Abstract
Larvae of Simulium (Pternaspatha) limay Wygodzinsky from two creeks from the Lanin National Park, Neuquen in Southwestern of Argentina were cytogenetically examined. No evidence of different cytoforms were observed. Polytene chromosome maps, idiograms, table with similarities of standard and known cytogenetically neotropical species are presented. S. limay could be distinguished from the standard species (Simulium venustum/verecundum) on the basis of different fixed inversions and an interchange of the chromosome arms; the last character is not found in the other neotropical simuliid species.
Introduction
The cytotaxonomy of the black flies from the Neotropical region based on the analysis of chromosomal rearrangements of salivary gland chromosomes is becoming better known. However, most of the literature is restricted to species involved in the transmission of the human onchocerciasis in South as well as in Mesoamerica (Conn 1988, 1990; Conn et al. 1989; Charalambous et al. 1993a, 1993b; Charalambous et al. 1996; Hirai 1985, 1987a, 1987b; Hirai and Uemoto 1983,1984; Hirai et al. 1994; Millest 1992; Shelley et al. 1986). Nevertheless, there are some other chromosomal studies performed in Neotropical Simulium species even though the species analyzed have not been yet reported as vectors of onchocerciasis (Chubareva et al. 1976; Duque et al. 1988; Campos and Muñoz de Hoyos 1990; Campos et al. 1996; Coscaron Arias 1998; Muñoz de Hoyos 1990, 1995).
From the 15 neotropical subgenera that are committed to the genus Simulium (Crosskey y Howard 1997) only the species within to S. (Notolepria), S. (Simulium), S. (Hemicnetha), S. (Chirostilbia), S. (Ectemnaspis) and S. (Trichodagmia)= S. (Grenierella) have been analyzed.
The systematics of the genus Simulium in the Neotropical realm is extremely complex and needs to be clarified (Coscarón 1987). For example among the taxa, S. (Pternaspatha) is a subgenus with subantarctic lineage possessing morphological characters which clearly can be separated from the remaining Neotropical genera (Coscarón and Coscarón Arias 1996) such as male dystistyle shape, different types of pupae trichomes or the great variation on female abdomen ornamentation. Accordingly, a chromosomal analysis of S. (Pternaspatha) could help to clarify the systematics of the genus.
S. (Pt.) limay, from Argentina, is endemic to the Subantarctic and Patagonia areas from the Southeastern Andes (Mendoza to Santa Cruz provinces) (Coscarón and Coscarón Arias 1995, 1996). It is an hematophagous insect but not considered to be a serious pest for men. It breeds in a diversity of habitats from creeks to big rivers in crystalline hard current waters in a cold to temperate area between 400 to 700 meters of altitude. It is sometimes sympatric with S. (P.) nigristrigatum and S. (P.) simile, Gigantodax marginalis, G. fulvescens, G. chilensis and Cnesia dissimilis. Preimaginal stages are found on submergent or fallen tree branches and leaves. According to previous reports, it belongs to nigristrigatum species group, and morphological characteristics admit the identity as a morphoespecies well differentiated (Coscarón and Wygodzinsky 1972; Coscarón 1991; Coscarón and Coscarón Arias 1996).
In the present report S. (Pt.) limay is cytologenetically described. The cytological features of this species could be useful to establish similarities and differences with other Neotropical species, and to infer which are the closest taxa of this subgenus. S. limay is studied from two main locality during different times of the year in order to see if there was a seasonal variation.
Materials and Methods
Larvae from the two last stages were collected from natural substrate. The sampling was done monthly from 1983 to 1984 from the Quitrahue and Chapelco Grande Brooks in the Lanin National Park (Neuquen Province, Argentina) (Fig. 1), in the northern part of the patagonian Andes with elevations ranging from 650 to 2,000 m.
Collection date and number of male and female larvae analized are given in tables 1 and 2.
Collection date of Simulium limay from Chapelco Grande brook.
Collection date of Simulium limay collected in Quitrahue brook.
Taxonomic determination of larval instars was performed following Coscarón (1989).
