Abstract
Aim
Persistent idiopathic dentoalveolar pain (PIDAP) is a chronic pain syndrome involving central sensitization; however, the underlying brain mechanisms remain poorly understood. This study was conducted using voxel-based morphometry (VBM) analysis in a magnetic resonance imaging (MRI) with the aim of investigating cortical plasticity (brain structural changes) in patients with PIDAP.
Methods
High-resolution T1-weighted MRI data were acquired from 24 patients with PIDAP and 25 age- and sex-matched healthy controls. Before treatment, patients underwent scanning and psychological assessments (Pain Catastrophizing Scale: PCS). VBM analysis was used to assess the between-group difference in gray matter volume.
Results
On VBM analysis, patients with PIDAP, compared with the controls, had significantly reduced gray matter volume in the right primary somatosensory cortex (S1), which corresponds to the oral area in the S1 region. Furthermore, the gray matter volume in this region was negatively correlated with the PCS scores.
Conclusions
These results highlight the association between cortical plasticity in the S1 (which may reflect altered somatosensory processing) and the PCS scores in patients with PIDAP, which suggests the involvement of central mechanisms in PIDAP pathophysiology.
Clinical Trial Registration
UMIN000029226.
This is a visual representation of the abstract.
Keywords
Introduction
Persistent idiopathic dentoalveolar pain (PIDAP), also known as atypical odontalgia, is classified as a nociplastic pain condition.1,2 PIDAP is defined as “persistent unilateral intraoral dentoalveolar pain, rarely occurring in multiple sites, with variable features but recurring daily for more than 2 hours per day for more than 3 months, in the absence of any preceding causative event.” 3 Clinically, PIDAP predominantly affects middle-aged women, and patients frequently experience a delay of several years between symptom onset and treatment initiation.4,5 These patients frequently present with comorbid anxiety and depressive symptoms, which may contribute to pain catastrophization.4,5 Additionally, PIDAP has been reported to coexist with other chronic pain conditions. 5 The absence of randomized controlled trials on PIDAP treatment has hindered the establishment of evidence-based treatment. 6 Currently, tricyclic antidepressants are the main treatment for PIDAP, 7 with amitriptyline being the primary drug of choice. 7 A previous crossover study investigating pharmacotherapy for PIDAP using amitriptyline revealed an improvement of approximately 30 points in the visual analogue scale (VAS) score. 8 Cognitive behavioral therapy, as an adjunct to antidepressant therapy, results in better outcomes than antidepressant therapy alone. 9
Despite the lack of clear evidence of actual or threatened tissue damage that would activate peripheral nociceptors or any indications of disease or lesions in the somatosensory system causing pain, nociplastic pain refers to persistent pain that arises from altered nociception. 10 Moreover, nociplastic pain is possibly associated with psychological factors.11–13 Specifically, several studies have reported a significant association between the Pain Catastrophizing Scale (PCS) scores and nociplastic pain.14,15 Nociceptive conditions are characterized by persistent symptoms that frequently persist for extended periods and, in some instances, may continue throughout the patient's lifetime. 1
Central sensitization may be an underlying cause of nociplastic pain, 10 and is defined as an increased responsiveness of nociceptive neurons in the central nervous system to normal or subthreshold afferent input, which clinically manifests as phenomena such as hyperalgesia and allodynia. Central sensitization is considered a key, albeit not exclusive, pathophysiological mechanism in nociplastic pain, and possibly contributes to the regional or widespread pain distribution and hypersensitivity that are observed in these conditions. 16 PIDAP is considered a nociplastic pain condition mediated by a centralized pathology; however, its pathophysiology remains poorly understood. 17 Although central sensitization is likely involved, other mechanisms, including peripheral sensitization and psychological factors, may also contribute to the clinical presentation of PIDAP and related nociplastic pain conditions. 16
Some studies have shown that increased pain sensitivity in nociplastic pain is associated with changes in pain-related brain regions, such as the insula, somatosensory cortex, and thalamus.18–22 Voxel-based morphometry (VBM) is an imaging technique that is used to assess changes in the brain anatomy. To illustrate these concepts, specific conditions such as fibromyalgia and chronic back pain can be examined, where similar brain changes have been observed. 1 Patients with fibromyalgia exhibit decreased gray matter volume (GMV) in the prefrontal cortex, amygdala, and anterior cingulate cortex. 23 Another study on fibromyalgia showed significantly lower GMV in the cingulate, insular, and medial prefrontal cortices and the parahippocampal gyrus. 24 In patients with chronic back pain, regional decreases in GMV were identified in the bilateral medial prefrontal cortex, extending to the anterior cingulate cortex and right medial prefrontal cortex, and reaching the orbitofrontal cortex. 25 These findings suggest that nociplastic pain induces neuroplastic changes in the brain.
