Abstract
Researchers emphasized acute lacunar stroke (ALS) patients suffer from poor social/physical outcomes, cognitive decline, and decreased quality of life. We hypothesized brain abnormalities may occur in ALS during this particular stage and may be associated with cognitive deficits upon evaluation. We investigated structural abnormalities in ALS using magnetic resonance imaging and voxel-based morphometry conducted on 28 healthy controls (HC) and 29 patients with ALS and proximal anterior circulation occlusion within 12 hours of symptom onset. Mini-Mental State Examination (MMSE) scores were used to evaluate cognitive dysfunction. Decreased gray matter (GM) in ALS vs. HC was predominantly in the superior frontal gyrus, inferior frontal gyrus, insula, superior temporal gyrus (STG), heschl gyrus, middle temporal gyrus (MTG), posterior cingulate cortex (PCC), hippocampus (HIP), and others. Positive correlation was found between GM density and MMSE scores in STG (r = 0.59, p = 0.0007), MTG (r = 0.46, p = 0.01), PCC (r = 0.42, p = 0.02), HIP (r = 0.4, p = 0.03), and medial prefrontal cortex (r = 0.5, p = 0.005). This study provided further information on pathophysiological/morphological mechanisms related to cognitive impairment in ALS and is the basis for further studies in aging-related diseases.
1 Introduction
Stroke is the second most common cause of death in the world. Acute lacunar stroke (ALS) is one of the major causes of disability in the elderly [1]. These strokes can injure the cortex or even deeper brain areas. A stroke in the thalamus, basal ganglia, or pons is called a lacunar stroke, and these areas have a distinct set of arteries that can become easily blocked causing significant infirmities. Due to the characteristics of these blood vessels, lacunar strokes occur differently from other strokes. Hypertension and atherosclerosis can result in lacunar strokes by directly damaging these arteries. Lacunar strokes can be identified by computed tomography (CT) or magnetic resonance imaging (MRI) brain scans (https://www.health.harvard.edu/a_to_z/lacunar-stroke-a-to-z).
Furthermore, researchers pointed out that 44%–74% of patients have some form of cognitive dysfunction following stroke [2]. Individuals with ALS have a higher risk of developing vascular cognitive impairment and dementia associated with functional brain reorganization [3, 4] leading to various cognitive deficits, difficult treatment/recovery [5], and poor social/physical outcomes. Most importantly, despite their high prevalence and negative consequences, cognitive deficits are relatively neglected in comparison to the treatment of post-stroke physical disabilities.
Anatomical damage in the brain was believed to affect corresponding cognitive functions, leading to behavioral impairments. Stroke also alters functional and physiological parts of brain regions distant from the site of anatomical damage (see the review by Carrera and Tononi, 2014 [6]). For example, some morphological findings in MRI include areas of infarction, white matter hyperintensities (WMH), and cerebral atrophy, leading to the development of vascular cognitive impairment [7–12]. Atrophy in the cerebrum has been associated with cognitive impairment following an episode of stroke, where gray matter (GM) atrophy was correlated with the severity of dementia [13]. Neuroimaging is thus crucial in collecting data about the disorder’s neuroanatomical substrates and playing an important role in the diagnosis and management of the disease [14]. Hence, it is important to study GM density alterations during the acute stage of stroke to improve the quality of life in patients and prevent the progression of mild ALS symptoms to vascular dementia in future studies [15].
Neuropsychological testing revealed that stroke has a significant impact on various cognitive abilities. Decreased GM volume was mostly detected in the thalamus with minor reductions found in the cingulate gyrus and frontal, temporal, parietal, and occipital lobes [15]. More specifically, functional neuroimaging [16] and noninvasive brain stimulation (NIBS) studies [17, 18] emphasized the role of the lateral prefrontal cortex (PFC) in cognitive functions such as retrieval of information from episodic memory. Simons and Spiers (2003) reviewed the importance of the interactions between the PFC and other brain regions during cognitive control processes such as selection, engagement, monitoring, and inhibition [19]. Epidemiological evidence indicated that minor strokes in the PFC detected by CT can result in cognitive impairments in vascular dementia subjects [20]. As a result, we aimed to investigate whether or not certain brain regions, such as the PFC, had altered GM density in mild ALS. We further explored the relationship between structural deficits and cognitive deficits in these subjects. To test our hypothesis, we focused on patients with ALS. This specific group of patients commonly displays deficient attention/executive function, but also shows varying degrees of impairment in other cognitive domains, including memory, language, and visuo-spatial function [1, 21]; the exact mechanism of associated morphological alterations remains unknown. Voxel-based morphometry (VBM) analysis [22] was used to evaluate the alterations in GM density [23–27]. Mini-Mental State Examination (MMSE) was used to evaluate cognitive function.
