Abstract
Cerebellar haemorrhage accounts for 5–10% of all intracerebral haemorrhages and leads to severe, long-lasting functional deficits. Currently, there is limited research on this stroke subtype, which may be due to the lack of a suitable composite neuroscoring system specific for cerebellar injury in rodents. The purpose of this study is to develop a comprehensive composite neuroscore test for cerebellar injury using a rat model of cerebellar haemorrhage. Sixty male Sprague-Dawley rats were subjected to either sham surgery or cerebellar haemorrhage. Twenty-four hours post-injury, neurological behaviour was evaluated using 17 cost-effective and easy-to-perform tests, and a composite neuroscore was developed. The composite neuroscore was then used to assess functional recovery over seven days after cerebellar haemorrhage. Differences in the composite neuroscore deficits for the mild and moderate cerebellar haemorrhage models were observed for up to five days post-ictus. Until now, a composite neuroscore for cerebellar injury was not available for rodent studies. Herein, using mild and moderate cerebellar haemorrhage rat models a composite neuroscore for cerebellar injury was developed and used to assess functional deficits after cerebellar haemorrhage. This composite neuroscore may also be useful for other cerebellar injury models.
Introduction
Cerebellar haemorrhage accounts for 5–10% of all intracerebral haemorrhage stroke cases, 1 affecting approximately 30,000 individuals each year in the U.S. 2 Cerebellar haemorrhage is one of the most devastating subtypes of intracerebral haemorrhage, with mortality rates of 40% 2 and large deficits in neurological function,3–6 with up to 50% of surviving patients retaining cognitive deficits for more than 30 days.7,8
Currently, treatment of cerebellar haemorrhage is limited to supportive care, and despite the poor prognosis, high incidence, and limited therapies of cerebellar haemorrhage, preclinical studies are almost non-existent. To date, five experimental studies have been published on cerebellar haemorrhage; however, no potential treatments have been investigated.9–13 A possible reason for the limited research on cerebellar haemorrhage is the lack of a well-described battery of neurobehavioral tests for evaluating acute functional outcome. Lekic et al.11–13 utilized a neuroscore comprising three individual tests which were subjectively scored, thus limiting the appeal of such an examination. While functional deficits were observed 30 days after cerebellar haemorrhage in the Morris water maze and rotarod tests, the acute neuroscore utilized did not indicate any functional deficits (compared to sham) by 0.2 U of collagenase, despite animals having a large haematoma volume and consequent brain edema. Neurobehavioral testing was not performed in any other experimental studies on cerebellar haemorrhage.
Therefore, the goal of this study was to develop a more comprehensive, robust composite neuroscoring system for cerebellar injury which (1) could identify functional deficits in animals which were subjected to cerebellar haemorrhage by 0.2 U of collagenase, and (2) capture the functional deficits associated with different severities of cerebellar haemorrhage.
Materials and methods
All experiments were conducted in compliance with the NIH Guidelines for the Use of Animals in Neuroscience Research. All studies were approved by the Institutional Animal Care and Use Committee at Loma Linda University. All experiments are reported in compliance with the ARRIVE (Animal Research: Reporting in Vivo Experiments) guidelines. Male Sprague-Dawley rats (300–350 g) were housed in a temperature and humidity controlled environment with a 12-h light/dark cycle and given ad libitum access to food and water.
Sixty male Sprague-Dawley rats (300–350 g) were subjected to either sham surgery (n = 20) or cerebellar haemorrhage (n = 20 for 0.2 U of collagenase, n = 20 for 0.6 U of collagenase). Thirty-six animals were randomly assigned to one of the three injury groups (Sham, 0.2 U of collagenase, and 0.6 U of collagenase) (n = 12/group) to assess the individual neurobehavioral tests. These animals, sacrificed for either haematoma volume (n = 6/group) or brain water content (n = 6/group), were randomly assigned to one of the two endpoints. Eighteen animals, randomly assigned to one of the injury groups (n = 6/group), were then evaluated for functional deficits, using the developed composite neuroscore, each day for 7-days post-injury before sacrifice for histological staining. Finally, six animals, randomly assigned to each group (n = 2/group), were allowed to live for 30-days post-injury before sacrifice for histological staining. Animals were randomly assigned to the groups and endpoints without bias 24 h before performing any surgical procedures.
The sample size required for the brain water content and haematoma volume data was based on a power analysis (SigmaPlot 11.0, SysStat, Germany) of previous data in the cerebellar haemorrhage model from our lab which indicated that six animals per group would be sufficient to test for statistically significant differences. This provided us with 12 animals per group on which we could test the individual neurobehavioral tests for functional dependence of cerebellar haemorrhage (24 h time-point, Figure 3). Upon developing the composite neuroscore for cerebellar haemorrhage (using the data from these thirty-six animals), we used a minimum detectable difference in means of 10 and a standard deviation of 5 (obtained from the data in Figure 3(e)), a power of 0.80, and an alpha of 0.05. This indicated that we would need to allocate six animals to each group for the seven-day composite neuroscore experiment to test for statistically significant differences.
Cerebellar haemorrhage model
The rat model of cerebellar haemorrhage was used as previously described.11–13 Briefly, rats were anesthetized with a mixture of ketamine (100 mg/kg) and xylazine (20 mg/kg). Animals were secured onto a standard rodent stereotactic frame. The head was disinfected, then the scalp was incised and a 1 mm burr hole was made (11.64 mm posterior and 2.4 mm right lateral to bregma). A 27-gauge Hamilton syringe was inserted through the burr hole into the brain to a depth of 3.5 mm. Using a microinfusion pump, 0.2 U/µL bacterial collagenase (VII-S, Sigma, St. Louis, MO) at a rate of 0.2 µL/min, two volumes of collagenase were administered to cause two different severities of cerebellar haemorrhage: 1 µL (mild haemorrhage) or 3 µL (moderate haemorrhage). After infusion, the needle remained in place for 10 min to reduce back-flow. Then the needle was removed, bone wax applied to seal the burr hole, the skin was sutured, and buprenorphine (0.01 mg/kg, subcutaneously) and saline (1 mL, subcutaneously) were administered. Sham surgery included all surgical procedures and needle insertion, but no injection.
Neurological evaluations
Composite neuroscore for cerebellar injury scoring criteria.
