Abstract
To provide guidance on the utilization and conservation of the forest defoliator Orgyia ericae parasitoids, we survey its parasitoid species. We identified the developmental stages associated with the different parasitoids, and the emergence pattern of adult parasitoids. Observations of hatching were conducted hourly from 7am to 12pm until all adults had emerged. Pupation time, emergence patterns, and the abundance of Exorista larvarum parasitoids were recorded in detail. The abundance and daily emergence patterns of Bracon (Habrobracon) sp., and Tetrastichus sp. were analyzed. Bracon (Habrobracon) sp. and E. larvarum both had only one emergence peak, whereas Tetrastichus sp. had two. The daily emergence of Bracon (Habrobracon) sp., Tetrastichus sp. and E. larvarum peaked at 3 to 5pm, 3 to 5pm and 7 to 8am, respectively. The E. larvarum pupation distribution was characterized by two peaks.
Introduction
Orgyia ericae (Germar, 1824) is a defoliator moth of the Lymantriidae family (Lepidoptera) (Xu 1980; Wu et al. 1982). It is distributed mainly in Heilongjiang, Jilin, Liaoning, Shaanxi, Gansu, Qinghai, and Shandong provinces, in the autonomous regions of Inner Mongolia and Ningxia in China, and in the former Soviet Union and Europe (Zhao 1978; Xiao 1992; Dapkus 2004, 2010). O. ericae is an important pest of trees. Given the economic and ecological importance of the species it attacks, O. ericae has a major impact in the regions where it is found. In recent years, it has been frequently reported to attack Hedysarum fruticosum Pall. var. mongolicum (Turcz. ex B. Fedtsch., 1902) (Fabaceae), Caragana, Hedysarum scoparium (Fisch. et C. A. Mey., 1841) (Fabaceae), Calligonum mongolicum (Turcz., 1832) (Polygonaceae), Ammopiptanthus mongolicus (Maxim. ex Kom.) (S. H. Cheng, 1959) (Fabaceae) and other desert plants, occurring in a large area of desert shrubbery of regions such as Inner Mongolia, Ningxia and Qinghai. These species are important for desert shrubbery in northwest China, and play a particularly key role in landscape stability and desertification control. They are resistant to drought, sand burial, wind erosion, sand, and barren soil, and grow rapidly. These plants can be used as fodder for livestock (except A. mongolicus), and as ingredients for Chinese medicine, also edible oils can be extracted from seeds of these plants (Liu et al. 1985; Liu et al.1987). Studies of the biological characteristics and life history of O. ericae have shown that O. ericae has two generations per year (Xu 1980; Xiao 1992; Wang & Liu 2002; Wang et al. 2009). Individuals overwinter as eggs in cocoons, which hatch in the middle of May. The newly hatched larvae undergo six instars of four to five days each, usually feeding on leaves. The dispersion of larvae depends on wind. Pupation begins in mid June on the branches of plants, and adults emerge after approximately ten days, in early July. The wingspan is 21-28mm for males, while females are wingless and release sex pheromones that attract mates to cocoons, where eggs are laid. Each female adult can produce, on average, approximately 250 eggs. Males have obvious phototaxis. The second generation of eggs begins to hatch in the middle of July, with pupation beginning in mid to late August and adult emergence in early September. The eggs overwinter in cocoons after mating. Although the biological characteristics and parasitoids of O. ericae (Wang & Liu 2002; Yu et al. 2007; Wang et al. 2008; Sun et al. 2008; Li et al. 2009), and the sex pheromone (Chen et al. 2010; Chen et al. 2011), nuclear polyhedrosis virus and polyhedrin gene sequence of of Orgyia ecricae (Zhang et al. 1991; Dai & Zu 1996; Zu & Dai 1997; Yang et al. 2006), have been intensely studied, there are currently no reports describing the emergence patterns of adult O. ericae parasitoids. To provide guidance on the utilization and conservation for parasitoids of O. ericae, we investigated O. ericae parasitoid species and the emergence pattern of adult parasitoids.
Materials and Methods
A total of 982 cocoons of O. ericae were collected from H. scoparium in Hangjinqi, Erdos, Inner Mongolia, on July 4, 2008. All cocoons were cultured and observed in two loosely airtight containers placed in a rearing room at 24±1°C, 50±10% relative humidity and a photoperiod of 14:10 (L:D).