After collection, larvae were fixed in absolute ethanol: acetic acid (3:1), and stored at 4° C until dissection under stereoscopic microscope. Chromosomal spreads were obtained using the Feulgen method described in detail elsewhere (Rothfels and Dunbar 1953; Rothfels et al. 1978). Slides were examined with an Olympus microscope under phase contrast. When necessary, photographs were taken by using Kodak Imagen Link black-and-white film.
From a total of 330 larvae, 184 were females and 146 males, were analyzed (Tables 1-2).
Chromosome nomenclature and mapping conventions observed followed previous recommendations (Gordon 1984; Rothfels and Dunbar 1953; Rothfels et al. 1978). Briefly, the chromosomes were identified using landmarks. They are numbered as I, II and III in decreasing order of size and each arm is S (short) or L (long) depending on their size. Major chromosomal features, namely Ring of Balbiani, para-Balbiani, nucleolar organiser, grey band, shield, frazzle, blister are designated as RB, pB, NO, gB, S, F, and B, respectively (Gordon 1984), are used to identify the chromosome arms. The entire complement is divided into 100 sections. The proportion of the complement assigned to an specific arm is expressed as mean percentage contribution of the arm in the total complement length after chromosomal measuring in at least 10 nuclei. The sections are numbered 1 to 100 beginning in IS and ending at IIIL. The proximal, central, and distal segments of a section are indicated by a, b, and c, respectively. Nomenclature of inversions followed previously established conventions (Basrur 1959; Bedo 1977; Gordon 1984; Rothfels et al. 1978). Mainly, they are indicated by brackets on illustrations