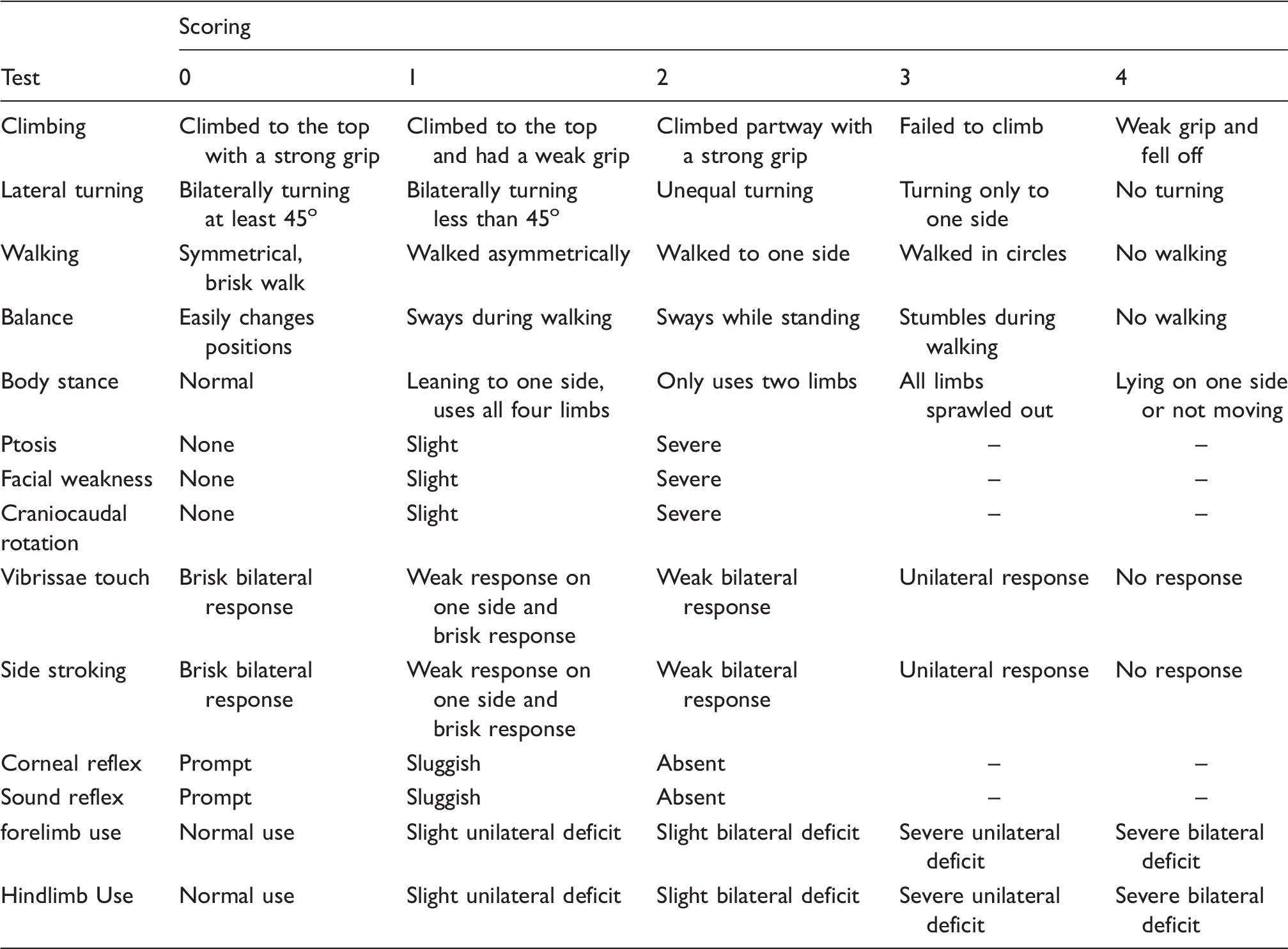
Note: A score of 0 indicates no neurological deficits. Maximum functional deficits score is 46.
Briefly, animals were placed into a novel rectangular cage. Spontaneous activity was observed for 5 min. Animals were then tested for reflex (vibrissae touch, side stroking, corneal reflex) using a cotton swab. The cotton swab was moved from the rear of the animal towards its head touching either the whiskers (vibrissae touch) or side between its hind and front limbs (side stroking). Corneal reflex was tested by advancing a cotton swab towards the animal’s eye until lightly touching the eye. Sound reflex was assessed by lightly rubbing observer’s fingers together behind the animal. The animal response was recorded for each reflex test.
The animal was then lifted out of the cage and placed with its front limbs on the table and the animal was allowed to walk (forelimb walking). The animal was then placed with all four limbs on the table, and the animal’s ability to walk and balance, its body stance, use of its front and hind limbs, and turning ability (lateral turning) were then observed and recorded. Next, the animal was placed onto a 45o gripping surface and allowed free movement to test for climbing ability for 1 min. Grip strength was assessed using a surface with a lip. Finally, the animal was observed for facial weakness (as indicated by facial drooping, asymmetry in facial expressions or jaw, and weakened response to a touch to the cheek), ptosis, dyspnea, and craniocaudal head rotation.
Haematoma volume
Eighteen rats (n = 6/group), immediately after testing neurobehavioral performance at 24 h, were used to quantify haematoma volume. 12 Briefly, deeply anesthetized rats underwent transcardial perfusion of 1 × PBS, then the brains were removed, the cerebellum isolated, and frozen. Cerebellar tissue was homogenized in 3 mL of PBS, sonicated, and then centrifuged; 200 µL of supernatant was added to 800 µL of Drabkin’s reagent (Sigma-Aldrich), incubated in the dark for 15 min, then the absorbance at 540 nm was recorded against a blank (200 µL of PBS in 800 µL of Drabkin’s reagent) using a spectrophotometer (Genesys 10 S, Thermoscientific).
Brain water content
Eighteen rats (n = 6/group), immediately after assessing neurobehavioral performance at 24 h, were used for measurement of brain water content. 12 Briefly, deeply anesthetized rats were decapitated, the brains removed, and isolated into right and left hemisphere, right and left cerebellar hemisphere, and brain stem. Each section of brain tissue was weighed for the ‘Wet weight’, then dried in an oven at 100℃ for two days, and reweighed for the ‘Dry weight’. Brain water content (%) = (Wet weight – Dry weight)/(Wet weight) × 100.
Histological staining
Histological staining was performed on 18 rats (n = 6/group) used in the 7-day study and six rats (n = 2/group) at 30 days post-injury. Deeply anesthetized rats were transcardially perfused with PBS, then 4% formalin. The brains were removed, dehydrated with 30% sucrose, then mounted for cryosectioning. The brains were sectioned into 30 µm slices (at − 10 Bregma), then Nissl stained using cresyl violet as previously described. 12
Statistical analysis
In this exploratory study, all tests were two-sided and no further adjustment for multiple comparison was done for the overall number of tests. After normality was confirmed (for haematoma volume and brain water content), data was analysed using one-way ANOVA with Tukey’s post hoc test and is expressed as mean (standard deviation, SD). For non-normal data (i.e. neurological tests’ and composite neuroscore at 24 h after injury), the data was analysed with Kruskall-Wallis with Dunn’s post hoc tests and are presented as the median and the interquartile range (25th percentile, 75th percentile). For the time course of functional deficits measured by the composite neuroscore for days 1–7 post-injury, the composite neuroscore was analysed using the Scheirer-Ray-Hare test (Real Statistics Resource Pack software, Release 4.3), a non-parametric two-way ANOVA test, followed by the Dunn’s post hoc test. This longitudinal data was also independently analysed using a repeated measures two-way ANOVA with Tukey post hoc test (GraphPad) after performing a square root transformation of the raw data (and confirming normality after transformation). The composite neuroscore time course experiment (Figure 4) is presented as the median and the interquartile range (25th percentile, 75th percentile). A p-value of less than 0.05 was considered statistically significant. GraphPad Prism 6 (La Jolla, CA, USA), SigmaPlot 11.0 (SysStat, Germany), and the Real Statistics Resource Pack software (Release 4.3) were used for graphing and analyzing all data.
To measure the intra-operator variability (κ-statistic, GraphPad, La Jolla, CA, USA), animals were categorized into either no deficits (0 ≤ composite neuroscore < 4), mild deficits (4 ≤ composite neuroscore ≤ 18), moderate deficits (18 < composite neuroscore ≤ 32), and severe deficits (32 < composite neuroscore ≤ 46). The range for no deficits was based on observations from sham and naïve animals’ performance on the developed composite neuroscore. The ranges of mild to severe deficits were determined by dividing the range of ‘deficits’ scores (4 to 46) into equal ranges.
Sensitivity and specificity of the developed composite neuroscore for cerebellar haemorrhage were analysed for appropriately identifying rats with and without functional deficits resulting from cerebellar haemorrhage.