Observations of O. ericae egg hatching as well as the identification of parasitoid species and measurements of their stage and abundance were conducted hourly from 7am to 12pm from July 7, 2008, until all adults had emerged. In order to represent a proportion of parasitism of all male and female cocoons, all cocoons collected were dissected on August 14, 2008. O. ericae cocoons were also classified with respect to sex, based on their morphological differences. Female cocoons present one mating hole, and differ in length and color: the female pupa is 15mm long and characterized by a yellow and brown color, whereas the male pupa is approximately 8mm long and it is dark brown.
To complete the next stage of their life cycle, the larvae of Exorista larvarum (Linnaeus, 1758) (Tachinidae) (S. M.-L.) must break out of the O. ericae cocoon and pupate. All the pupae of E. larvarum were collected in vitro, and the time of pupation and emergence, as well as the quantity of parasitoids, were recorded. We used LSD multiple comparisons to analyze and identify where differences in the peaks of emergence occurred.
Results
Parasitoid species
Out of the 982 cocoons of O. ericae, 485 male cocoons and 384 female cocoons were parasitized, representing a parasitism of 88.18% and 88.89% for male and female cocoons, respectively. Eight parasitoid species were identified, including the following Hymenoptera, numbers within brackets indicate their abundance: Bracon (Habrobracon) sp. (Braconidae) (174), Itoplectis viduata (Gravenhorst, 1829) (Ichneumonidae) (S. M.-L.) (11), Pimpla disparis (Viereck, 1911) (Ichneumonidae) (S. M.-L.) (3), Eutanyacra picta (Schrank, 1776) (Ichneumonidae) (S. M.-L.) (3), Tetrastichus sp. (Chalcidoidea) (S. M.-L.) (180), Aprostocetus sp. (Chalcidoidea) (S. M.-L.) (1), Brachymeria lasus (Walker, 1842) (Chalcidoidea) (S. M.-L.) (2), and the Diptera Exorista larvarum (Linnaeus, 1758) (Tachinidae) (445).
Adult emergence period of parasitoids
Bracon (Habrobracon) sp. and Tetrastichus sp. were the most frequent Hymenoptera parasitoid species in the sample. By contrast, only 11 adults of I. viduata emerged, and the remaining species of parasitic wasp were represented by three or less individuals. There were 445 adults of E. larvarum in our sample.
For Bracon (Habrobracon) sp. it took 11 days for adults to begin to emerge and emergence occurred from the 7th to the 22nd July (Fig.1). Peak emergence corresponded to 54 adults (31% of the total number of Bracon sp. individuals that emerged). Eclosion took 16 days for Tetrastichus sp. and occurred from the 9th to the 27th July (Fig.1). Emergence in this species was characterized by a bimodal pattern of emergence, with the first peak of emergence being slightly higher than the second. Adults of E. larvarum emerged from mid July to early August (Fig.1). There were three small peaks of emergence concentrated in mid and late July, followed by a low rate of emergence until the 10th August.
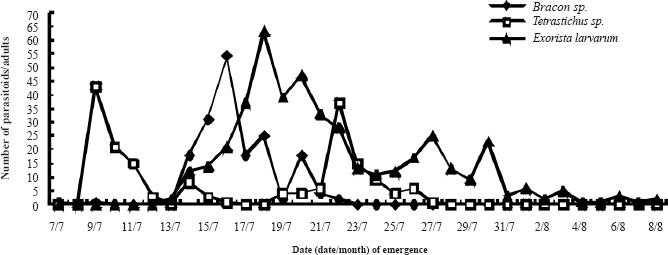
Adult emergence periods of Bracon (Habrobracon) sp., Tetrastichus sp. and E. larvarum adults.
Daily emergence rhythm of parasitoids
The emergence of Bracon (Habrobracon) sp. peaked between 11 and 12am and between 3 and 5pm, with most emergences occurring between 3 and 5pm (Fig. 2). Tetrastichus sp. emerged most frequently from 7am to 8pm, with two emergence peaks at 8 to 10am and 3 to 5pm, with the second peak being slightly higher (Fig. 2). The daily emergence of E. larvarum was characterized by a high peak of emergence from 7am to 8pm and several low peaks of emergence throughout the day (Fig. 2).
