Abstract
The predation and parasitism of Cirrospilus neotropicus (Hymenoptera: Eulophidae) was evaluated on third instar Phyllocnistis citrella (Lepidoptera: Gracillariidae) larvae reared on Citrus limonia under controlled conditions (25 ± 1°C; L:D 12:12 h). Two groups of parasitoid females were evaluated: one in which pupae were collected from citrus orchards (field females) at Eldorado do Sul, RS, Brazil (30°29'S, 51°06′W), and the other from a laboratory colony (laboratory females). After mating, each female was maintained in an individual container and fed honey, pollen and 12 P. citrella every 48 hours. In both groups the behaviors of predation (host feeding), oviposition and superparasitism were observed. The average percent mortality caused by field females (48.71 ± 3.29%) was significantly higher than that caused by laboratory females (35.12 ± 3.45%)(H = 4.74; P = 0.01). Mortality due to predation was 19.15 ± 1.75% in field females and 25.11 ± 3.82% in laboratory females, with no significant difference (H = 1.57; P = 0.21). The average percent parasitism without predation did not differ significantly between field (49.15 ± 2.89%) and laboratory females (42.33 ±4.07%)(H=0.53; P=0.47). Superparasitism represented 6.92 ± 1.21% of the total hosts killed by field females, and 3.51 ± 0.99% by laboratory females. The results show the influence of P. citrella in the death of C. neotropicus caused by parasitism and predation.
Introduction
The citrus leafminer, Phyllocnistis citrella Stainton, 1856 (Lepidoptera: Gracillariidae), is a microlepidopterous species, native from Southeast Asia (Clausen 1931). Since 1993, P. citrella has been rapidly colonizing citrus orchards in diverse regions worldwide (Hoy and Nguyen 1997). In Brazil, it was first recorded in 1996, in the region of Limeira of the state of São Paulo, and in a few months was able to spread to all other citrus growing areas of the country (Cônsoli 2001). The citrus leafminer is considered a menace to citriculture due to the economical losses that it causes by mining the leaves. In addition to the reduction of the photosynthetic area, mining may facilitate the access of the citrus canker bacteria (Chagas and Parra 2000; Ujiye 2000; Chagas et al. 2001).
Among the alternatives to control the leafminer, the release of the exotic parasitoid Ageniaspis citricola Logvinovskaya, 1983 (Hymenoptera: Encyrtidae) has been carried out in Brazil and in various other citrus-growing countries around the world (Neale et al. 1995; Pomerinke and Stansly 1998; Paiva et al. 2000; Urbaneja et al. 2002). Nonetheless, the action of native parasitoids on the leafminer has been verified in diverse citrus growing regions (LaSalle and Peña 1997; Penteado-Dias et al. 1997; Paiva et al. 1998; Sá et al. 1999).
In Brazil, a noteworthy number of indigenous parasitoids were recorded associated with P. citrella. Among these, are the eulophids of the genera Cirrospilus, Elasmus, Sympiesis and Chrysocharis (Jahnke et al. 2005; Efrom 2006). Jahnke et al. (2006) and Efrom (2006), in studies carried out in Montenegro County, Rio Grande do Sul, in a four-year survey, found Cirrospilus neotropicus Diez & Fidalgo, 2003 (Hymenoptera: Eulophidae) as the most frequent species among the native parasitoids. Only the exotic A. citricolla, previously released in a nearby area to the study orchard, was more frequent. The same phenomenon was observed in Argentina, where C. neotropicus kept a parasitism percentage of 19% on P. citrella, only lower than that of A. citricola (54%) (Diez et al. 2000).
The ectoparasitoid C. neotropicus was recorded parasitizing P. citrella practically all over Latin America. However, there are no records of the native hosts of this parasitoid. The first records of this species were done in Cuitlahuac and in Colima, Mexico in 1996 (Bautista-Martinez et al. 1996; Perales-Gutiérrez et al. 1996), but the species was only described in 2003 by Diez and Fidalgo (2003).
Information regarding bionomic aspects of C. neotropicus and its contribution to P. citrella mortality is very scarce. Desirable characteristics of a biocontrol agent are described by Jervis and Kidd (1996) as synchronism with the host and seeking efficiency. According to Garcia (1991), the viability of parasitoid progeny may vary depending on the suitability both of the host of the immature female and of the plant upon the host had developed. Thus, females that developed in orchard plants in the field may present offspring with higher survival rates than those that were grown in artificial conditions. Therefore, this work endeavored to study the behavior of predation and parasitism of C. neotropicus females aiming to gather data to evaluate the potential of this parasitoid as a candidate to biological control agent of P. citrella.