Results
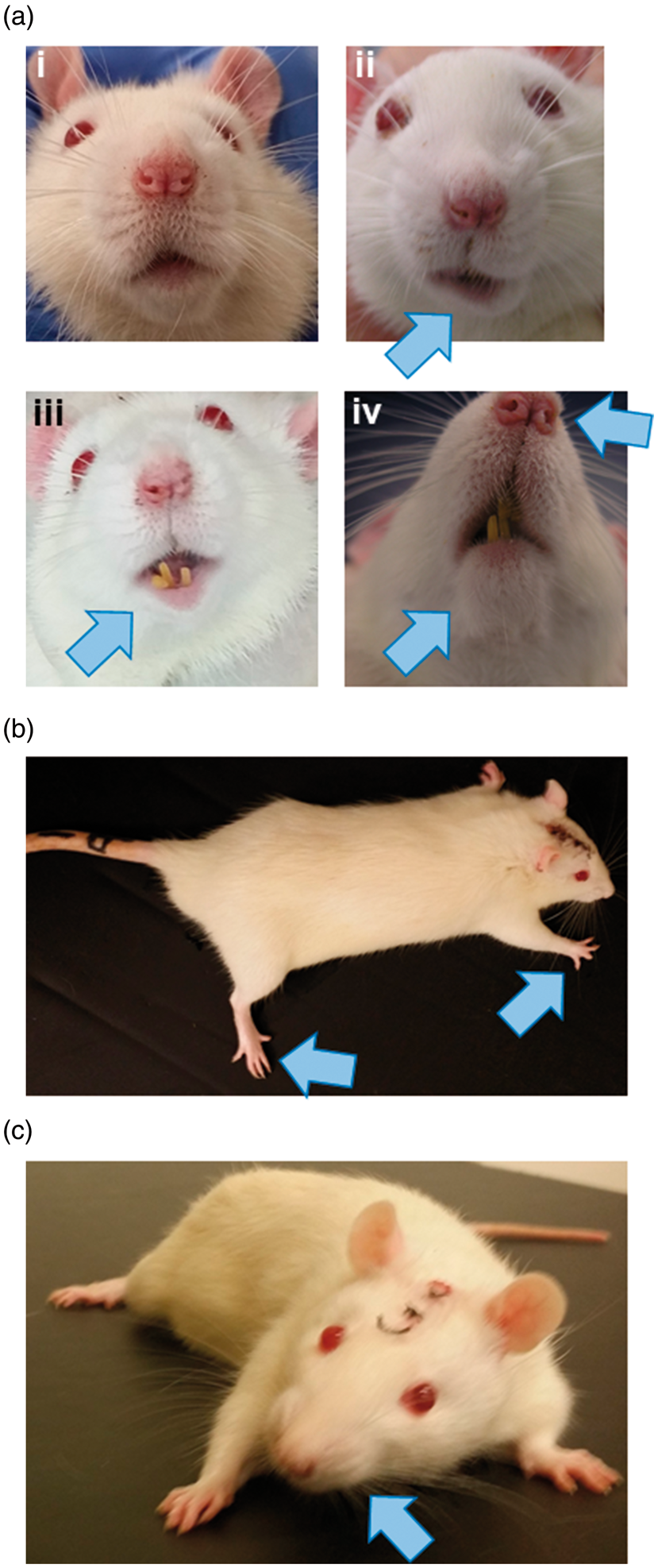
No mortality was observed in this study. A Supplemental Video is available online showing acute functional deficits in rats after cerebellar haemorrhage. Figure 2 shows representative deficits in the facial weakness, limb use, and craniocaudal head rotation tests.
Cerebellar haematoma
Injection of collagenase into the cerebellum of rats resulted in haematomas 24 h post-ictus (Figure 1(a)), and a larger haematoma volume than Sham (sham vs. cerebellar haemorrhage (0.2 U): p = 0.005, sham vs. cerebellar haemorrhage (0.6 U): p < 0.001, cerebellar haemorrhage (0.2 U) vs. cerebellar haemorrhage (0.6 U): p < 0.001) (Figure 1(b)). 0.6 U of collagenase caused a bigger haematoma than 0.2 U of collagenase (Figure 1(b)) (Table 2).
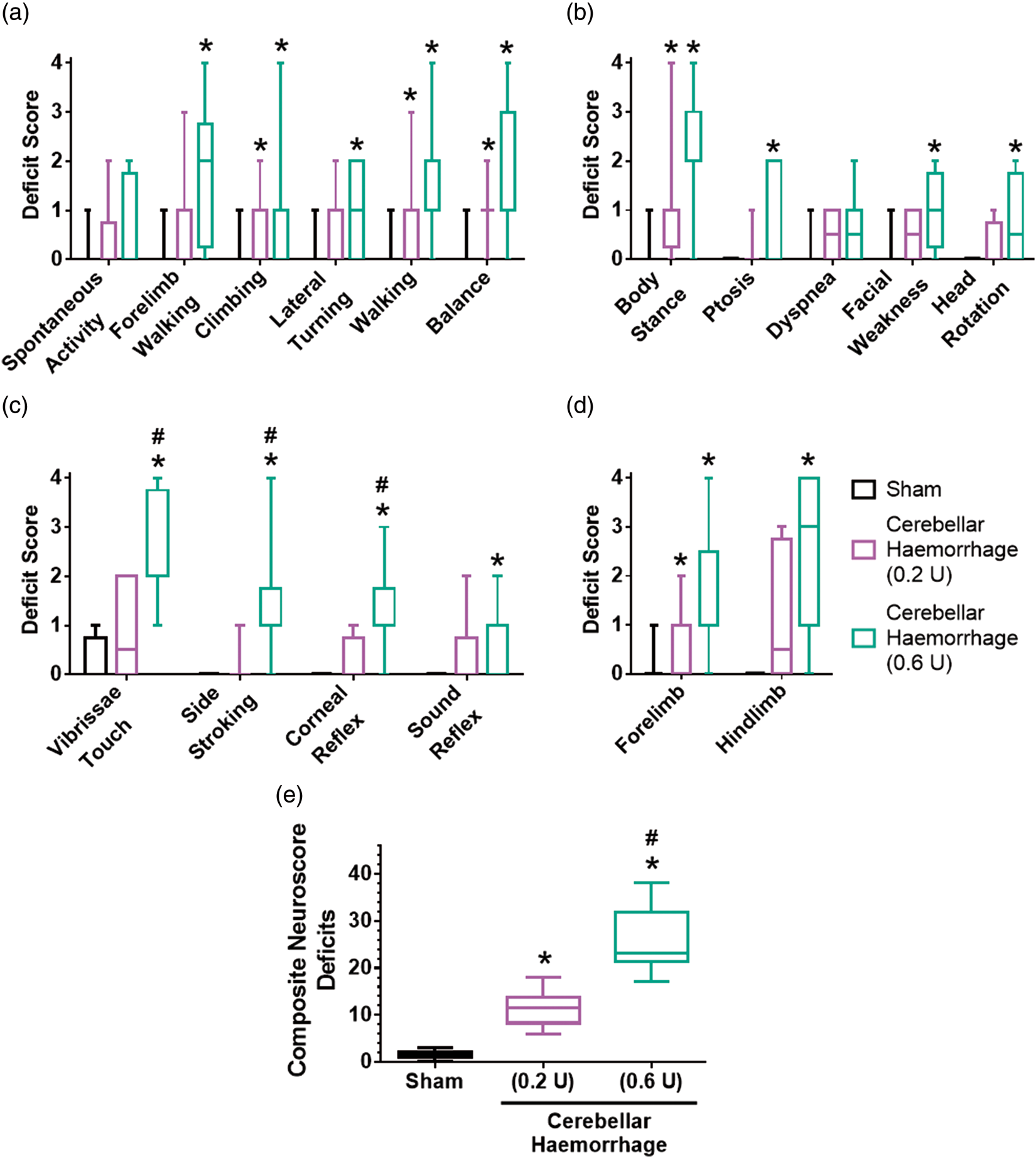
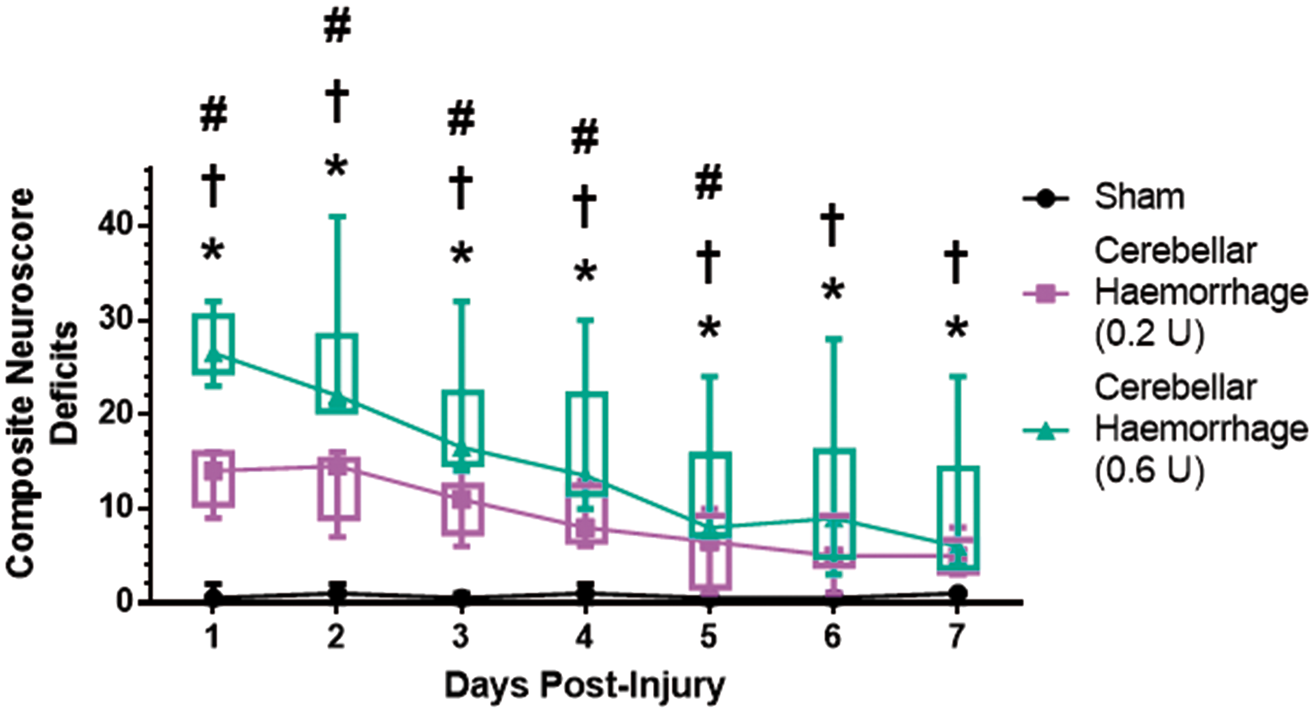
Cerebellar haemorrhage rat model 24 h post-ictus. (a) Cerebellar haematoma (blue arrow). ‘R’ indicates the right side of the brain. (b)Cerebellar injection of collagenase causes a haematoma (mild cerebellar haemorrhage: 0.2 U of collagenase; moderate cerebellar haemorrhage: 0.6 U of collagenase). n = 6/group. Haematoma volume and brain water content. *p < 0.05 vs. sham, #p < 0.05 vs. cerebellar haemorrhage (0.2 U). Note: Values are expressed as mean (SD). n = 6/group/measure. Exact p-values are reported in the Results Section. Acute ataxic deficits after cerebellar haemorrhage. (a) Facial weakness 24 h post-ictus. Sham animals do not show signs of facial weakness (i). Rats with cerebellar haemorrhage (ii–iv) are prone to facial weakness (blue arrows) characterized by facial drooping, asymmetry in facial expressions or jaw, and weakened response to a touch on the cheek. (b) Ipsilesional limb use rigidity (blue arrows) persists up to seven days post-ictus. (c) Craniocaudal head rotation (blue arrow) persists up to seven days post-ictus. Neurological tests 24 h after cerebellar haemorrhage. (a) Coordination and balance deficits were assessed using spontaneous activity, forelimb walking, climbing, lateral turning, walking, and balance tests. (b) Posture and strength deficits were assessed by the body stance/tone, ptosis, dyspnea, facial weakness, and craniocaudal head rotation tests. (c) Reflex deficits were assessed using the vibrissae touch, side stroking, corneal reflex, and sound reflex tests. (d) Limb use deficits were assessed using the forelimb use and hindlimb use tests. (e) Composite neuroscore deficits 24 h after cerebellar haemorrhage. Mild cerebellar haemorrhage resulted in significantly more deficits than