2 Materials and methods
All research procedures were approved by the Institutional Review Board of the First Affiliated Hospital of the Medical College and were conducted in accordance with the ethical standards as laid down in the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards. Informed consent was obtained from all individual participants included in the study. The datasets generated during and/or analyzed during the current study are not publicly available because this is a pilot study and we are still conducting further research on them, but are available from the corresponding author on reasonable request.
2.1 Participants
A group of 29 patients (14 males, and 15 females) were diagnosed with ALS for the first time based on clinical presentation, with proximal anterior circulation occlusion within 12 hours of mild symptom onset, physical examination, and MRI test at the intensive care unit of the Department of Neurology between May 2014 and November 2015. Patient’s average age was 57.6 ± 0.21 years. Most of the patients suffered from hypertension and were taking medications for their high blood pressure. No other underlying conditions were found in the ALS group.
The inclusion criteria were as follows: (1) presented with non-disabling ischemic stroke and complaint of mild symptom onset > 4.5 hours or received intravenous recombinant tissue-type plasminogen activator given within 4.5 hours, which didn't have any effect; (2) had anterior circulation proximal occlusion or thrombolysis scaled 0 to 1 based on time-of-flight magnetic resonance angiography; and (3) underwent 3T MRI < 12 hours after onset. In this study, MRI evidence for a lacunar infarct was defined as “an area of abnormal signal intensity in a vascular distribution that lacked mass effect” [28]. Non-hemorrhagic infarcts in cortical gray matter and deep structures were hyperintense in T2-weighted images than in normal gray matter. Non-hemorrhagic infarcts in white matter (WM) were similarly defined, except that they were hypointense on T1-weighted images, thus distinguishing them from diffuse WM disease. When evaluating WMH by an experienced neuroradiologist, focal abnormalities such as infarctions with low T1-weighted signal intensity, were ignored because abnormalities < 3 mm could not be reliably detected, so all infarcts in these analyses had to be ≥ 3 mm. Lacunes were defined as subcortical infarcts 3 to 20 mm in size [29].
Hemorrhagic lesions had a heterogeneous increased signal on T1-weighted images, heterogeneous decreased signal on T2-weighted images, or both. WM changes were graded in a standard fashion on a 9-point scale as described by Manolio et al. (1994) [30]. The reference standards are briefly described as follows: no white matter signal abnormalities (grade 0); discontinuous periventricular rim or minimal “dots” of subcortical white matter (grade 1); thin continuous periventricular rim or few patches of subcortical WMH (grade 2); thicker continuous periventricular rim with scattered patches of subcortical WMH (grade 3); thicker shaggier periventricular rim with mild subcortical WMH, which may have minimal confluent periventricular lesions (grade 4); mild periventricular confluence surrounding the frontal and occipital horns (grade 5); moderate periventricular confluence surrounding the frontal and occipital horns (grade 6); periventricular confluence with moderate involvement of the centrum semiovale (grade 7); periventricular confluence involving most of the centrum semiovale (grade 8); and all white matter involved (grade 9) [30].
The exclusion criteria included: (1) individuals with impaired consciousness, language, cognition and memory, or history of organic encephalopathy; (2) patients with partial clinical data; (3) subjects diagnosed with ischemic heart diseases, severe hepatic and renal dysfunction, acute or chronic infectious diseases, other stress-related diseases, thyroid dysfunction, or type II diabetes mellitus; and (4) those with ALS caused by two or more problems including arteritis, drugs, tumors, and hematological system diseases, or unknown reasons.