Material and Methods
Individuals of C. neotropicus emerged from pupae and collected from citrus orchards (“field females") located in Eldorado do Sul County (30°29′S, 51°06′W), Rio Grande do Sul State, Brazil, and individuals obtained from the first generation reared in laboratory (laboratory females) were utilized. Soon after emergence each female was transferred to a glass test tube (diameter = 3 cm; length = 10 cm) and kept with five males in a climatic chamber (25 ± 1°C, L:D 12:12 h) for five days to promote mating. Adults were fed with honey and pollen, and water was provided with a wetted cotton placed in the bottom of the glass tube (Urbaneja 2000). Posteriorly, in the same climatic chamber, females were individualized in Plexiglas boxes (12 cm x 12 cm x 3cm) with leaves of Citrus limonia Osb. attached to water-agar (WA) culture medium, to keep them fresh enough during two days. Each leaf had 12 unparasitized third instar larvae of P. citrella, originated from a laboratory culture. At two-day intervals, citrus leaves were replaced with fresh leaves containing 12 third instar larvae. This procedure was repeated until the death of the females. We considered third instar larvae those measuring between 3 to 4 mm in length according to Cônsoli et al. (1996) and Lim and Hoy (2005).
The leaves bearing P. citrella larvae were removed from the Plexiglas box, checked using a stereomicroscope, and the condition of each host larvae recorded after exposition to parasitoids. Leaves with parasitized larvae were kept singly in Petri dishes (diameter = 10 cm; height = 1.5 cm) containing 2% WA medium and maintained in the same climatic chamber. Host larvae without parasitism were removed. The number of larvae killed by host feeding, oviposition or superparasitism were recorded, and the mean percentage at each sampling occasion, was estimated. Parasitism and predation (host feeding) were examined on 23 females of C. neotropicus from the field group, and on 22 females from the laboratory group. The females from the field group were exposed to 2.304 larvae of P. citrella and those from of the laboratory group to 1.752 larvae.
The statistical analysis was performed on the percentage arcsin- transformed data and tested to normality using the Shapiro-Wilk test. The porcentage values of the simple parasitism, superparasitism, and feeding stings were compared by one-way analysis of variance (ANOVA) or by the Kruskal-Wallis test. The association between variables was calculated using Pearson correlation coefficient. The significance level adopted was 0.05. All calculations were performed using the computer programs BioEstat® 4.0 and Microsoft Office Excel® 2000.
Results
C. neotropicus females insert its ovipositor into the body of P. citrella larvae paralyzing it before laying eggs outside its body. Regarding the distance from the host body, it was recorded that 2.56% of the total eggs deposited by females in the field group and 4.47% layed by females in the laboratory group, were located 5 mm further from the body of the host. Females in the field group were resposible for a significantly higher mean percentage of total mortality (48.7 ± 3.3%) than the females from the laboratory group (35.1 ± 3.4%) (H = 4.74; df = 1; P = 0.01). The total mean number of individuals killed by field females was 51.13 ± 7.5, varying from 11 to 120, meanwhile for the laboratory females the average was 26.5 ± 9.4, fluctuating from five to 82, being the former significantly higher (F = 10.45; df =1; P<0.01).
In both groups of parasitoids, host feeding, host feeding + oviposition, only oviposition, and superparasitism were observed. Forty eight hours after the first exposure to the host, 96% of the field females and 100% of the laboratory females had already oviposited. Considering the 1.176 larvae of P. citrella killed by field females, 258 of them suffer host feeding, 397 host feeding + oviposition and 521, only oviposition. In relation with the 583 killed larvae by the laboratory group, 130 suffered host feeding, 191 host feeding + oviposition and 262 only oviposition. There were no significantly differences between the field (31.7± 2.2%) and laboratory female groups (32.6±3.4%) regarding the host feeding and parasitism rates (H = 0.08, df = 1, P = 0.77). The mean percentage of hosts killed by host feeding only, also did not differ significantly between field (19.5 ± 1.7%), and laboratory females (25.1 ± 3.8%) (H = 1.57; df = 1; P = 0.21) (Table 1).
