on memory and cognition, i = significantly different when compared with same trial of group A at p < 0.05, ii = significantly different when compared with same trial of group B at p < 0.05, α = significantly different when compared to group A, β = significantly different when compared to group B; (C)Effect of AlCl3 & APDLF on memory and cognition.
3.2.2 AEPDLF Preserves Memory and Cognition Following AlCl3 Neurotoxicity
3.2.2.1 Baited arm:
There were no significant differences (p = 0.0879, F = 3.040) in the comparisons made between trial 1 of the experimental groups. When comparing the entries in trial 2 among the experimental groups significantly fewer
3.2.2.2 Non-baited arm:
There were no significant differences in the comparisons of entries made between trials 1 (p = 0.4645, F = 0.8852), 2 (p = 0.2936, F = 1.378), or 3 (p = 0.0347, F = 4.630) of the experimental groups. Moreover, inter-group comparisons revealed no significant differences
3. 3 AEPDLF Increased Superoxide Dismutase Activity Levels Following AlCl3 Neurotoxicity
There were significantly reduced (p = 0.0192, F = 9.41) SOD activity levels in groups B (p = 0.0303) and C

Effect of AlCl3 & APDLF. (A) Effect of AlCl3 & APDLF on SOD activity levels; (B) Effect of AlCl3 & APDLF on Catalase activity levels.
3.4 Role of AEPDLF on Catalase Levels Following AlCl3 Neurotoxicity
There were no significant differences in CAT activity levels (p = 0.0918, F = 6.24) when comparisons were made (Fig. 3B).
4 Discussion
The diversity of antioxidants and phenolic contents of different varieties of date palm accounts for its protective and ameliorative potency [26]. Some of the metabolites that combat ROS are carotenoids, phenols, flavonoids and proanthocyanidins. The presence of these secondary metabolites indicate the antioxidant capacity of the date fruit that was explored in the present study, which was corroborated by the findings of Kchaou et al., [27], who assessed the aqueous, ethanolic, acetonic and methanolic extracts of six date cultivars, including the Deglet Noor variety, and reported that extractions with polar solvents yielded various ranges and levels of antioxidant activities and phenolic content
On physical observation, exploratory activity parameters such as rearing, grooming and sniffing were observed in control rats but were reduced in experimental rats. In addition, rats in groups B, C, D and E demonstrated restlessness during drug administration. These findings might point to a heightened state of anxiety state in the experimental rats relative to the control; metal-triggered neurotoxicity has been reported to result in these characteristics [28, 29]. Therefore, the observed increase in aggression and anxiety and reduction in exploratory activity might be an early indicator of aluminium-induced neurotoxicity. Aluminium chloride is an acid with corrosive properties and might have caused corrosive injury to the gastrointestinal wall of experimental rats. Aluminium is also known to replace zinc, a biomolecule responsible for maintenance of gut epithelium and intestinal mucosal barrier functions [30]. Corrosive injury and zinc replacement might thus be the reason for the initial reduction in food intake and absorption observed in female rats during the initial days of the experiment. Furthermore, watery faecal pellets were noted, corroborating the altered food metabolism. These features may be responsible for the weight reduction noted in this study.
There was severe weight reduction in the experimental rats, which was likely caused by defective feeding. This defective feeding might have resulted from loss of appetite, supported by an earlier report that the fatality of aluminium-induced neurotoxicity is associated with weight loss [31]. This effect was noted in the AEPDLF-treated female rats but was worse in the untreated (group B) rats. Although, weight loss is generally caused by reduced food intake, impaired metabolism or reduced nutritional efficiency (i.e reduction in the ability to eat a sufficient amount of food or an inability of the body to utilise available nutrients from the consumed food because of altered metabolic pathways), aluminium uptake via the blood stream and its subsequent disruption of glucose metabolism pathways might also explain the observed weight loss [32, 33]. However, occurrence of very similar findings among groups C, D, and E, which received graded doses of AEPDLF, might indicate that a critical dose of AEPDLF is required to reverse weight loss secondary to AlCl3 toxicity. Furthermore, endocrine disruption/ imbalance has been predicted as a causative factor for many symptoms such as weight reduction, loss of appetite and emotional imbalance in females [34]. Although this study did not include any hormonal assays, the noted weight reduction was adverse.