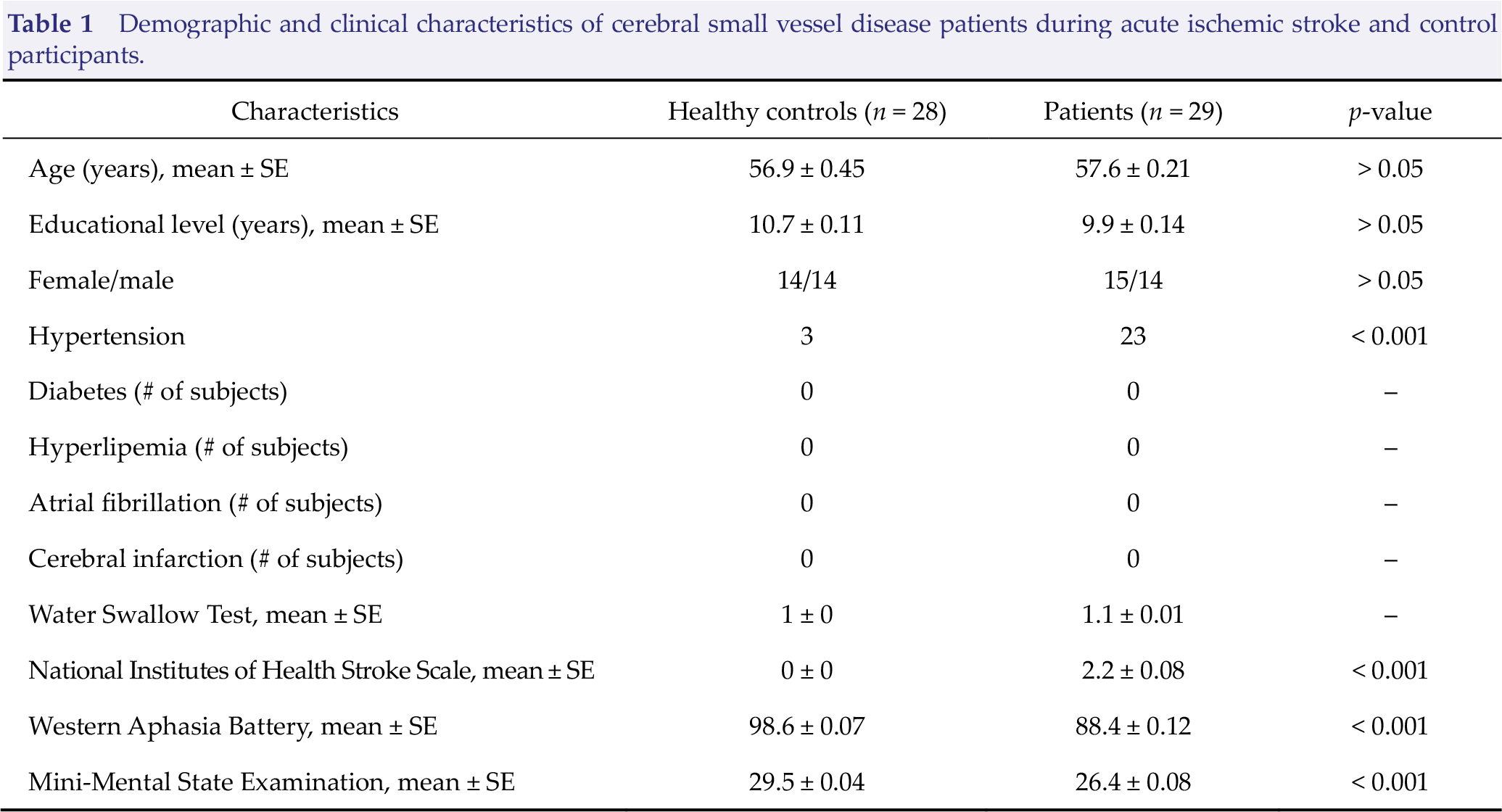
In addition, 28 healthy controls were also recruited and underwent thorough physical examinations simultaneously (average age: 56.9 ± 0.45 years). All of the participants recorded their medical history conditions such as diabetes, hypertension, hyperlipidemia, atrial fibrillation, and cerebral infarction. All subjects were given the Western Aphasia Battery (WAB) and the composite indices were extracted as the aphasia quotient to assess the severity of aphasia [31]. Swallowing function was evaluated by the Water Swallow Test. The National Institutes of Health Stroke Scale (NIHSS) and MMSE were also done. This is all described in Table 1.
Demographic and clinical characteristics of cerebral small vessel disease patients during acute ischemic stroke and control participants.