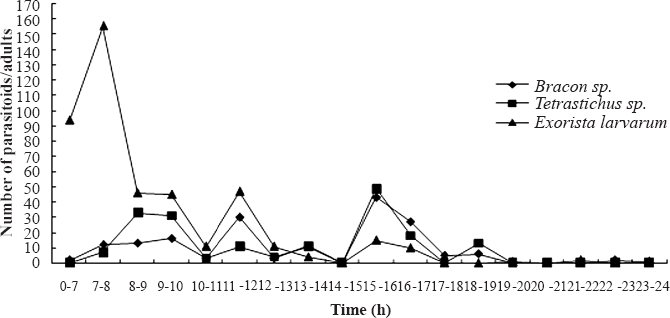
Daily emergence patterns of Bracon (Habrobracon) sp., Tetrastichus sp. and E. larvarum adults.
Pupation range of Exorista larvarum
E. larvarum pupation lasted from 7 to 34 days and two emergence peaks were observed, accounting for respectively 40% (first peak) and 48% (second peak) of all emergences (Fig. 3).
Our results show that the parasitoid species Bracon (Habrobracon) sp., Tetrastichus sp. and E. larvarum emerged at different times in July, with distinctive differences in the timing of their peaks of eclosion. The consecutive peaks of eclosion may be the consequence of long-term co-evolutionary competition processes among these three parasitoid species, which would have resulted in the minimization of inter-species competition and parasitation of the same host at different periods. This conjecture may also explain how these parasitoid populations are maintained in balance.
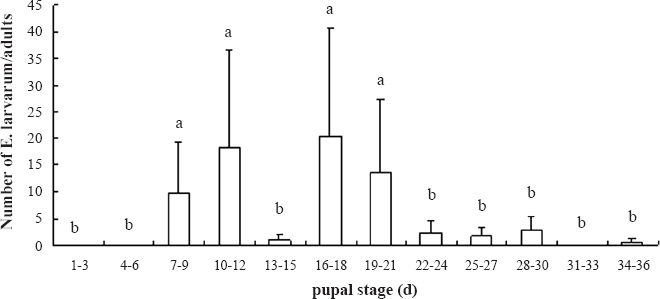
Pupation time of E. larvarum in days (Means ± SE, and the different letters indicate significant differences (P±0.05) based on LSD multiple comparisons).
Although this study has confirmed that the emergence of Tetrastichus sp. adults is characterized by two peaks, it is still not clear whether Tetrastichus sp. is a hyperparasitoid insect. Conversely, a large number of E. larvarum adults emerged between midnight and 7am, but there are no data on the number of eclosions per hour, precluding the determination of specific emergence times. In addition, because the number of individuals belonging to I. viduata, P. disparis, E. picta, Aprostocetus sp. and B. lasus was low, we were unable to determine their patterns of eclosion, an issue that should be addressed in future studies.
Most adults of O. ericae emerge in early July, mate and lay eggs. Newly hatched larvae of the second generation emerged in mid July, and mature larval pupated in mid and late August. Adults emerged in early September. Based on the life cycle and habit of O. ericae, Tetrastichus sp. might parasitize eggs of O. ericae, Bracon (Habrobracon) sp. and E. larvarum might attack larvae of O. ericae. Similar results were reported by Haiyan Li et al. (2009), whose anatomical observations of the parasitoids of egg cocoons of O. ericae showed that each developmental stage of O. ericae is parasitized by a different parasitoid species. These findings could permit the selection of the most appropriate parasitoids according to the developmental stage of the host, as well as the selection of suitable prevention and control periods based on adult emergence patterns of parasitoids and the biological characteristics of the host.
Chemical control and cocoon extirpation are commonly used to prevent and control O. ericae (Yu et al. 2007; Sun et al. 2008; Wang et al. 2009; Luan et al. 2010). However, chemical control and cocoon extirpation would also adversely affect parasitoid population numbers, reducing the effectiveness of this natural control of O. ericae. Therefore, it might be appropriate to reduce these artificial control measures during adult emergence periods of parasitoids. Simultaneously, through investigations of O. ericae parasitoid species and the emergence patterns of adult parasitoids, it might be possible to choose a suitable time for the release of parasitoid species into the field to control population numbers of O. ericae.
Footnotes
Acknowledgments
This research was supported by the Program of the Key Science and Research projects of China's State Forestry Administration (Grant No.2006-46), the Fundamental Research Funds for the Central Universities (BLYX200919) and National Natural Science Foundation of China (30730075). We gratefully acknowledge Professor Wang Yi-Ping from Zhejiang Forestry University for helping us to identify Bracon (Habrobracon) sp. (Braconidae) parasitoids.
