Abstract
The digestive fluid of the coffee berry borer, Hypothenemus hampei, larvae contains aspartic proteases, which show greatest activity at pH 3,0 and a temperature of 40°C. By using isoelectric focusing gels, it was possible to observe two well-defined bands in the intestinal tract. The band of highest activity was found very close to the anodic side and showed a pI of 5,2. The addition of 0,1 µM of Pepstatin A caused a 70% inhibition of protease activity and a 90% inhibition when the concentration was 1µM. The strong inhibition of the protease activity in the intestinal tract of H. hampei after addition of Pepstatin A, allowed us to identify this protease as an aspartic-type protease. Other results permitted us to classify this protease as Cathespin D given that when BSA was used as a substrate the enzyme only showed 20% of the total activity with respect to hemoglobin. Incubation of the αAI inhibitor from Phaseolus vulgaris with increasing amounts of this intestinal enzyme from the coffee berry borer, showed that the biological activity of the inhibitor was significantly reduced due to the proteolytic activity. This work constitutes the first electrophoretic evidence of αAI proteolysis of P. vulgaris by endogenous proteases from H. hampei.
Introduction
The coffee berry borer, Hypothenemus hampei (Ferrari) (Coleoptera: Curculionidae: Scolytinae) is not only a major pest in Colombia and other countries, but also a serious economic pest of coffee in many coffee-growing areas of the world (Murphy and Moore 1990; Reid 1983). Insects, as well as other organisms, use proteinase for digestion of proteins that are ingested with their food (Xu and Qin 1994). Usually, the proteinase type is determined by the pH range of activity, by its similarity to well-characterized proteinases and by its sensitivity or susceptibility to different inhibitors (North 1982). Several insect proteases have already been described, isolated and characterized, some of which occur as mixtures of different isozymes, and which are generally similar in their catalytic activity to those found in vertebrates (Blanco et al. 1996; Applebaum 1985). However, some proteinases present major differences in their more important characteristics such as: location, optimal pH activity, kinetic constants and thermostability (Blanco et al. 1996).
Most of the midgut proteolytic enzymes in coleopteran insects have been shown to be cysteine proteinases (Murdock et al. 1987), or aspartic proteinases often found in combination with other proteinases and which are very active in acid environments, pH normally below 4,5 (Jongsma and Bolter 1997).
Characterizing the major protease activity present in the digestive tract of H. hampei larvae is important not only to understand the protein digestion, but also to know how susceptible to digestion the αA1 from Phaseolus vulgaris is. Interest in this area has been stimulated by the finding that insect digestive proteases have potential to digest αAmylase inhibitors and to increase by this way their natural resistance to these plant inhibitors (Ishimoto and Chrispeels 1996).
This paper describes the use of specific and general substrates, with a specific inhibitor, to determine the type of protease in the intestinal tract of larvae of H. hampei and to assess the role of this protease in the αAI digestion. We believe the present paper to be not only the first report but also the first electrophoretic evidence by using zimograms of αAI proteolysis with endogenous aspartic proteases from H. hampei.
Materials and Methods
Sources of plant inhibitors and insects
Hypothenemus hampei was obtained locally in Colombia and cultivated in an insectary at Cenicafé in Chinchiná, Colombia. P. vulgaris cv. L. radical is a local commercial bean in Manizales, Colombia.
Extraction procedures of insect proteases
Fifty insect guts carefully obtained by dissection of whole coffee berry borer larvae were homogenized in 50 µl of citrate buffer 0,05M pH 3,0, filtrated and centrifuged at 10.000g for 30 min at 4°C. The supernatant was freeze-dried and the resulting powder was stored at -80°C and used as a source of enzyme.
Purification of αAI-1
Seeds (20 g) of P. vulgaris were ground in a coffee grinder and the powder mixed with 250 ml of distilled water and stirred in the cold room from 6h. The slurry was centrifuged for 30 min at 8000g and the pH of the supernatant was adjusted to 4,0 with 1 N HCl. The precipitated proteins were removed by centrifugation (30 min at 8000g) and the pH of the supernatant adjusted to pH 6,7 with 1 M KPO4. If there was a precipitate it was removed again by centrifugation. The proteins in the clear supernatant were precipitated by the gradual addition of saturated ammonium sulfate (4,1M) to a final concentration of 85% saturation.
After standing on ice for 4 h, the proteins were collected by centrifugation for 30 min at 8000g. The pellet was resuspended in water, dialyzed against water for 36 h and then against 20 mM KPO4 pH 6,7 for at least 12 h. The protein solution was loaded on a DEAE cellulose column (120 x 16 mm) and the column washed with 200 ml of the same phosphate buffer. The column was eluted with a linear NaCl gradient (0 to 100 mM) and absorbance at 280 nm and αAI activity were measured in the eluted fractions.