did sham surgery, and moderate cerebellar haemorrhage had an even worse performance. n = 12/group. *p < 0.05 vs. sham, #p < 0.05 vs. cerebellar haemorrhage (0.2 U). Box and whiskers plot: horizontal line is the median, box is the interquartile range, whiskers are the minimum and maximum values. Composite neuroscore for cerebellar injury. Composite neuroscore deficits over seven days post-injury. Box and whiskers plot: symbol is the median, box is the interquartile range, whiskers are the minimum and maximum values. n = 6/group. *p < 0.05 for sham vs. cerebellar haemorrhage (0.2 U), †p < 0.05 for sham vs. cerebellar haemorrhage (0.6 U), #p < 0.05 for cerebellar haemorrhage (0.2 U) vs. cerebellar haemorrhage (0.6 U).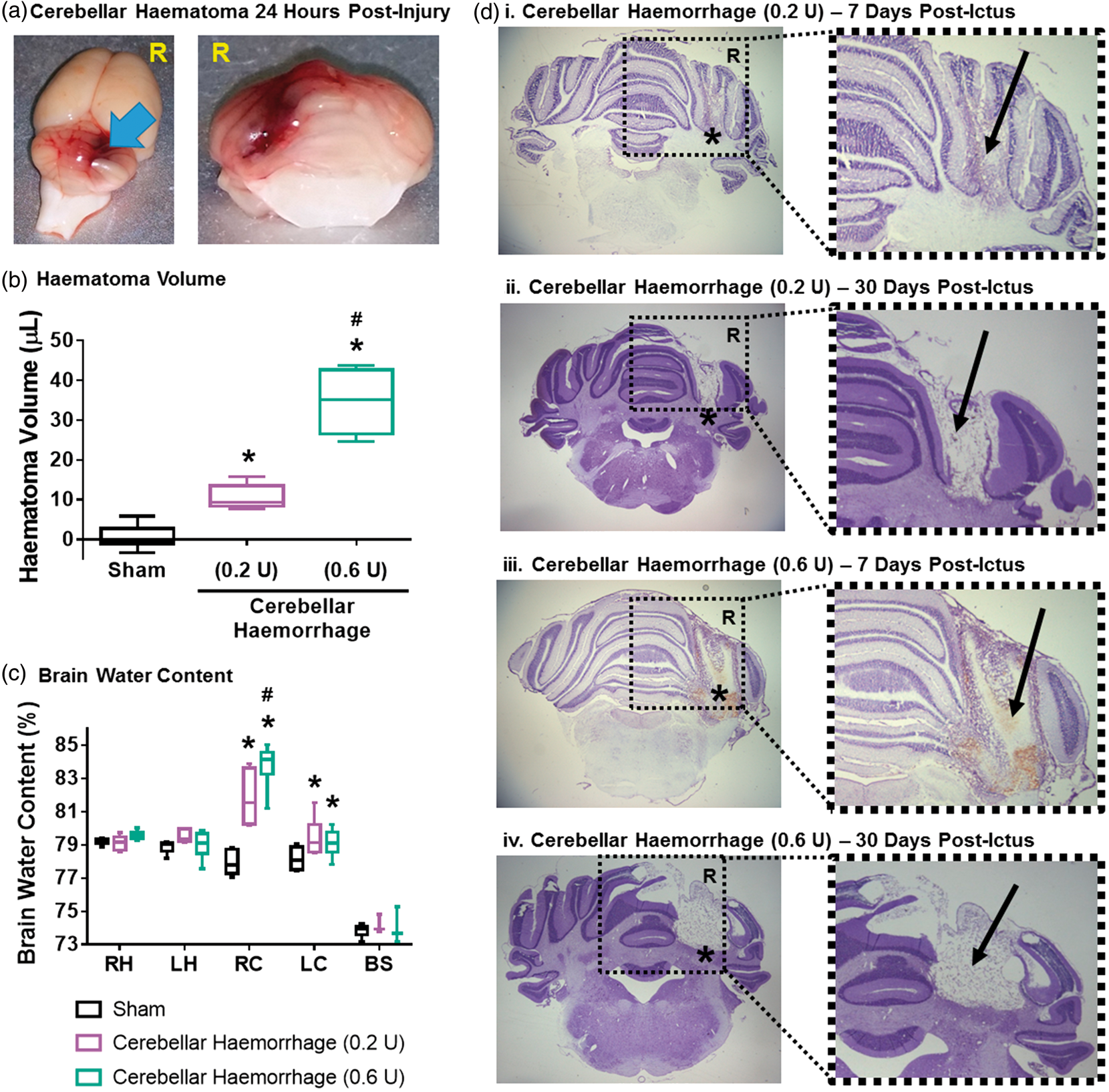
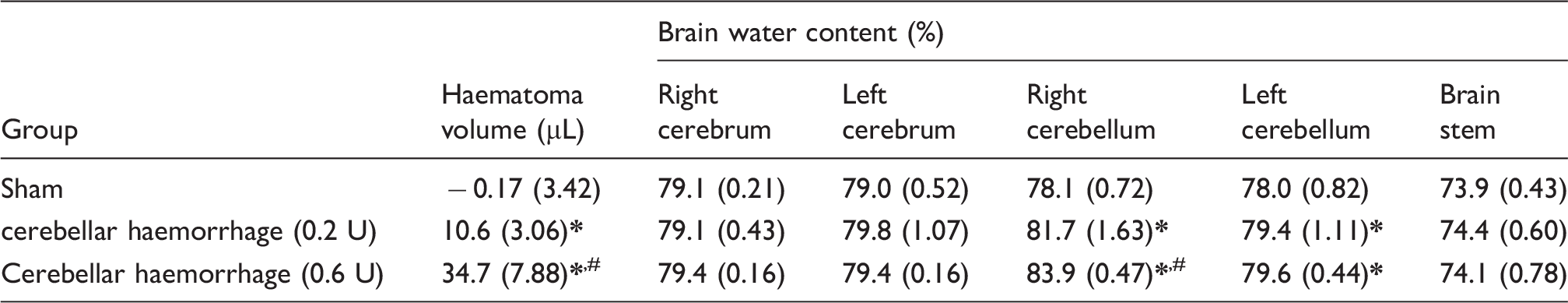
Collagenase infusion also led to in an increased water content of the right cerebellums compared to that of Sham (sham vs. cerebellar haemorrhage (0.2 U): p < 0.001, sham vs. cerebellar haemorrhage (0.6 U): p < 0.001), with a significantly higher right cerebellar water content for the moderate cerebellar haemorrhage model (0.6 U) compared to that of the mild cerebellar haemorrhage (0.2 U) (cerebellar haemorrhage (0.2 U) vs. cerebellar haemorrhage (0.6 U): p = 0.048) (Figure 1(c)). Both collagenase doses for cerebellar haemorrhage lead to significantly higher water content in the left cerebellum compared to that of sham (sham vs. cerebellar haemorrhage (0.2 U): p = 0.003, sham vs. cerebellar haemorrhage (0.6 U): p < 0.001) (Table 2, Supplemental Table VI).
Neurobehavior: Coordination and balance tests
Twenty-four hours after cerebellar haemorrhage, rats were subjected to six functional tests evaluating coordination and balance: spontaneous activity, forelimb walking, climbing, lateral turning, walking, and balance (Figure 3(a)). Cerebellar haemorrhage using 0.2 U of collagenase produced significant deficits compared to sham animals’ performance in the climbing, walking, and balance tests (climbing: p = 0.0185, walking: p = 0.0351, balance: p = 0.0025). Cerebellar haemorrhage using 0.6 U of bacterial collagenase led to significant deficits in the forelimb walking, climbing, lateral turning, walking, and balance tests compared to the performance in animals subjected to only sham surgery (forelimb walking: p = 0.0032, climbing: p = 0.0142, lateral turning: p = 0.0241, walking: p < 0.001, balance: p < 0.001). No statistical significance was observed between any groups for spontaneous activity (sham vs. cerebellar haemorrhage (0.2 U): p > 0.9999, sham vs. cerebellar haemorrhage (0.6 u): p = 0.3615, cerebellar haemorrhage (0.2 u) vs. cerebellar haemorrhage (0.6 U): p = 0.9017).