2.2 Data acquisition
This experiment was carried out in a 3.0-Tesla Signa GE scanner (GE Healthcare, Milwaukee, WI) with an 8-channel phase array head coil. For each subject, a high-resolution structural image was acquired by using a three-dimensional MRI sequence with a voxel size of 1 × 1 × 1mm3 using an axial fast spoiled gradient recalled (FSGPR) sequence with the following parameters: repetition time (TR) = 1,900 ms; echo time (TE) = 2.26 ms; data matrix = 256 × 256; field of view (FOV) = 256 × 256 mm2; slice thickness = 1 mm; no slice gap; number of slices = 140.
2.3 Imaging data processing
Two professional radiologists examined all subjects’ structural images to exclude the possibility of clinically silent lesions. Structural data were analyzed with the FSL-VBM protocol with the FMRIB Software Library (FSL) 4.1 software (http://fsl.fmrib.ox.ac.uk/fsl). First, the brain extracting tool (BET) was used to extract the whole brain from all T1 images [32]. Second, tissue-type segmentation was carried out using the FMRIB’s automated segmentation tool (FAST) V4.1 [33]. The resulting GM partial volume images were then aligned to MNI152 standard space using the FMRIB’s linear image registration tool (FLIRT) [34, 35], followed by nonlinear registration using FMRIB’s nonlinear image registration tool as optional (FNIRT). Third, the resulting images were averaged to create a study-specific template to which the native GM images were nonlinearly re-registered. The optimized protocol introduced a modulation for the contraction/enlargement caused by the nonlinear component of the transformation: each value of the voxel in the registered GM image was divided by the Jacobian of the warp field. Fourth, the modulated GM images were then smoothed with an isotropic Gaussian kernel with a sigma of 4 mm. Finally, regional alterations in GM were assessed using permutation-based nonparametric testing with 5000 random permutations. Correction for multiple comparisons was carried out using a cluster-based thresholding method, with an initial cluster forming a threshold at t = 2.0.
2.4 Statistical analysis