Neurobehavioural assessments of anxiety, locomotion and exploratory activities in rats can be conducted in many ways; however, the most common method is the open field maze. The considered parameters can be used to measure the functionality of motor areas of the brain. In the present study, there was reduced locomotion in the experimental rats, with fewer line crossings in groups B, C, D, and E than in group A. Aluminium causes changes in organelles by binding to phosphate groups on the cell membrane thereby disrupting cell membrane permeability. This leads to degenerative changes in cells which can cause the neurobehavioural deficits observed in this study. The structural degeneration of neurons causes disrupted hippocampal formations and synaptic imbalances, leading to memory impairment. Alumnium also distorts the homeostasis of biodegradable, non-toxic, biochemically necessary metals. It therefore causes physiological and morphological defects at four interconnected levels: biomolecular disruption, impairments/irregularities in organelle functions, neural cell alterations, and conformational changes. These derangements are cofactors for behavioural anomalies.
Our findings also revealed the potency of AlCl3 in causing cognitive and memory impairments, with fewer baited arm entries and more non-baited arm entries by the AlCl3-induced rats. Notably, this was mitigated by AEPDLF administration. The major cause of inflammatory and oxidative responses to aluminium toxicity in the brain are changes in blood-brain barrier permeability as a result of the degeneration of endothelial cells and pericytes - essential components of the blood-brain barrier that regulate both molecular transport and vascular permeability across the barrier. This cellular degeneration is accompanied by damaged mitochondrial structures and membrane disruptions; which have important roles in neuropathological diseases [35]. Mitochondrial (membrane) impairment, an effect of aluminium toxicity, increases mitochondrial permeability, which then increases calcium influx, thereby affecting energy-dependent processes, including protein production machinery (e.g., special transport proteins). Changes in calcium ion levels might lead to endoplasmic reticulum homeostasis perturbation which then affects protein folding and causes endoplasmic reticulum stress, a condition that has been implicated in neurological disorders [36]. Degeneration of the rough endoplasmic reticulum results in reduced protein synthesis/segregation leading to Nissl bodies' disintegration. This might explain the disruption of biological processes around learning, memory and signal conduction. Aluminium toxicity also leads to alterations in brain neurochemistry (e.g., neurotransmitter functions) which might result in reduced locomotive activity, impaired muscle coordination, and cognitive and memory deficits.
Aluminium effects biomolecular disruption by causing imbalances in lipid, protein, glucose, metals, hormone (e.g., parathyroid), and ions (e.g., transferrin, albumin and citrate) homeostases [37, 38, 39]. Oxidative damage is also caused by impairing the oxygen-carrying capacities of cells, thereby suppressing antioxidant production/ activity. Consequentially, oxidative imbalance leads to hydroxyl radical production which might then cause the oxidation and modification of lipids, proteins, carbohydrates, and DNA. This will inferably impair metabolic pathways, reduce/inhibit antioxidant enzymes levels, affect cellular organelles and impair cellular structure. Moreover, ROS generation leads to lipid peroxidation, matrix metalloproteinase inhibition, adenosine triphosphate reduction, neurotransmitter dysfunction, and neural death. It also prevents DNA and RNA formation and, DNA repair, while inhibiting protein phosphatase 2 activity resulting in tau and neurofilament protein hyperphosphorylation [40]. Aluminium toxicity leads to increased biosynthesis of the transferrin receptor, prevention of ferritin production, and increased iron in the cytoplasmic pools of neurons leading to oxidative stress [41]. Oxidative damage is mediated by the generation of free ROS which can directly affect endogenous antioxidant enzymes. SOD and CAT are first line innate antioxidants capable of mopping oxygen radicals thereby enhancing the survival of cells and their organelles [42]. The antioxidative role of SOD occurs within the mitochondria of cells. This study observed a significantly lower SOD activity in brain homogenates from female rats in group B compared with group A and when compared with group E. This reduction in antioxidant levels/SOD activity, linked to AlCl3 toxicity in the brains of rats in groups B, C, D, and E is likely because of an overabundance of oxidants, beyond the antioxidative capacity of antioxidants [43]. The hallmarks of an oxidative environment include decreased membrane fluidity, increased lipid peroxidation, oxidised high-density lipoproteins, and an altered redox status, and these dysfunctions have all been linked to aluminium toxicity [44]. They might therefore explain some of the consequences of aluminium neurotoxicity that were observed in the experimental rats. Catalase is another antioxidant that constitutes a first line of defence; it helps to mop up any remaining oxidants left by SOD during redox reactions, and is localized to peroxisomes. Peroxisomal involvement in ROS generation and its integrity to scavenge H2O2 and other oxygen metabolites has been reported previously [45]. However, for healthy homeostasis the regulation of radical-producing and -scavenging enzymes must be balanced or even enhanced by the production of antioxidant enzymes beyond ROS failure; this can result in peroxisomal disorders [46]. This study noted lower CAT activity levels in AlCl3-treated rats than in controls indicating a reduction in CAT levels caused by AlCl3 toxicity in the brains of the rats.