Neurobehavior: Posture and strength tests
The body stance/tone, ptosis, dyspnea, facial weakness (Figure 2(a)), and craniocaudal head rotation (Figure 2(c)) tests were used to assess posture and strength (Figure 3(b)). Cerebellar haemorrhage with 0.2 U of collagenase causes deficits in only the body stance/tone test compared to sham performance (p = 0.0469), while 0.6 U of collagenase also led to deficits in the ptosis, facial weakness, and craniocaudal head rotation tests compared to the performance of sham animals (body stance/tone: p < 0.001, ptosis: p = 0.0249, facial weakness: p = 0.0066, craniocaudal head rotation: p = 0.0120). Neither mild nor moderate cerebellar haemorrhage caused significant dyspnoea (sham vs. cerebellar haemorrhage (0.2 U): p = 0.3734, sham vs. cerebellar haemorrhage (0.6 u): p = 0.1918, cerebellar haemorrhage (0.2 u) vs. cerebellar haemorrhage (0.6 U): p > 0.9999).
Neurobehaviour: Reflex tests
Reflex was assessed using four tests: vibrissae touch, side stroking, corneal reflex, and sound reflex (Figure 3(c)). Cerebellar haemorrhage with 0.2 U collagenase did not induce significant deficits (compared to sham) in any of the reflex tests (vibrissae touch: p = 0.5392, side stroking: p > 0.9999, corneal reflex: p = 0.7734, sound reflex: p = 0.4755). However, 0.6 U of collagenase led to significant deficits in all four reflex tests compared to the performance of sham (vibrissae touch: p < 0.001, side stroking: p < 0.001, corneal reflex: p < 0.001, sound reflex: p = 0.0027), and also caused significantly more deficits than 0.2 U of collagenase in the vibrissae touch, side stroking, and corneal reflex tests (vibrissae touch: p = 0.0054, side stroking: p = 0.0014, corneal reflex: p = 0.0061, sound reflex: p = 0.1676).
Neurobehavior: Limb use tests
The deficits in limb use were determined using two tests: forelimb use and hindlimb use (Figures 2(b) and 3(d)). Cerebellar haemorrhage using 0.2 U of collagenase led to statistically significant deficits in the forelimb use test compared to Sham performance (forelimb use: p = 0.0312, hindlimb use: p = 0.1074), whereas 0.6 U of collagenase caused significant functional deficits in both the forelimb use and hindlimb use test compared to the performances of sham (forelimb use: p < 0.001, hindlimb use: p < 0.001). No differences were observed for either forelimb or hindlimb use between the performances of the two cerebellar haemorrhage models (forelimb use: p = 0.7464, hindlimb use: p = 0.0720).