or photographs and are numbered arbitrarily starting from one. The fixed (interspecific) inversions are designated by underlining the inversion number. Floating inversions are designated by an inversion number not underlining. Sex-linked polymorphisms were analyzed by comparing the frequencies of inversions in males and females.
The standard banding pattern for comparison with S. (Pt.) limay was Simulium venustum/verecundum (Rothfels et al. 1978). This species was chosen as it has the standard map for the subgenus Simulium and it is one of the species of Simulium genus better cytologically studied and frequently used for comparison in the New World.
Results
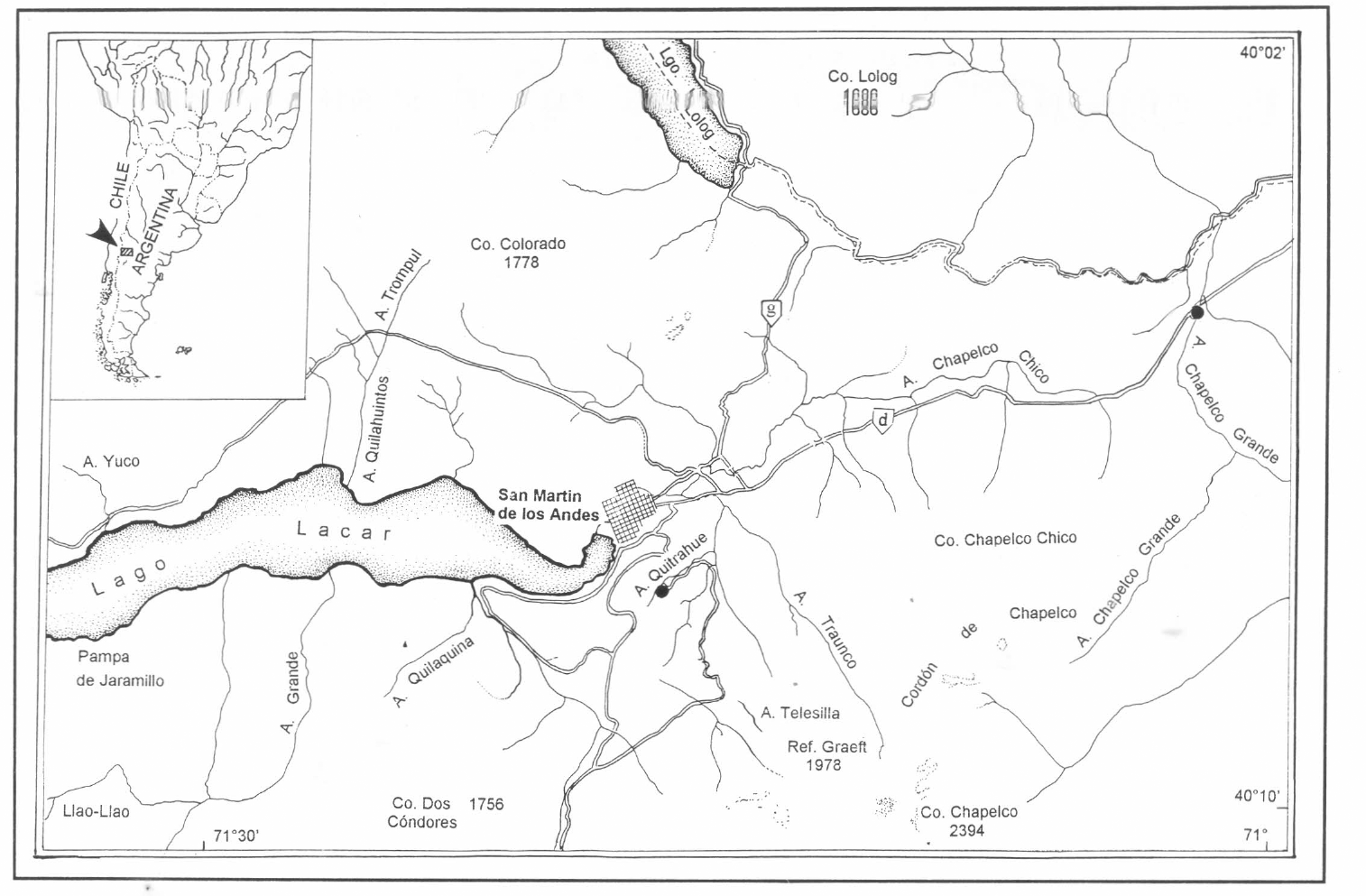
Quitrahue and Chapelco Grande Brooks (Lanín National Park, Neuquen, Argentina).
Description of S. limay (Figs. 2-4)
The arms of the chromosomes were identified using the landmarks of S. venustum/verecundum (Fig. 2).
Simulium limay shows an interchange of the whole arm between chromosomes II and III. The arms are associated as shown: IIIL-IIL; IIIS-IIS. Because of this arm-association, the "chromosomes" show great difference in size with "chromosome III" being very small because it is composed of two short arms: IIS+IIIS (Fig. 3).
No inversions or polymorphisms differentiating the sexes were observed. No cytological differences were observed during the different seasons of the year.
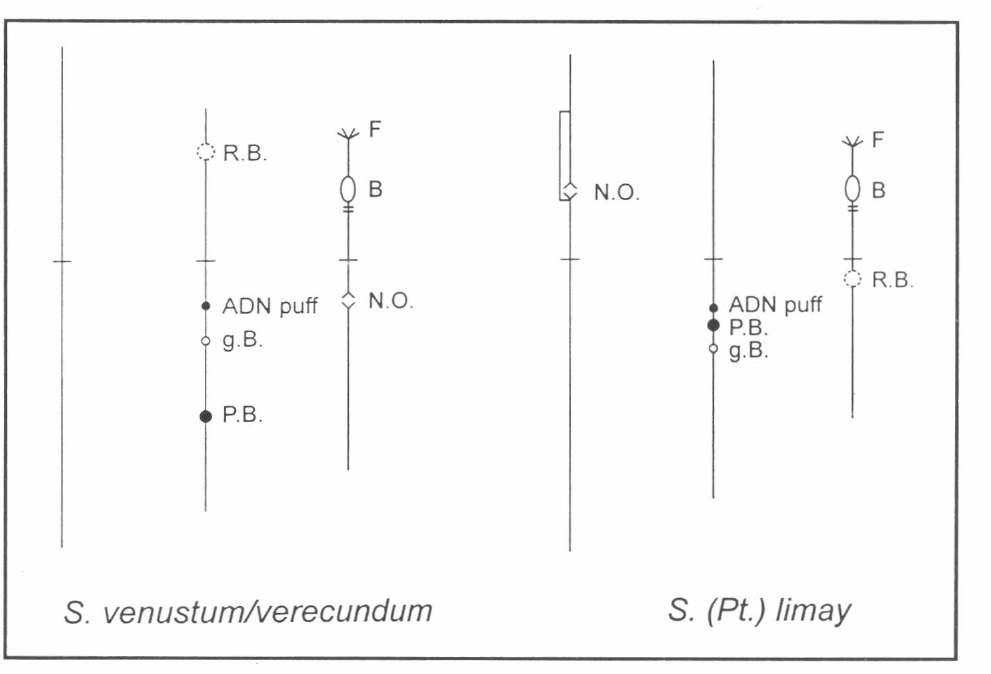
Idiograms of Simulium venustum/verecundum and S. limay. Interspecific inversions shown by brackets to the left of chromosome arms. Abreviations: RB, ring of Balbiani; pB, parabalbiani; NO, nucleolar organizer; gB, grey band; F, frazzled; B, blister; ADN puff; C, centromere.