SPSS (version 22; SPSS, Chicago) software was used for the demographic analysis between patient and control groups. Independent-sample t-test was applied to analyze the demographics, and a Chi-square test was used to compare gender differences. A Pearson correlation coefficient analysis was conducted to determine the correlation between GM density and MMSE scores in the patient group.
3 Results
There were no age, education level, and gender group differences between groups as shown in Table 1. The brain regions that had decreased GM density included the superior frontal gyrus (SFG), anterior cingulate cortex (ACC), inferior frontal gyrus (IFG), insula (INS), superior temporal gyrus (STG), heschl gyrus (HES), middle temporal gyrus (MTG), posterior cingulate cortex (PCC), gyrus rectus (REC), medial frontal gyrus (MFG), hippocampus (HIP), post-central gyrus (POST), paracentral lobule (PCL), and superior middle gyrus (SMG). These are shown in Fig. 1. All results were family-wise error (FEW) corrected for multiple comparisons.
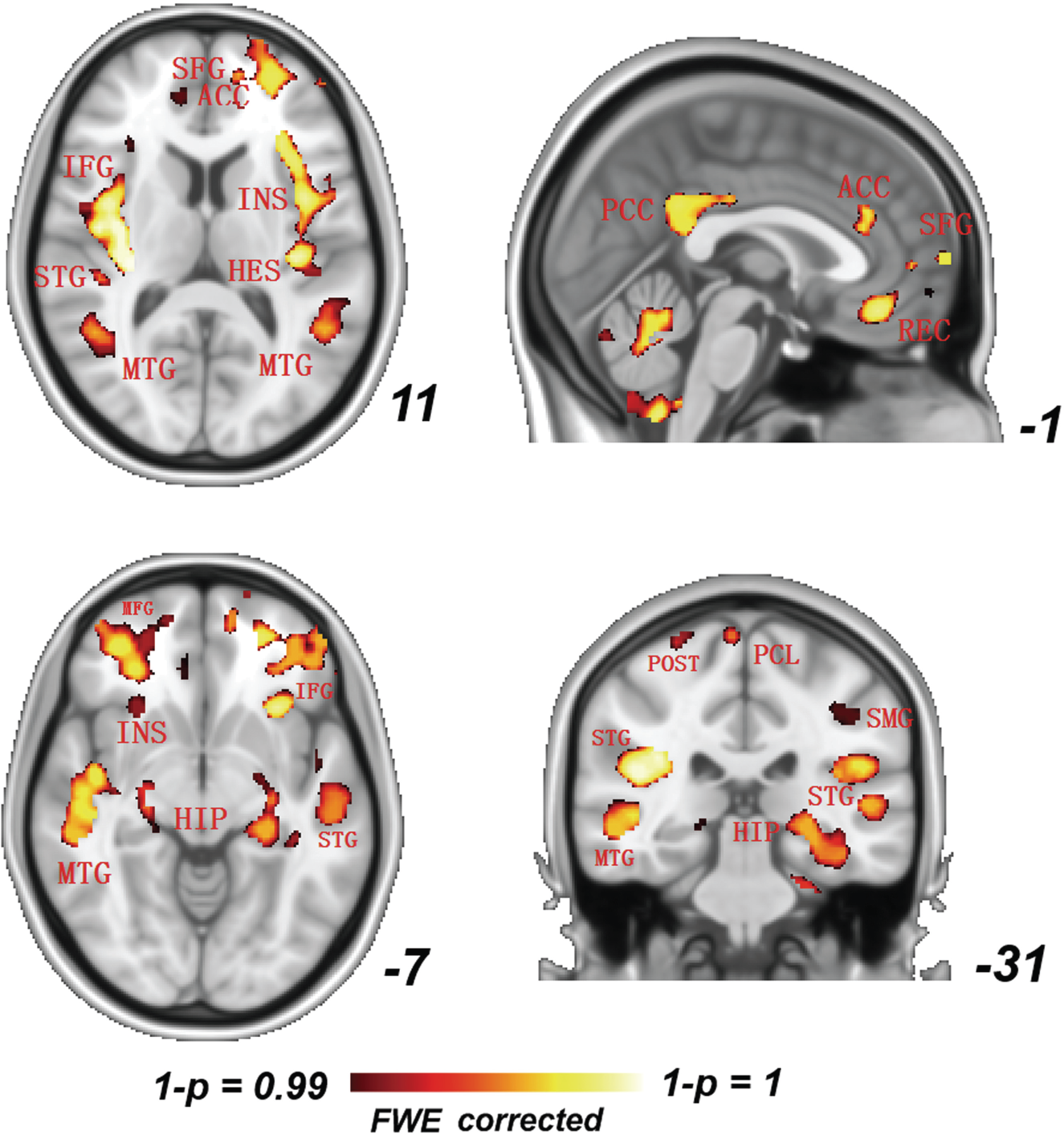
A significant GM reduction (p < 0.05) as assessed by voxel-based morphometry (FWE corrected, p < 0.05) in the ALS group (n = 29, 15 males; average age 57.6 ± 0.21 years) versus healthy controls (HC) (n = 28, 14 males; average age 56.9 ± 0.45 years), especially in the temporo-occipital junction including the superior frontal gyrus (SFG), anterior cingulate cortex (ACC), insula (INS), heschl gyrus (HES), middle frontal gyrus (MFG), superior temporal gyrus (STG), middle temporal gyrus (MTG), posterior cingulate cortex (PCC), gyrus rectus (REC), hippocampus (HIP), post-central gyrus (POST), paracentral lobule (PCL), superior middle gyrus (SMG), and inferior frontal gyrus (IFG). Slices are marked in x, y, z Talairach coordinates.
In patients, we found a positive correlation between GM density and MMSE scores as shown in Fig. 2. The brain regions that were affected included the STG (r = 0.59, p = 0.0007), MFG (r = 0.46, p = 0.01), PCC (r = 0.42, p = 0.02), HIP (r = 0.4, p = 0.03), and mPFC (r = 0.5, p = 0.005). All correlation results were false discovery rate (FDR) corrected. Inter-group comparisons between GM and MMSE scores were not done in the HC group.