Composite neuroscore for cerebellar injury
Composite neuroscore data.
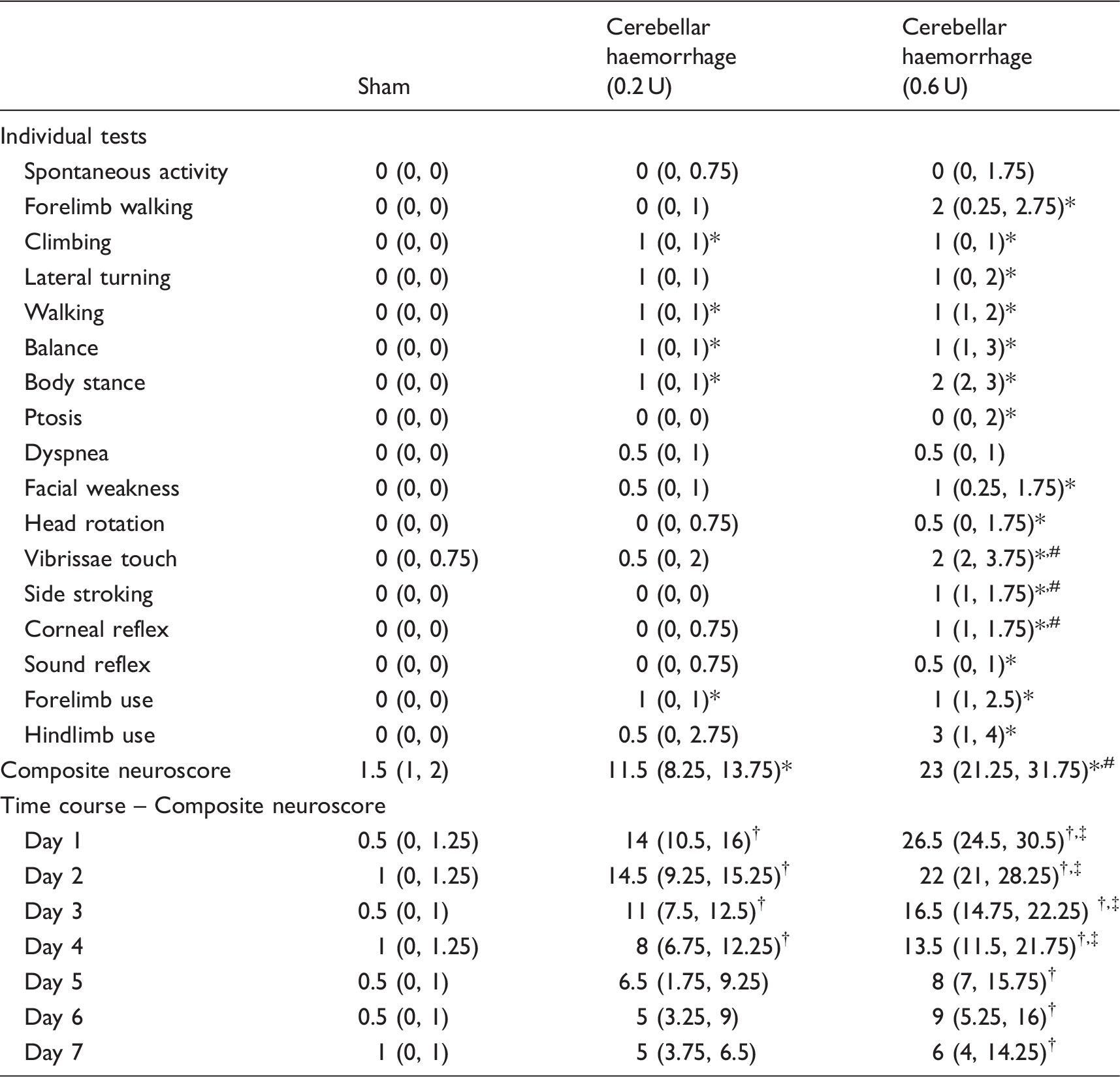
Note: Values are expressed as median (interquartile range 25th percentile, 75th percentile). Individual neurobehavioural tests (n = 12/group): *p < 0.05 vs. sham, #p < 0.05 vs. cerebellar haemorrhage (0.2 U). Time course (n = 6/group/day): †p < 0.05 vs. sham on the same day, ‡p < 0.05 vs. cerebellar haemorrhage (0.2 U) on the same day. Exact p-values are reported in the Results Section.
Composite neuroscore intra-operator variability, sensitivity, and specificity
Animals were categorized into either no deficits (0 ≤ composite neuroscore < 4), mild deficits (4 ≤ composite neuroscore ≤ 18), moderate deficits (18 < composite neuroscore ≤ 32), and severe deficits (32 < composite neuroscore ≤ 46). The range for no deficits was based on observations from sham and naïve animals’ performance on the developed composite neuroscore. The ranges of mild to severe deficits were determined by dividing the range of ‘deficits’ scores (4 to 46) into equal ranges. Within these categories, the intra-operator variability to place animals into the correct categories was calculated to be κ = 0.880 (95% confidence interval: 0.756 to 1.000). All sham animals were correctly identified in the ‘no deficits’ category. Animals with cerebellar haemorrhage by 0.2 U of collagenase were all identified as having mild deficits. Animals with cerebellar haemorrhage by 0.6 U of collagenase were identified as having either moderate (9/12) or severe (3/12) functional deficits in the composite neuroscore.
For sensitivity and specificity analysis, animals were categorized as either no injury or cerebellar haemorrhage based on either no functional deficits (neuroscore < 4) or functional deficits (neuroscore ≥ 4) in the composite neuroscore test. The sensitivity and specificity at 24 h post-injury were calculated, using the above cutpoint, to be 100% (24 true-positives in a total of 24 injured animals) and 100% (0 false-negatives in a total of 12 sham injured rats), respectively. An ROC curve and table of sensitivities and specificities have been added to the Online Supplementary Material (Supplemental Figure V, Supplemental Table V).