Protein determination
Protein was determined by the Coomassie procedure (Bradford 1976). Bovine serum albumin was used as standard protein.
Electrophoretic separation and enzyme zymogram in isoelectrofocusing (IEF gels)
For all gels we used the PhastSystem electrophoresis unit (Pharmacia) following the manufacturer instructions. The isoelectric point was calculated by using standard proteins with known isoelectric points and with software (ImageMaster VDS) provided by Pharmacia Biotech. For the protease zymogram, the proteins were first separated in nondenaturing conditions on IEF (3,0-9,0) Phastgels. After electrophoresis, the IEF gel was lightly rinsed with distilled water and incubated for 3 hours at 35°C with Citrate buffer 0,05M pH 3,0 containing 1% of hemoglobin. The gel was rinsed with water and stained with Coomassie Blue (Electrophoresis solution) during 30 min at 45°C and cleaned with water / methanol / acetic acid (60:30:10) solution. Protease activity appears on the polyacrylamide gel as clear bands on a blue-colored background. The gel was washed once again with water and then photographed.
Electrophoretic separation and inhibitor zymogram in isoelectrofocusing (IEF)
For the inhibitor zymograms, the IEF gels were incubated in 1,5% soluble starch in 10 mM sodium citrate, pH 5,0, containing 10 mM NaCl and 20 mM CaCl2 for 1h at 4°C. The starch-gel was rinsed with water and incubated for 20 min at 30°C, rinsed and incubated with human salivary amylase dissolved in the same buffer. Staining with KI/I2 (I2: 0,5% and KI: 5%) produced dark blue bands on a light background (Valencia et al. 2000).
Proteolytic activity
Total proteolytic activity was assayed according to the method of Lenney (1975) and Blanco Labra et al (1996). The bovine hemoglobin was prepared in water and denaturalized at pH 1.0 with a HCl solution 6,0N. This solution was dissolved with Citrate buffer pH 3,0 in order to get a 0,083% hemoglobin final solution. To carry out the activity tests, 40 µl of the enzyme solution was incubated for 3 hours at 30°C with 300 µl of Citrate buffer (0,05M pH 3,0) and 1,3 ml of hemoglobin solution (0,083%). The reaction was stopped by the addition of 2,5 ml of Trichloroacetic acid solution 0,34N. Then, samples were put in ice for 10 minutes and centrifuged at 10000g for 10 minutes. One enzymatic activity unit was defined as a decrement of 0,001 in absorbance at 280 nm. The absorbance was determined by using a UV/VIS spectrophotometer (UNICAM).
Proteolytic inhibition by pepstatin
Proteolytic inhibition was evaluated following preincubation of the enzyme preparation with pepstatin A, a synthetic inhibitor. This specific inhibitor was added to enzyme preparation at a final concentration of 0,1µM, 0,4µM, and 1µM and allowed to stand at 25°C for 15 min before adding the substrate. Following preincubation the remaining activity was assayed as described above.
Proteolytic digestion of αAI
Proteolytic digestion of αAI (Phaseolus vulgaris) by acidic proteases present in the digestive fluid of H. hampei, was assayed as follows: An aliquot (1 µl) of the pure αAI (2 μg/µl) was incubated for two hours at 30°C with 1 µl of crude enzyme extract containing 3,92, 1,96, 0,98 or 0,0 enzymatic activity units. The reaction was stopped by adding Pepstatin A to this reaction solution at a final concentration of 0,1 µM. 1 µl of this solution containing the αAI was separated by using an isoelectrofocusing gel (IEF 3-9). Inhibitor zymogram was developing as described above.
Results and Discussion
Characteristics of intestinal tract proteases
In preliminary experiments we determined some biochemical properties of the digestive proteases of the coffee berry borer larvae. We found, that the enzyme has an optimum pH between 2,5 and 3,5 and it holds an 80% of their enzymatic activity when it is heated up to 50°C. In addition, when different buffer solutions were assayed, it was found that 0,05 M Citrate buffer, pH 3,0, was the best buffer solution to assay the proteolytic activity (Data not shown). Similar to most insect proteases, the enzyme activity found in coffee berry borer has similar values to those registered previously for other Coleoptera insects. For example, Tribolium castaneum (Herbst) shows an optimum proteolytic activity at pH between 3,0 and 6,9 (Blanco et al. 1996; Murdock et al. 1984; Birk et al. 1962); between 3,3 and 6,0 for Callosobruchus maculatus (F.) (Silva y Filho 1991; Murdock et al. 1984; Kitch y Murdock 1986; Gatehouse et al. 1985; Campos et al. 1989); between 5,5 and 6,5 for Acanthoscelides obtectus Say (Wieman y Nielsen 1988); and between pH 3,5 and 5,5 for Zabrotes subfasciatus (Boh) (Lemos et al. 1990). The highest proteolytic activity founded at pH 3,0 in H. hampei clearly indicates that this enzyme is an acid protease (Aspartic protease).