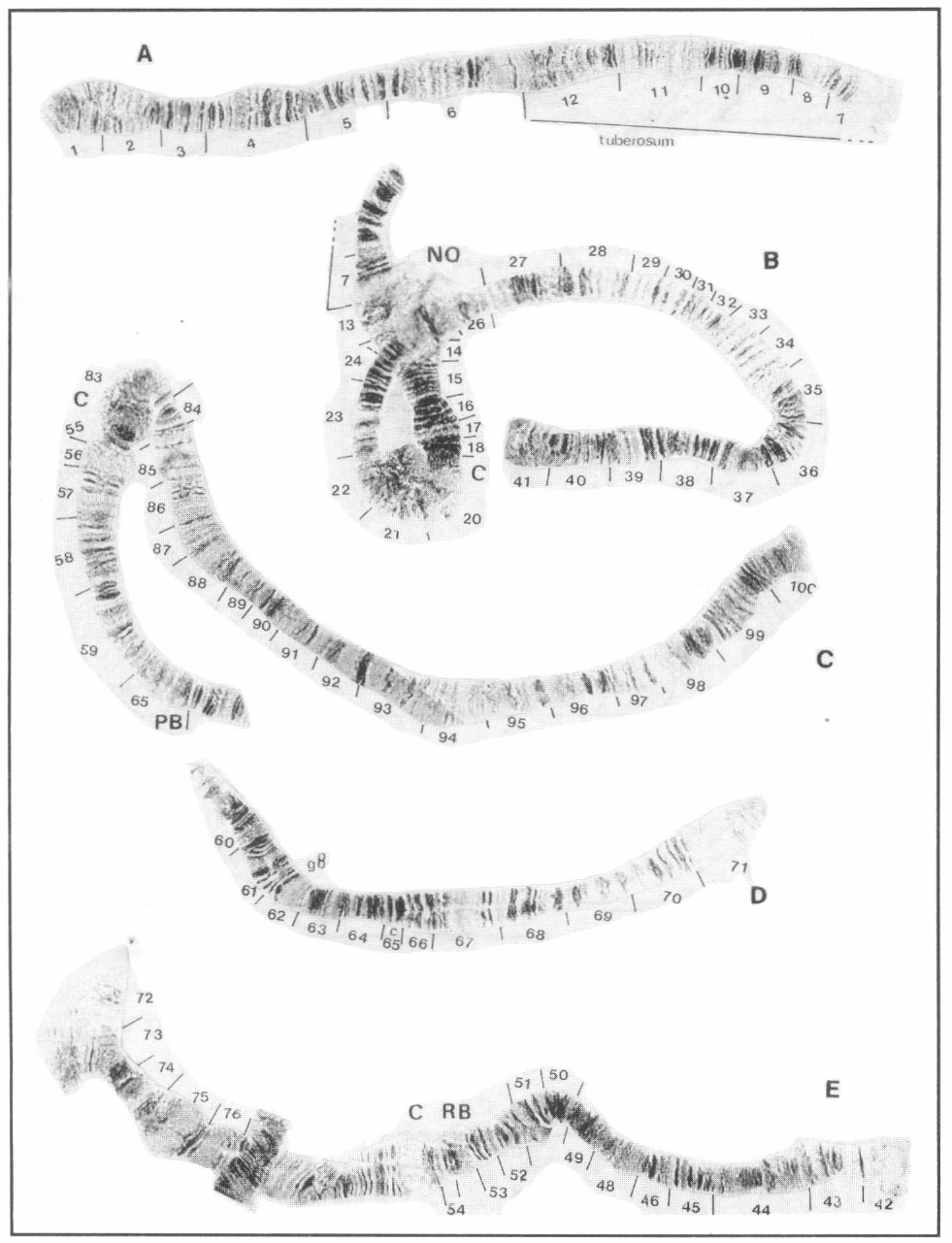
S. limay A: chromosome IS (part), B: chromosome IS (part) and chromosome IL, C: "chromosome II" (IIIL + IIL), D: "chromosome III" (part IIL), E: "chromosome III" (IIIS + IIS). Abbreviations: RB, ring of Balbiani; pB, parabalbiani; NO, nucleolar organizer; C, centromere; c, distal portion of subsection.