Furthermore, the primary somatosensory cortex (S1) plays a crucial role in sensory processing and pain perception, and changes in S1 have been reported in patients who are experiencing chronic pain.26,27 The insula, cingulate cortex, prefrontal cortex, anterior cingulate cortex, and S1 contribute significantly to pain processing and modulation.23–25 However, compared with other brain regions, the S1 may exhibit a distinct neural activity pattern in response to pain. 26 As PIDAP is specifically localized to the face, 3 it is likely influenced by somatotopy in S1, which is strongly associated with chronic pain conditions.
Despite the classification of PIDAP as a nociplastic pain condition, 28 analogous structural neuroimaging studies have not been conducted yet. Thus, we hypothesized that if prolonged pain is closely associated with brain reorganization, then patients with PIDAP would have GMV alterations in their brains. By elucidating the neurobiological underpinnings of PIDAP, this study aimed to contribute to a better understanding of its pathophysiology and potentially guide the development of more targeted and effective treatment strategies for this challenging orofacial pain disorder.
Methods
General design and participants
Two study groups (patients and controls) were recruited for this study. The patient group consisted of 24 individuals treated for PIDAP at an outpatient orofacial pain clinic at Shimizu Hospital (mean age: 54.7 ± 12.8 years; seven men and 17 women). Data were expressed as the means ± standard deviation, unless otherwise indicated. The average time from PIDAP onset to the initial visit to the outpatient orofacial pain clinic at Shimizu Hospital was 3.82 ± 5.05 years. Before visiting the outpatient orofacial pain clinic at Shimizu Hospital, 22 patients had received treatment at previous clinics, and among them, 17 had undergone invasive treatments (e.g. root canal therapy or tooth extraction). The average pain intensity in the patient group over the past 4 weeks was 6.77 ± 1.90 on an 11-point numerical rating scale (0–10). Comorbidities included insomnia in six patients; asthma and periarthritis of the shoulder in two patients each; and angina pectoris, anxiety disorder, autonomic dysfunction, glaucoma, Hashimoto's thyroiditis, posttraumatic stress disorder, sarcoidosis, and vertigo in one patient each. Pharmacotherapy, primarily amitriptyline, was administered.29,30 Epidemiologically, PIDAP is more common in women, but information regarding the menstrual cycle was not collected in this study. Although pain sensitivity can fluctuate according to the menstrual cycle, PIDAP is defined as a chronic pain condition that persists for at least 3 months, and the pain is continuous and spontaneous, independent of cyclic or functional triggers. 31 Therefore, we considered that menstrual phase data would have limited relevance for the interpretation of our results.
Healthy control participants were recruited through community bulletin boards at local medical institutions from 22 January 2021 to 31 August 2022. Exclusion criteria for controls included chronic pain conditions and any medical conditions that required treatment within the past year. The control group was closely matched to the patient group using 5-year age brackets and sex stratification. The control group comprised 25 healthy volunteers (mean age: 54.1 ± 12.7 years, seven men and 18 women; Table 1). Prior to treatment initiation, the patients underwent magnetic resonance imaging (MRI) and completed a psychological assessment questionnaire.
Characteristics of controls and patients.

The controls were age- and sex-matched healthy individuals. The patients received orally administered amitriptyline. Based on the average pain that was experienced over the past 4 weeks, the average pain intensity was assessed on an 11-point numerical rating scale (0–10).