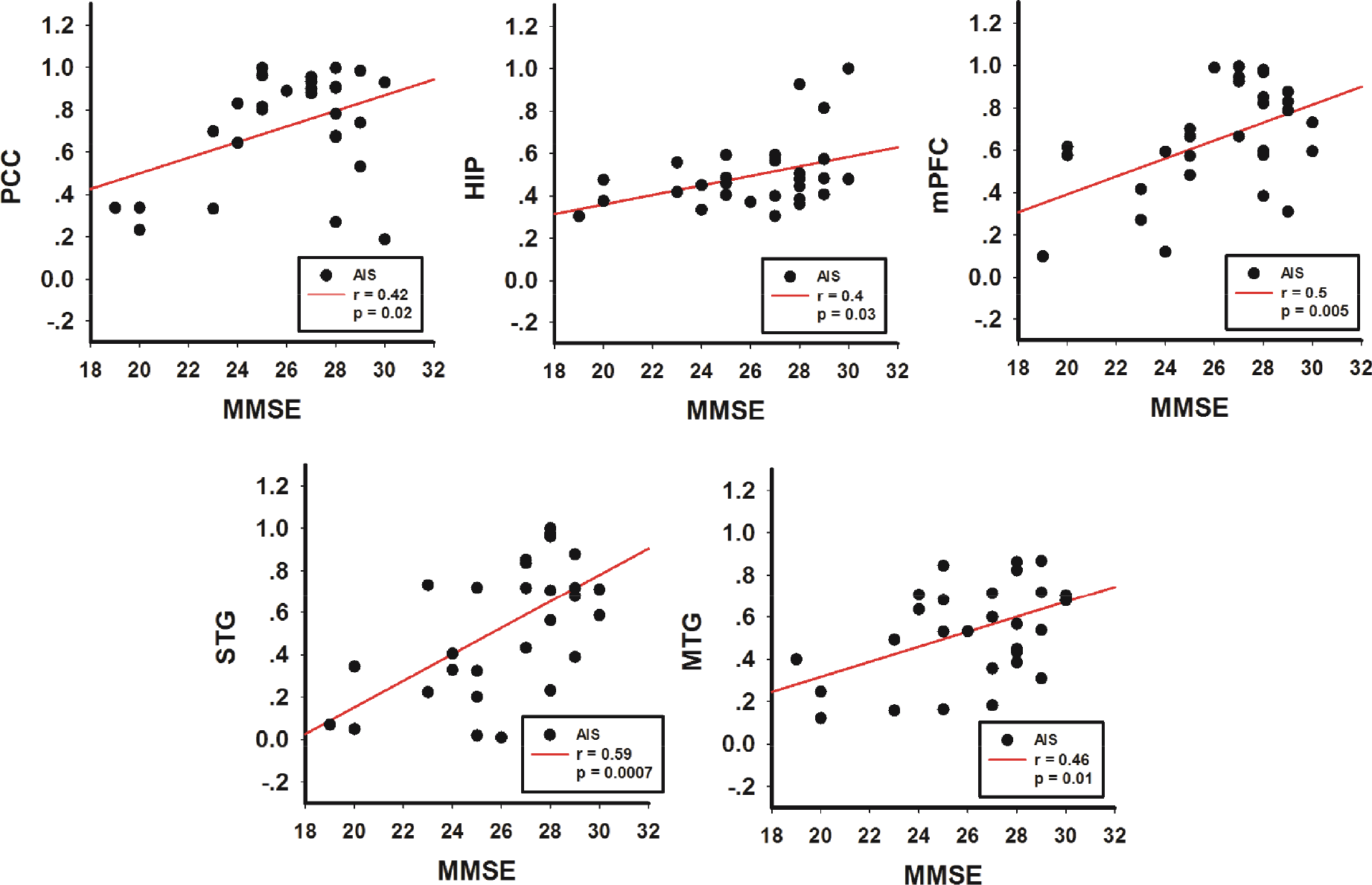
Correlation analysis in the ALS group (n = 29, 15 males; average age 57.6 ± 0.21 years) versus HC group (n = 28, 14 males; average age 56.9 ± 0.45 years) showed a positive correlation between GM density (y-axis) and MMSE scores (x-axis) in ALS in the posterior cingulate cortex (PCC) (r = 0.42, p = 0.02), hippocampus (HIP) (r = 0.4, p = 0.03), medial prefrontal cortex (mPFC) (r = 0.5, p = 0.005), superior temporal gyrus (STG) (r = 0.59, p = 0.0007), and middle temporal gyrus (MTG) (r = 0.46, p = 0.01) (FDR corrected).