Diverse studies have revealed the pro-oxidant properties of aluminium in the rat brain by inhibiting SOD and CAT [47, 48] which was further confirmed in this study by reduced brain SOD and CAT levels as they are considered bio-markers of oxidative stress. However, the adverse effects of aluminium toxicity on the enzymatic and antioxidant capacities of the brain was ascertained by a significant decrease in SOD and CAT levels in the AlCl3-treated (group B) rats. Moreover, although treatment with AlCl3 resulted in decreased SOD and CAT levels, these changes were ameliorated by AEPDLF administration. The observed dose-dependent increases in SOD and CAT activities in groups C, D, and E (with AEPDLF administration) suggest the potency of AEPDLF to enhance the removal of free oxygen radicals from the brain, thereby protecting it from oxidative stress by promoting SOD and CAT production (or by supplying exogenous SOD and CAT) and/or activity in the brain. Several previous reports have demonstrated the ameliorative properties of date fruit. For example, a significant decrease in malondialdehyde levels and increased SOD and glutathione activities have been reported after date fruit administration [49]. Similarly, Kalantaripour et al., [50] reported a reduction in neuronal damage in rats treated with date fruit extract, and suggested that it was achieved through decreased lipid peroxidation and the restoration of altered antioxidant enzymes caused by ROS in the brain. Oxidative stress is a primary contributor to neurodegenerative diseases and neurotoxicity. Thus, preventing its build-up and reducing its production may help to prevent and/or repair damaged nerve cells while protecting viable ones. We posit that AEPDLF may significantly reduce behavioral and cognitive function deficits in rats through the activation of anti-oxidative stress pathways. The chemical and phytochemical constituents found in AEPDLF serves as an exogenous supply of potent antioxidants for reducing oxidative damage, thus controlling the progression of oxidation in neuronal cells and restoring homeostatic and neurotransmitter balance. Moreover, the presence of bioactive compounds such as flavonoids and alkaloids may stimulate brain repair mechanisms and support neuronal survival. Although we did not conduct any neurotransmitter assays, we note that one of the protective roles of AEPDLF might include the preservation of ions involved in neurotransmission (e.g., calcium) in the brain.
The results of this study extrapolate a reversal or reduction of the alterations in various mechanisms involved in AlCl3 neurotoxicity; however, more research is needed to unravel the precise mechanisms behind its various effects and more clinical research in humans is needed. Nonetheless, our findings suggest that AEPDLF may serve as a potential drug for the treatment of cognitive impairment.
5 Conclusion
This study showed that AlCl3 effected oxidative capacities in adult female Wistar rats. Notably, our findings highlighted the little but significant antioxidative efficacy of AEPDLF in ameliorating the molecular and neurobehavioural responses to AlCl3. Our findings also emphasised the effectiveness of AEPDLF at dosages above 1000mg/kg bw/d against aluminium toxicity in female rats.
Footnotes
Acknowledgements
None.
Funding information
None.
Author contribution
OKE Janet Olufunmilayo and AYANNUGA Olugbenga Ayodeji: conceptualization, design and data organization. OKE Janet Olufunmilayo: project administration, writing - original draft, data collection and collation.
AYANNUGA Olugbenga Ayodeji: reviewing, editing and revising.
AGBOOLA James Oluwabusayo: Animal rearing and neurobehavioural assessment.
All authors commented on previous versions of the manuscript and approved the final version.
Conflict of interest
The authors declare there is no conflict of interest.
Data Availability Statement
The data that support the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
Ethical Statement
This article is a practice-oriented case study description that made extensive use of experimental animals. Ethical clearance for this study with number IPH/OAU/12/1957 was obtained from Health Research Ethics Committee (HREC) of the Institute of Public Health, Obafemi Awolowo University, Ile Ife (HREC, IPH OAU).
Informed Consent
Not applicable.