According to their Edinburgh Handedness Inventory scores, all participants were right-handed. Pain was localized to the left side of the face in eight patients, the right side in two patients, and bilaterally in 14 patients. All participants received monetary compensation for their time. This study was approved by the Ethics Committee of Shimizu Hospital, Shizuoka, Japan (UMIN000029226). All participants completed the Stanford Sleepiness Scale, a seven-grade assessment tool that measures drowsiness. 32 All participants provided written informed consent prior to the MRI scan and completed a psychological questionnaire.
Imaging measurement protocol
All MRI data were acquired using a 1.5-Tesla (T) scanner (Ingenia; Koninklijke Philips, N.V., Amsterdam, The Netherlands) at Shimizu Hospital. Each participant's head was stabilized within a 20-channel phased-array head coil. The total MRI scan time was 20 minutes and 23 seconds.
VBM
A whole-brain high-resolution T1-weighted anatomical MRI scan was performed for each participant using a magnetization-prepared rapid acquisition gradient echo (echo time = 2.28 ms, repetition time = 2500 ms, field of view = 256 mm × 256 mm, flip angle = 9°, matrix size = 1 mm × 1 mm × 1 mm, slice thickness = 1 mm, and slice numbers = 160 coronal slices).
Data processing and statistical analyses
Psychological questionnaire and pain assessment
Prior to treatment, the patients responded to the PCS, and the results were compared between the patient and control groups using a two-tailed two-sample t-test. The statistical threshold for significance was set at p < 0.05.
Pain intensity was evaluated using the VAS.
VBM
Statistical Parametric Mapping 12 (revision 7487; Wellcome Trust Centre for Neuroimaging; www.fil.ion.ucl.ac.uk/spm) was applied.33,34 Structural images were corrected for bias field inhomogeneity and spatially normalized using diffeomorphic anatomical registration through exponentiated Lie algebra on the Montreal Neurological Institute template. The tissues were classified as gray matter, white matter, or cerebrospinal fluid. During modulation, nonlinear deformation was used for normalization, ensuring that the voxel intensities reflected the regional GMVs adjusted for individual brain sizes. Subsequently, the images were smoothed using a Gaussian kernel with a full width at half a maximum of 8 mm. Statistical analyses included correction for total intracranial volume to normalize global brain-volume differences across participants.
After preprocessing the structural images, a whole-brain analysis was conducted. GMV differences between patients and controls were further examined using a two-tailed two-sample t-test. The statistical threshold for significance was set at a p-value of < 0.05, corrected for family wise error (FWE) at the voxel level. In addition, we conducted regression analyses using both PCS and VAS scores with GMV values to ascertain the relationship between psychological factors, pain intensity, and structural changes in the brain. Multiple comparisons were corrected using the Bonferroni method for the two regression analyses (PCS-GMV and VAS-GMV correlations), with a significance threshold set at p < 0.025.
Results
Psychological questionnaire and pain
The pain intensity in patients was 66.0 ± 22.9 on the VAS. The PCS scores were 37.9 ± 7.4 for patients and 19.0 ± 11.4 for controls. A significant difference was found in PCS scores (p < 0.001) between controls and patients (Table 2). Meanwhile, no significant correlation was found between pain intensity and PCS scores [R2 = 0.100, p = 0.132].
Differences between controls and patients.

VAS: Visual Analogue Scale; PCS: Pain Catastrophizing Scale.
Pain was evaluated using the VAS. A significant difference was observed in the PCS scores between controls and patients.
VBM results
Compared with controls, patients showed a significantly smaller GMV in the right primary somatosensory cortex (S1) [top peak = (52, −33, 38); FWE-corrected p = 0.032, voxel level; number of voxels = 4] [controls > patients] (Figure 1). No significant clusters were found in the opposite direction [patients > controls]. In the regression analysis of PCS scores, pain intensity, and GMV values of the S1 cluster in patients and controls, a significant negative correlation was found between PCS scores and GMV values of S1 [R2 = 0.110, p = 0.020] (Figure 2) whereas no significant correlation was observed between the VAS scores and GMV values [R2 = 0.047, p = 0.311].