The mean percentage of killed hosts by parasitization only did not differed between the field (49.2 ± 2.9%) and the laboratory groups (42.3 ± 4.1%) (H = 0.53; df = 1; P = 0.47). A difference was found between the percentage of host larvae showing host feeding and a single egg (H = 7.36; df = 1; P < 0.01) and the percentage of those with host feeding and two eggs (H = 5.79; df = 1; P < 0.01) (Table 1).
There was no significant difference in the mortality of host larvae, (H = 0.52; df = 1; P = 0.47) with feeding stings, between the field (50.9 ± 2.9%) and the laboratory groups (57.7 ± 4.1%). The feeding stings occurred with or without parasitism. The feeding stings, caused by a host feeding, resulted in a darkened coloration in the body of the host at the lesion site, and showed a turgid aspect with a spot and haemolymph exudation. A decreasing number of host feeding associated with female age was recorded for both groups (Fig. 1). There was no difference in the percentage of total number of larvae killed by parazitation between field females (80.01%) and laboratory females (79.9%) (H = 1.42; df = 1; P = 0.23).
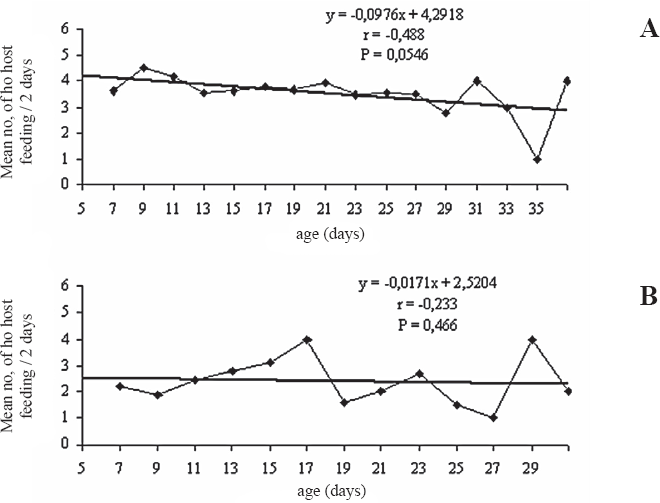
Mean number of Phyllocnistis citrella (Lep.: Gracillariidae) larvae with host feeding performed by females of Cirrospilus neotropicus (Hym.: Eulophidae), after the 5th day of life, recorded at 48 h intervals, (A) field females and (B) laboratory females (25 ± 1 °C; L:D 12:12 h).
All females of the laboratory generation made the first oviposition up the second day of host exposition, and only one from the field generation oviposited after the fourth day. For field females, the number of parasitized hosts decreased as their age increased (r = -0.72; P < 0.01). This decrease was not significant for laboratory females (r = 0.13; P = 0.67) (Fig. 2). The mean percentage of larvae with oviposition only was higher than the superparasitism in both female groups, being 90.6 ± 1.6% for the field females and 85.9 ± 6.4% for the laboratory females (H = 1.79; df = 1; P = 0.18) (Table 1).
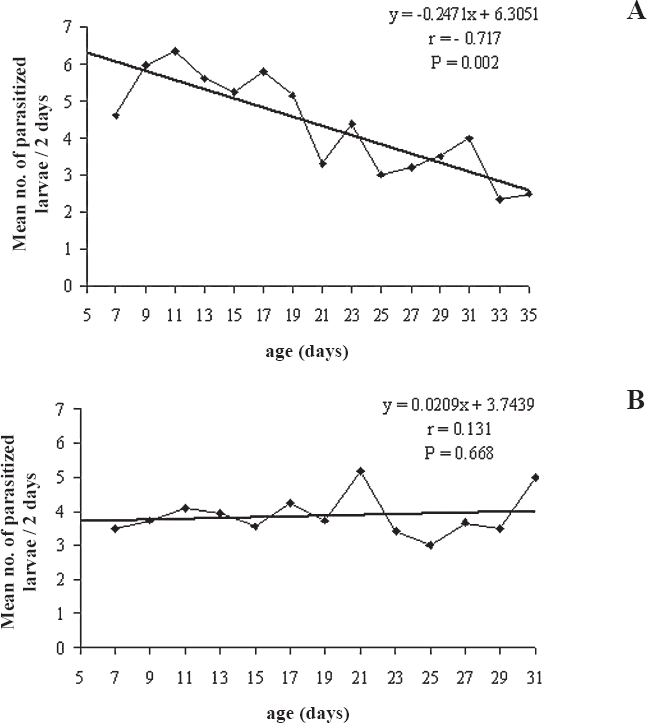
Mean number of Phyllocnistis citrella (Lep.: Gracillariidae) larvae parasitized by females of Cirrospilus neotropicus (Hym.: Eulophidae), after the 5th day of life, recorded at 48 h intervals. (A) field females and, (B) laboratory females (25 ± 1°C; L:D 12:12h).