Similar results have been reported in other Coleoptera insects (Vundla et al. 1992; Barret 1977; Houseman and Downe 1982; Okasha 1968; Smith and Birt 1971).
The gradual loss of proteolytic activity that was observed in H. hampei over 50°C, has also been registered for proteinases coming from other organisms such as Mujil auratus (Bonete et al. 1984), Tribolium castaneum (Blanco et al. 1996), and Trichoplusia ni (Pritchett et al. 1981), indicating its thermal instability.
Electrophoretic zymograms and pI determination
To find out if coffee berry borer insect can synthesize different isoforms of acidic proteases, we analyzed enzymatic extracts from its intestinal tracts on native 16% polyacrylamide gels and develop them as zimograms. We observed two well-defined bands in the intestinal tract of H. hampei larvae (Fig. 1).

Zymogram of proteolytic activity from the digestive tract of H. hampei by using native electrophoresis on 7,5% homogenous Phastgel. After electrophoresis the gel was incubated with 1% hemoglobin solution in buffer Citrate pH 3,0, during 2 h at 35°C.
Similar analysis of the acidic proteases after separation of the proteins present in the intestinal tract on an isoelectric-focusing gels (IEF 3-9) showed the presence of a single major band very close to the anodic side with a pI of 5,2 (Fig. 2). Again, this result with H. hampei proteases was in general agreement with those reported for other insect acidic proteases, most of which fall in the acidic region of the IEF gel. For example, the two-reported aspartic proteases of Tribolium castaneum had isoelectric points between 2,8 and 3,5, respectively (Blanco et al. 1996; Barret 1977). The presence of this enzyme in the intestinal lumen of the insect shows the digestive nature of the enzyme.
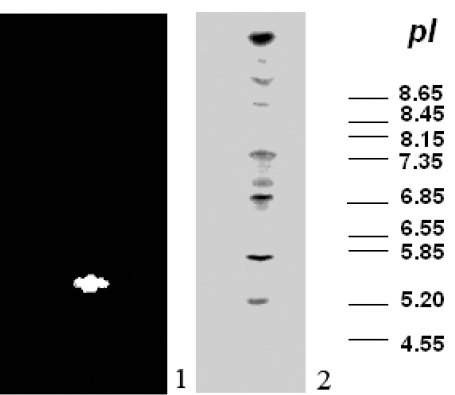
Determination of the pI of the major aspartic protease from intestinal tract of H. hampei by using an IEF Phastgel (3-9). Lane 1, represents the major aspartic protease H. hampei larvae. Lane 2, displays the pI markers.
Enzymatic inhibition by pepstatin A
Pepstatin A is a potent inhibitor of aspartic proteases and we prove its effect on H. hampei acidic proteases. So, to determine if the enzyme activity that is present in the digestive tract of coffee berry borer, could be inhibited by Pepstatin A, we assayed an intestinal extract for protease activity after incubation with increasing amounts of Pepstatin A. The results (Fig. 3) showed that addition of 0,1µM of Pepstatin A caused a 72% inhibition of protease activity and a 91% inhibition when 1µM of inhibitor was assayed. The strong inhibition caused on the protease activity in H. hampei after addition of Pepstatin A, allow us to conclude that the endogenous proteolytic activity in the intestinal tract of coffee berry borer is aspartic protease type.
In order to determine if the insect protease was Pepsin or Cathepsin D type, we assayed the enzyme activity by using hemoglobin and bovine serum albumin (BSA) as two different substrates. The results showed that this proteolytic enzyme hydrolyze hemoglobin more efficiently than BSA. Only 20% of the total activity on hemoglobin was found when BSA was used as substrate (data not shown).
According to this, it was possible to classify this enzyme as a protease cathepsin D type. The results of our study are consistent with the identification of a cathepsin protease activity in intestinal tracts of Tribolium species (Blanco et al. 1996).