Patient-specific structural anatomy (control vs. patient). The location of significantly smaller clusters in the right S1 of patients is shown, with the statistical threshold for significant differences set at a family wise error (FWE)-corrected p < 0.05 at the voxel level. S1, primary somatosensory cortex.
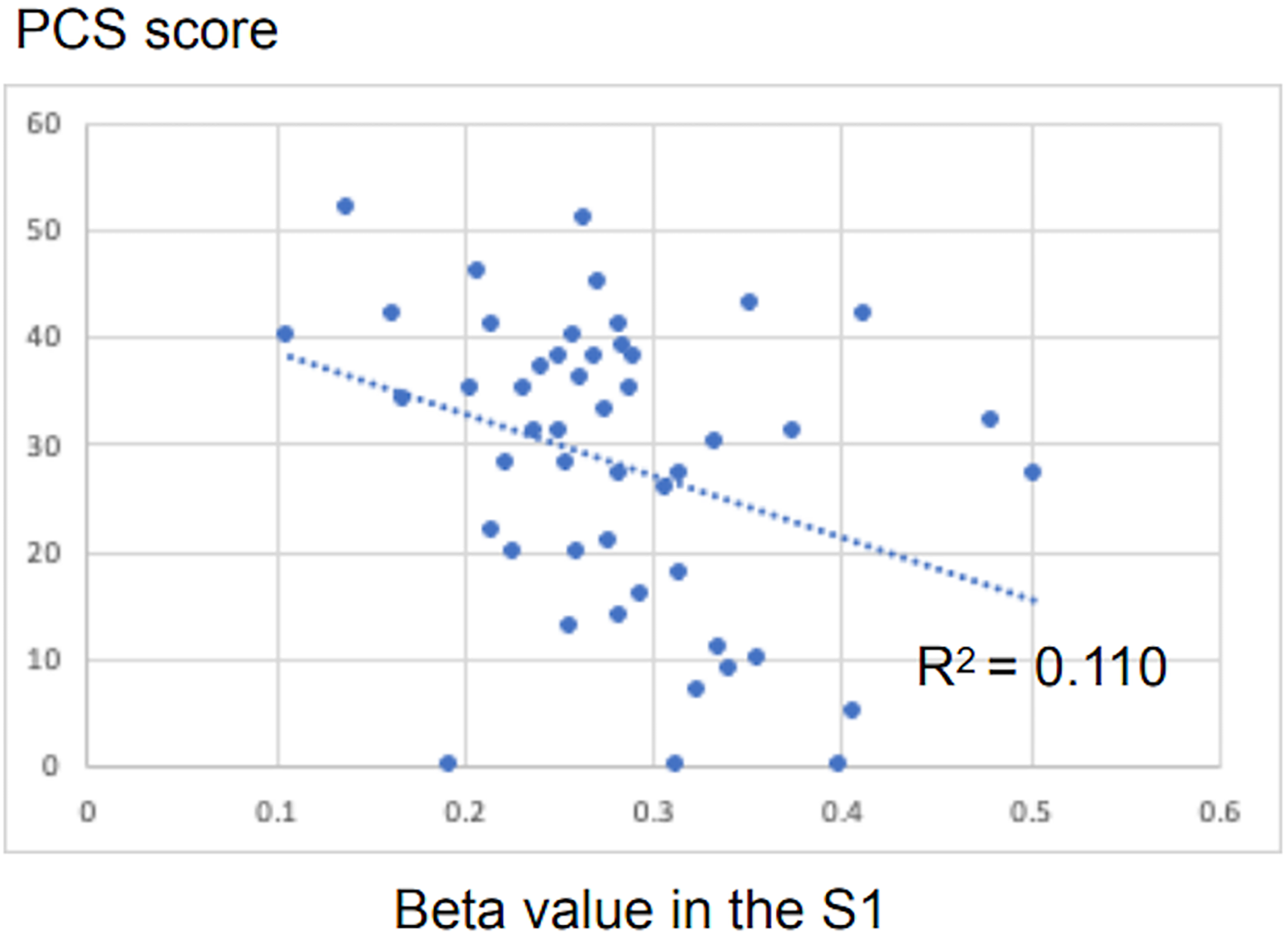
Correlation between PCS scores and GMV values. This scatterplot illustrates a significant negative correlation (R2 = 0.110, [p = 0.020]) between the PCS scores and the mean beta values of the right S1 cluster in patients and controls. S1, primary somatosensory cortex. PCS: Pain Catastrophizing Scale; GMV: Gray Matter Volume.