When superparasitism occurred, more than one larva hatched. Even though, the emergence of a single parasitoid per host was verified. The mean percentage of superparasitism found for field females (6.9 ± 1.2%) was higher (H = 5.2; df = 1; P = 0.02) than of the observed for laboratory females (3.5±0.9%). Total percentage of superparasitism in the field generation was 11.7% and 7.1% in the laboratory colony. Superparasitism was observed in both groups since the first observation, carried out two days after the initial exposure to the hosts. Three days after the first oviposition, the total superparasitism average numbers increased especially in the field group (Fig. 3). After the 25th day of observation, a reduction in both groups was observed. The maximum mean percentage recorded at two days interval represented 20% and 5% of the host total offered, in the field and in the laboratory groups respectively.
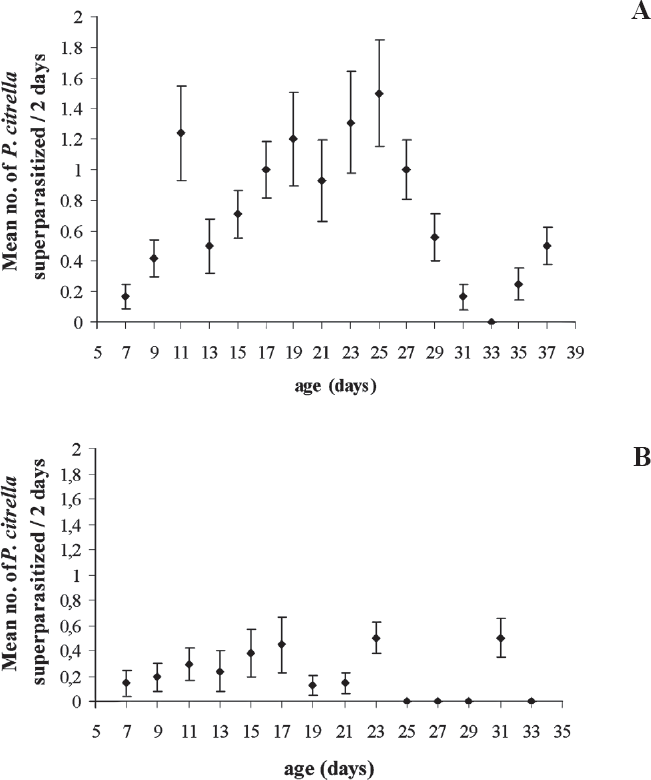
Mean number and standard error of Phyllocnistis citrella (Lep.: Gracillariidae) larvae superparasitized by females of Cirrospilus neotropicus (Hym.: Eulophidae), after the 5th day of life, recorded at 48 h intervals. (A) field females and, (B) laboratory females (25 ± 1°C; L:D 12:12h).
Female host feeding behavior was more frequent than superparasitism. All females from both groups showed host feeding, while only ten females from the laboratory generation and 20 from the field showed superparasitism, laying a minimum of two and a maximum of five eggs per host (Table 1).
There was a significant difference in the total mean number of eggs laid every two days by the field females (5.1) and by the laboratory females (4.0) (H = 11.77, df = 1, P = 0.04). The maximum mean number of eggs deposited by the field females, at each sampling occasion, was 7.7, at the 27th day of life, and 5.3, at the 19th, by the laboratory females (Fig. 4). The number of eggs laid every two days decrease significantly with the ageing of the field females (r = -0.52; P = 0.03), meanwhile, this correlation was not significant for the laboratory females (r = -0.23; P = 0.47) (Fig. 4). The majority of females in both groups oviposited until the last day of their lives, suggesting that there is not a post-oviposition period in this species. The mean longevity was 21.8 days for the field generation, and 16.8 days for the laboratory generation. In the first half of their lives, the field females laid 49.3% of their total egg load. Meanwhile, the laboratory females laid 28.3% in this period, indicating a higher oviposition activity in the second half of their lives.