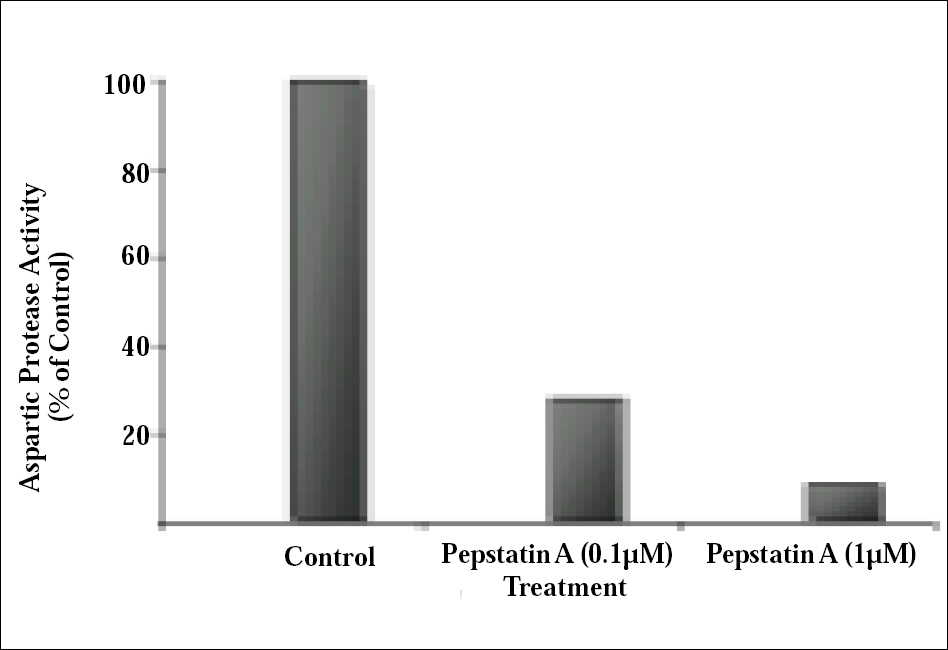
Effect of Pepstatin A on aspartic protease activity of coffee berry borer (H. hampei) gut extract. Data are presented as percentage of control containing no inhibitor.
Proteolysis of αAI by larval proteases
To determine if the ΑΙ-1 could be digested by the acidic proteases found in the intestinal tract of coffee berry borer, we assayed an exact quantity of αAI-1 for inhibitory activity after incubation with increasing amounts of acidic protease activity. The results (Fig. 4) showed that the inhibitory activity of αAI was significantly reduced when the inhibitor was incubated with acidic proteases from the intestinal tract of H. hampei. 3,92 units/µl of aspartic proteases from coffee berry borer nearly destroyed the total biological activity of the αAI from P. vulgaris after two hours of incubation. Previous works of proteolysis of αAI by larval enzymes, showed that Z. subfasciatus larval enzymes can cleave αAI-1 to smaller molecules (Ishimoto and Kitamura 1992). Ishimoto and Chrispeels (1996) have reported a similar result when seed extracts of Phaseolus vulgaris were incubated with larval midgut extracts of Z. subfasciatus, Acanthoscelides obtectus, and Callosobruchus chinensis, and the resulting αAI polypeptides were visualized on an immunoblot after SDS-PAGE. Although it is well known that insect gut proteases could digest different proteins that are intake by the insect during feeding, it doesn't exists electrophoretic evidence on this phenomenon when the well-characterized αAI is used as target; especially if the biological activity of the amylase inhibitor is monitored.
Because of this evidence, use of transgenic insect-resistant plant expressing αAI requires not only a complete characterization of the digestive proteases in the insect gut but also, requires to know if the inhibitor can act at the pH found in its digestive tract and, if it is resistant to attack by insect gut proteases.

Proteolytic digestion of αAI-1 (Phaseolus vulgaris c.v. Radical) by H. hampei larval acidic protease(s). Purified αAI-1 was incubated with increasing amounts of larval midgut acidic protease from Coffee berry borer. Lane 1, 3,92 units/µl. Lane 2, 1,96 units/µl. Lane 3, 0,98 units/µl. Lane 4, Control (Untreated AI-1). An aliquot (1 µl) of the digestion solution containing the remaining αΑΙ-1 (1 µg/µl) was loaded in each well and run on an IEF (3-9) gel.
Based on the experiments presented here, we think that in order to increase the protective efficacy and durability of αAI in transgenic plants, it is envisaged that “Packages” of different genes must be introduced into plants. The component of such packages should each act on different targets within the insect. Protease inhibitors should be particularly valuable especially because they would protect other introduced gene products from premature and active digestion in the insect gut.
Conclusions
The results in this paper provide further insight into the relation between a-amylase inhibitors and the digestive proteases that are present in the insect target.
The data presented here lead to three important conclusions. First, the endogenous proteolytic activity in the digestive tract of H. hampei is aspartic protease (Cathepsin D) type. Second, the inhibitory activity of αAI could be significantly reduced when the inhibitor is incubated with acidic proteases from the intestinal tract of coffee berry borer. Third, protease inhibitors should be particularly valuable especially because they would protect other introduced gene products from premature and active digestion in the insect intestinal tract.
Footnotes
Acknowledgements
The authors gratefully acknowledge Colciencias and Cenicafé (Colombia) for financial support. We also thank the Entomology Laboratory of Cenicafé for their assistance with the insects.