4 Discussion
The primary purpose of this study was to identify the brain regions showing GM atrophy that would affect the cognitive outcome of patients with ALS as well as to better understand the underlying pathophysiology. First of all, patients with ischemic stroke in particular are at an inherent risk for vascular cognitive impairment, which can be mild to severe [ 36 ]. General cerebral atrophy has also been associated with the development of cognitive impairment after stroke. The exact mechanism of this atrophy is not known but may reflect secondary degeneration after ischemic events, as may be the case for patients with ALS. In particular, we believe that the GM abnormalities we found were related to the mild behavioral symptoms seen in this special group of patients. In our study, the vast majority of GM reduction in the ALS group was in the cerebellar, temporal, occipital, frontal, and parietal areas (see Fig. 1 for specific regions). This reduction in GM density reflected group differences in the MMSE (Table 1) which provides a generalized rather than a specific assessment of orientation, attention, working memory, visuo-spatial abilities, language, and declarative memory. Since MMSE scores measure overall cognitive performance [37] rather than specific aspects of cognitive function, we found that altered GM density was correlated with these results, indicating an association with poor cognitive performance in ALS patients (refer to Fig. 2). The MMSE also rates the odds of dementia, level of education, and severity of cognitive impairment [37]. Most importantly, the nature of these relationships suggests that as cerebellar, temporal, occipital, frontal, and parietal lobe integrity is affected, cognitive performance suffers [37]. Therefore, even though the ischemic damage was acute, such an event affected major portions of the brain, as presented in our results.
There were several key neural regions in our work that will be explained in more detail regarding the mechanism of cognitive dysfunction, even though we only had structural information rather than functional data. Based upon our hypothesis, the mPFC rather than the PFC was affected. This meant that a decrease in GM density in the mPFC could be associated with the involvement of mPFC in both memory and decision-making [38], reflecting the poor cognitive function results on the MMSE (Table 1).
The MTG is assumed to perform a key function in emotional recognition and cognitive control, such as attention to emotion, identification, or regulation of emotion [39]. Two previous fMRI studies [40, 41] demonstrated that acute and chronic ischemic stroke patients had altered prefrontal activity, sharing a similar pathophysiology with ALS. The alteration in the prefrontal cortex has been implicated in an altered prefrontal-based attentional network in stroke patients [42].
Similarly, the PCC is involved in visual, sensorimotor, and attentional information [43] and memory retrieval [44, 45], which was affected in our patients taking the MMSE. One research group stated that stroke patients exhibited working memory damage and poor cognitive performance with visual stimuli [46], sharing similar pathophysiology with transient ischemic attack (TIA) and ALS patients. According to the results of the cognitive assessment scores of the MMSE (Table 1), ALS patients performed poorly in verbal and working memory compared with healthy controls (data not shown).
GM atrophy in the bilateral ACC and was thought to reflect abnormal information processing from prior experiences in the form of memories because of ischemia in ALS. The ACC is involved in numerous cognitive processes such as executive, attention, social cognitive, affective, and skeleton-motor functions [47]. Previous functional studies reported that TIA patients can suffer from damage of executive function as well [48, 49], and the ACC is a critical neural substrate for execution control [50]. This region is particularly sensitive to ischemic injury [51]. The results of GM reduction of the ACC (Fig. 1) implicated executive impairments in ALS. Thus, several other studies reported ACC dysfunction in stroke patients [46, 52], which may be a key reason for post-stroke depression or working memory damage in these individuals.
According to Chen et al. (2014), asymptomatic lacunar infarct patients showed more severe GM atrophy in the insula, ACC, caudate, and superior temporal pole than controls [53]. Another group found that vascular mild cognitive impairment subjects exhibited significantly reduced GM volumes [21]. This was predominant in the frontal regions, such as the bilateral dorsolateral prefrontal cortex (DLPFC), the orbital portion of the SFG, and the left supplemental motor area (SMA), as well as the bilateral PCC [21], similarly to our study findings. GM volumes were significantly correlated with performance in the Trail Making Test (part B) in the bilateral DLPFC and PCC, the clock drawing test in the right orbital portion of the SFG, and Rey–Osterrieth complex figure test (CFT)-delayed recall in the right PCC [21]. On the other hand, another study found no independent associations between MRI measures and cognitive assessment [54].