Discussion
In this study, we investigated brain structural changes in patients with PIDAP using VBM and obtained two main findings. First, patients with PIDAP showed significantly reduced GMV in the right S1 compared with healthy controls. Second, a negative correlation was observed between PCS scores and GMV values in S1.
Our observation of reduced GMV in the S1 of patients with PIDAP aligns with previous research indicating structural brain changes in chronic pain conditions. 35 For example, in unilateral chronic pain caused by herpes simplex infection, the distance between the thumb and little finger representation in S1 is smaller on the painful side than on the healthy hemisphere. 36 In trigeminal neuralgia, thinning of S1 is observed in the facial representation areas. 37 The significant reduction in GMV within the S1 region observed in patients with PIDAP corresponds to the areas representing the face. 38 Furthermore, our study implied that changes in S1 were only observed on the right side, partly because of the location of the symptoms in the present case. Among the patients, eight presented with symptoms on the left side, two on the right side, and 14 on both sides, with a higher number of patients exhibiting left-sided facial pain. Consequently, changes in the right S1 were detected in patients with PIDAP, likely influenced by the higher number of left-sided facial perceptions in these patients. Similar findings have been reported in studies on trigeminal neuralgia, where more significant brain structural changes were noted in the hemisphere opposite to the site of the symptoms. 37
Our findings in patients with PIDAP differ from those observed in other nociplastic pain conditions such as fibromyalgia and chronic back pain. Although a reduction in GMV is typically noted in regions such as the prefrontal cortex, anterior cingulate cortex, and insula in these disorders, localized changes were particularly evident in the right S1 in patients with PIDAP. This specificity underscores the unique pathophysiology of PIDAP as an orofacial pain disorder. The most distinctive feature of PIDAP, compared to other nociplastic pain conditions, is its localization to the orofacial region. The S1 area, representing the orofacial region, occupies a substantial cortical territory.38–40 This extensive representation may account for the structural changes in S1, as observed in PIDAP. The S1 plays a critical role in sensory processing and pain perception, and the structural changes observed in the S1 may have contributed to the altered pain-processing characteristics of PIDAP, such as hyperalgesia and allodynia. These findings are consistent with the concept of central sensitization, which is a key mechanism of nociplastic pain.41,42 The observed GMV reduction in the right S1 (oral somatotopic area) aligns with the imaging findings in other chronic pain conditions, where structural brain changes were consistently reported as a consequence of prolonged nociceptive input.35–38 In PIDAP, the chronic spontaneous pain (persisting ≥ 3 months) may lead to neuroplastic adaptations in S1, reflecting altered somatosensory processing. Furthermore, recent studies suggest that chronic pain induces functional coupling between the somatosensory cortex and the default mode network (DMN), which mediates the embodiment of pain into self-perception. 43 This aberrant S1-DMN connectivity might mediate the structural changes observed in our study, as the brain reorganizes to integrate chronic pain into the self-representation system. Similar to fibromyalgia and chronic back pain, GMV reductions in PIDAP are likely a downstream effect of chronic pain rather than a causative factor. The diagnostic criteria for PIDAP (persistent pain ≥3 months without functional triggers) support this interpretation, as structural changes emerge after prolonged nociceptive input. Similar GMV reductions have been reported in many nociplastic pain conditions, and these changes are a potential consequence of chronic pain rather than a disease-specific biomarker.35–38
The negative correlation between PCS scores and the S1 GMV is a particularly noteworthy finding in our study. Interestingly, this relationship was observed in the absence of a significant correlation between the S1 GMV and pain intensity. The negative correlation between PCS and the S1 GMV has been confirmed in multiple studies.44,45 Additionally, associations between personality traits and chronic pain have been identified. 46 PCS also mediates the relationship between neuroticism and heightened pain sensitivity. 47 This relationship suggests a complex interaction between brain structure, pain perception, and psychological factors in patients with PIDAP. 48 These findings support the biopsychosocial model of chronic pain, emphasizing the need to address both neurobiological and psychological aspects in the management of PIDAP. 48