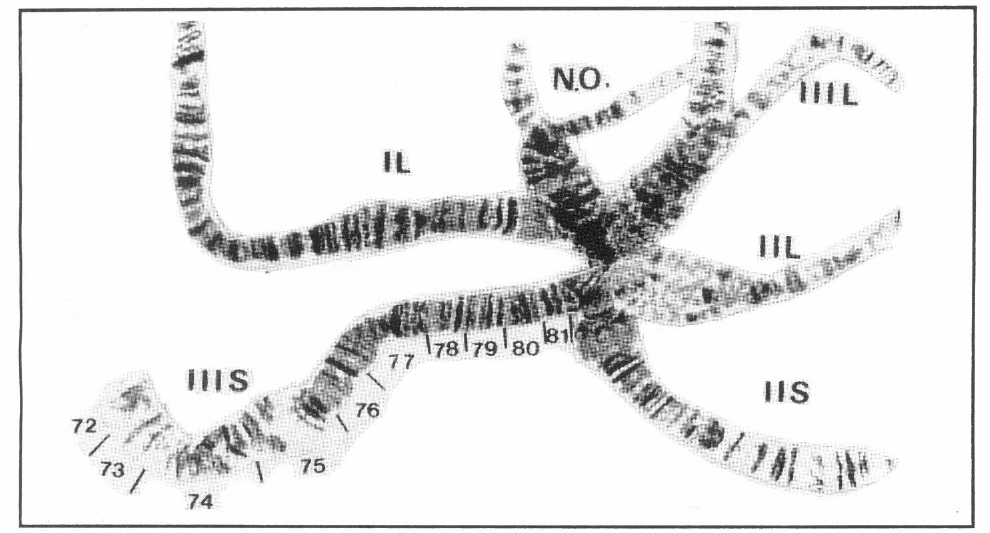
S. limay general view of the chromosomes illustrating chromosome IIIS.
Discussion
The results demonstrate that the appearance of the centromere, the beginning of IS, IL and IIIL, the frazzled end of IIIS of S. limay are chromosome features shared with S. venustum/verecundum. As it is the "A" ending for IS stated by Landau (1953) for some species of Simulium like Simulium damnossum and Eusimulium and also present in Gymnopais, Twinnia, Prosimulium spp., etc (Rothfels et al. 1978).
The unique inversion observed (IS-
On the other hand, several characters of S. limay have been observed to differ from S. venustum/verecundum. Among them the arm interchange between chromosome II and III, producing a small chromosome, the presence of N.O. in chromosome I (the change of position of the N.O. is common to other members of the family (Procunier et al. 1985), the interspecific inversion in IS, the translocation of the paraBalbiani within IIL and the inversion involving a landmark of IIIS.