Composite neuroscore testing over seven days post-cerebellar haemorrhage
When animals with cerebellar haemorrhage are evaluated each day for seven days post-ictus, the composite neuroscore for cerebellar injury shows that the mild cerebellar haemorrhage group recovers faster than the moderate cerebellar haemorrhage group (Figure 4). Use of the Scheirer-Ray-Hare statistical method to analyse, this longitudinal data conclude that cerebellar haemorrhage causes significantly more deficits in the composite neuroscore than did sham surgery (H = 89.40, p < 0.001), with marginal variation in the time factor (H = 12.12, p = 0.059). Transforming the raw composite neuroscore data (square root transformation) followed by a repeated measures two-way ANOVA with Tukey post hoc test reaches a similar conclusion; the group factor is a major source of variation (F = 72.51, p < 0.001, % of total variation: 72.1) as well as time variation (F = 17.74, p < 0.001, % of total variation = 8.6). A multiple comparisons test after the repeated measures two-way ANOVA test indicated that cerebellar haemorrhage caused significantly more composite neuroscore deficits than that observed in Sham rats up to seven days post-ictus (Day 1 sham vs. cerebellar haemorrhage (0.2 U): p < 0.001, Day 1 sham vs. cerebellar haemorrhage (0.6 u): p < 0.001, day 2 sham vs. cerebellar haemorrhage (0.2 u): p < 0.001, day 2 sham vs. cerebellar haemorrhage (0.6 U): p < 0.001, Day 3 sham vs. cerebellar haemorrhage (0.2 u): p < 0.001, day 3 sham vs. cerebellar haemorrhage (0.6 u): p < 0.001, day 4 sham vs. cerebellar haemorrhage (0.2 U): p < 0.001, Day 4 sham vs. cerebellar haemorrhage (0.6 U): p < 0.001, Day 5 sham vs. cerebellar haemorrhage (0.2 u): p < 0.001, day 5 sham vs. cerebellar haemorrhage (0.6 u): p < 0.001, day 6 sham vs. cerebellar haemorrhage (0.2 u): p < 0.001, day 6 sham vs. cerebellar haemorrhage (0.6 u): p < 0.001, day 7 sham vs. cerebellar haemorrhage (0.2 u): p < 0.001, day 7 sham vs. cerebellar haemorrhage (0.6 U): p < 0.001). Additionally, significant differences in the composite neuroscores of cerebellar haemorrhage by 0.6 U of collagenase and that of cerebellar haemorrhage by 0.2 U of collagenase were observed on Days 1–5 post-ictus (Day 1 cerebellar haemorrhage (0.2 U) vs. cerebellar haemorrhage (0.6 U): p < 0.001, Day 2 cerebellar haemorrhage (0.2 U) vs. cerebellar haemorrhage (0.6 U): p = 0.0016, Day 3 cerebellar haemorrhage (0.2 U) vs. cerebellar haemorrhage (0.6 U): p = 0.0183, Day 4 cerebellar haemorrhage (0.2 U) vs. cerebellar haemorrhage (0.6 U): p = 0.0374, Day 5 cerebellar haemorrhage (0.2 U) vs. cerebellar haemorrhage (0.6 U): p = 0.0469, Day 6 cerebellar haemorrhage (0.2 U) vs. cerebellar haemorrhage (0.6 U): p = 0.0659, Day 7 cerebellar haemorrhage (0.2 U) vs. cerebellar haemorrhage (0.6 U): p = 0.2982). (Table 3, Supplemental Tables VII and VIII).
Histological damage
Histological staining of rat brains after cerebellar haemorrhage shows haematoma-induced damage in the right cerebellum 7 days after injury (Figure 1(d): i, iii) and brain atrophy 30 days post-ictus (Figure 1(d): ii, iv). The damage caused by 0.6 U of collagenase was primarily located in the cerebrocerebellum but extended into the ipsilateral portion of the spinocerebellum (paravermis). Additionally, the damage extended into the vermis in about half of the animals subjected to moderate cerebellar haemorrhage. About one-half of the rats with moderate cerebellar haemorrhage also had damage to the vestibulocerebellum. Mild cerebellar haemorrhage caused damage mainly to the cerebrocerebellum with less than half of the animals having only minimal damage to the spinocerebellum (paravermis). No rats subjected to mild cerebellar haemorrhage showed damage to either the vermis or the vestibulocerebellum.
Discussion
Cerebellar haemorrhage is characterized by a haematoma within the cerebellum and is often associated with brain oedema, leading to functional deficits. To date, preclinical, basic science research is lacking on cerebellar haemorrhage. This may be, in part, due to a lack of a specific functional neuroscore test to evaluate post-haemorrhagic neurobehaviour in rodents. Cossu et al.9,10 developed the blood injection model of cerebellar haemorrhage in rats, but functional deficits were not evaluated. Lekic et al. injected bacterial collagenase to induce cerebellar haemorrhage in rats. Lekic et al.11–13 tested acute neurological function following injury using three indices combined into a modified Luciani neuroscore: body tone, limb extensions, and dyscoordination. Using the collagenase model, Lekic et al.11–13 observed acute functional deficits in their neuroscoring system for cerebellar haemorrhage created with 0.6 U of collagenase, but not 0.2 U of collagenase. Despite 0.2 U of collagenase causing a large haematoma, no reason was mentioned as to why 0.2 U of collagenase failed to induce functional deficits.11–13 Interestingly, 0.2 U of collagenase is similar to the dose used to produce intracerebral haemorrhage, which indeed results in a large haematoma and functional deficits.14–16 Although neurological deficits were observed for cerebellar haemorrhage (specifically 0.6 U of collagenase), the limited number of tests utilized (three) and their subjectively are likely to reduce sensitivity to injury severity, making it more difficult to distinguish between haemorrhage severities, as well as treatment effects. Furthermore, there are a variety of other neurological examinations used for cerebellar injuries (including ischemia 17 and traumatic injury18,19) which are likewise limited or otherwise cost ineffective (i.e. require pretraining, are long term tests, or require expensive equipment). Thus, the development of a more comprehensive, objective composite neuroscoring system, which is cost effective and easy to perform, was the goal of this study.
Performance of rats on individual neurobehavioral tests
In order to establish a composite neuroscoring system, several tests were performed to evaluate neurological function related to cerebellar injury: coordination and balance, posture and strength, reflex, and limb use.3,4,6,7,20 All of the tests used were economical, easy and fast to perform, and required no pretraining. Animals were subjected to each of the 17 tests, which were then examined for sensitivity to cerebellar injury (specifically cerebellar haemorrhage) and its severity.
Despite cerebellar haemorrhage inducing significant deficits in several of the coordination/balance, posture/strength, and limb use tests, none of these tests alone were sensitive enough to distinguish between the two severities of cerebellar haemorrhage induced in rats. However, three of the reflex tests were able to identify differences between the two cerebellar haemorrhage severities, but this is due to the lack of reflex deficits in animals receiving mild cerebellar haemorrhage. Therefore, in order to develop a robust and rigorous neurobehavioral examination which is sensitive to cerebellar haemorrhage severity, several tests were combined to create a composite neuroscore for cerebellar injury.
Correlating cerebellar injury with functional deficits
Cerebrocerebellum
The cerebrocerebellum regulates movement, gait, and limb use,3,4,6 so the damage (necrosis and atrophy) to the cerebrocerebellum, which occurred in both cerebellar haemorrhage severities, may have led to deficits in the forelimb walking (i.e. altered forelimb use), walking (i.e. altered gait and circling), lateral turning (i.e. circling), and climbing (i.e. grip strength and climbing ability) neurobehavioural tests. Since the control of movements and limb use is regulated by the cerebrocerebellum, and seeing that both severities of the rat cerebellar haemorrhage model induce damage to the cerebrocerebellum, it is no surprise that animals subjected to either severity of cerebellar haemorrhage tended to have significant functional deficits in many of the coordination and balance, and limb use tests. Additionally, many of the rats with cerebellar haemorrhage displayed ipsilateral turning or circling (i.e. the rats could not turn towards the contralateral (left) side).
Clinically, cerebellar haemorrhage includes the dentate nucleus (part of the cerebrocerebellum). The location of the haemorrhage in our rodent model is centred at − 11.64 mm posterior to bregma and 2.4 mm right lateral from the midline, and 3.5 mm deep. This location, as confirmed using The Rat Brain in Stereotaxic coordinates (Figures 63–66), is adjacent to the dentate nucleus. Therefore, the dentate nucleus is damaged by the blood (downstream injury from red blood cell lysis), but also from swelling and compression (due to increased cerebellar water content). So, it expected that rats subjected to cerebellar haemorrhage by collagenase have functional deficits including coordination and timing of movement since the cerebrocerebellum (dentate nucleus and the overlying cortex) is directly damaged.
Spinocerebellum
The spinocerebellum (comprises the vermis and paravermis) receives sensory inputs and is responsible for regulating proprioception, locomotion, muscle tone, nystagmus, and posture.3,4,6 Thus, damage to the spinocerebellum may lead to deficits in the walking (i.e. altered gait), balance, body stance (i.e. limbs sprawled out, stumbling, not moving), craniocaudal head rotation, vibrissae touch, side stroking, sound reflex, and forelimb and hindlimb use (i.e. rigid limbs) neurobehavioral tests.