Our findings have several clinical implications that warrant further discussion. The observed structural changes in the S1 provide a neurobiological basis for the persistent nature of pain in PIDAP, potentially explaining the limited efficacy of peripheral interventions. These results underscore the necessity of centrally acting treatments, such as tricyclic antidepressants, which remain the cornerstone of PIDAP management. 49 The correlation between brain structure and psychological factors emphasizes the potential benefits of integrated treatment approaches that address both the neurobiological and biopsychosocial aspects of PIDAP. 34 This aligns with the results of previous research demonstrating improved outcomes through the combination of pharmacological and cognitive behavioral interventions. 48
Limitations and future directions
This study has some limitations that should be considered when interpreting the results. First, the relatively small sample size may limit the generalizability of our findings. Second, the cross-sectional nature of the GMV analysis prevents drawing definitive conclusions regarding causality, leaving the relationship between observed structural changes and PIDAP unclear. Finally, the use of a 1.5-T MRI scanner may have limited our ability to detect more subtle structural changes in the brain.
To overcome these limitations and further enhance our understanding of PIDAP, future research should focus on longitudinal studies with larger sample sizes that track structural brain changes over time and in response to treatment. 35 Moreover, the use of higher-resolution neuroimaging techniques, such as 3-T or 7-T MRI, could enhance the detection of more subtle structural alterations. Future studies should also investigate changes in functional connectivity along with structural changes to gain a more comprehensive understanding of brain reorganization in patients with PIDAP.
Conclusion
In conclusion, our study is the first to provide evidence of structural brain changes in patients with PIDAP, specifically the reduced GMV in the right S1 region. Furthermore, the GMV in this region was negatively correlated with the PCS scores. These findings contribute to our understanding of the central mechanisms underlying PIDAP and highlight the interplay between brain structure, pain perception, and biopsychosocial factors.35,37 These results have important clinical implications, suggesting that treatment strategies for PIDAP should target both central pain mechanisms and psychological aspects. Our results support the classification of PIDAP as a nociplastic pain condition and emphasize the need for comprehensive multimodal treatment approaches. 37
Clinical implications
Cortical plasticity in the S1 region of patients with PIDAP, associated with PCS scores, suggests that psychological interventions targeting pain catastrophizing may be crucial for effective PIDAP management. S1 plastic changes may reflect altered somatosensory processing, indicating central mechanisms in PIDAP pathophysiology. These findings imply that PIDAP involves nociplastic pain pathology.
Footnotes
Acknowledgements
We extend our gratitude to all participants involved in this study. We sincerely thank Takamori Kouji and Michibata Aya from the Department of Oral Surgery, Shizuoka City Shimizu Hospital, for their valuable suggestions. We are also grateful to Takii Tsuyoshi and Nagai Katsuaki of the Department of Radiation Technology, Shizuoka City Shimizu Hospital, for their technical assistance with magnetic resonance imaging.
Authors’ contributions
TM, OY, and KH analyzed the data and wrote the manuscript. TM, OY, and IM designed and conducted the study. TM, OY, and IM conducted experiments. SS supervised the study and edited the manuscript. All authors contributed to the editorial revisions in the manuscript. All authors have read and approved the final version of the manuscript.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval and consent to participate
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Japan Society for the Promotion of Science KAKENHI [grant numbers 20K15923 and 22K09087].