Comparison of one of the most obvious landmark from the Neotropical Simulium species reveals the existence of some similarities among several species (Table 3). In S. limay the N.O. occupies a basal position in IS, resembling the localization of this chromosome marker as in S. exiguum (Procunier et al. 1985; Charalambous et al 1993 a, b), S. furcillatum (Campos and Muñoz de Hoyos 1990), Simulium "C" (Duque et al. 1988); S. ochraceum (Hirai and Uemoto 1983; Hirai et al. 1994; Millest 1992); S. pertinax (Campos Gaona et al. 1996); S. bicornutum and tunja as well as S. lewisi lutzianum (Miranda and Muñoz de Hoyos 1995). It should also be mentioned that the R.B. is located in S. limay in IIS occupying a proximal position to the centromere as it has been also observed in S. furcillatum (Campos and Muñoz de Hoyos 1990), S. metallicum "H" and "I" (Conn et al. 1989), S. pertinax (Campos Gaona et al. 1996) S. bicornutum and S. tunja (Miranda and Muñoz de Hoyos 1995).
In S. limay the pB and the gB usually are two characteristic landmarks for IIL. The PB is proximal to the centromere as it has been previously reported for S. metallicum "A", "C", "D", "E", "F", "G" and "K" (Conn et al. 1989) and "L" (Arteaga and Muñoz de Hoyos 1999).
The presence of the IIIS frazzled end in S. limay is a common character within the Simulium species cytogenetically analysed. It has been observed in S. furcillatum (Campos Gaona y Muñoz de Hoyos 1990), S. metallicum "A" to "K" (Conn et al. 1989), S. muiscorum (Muñoz de Hoyos 1990) and S. ochraceum (Hirai et al. 1994; Millest 1992). Accordingly to it, it could be suggested that almost all known Neotropical Simulium species share several landmarks, and that chromosome III is the most homosequential with the standard pattern, especially IIIL.
Chromosomal characters of the known neotropical species
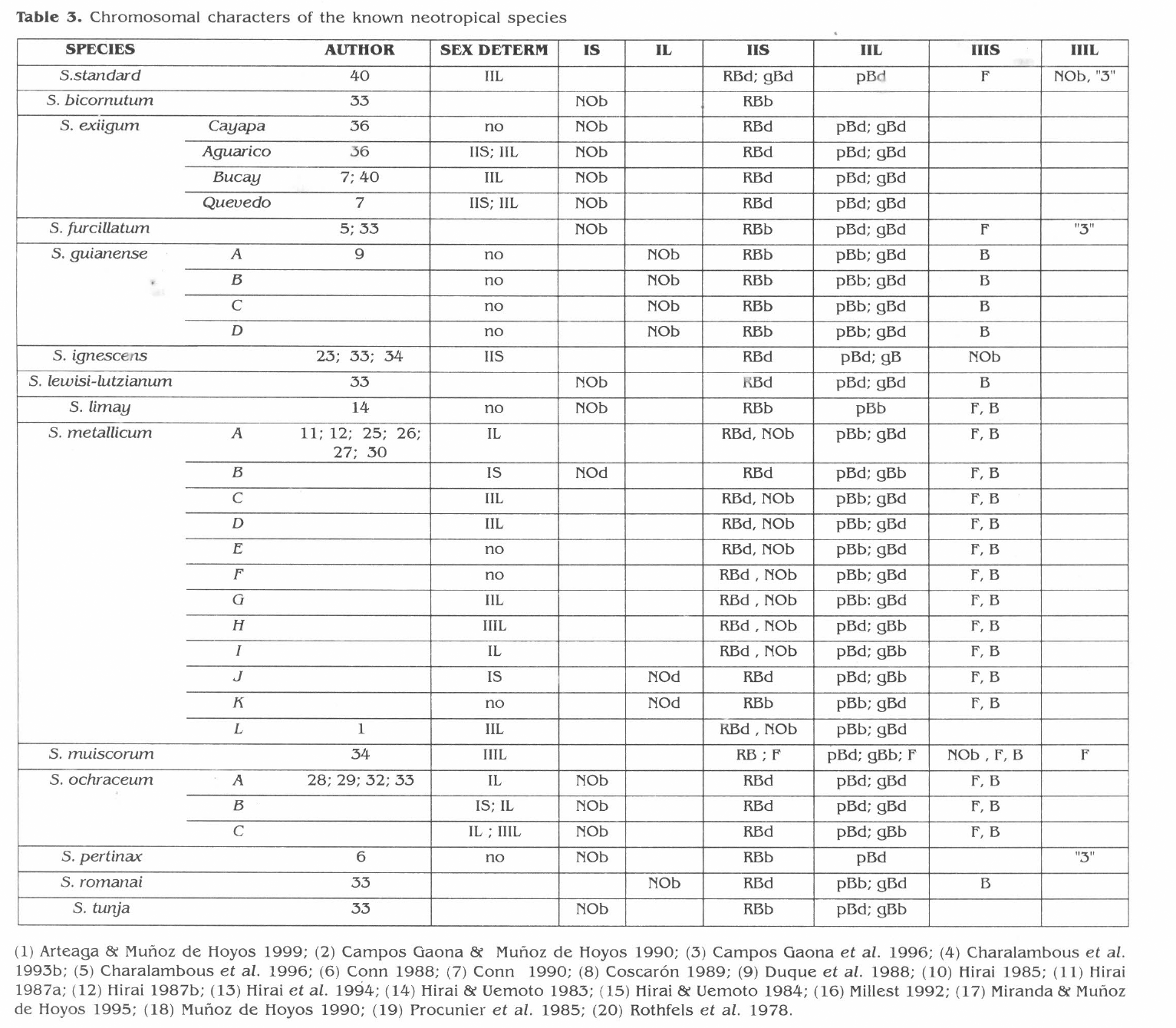
In S. limay no inversions or polymorphisms differentiates sexes. These results are in good agreement with previous reports demonstrating that the genetic X and Y chromosomes appear alike. Procunier et al. (1985) have observed no chromosomal rearrangements between sexual chromosomes in specimens from Simulium exiguum (Cayapa), and similar observations have been found by Duque et al. (1988) in Simulium "C", and in S. metallicum "E", "F" and "K" by Conn et al. (1989), S. guianense (Charalombous et al. 1996) and S. pertinax (Campos Gaona et al. 1996), suggesting that X,Y, is, therefore, a primitive character of this species group (Rothfels 1987).