The association with the MMSE, a psychological screening test that is related to cortical neurodegenerative disease such as Alzheimer’s disease [42], is an important finding. However, one study in TIA patients found no correlation between decreased GM and Montreal Cognitive Assessment (MoCA) scores and clinical parameters [55], even though the MoCA is similar to the MMSE. These patients tended to have lower MoCA scores compared to healthy controls, which indicated that the affected group showed poor performance in cognitive function [55], just like in patients with ALS. They also showed widespread morphological atrophy in the default mode network (DMN) despite the absence of a cerebral infarction stating that ischemic injury may induce structural abnormalities and eventually contribute to functional impairments in these patients [55], as seen in the ALS group.
For example, Guo et al. (2014) showed that TIA patients had altered regional spontaneous activities in the frontal cortex, which were positively correlated with cognitive assessment scores [56], as depicted in our results. One possible cause of this cognitive dysfunction was considered to be structural cerebral damage resulting from thromboembolism and cerebral hypoperfusion [57, 58]. These issues may have been caused by pre-existing hypertension, which was also a confounding factor we could not control for in our study. This needs to be investigated further in our continuing research. Furthermore, a Sydney stroke study conducted mainly in stroke/TIA subjects suggested that these patients showed atrophy in the AMY, especially those with cognitive impairment [59]. We did find that the AMY was affected as shown in Fig. 1. Intrinsic activity in the HIP is associated with episodic memory function [60]. Just like in our study for ALS, preferential volume loss was found in the bilateral MFG in TIA patients compared with controls [55]. However, these studies only focused on the structural abnormalities in the local brain areas using an ROI-based method, and alterations in the whole-brain GM density in TIA remain unclear.
Overall, our VBM results demonstrated significant GM volume reductions in patients with ALS with cognitive domain impairment compared with those without cognitive impairment, which were seen with smaller reductions found particularly in the cingulate gyrus. However, we did not see involvement of the thalamus (THAL) in our results even though it is associated with cognitive impairment after ischemic stroke [61]. In fact, thalamic volumes add significant information to the classification of cognitive impairment beyond information provided by demographic, stroke-related, and cognitive performance measures [15]. For example, involvement of the THAL in cognitive disturbances has been proven, resulting in vascular syndromes that cause impairments in sensory function, motor abilities, language, executive function, and long-term memory [61]. Therefore, we are uncertain why the THAL was not damaged in patients with ALS and further investigation needs to be done. In future studies, a larger functional and structural comparison study of different types of strokes and stages (transient, acute, and chronic) should be done to depict a complete neuropsychological explanation of the extent of cognitive impairment using a panel of various evaluations.
5 Limitations
There were several important limitations in this study. Obviously, a larger group size should be used in future experiments. One confounding factor in the study was the fact that 3 normal healthy controls had hypertension. Inter-group comparisons between GM and MMSE scores should also be done in the HC group. Post-stroke cognitive symptoms are difficult to explain via location of the infarction, but depend on impairment of remote cortical regions. A possible explanation for these remote effects can be the disruption of neuronal function at remote cerebral regions belonging to certain functional networks [62–64]. The size and location of the lesion are subjects of discussion in stroke research. However, this was a limitation of this study because we did not delve into this matter, but will do so in future investigations. On the other hand, if the lesion were in a crucial region like the PCC, the consequences would have deleterious effects in the short and long run. In future studies, how ALS affects hemisphere-specific processes such as language can be assessed further with more specialized cognitive tests as well as fMRI tasks.
6 Conclusion
VBM revealed a significant GM density reduction in patients with ALS as seen in TIA [55] and chronic stroke patients [4]. Our correlation results meant that decreased GM density was associated with MMSE scores in patients with ALS. Thus, based on previous research [4, 55, 61], widespread GM reduction due to ischemic injury could have caused structural abnormalities contributing to functional and cognitive impairments in all forms of stroke especially in ALS. As a result, our morphological findings supported and helped define the neurophysiological mechanisms of cognitive impairment in patients with ALS.
Footnotes
Ethical approval
The author asserts that all procedures contributing to this work comply with the ethical standards of the relevant national and institutional committees on human experimentation and with the Helsinki Declaration of 1975, as revised in 2008.
Consent
Informed consent was obtained from all patients.
Conflict of interests
The author declares that she has no conflict of interests to disclose.