Damage to the paravermis is invariably linked with limb use deficits, with the primary underlying cause being limb rigidity. 4 In the rat model of moderate cerebellar haemorrhage, forelimb and hindlimb use deficits were observed in the majority of the animals, with the typical presence of limb use deficits being rigid limbs (Figure 2(b), Supplemental Video). Additionally, the functional deficits observed in the limb use tests were primarily unilateral (affecting only the ipsilesional limbs). Less than 25% of the rats subjected to mild cerebellar haemorrhage had any signs of limb rigidity, and those which showed limb rigidity only displayed hindlimb rigidity.
Interestingly, moderate cerebellar haemorrhage in the rat caused significant deficits in the reflex tests (vibrissae touch, side stroking, and sound reflex), whereas the mild cerebellar haemorrhage rodent model did not have many deficits in any of these reflex tests. This is likely caused by damage to the spinocerebellum, and particularly the vermis; about 50% of the moderate cerebellar haemorrhage rats had damage extend into the vermis, whereas no rats with mild cerebellar haemorrhage had damage within the vermis.
Vestibulocerebellum
The primary function of the vestibulocerebellum is to regulate equilibrium and movement of the eyes3,4,6 which may lead to the deficits observed in the balance, body stance (i.e. leaning or being unable to display normal posture), corneal reflex, ptosis, and facial weakness tests.3,4,6 In the moderate cerebellar haemorrhage rat model, about half of the animals contained damage to the vestibulocerebellum, which likely led to the significant functional deficits in these neurobehavioral tests, most notably being the ptosis, facial weakness, and corneal reflex tests, which are primarily linked with vestibulocerebellar damage. Interestingly mild cerebellar haemorrhage also had significant functional deficits in the balance and body stance tests; however, this may be explained by (1) altered gait induced by cerebrocerebellar damage, (2) limb deficits induced by spinocerebellar damage, or (3) compression of the vestibulocerebellum due to increased cerebellar water content (which did not lead to any gross histological damage to the vestibulocerebellum).
Although the vestibular nuclei are located in the pons, the vestibulocerebellar pathway extends from the brain stem to the inferior cerebellar peduncle. The large amount of histological damage (seen in Figure 1(d)) shows that the vestibulocerebellar pathway is likely to be affected (increased cerebellar water content leads to compression of the flocculonodular lobe), leading to functional deficits in balance and body stance/tone.
Towards the development of a composite neuroscore for cerebellar injury in rodents
The use of composite neuroscores is not new. Indeed, a number of composite neurobehaviour scoring systems have been developed and used for evaluating neurological function after stroke in rats.21–23 Although many of these composite scores were developed for ischemic stroke models, several composite neuroscores were modified to study functional outcome after intracerebral haemorrhage and subarachnoid haemorrhage models.16,24,25 A major reason for the use of a composite neuroscoring system is the lack of sensitivity by individual tests. However, when the appropriate tests are combined into a composite score, sensitivity to injury severity increases. 26
To this end, a composite neuroscore was developed for cerebellar injury and used to evaluate functional outcome over the first seven days after cerebellar haemorrhage. A neuroscoring system based on Luciani’s observations of patients with cerebellar injury 20 served as the basis of the composite neuroscore developed within. Cerebellar lesions in patients have been reported to cause decreased muscle tone, reduced strength, and dyscoordination.7,20 Based on these functional symptoms identified by Luciani, 20 the composite neuroscore was developed to evaluate: (1) coordination and balance using the climbing, lateral turning, walking, and balance neurobehavioral tests, and (2) posture, strength, and muscle tone using the body stance/tone, ptosis, facial weakness, and craniocaudal head rotation neurobehavioral tests.
In addition to Luciani’s observations, our composite neuroscore for cerebellar injury also incorporated other functional deficits observed in patients with cerebellar injury;5,7,27 a number of other ataxic deficits and hypoesthesia/reflex deficits have been reported in patients with cerebellar injury.5,7,27 Therefore, the composite neuroscore for cerebellar injury also incorporated reflex and sensation (with the vibrissae touch, side stroking, corneal reflex, and sound reflex neurobehavior tests) and ataxic deficits, specifically limb use (with the forelimb use and hindlimb use tests). By incorporating these tests evaluating specific functional deficits which have been reported in cerebellar injury patients, the composite neuroscore was developed and is capable of identifying worsened neurobehavioral performance in rats subjected to cerebellar haemorrhage compared to that of sham animals. Furthermore, this composite neuroscore was also able to differentiate the neurological function of mild cerebellar haemorrhage from that of moderate cerebellar haemorrhage, indicating that our composite neuroscore is sensitive to cerebellar injury and its severity. This suggests that the composite neuroscore for cerebellar injury may provide the platform on which functional recovery after treatment of cerebellar damage can be tested. However, this remains to be studied.
Study limitations
One limitation of the present study is the severity of cerebellar haemorrhage induced in rodents. No mortality was observed in this study despite a significant portion of the cerebellum containing a haematoma. Thus, even using 0.6 U of collagenase to induce cerebellar haemorrhage may not produce a severe cerebellar haemorrhage similar to that observed in the clinic. In future studies, we will examine greater doses of collagenase for inducing cerebellar haemorrhage to more closely mimic the human presentations.
Secondly, rats recovered fairly rapidly in the composite neuroscore developed within. This is not uncommon with composite neuroscore examines for other brain injury models. Indeed, rodents recover remarkably well from middle cerebral artery occlusion,28,29 subarachnoid haemorrhage, 30 intracerebral haemorrhage, 29 and surgical brain injury. 31 Thus, the rapid recovery observed during the first week after cerebellar haemorrhage is acceptable (and not a flaw of the composite neuroscoring system). Also, based on the data within, our composite neuroscore for cerebellar haemorrhage may be used for up to one week post-cerebellar haemorrhage, after which more sensitive, long-term behavioural tests should be used (such as rotarod, foot fault, and beam balance).
Another limitation of this study is the difference between statistical significance and clinical significance. The time course study of composite neuroscore deficits after cerebellar haemorrhage for one week post-ictus indicated that cerebellar haemorrhage by 0.2 U or 0.6 U of collagenase led to statistically different composite neuroscores compared to that of sham animals on each day after injury. However, it should be noted that while the p-values are less than 0.05, the actual functional deficits score’s on Days 5–7 for the mild cerebellar haemorrhage model (0.2 U of collagenase) may not be of clinical significance; the same can be said of the composite neuroscore deficits for cerebellar haemorrhage by 0.6 U of collagenase on Days 6 and 7 post-injury.
Future studies will also use this newly developed composite neurobehavioural scoring system to investigate potential therapies for cerebellar haemorrhage, as well as the identify any correlation between functional deficits induced by cerebellar haemorrhage and the primary pathological outcomes of said injury (i.e. haemorrhage volume and brain edema).
Conclusions
Herein, a composite neuroscore for cerebellar injury was created, using 14 economical and time efficient tests, and it was found to be more sensitive to cerebellar haemorrhage severity than the individual neurobehaviour tests. Since the goal of preclinical studies is to be clinically translational, functional testing is of the utmost importance. The composite neuroscoring system for cerebellar injury, developed within, will allow for treatment effects to be better evaluated in the rat models of cerebellar injury, increasing translation of such preclinical experiments.
Footnotes
Funding
This work was supported by a National Institutes of Health Research Program Projects grant to J.H. Zhang (NIH grant P01 NS082184).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
DWM, JT, and JHZ contributed to study design. DWM, DN and TL contributed to data collection. DWM, HK, GW, and LH contributed to the discussion and drafting. DWM and JHZ made revisions.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