In S. limay an interchange of chromosome arm was observed so that IIIL-IIL, IIIS-IIS are associated. This interchange has not been observed in other Neotropical simuliid species but does appear in Twinnia hirticornis (Gymnopaidinae) (Rothfels 1980). The fact that the same interchange of chromosome arms occurs in S. limay and Twinnia spp. could reflect a close phylogenetical relationship. However, they belong to different tribes (sensu Crosskey and Howard 1997). It is more likely that, this interchange of arms is probably a homoplasic character occurring in different taxa. S. limay has a morphology which is different from the rest of the Neotropical species and its distribution is closed to the Subantarctic fauna (Coscarón and Coscarón Arias 1995, 1996), which has a close relationship with the Australian fauna (Crisci et al. 1991). Therefore, S. limay was compared with Austrosimulium bancrofti (Ballard y Bedo 1991) but no evidences was found of close phylogenetical relationship among them. Comparing S. limay with S. ornatipes and S. neornatipes (Bedo 1977) it was observed that they share a proximal position of the pB in IIL and the frazzled end of IIIS. However, these characters alone are not enough to suggest a close relationship between S. limay and the australian species.
In conclusion, the interchanges observed in the arms of the chromosomes from S. limay may reinforce the opinion based on morphology that S. (Pternaspatha) is an independent subgenus, (Coscarón y Wygodzinsky 1972; Wygodzinsky and Coscarón 1967; Coscarón 1987) and justifies S. Pternaspatha as a valid supraespecific taxon. With the present information (Table 3) there is not enough chromosomal information that allows to establish that any neotropical or australian species are closest to S. (Pternaspatha) and to infer a phylogenetic relationship.
Footnotes
Aknowledgements
The author would like to acknowledge the helpful suggestions of Magda Charalombous (Department of Entomology of The Natural History Museum, London, U.K.) and Paulina Muñoz de Hoyos (Universidad Nacional de Colombia, Facultad de Ciencias, Instituto de Ciencias Naturales). Also to Prof. Nelida Caligaris (Facultad de Ciencias Naturales y Museo, Universidad Nacional de La Plata) for her drawings.