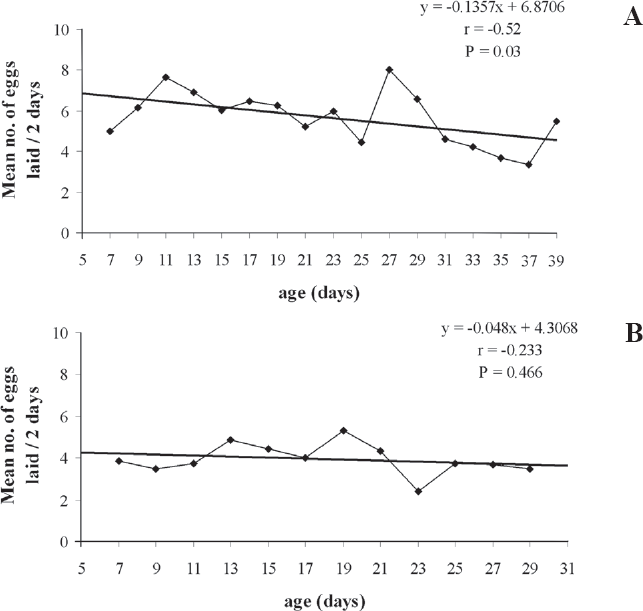
Mean number of eggs laid on Phyllocnistis citrella (Lep.: Gracillariidae) larvae by females of Cirrospilus neotropicus (Hym.: Eulophidae), after the 5th day of life, recorded at 48 h intervals. (A) field females and, (B) laboratory females (25 ± 1°C; L:D 12:12h).
Mean percentage ± SE of different conditions of third instar larvae of Phyllocnistis citrella (Lep.: Gracillariidae) killed by females of Cirrospilus neotropicus (Hym.: Eulophidae) from field (n=23) and laboratory (n=22) (25 ± 1 °C; L:D 12:12 h).
Data within the same row followed by the same letter means that are not significantly different (P = 0.02).
Discussion
Host paralyzation before oviposition, a characteristic of idiobiontic parasitoids, is also listed for other eulophids such as Cirrospilus pictus (Ness, 1834), Cirrospilus diallus (Walker, 1838), Cirrospilus vittatus (Walker, 1838) and Cirrospilus near lyncus (Urbaneja 2000; Urbaneja et al. 2002; Lo Pinto et al. 2005). A percentage of 21.55% eggs of C. pictus and C. diallus, deposited at a distance of 6 mm from the host (P. citrella) was observed by Lo Pinto et al. (2005). Total mortalities similar to these observed for C. neotropicus were found, in laboratory conditions, by Lo Pinto et al. (2005) for C. pictus (30%) and C. diallus (35%) at similar host densities.
Host feeding as a factor of host mortality is a very common behavior in eulophids, as well as in other groups because the body of the host represents a rich protein source, which gives the females energy for oviposition (Christie and Parrela 1987; Ridgway and Mahr 1990; Jervis and Kidd 1996; Urbaneja 2000; Urbaneja et al. 2002; Lim and Hoy 2005; Lo Pinto et al. 2005). The behavior of host feeding may be related to the reproductive strategy of synovigenic, where, at emergence, the eggs are not mature and need proteins from the females to reach a mature stage (Van Driesche and Bellows Jr. 1996).
Ridgway and Mahr (1990) observed that the eulophid Sympiesis marylandensis Girault, 1917, kills its host, Phyllonorycter blancardella (F., 1761) (Lepidoptera: Gracillariidae), by inserting its ovipositor in an exploratory behavior without necessarily laying eggs, but which consequently lead to host paralyzation. According to these authors, the paralyzed larva has a different appearance when compared to that which suffered host feeding: the former showing a yellowish coloration and the latter, a darkened coloration, similar to that observed for P. citrella exposed to C. neotropicus. Thus, it is possible that in the present study, some hosts that were considered killed by host feeding were indeed killed by paralyzation, considering that no difference was observed between these conditions. Lim and Hoy (2005) mentioned that in experiments where Semiolacher petiolatus Girault, 1917 parasitized P. citrella, some figures attributed to host feeding were, possibly, due to inserting its ovipositor which might lead to overestimate of the former figures.
The recorded decrease in the number of host feeding and in the number of laid eggs, which occurred with female ageing, could be related to the reduction in their energy consumption. Also, it is possible that this reduction was not noticeable in the laboratory females due to their lower mean longevity (16.8 days), when compared to the field females (21.8 days). Therefore, the former would not have demonstrated all their fecundity potential. The solitary parasitism, recorded in a greater percentage relative to other behaviors, may be beneficial to idiobiontic and solitary parasitoids such as C. neotropicus. According to Quicke (1997), Jervis and Kidd (1996) and Godfray (1994) the solitary parasitism is advantageous to this group because usually either the female parasitoid kills the host when ovipositing or the parasitoid larva does it soon after eclosion. Therefore, the immature individual has a limited resource to its development, which may not allow for the successful development of another individual. This does not occur in koinobiont parasitoids, because the host remains alive and growing. Solitary parasitoids demonstrate a remarkable intra-specific competition, eliminating the resource competitors in the same host (Jervis and Kidd 1996). In the present study, the emergence of a single individual, from larvae superparasitized, supports the data of Urbaneja (2000) that did not verify the survival of a second parasitoid in the same host, in the case of C. near lyncus. In both cases, probably occurred competition for resources and cannibalism. Cannibalism is a very common behavior in solitary parasitoids (Viggiani 1964; Urbaneja 2000; Urbaneja et al. 2002; Goubault et al. 2003; Lim and Hoy 2005; Lo Pinto et al. 2005). On the other hand, Urbaneja et al. (2002) observed, for C. vittatus, that in conditions of superparasitism, up to four eggs per P. citrella larva, there was the emergence of more than one parasitoid individual per host.
For the eulophid S. petiolatus superparasitism in P. citrella larvae was observed in citrus orchards in Sicily, Italy (Rizzo and Mineo 2000). Lim and Hoy (2005) verified this behavior for this same species, in laboratory conditions, when 30 third instar larvae of P. citrella per day were offered to each female. According to these authors, the confinement in laboratory conditions would have stimulated superparasitism in S. petiolatus. Superparasitism, when carried out by a single female, is advantageous in assuring its progeny survival and avoiding the parasitization of the same host larva by other female parasitoids (Godfray 1994). Nevertheless, this author points out that this behavior may be a consequence of female inability to distinguish already parasitized larvae. Percentages of parasitism higher than the recorded in the present study were found by different authors: Urbaneja (2000), in laboratory conditions (25°C; 70% RH; L:D 16:8 h), recorded 34.3% of total parasitism of C. near to lyncus on P. citrella larvae; Urbaneja et al. (2002), 42.8% for C. vitatus; and Lo Pinto et al. (2005), 27% for C. dialus and 37.2% for C. pictus.
Host feeding had also been recorded by other authors more frequently than superparasitism. Lo Pinto et al. (2005) in controlled conditions (26 ± 1°C; 65 ± 5% RH; L:D 16:8 h) recorded for C. diallus and C. pictus that about 70% of the mortality was caused by stings. Urbaneja (2000), in studies on C. near to lyncus, at 25°C, observed the following mean percentages: 47.78 ± 7.87 of total parasitism, 21.0 ± 5.7 of host feeding and 68.1 ± 9.3 of total mortality (host feeding % + parasitism %), with a high frequency of parasitism behavior. The eulophid ectoparasitoid, Diglyphus begini (Ashmead, 1904), caused mortality of Liriomyza huidobrensis (Blanchard, 1926) (Diptera: Agromyzidae), either by parasitism or by predation, in commercial cultures of ornamental plants and in green houses, in Colombia, totalizing 40% of host predation (Cure and Cantor 2003). The predation activity of various eulophids, in laboratory conditions, has been frequently recorded (Urbaneja 2000; Urbaneja et al. 2002; Lim and Hoy 2005; Lo Pinto et al. 2005). However, in field conditions, this behavior had not been recorded for Eulophidae in the literature.
The field females, which went through their larval phase in hosts that have developed in adult plants, probably larger than plants kept in laboratory, might have originated more vigorous individuals, which would explain the higher mean number of eggs recorded for this group. Urbaneja (2000) observed in C. near lyncus females, at 25°C, that the number of eggs laid increased until the 18th day, similar to that recorded for C. neotropicus in the laboratory generation. Nevertheless, the author found a higher mean value (10.5 eggs/day) and a steep decrease after this period. Urbaneja et al. (2002), studying C. vittatus, observed that the oviposition pattern was dependent on female age thus, supporting what was observed in the field females of C. neotropicus. Urbaneja et al. (2002) verified that all females began oviposition until the second day of life. From the sixth until the 16th day, females produced a mean oviposition rate of 5 eggs/day, and they kept laying eggs until the last day of their lives. The present study allowed us to demonstrate that females of C. neotropicus of both groups were able to kill P. citrella larvae, either by parasitism or predation.
Footnotes
Acknowledgments
To Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) for fellowships conceded to first, second and last authors.
