Abstract
Radiation therapy is an effective cancer treatment used in over 50% of cancer patients. Preclinical research in radiobiology plays a major role in influencing the translation of radiotherapy-based treatment strategies into clinical practice. Studies have demonstrated that various components of tumors and their microenvironments, including vasculature, immune and stem cells, and stromal cells, can influence the response of solid tumors to radiation. Optically enabled imaging techniques used in experimental animal models of cancer offer a unique and powerful way to quantitatively track spatiotemporal changes in these tumor components in vivo at macro-, meso-, and microscopic resolutions following radiotherapy. In this review, we discuss the role of both well-established and emerging intravital microscopy techniques for studying tumors and their microenvironment in vivo, in response to irradiation. The development and application of new animal models, small animal microirradiation technologies, and multimodal optically enabled intravital microscopy techniques are emphasized within the framework of preclinical radiobiology research. We also comment on the potential influence that these newer imaging techniques may have on the clinical translation of new preclinical radiobiology discoveries.
OVER THE COURSE of cancer treatment, more than half of cancer patients receive radiation therapy. 1 Despite the effectiveness of radiation therapy, resistance to radiation and local recurrence within the primary site remain clinical challenges, negatively affecting treatment outcome. 2 Although the direct mechanisms of resistance and recurrence are unclear, the characteristics of the tumor microenvironment and tumor cell population affect the response to, and consequences of, radiation therapy. 2 The solid tumor microenvironment, characterized by its often intrinsic hypoxic state, is composed of vasculature and various cells, including immune and stromal cells. Moreover, tumors may contain self-renewing cancer stem cells (CSCs), which contribute to the heterogeneous tumor cell population. 3 These different components within the tumor and its microenvironment alter tumor responsiveness to radiation treatment and may have an impact on tumor recurrence and metastasis. 2 As ongoing preclinical studies elucidate the influence of tumors and their microenvironments on radiation therapy outcome, there is increased interest in exploiting these biological mechanisms to improve therapeutic efficacy. For example, tumor hypoxia and vasculature have been studied as therapeutic targets in combination with radiation therapy, with some studies advancing to clinical trials.1,4,5 Furthermore, radiation therapy combined with agents that target the CSC population may improve therapeutic benefit. 6
The goal of enhancing the antitumor effectiveness of radiation therapy, alone or in combination with other treatments, has helped invigorate modern preclinical radiobiology research. The promise of (1) identifying new therapeutic targets within the complex tumor milieu and (2) evaluating novel therapeutic combinations on multiple cellular components has initiated a shift from conventional experimental assays 7 to more advanced imaging techniques for studying solid tumors and their microenvironments in experimental cancer models in vivo. Conventional experimental assays such as tumor growth delay and clonogenic survival assay 8 cannot reveal the radiobiological response of individual tumor components within the living tumor over time. In addition, ex vivo tissue histopathologic approaches are limited by the static information they provide at any single time point in an otherwise dynamically changing tumor microenvironment. Advanced imaging techniques have been used to complement these conventional assays to provide dynamic information about the complex tumor microenvironment. For example, magnetic resonance imaging (MRI) and positron emission tomography (PET) have been used in the context of radiation research, 9 mainly with in vivo applications such as metabolic imaging, 10 perfusion imaging, 11 and oxygenation measurements. 12 Although such methods provide useful information at a macroscopic level, they do not sufficiently convey information about the tumor microenvironment at the meso- and microscopic levels in vivo. Optical imaging methods are best suited for assessing radiation response at functional, cellular, or molecular levels due to their specificity, sensitivity, high resolution, multiplexing capability, and low cost. 13 Intravital microscopy, defined as the imaging of living animals at microscopic resolution, 14 is particularly useful for assessing dynamic changes over time. Intravital microscopy is largely based on fluorescence imaging, although other techniques, such as optical coherence tomography (OCT) and photoacoustic imaging, are increasingly used alone or in combination to visualize various components within the tumor microenvironment in situ. When these imaging techniques are combined with an optically transparent glass window chamber (WC), surgically implanted in various parts of the mouse anatomy depending on the research question, they enable serial, quantitative microscopic imaging of cellular and molecular changes in tumors or organs over time. This intravital microscopy method allows the unprecedented study of the interdependence of critical biological features in living experimental systems. Although widely used to investigate tumor biology, and despite early pioneering work in the field, 13 the full potential of intravital imaging to investigate cancer radiobiology remains unexplored.
The purpose of this review is to discuss the contemporary role of optically enabled intravital microscopy in preclinical radiobiology research, particularly for studying the radiation response of solid tumor and normal tissue systems. We cover recent notable developments in new animal experimental tumor models and small animal x-ray microirradiation techniques, which enable complementarity with intravital microscopy techniques for the in vivo study of radiobiological response in animal models of cancer. We also discuss recent topics in the field of radiobiology and review ways in which intravital microscopy has been used to help uncover new insights into the temporal mechanisms of action and interdependency of tumor components in response to radiation in situ. Specifically, we survey recent peer-reviewed literature on the use of intravital optical imaging techniques to study tumor components and the tumor microenvironment, including vasculature, hypoxia, CSCs, and the “invasive” and “immunologic” niches—crucial regulators of tumor radiation response (Figure 1). Finally, we comment on the evolving role that intravital microscopy may play in driving new therapeutic discoveries toward clinical translation in the field of tumor radiobiology.

Radiation-induced changes to tumor microenvironment can be assessed using intravital imaging techniques. (A), Intrinsic factors such as tumor hypoxia and the presence of cancer stem cells may contribute to radioresistance and tumor invasion. (B), Radiation induces changes to the tumor microenvironment, which may further contribute to radioresistance and tumor invasion. Particularly, this review focuses on those components that can be assessed using intravital microscopy: vasculature, hypoxia, cancer stem cells, immunomodulation, and invasion. A high dose of radiation causes vascular damage, altering perfusion to the tumor. In addition, bone marrow-derived cells may be recruited to the site of vascular damage via hypoxia-inducible factor 1 (HIF-l)-dependent cytokine signaling and contribute to vasculogenesis, further altering vascular structure and function. T cells and dendritic cells are also recruited, further attracting immune cells to the irradiated site. Irradiation also alters tumor and stromal components through cytokine signaling such as that mediated by transforming growth factor β (TGF-β), contributing to epithelial mesenchymal transition (EMT) and extracellular matrix remodeling, which promote invasion and metastasis.
Animal Models for Intravital Imaging
Longitudinal intravital microscopic imaging of biological systems is enabled by experimental animal models specifically designed for interrogation by optical microscopy. The first such technique, described 90 years ago, used a transparent WC in rabbits' ears to study living tissue. 15 Following this pioneering study, the WC approach was adapted for use in the dorsal skinfold (Figure 2, A and B) in 1943 to study tumor growth and vascular development in mice. 16 Since then, the dorsal skinfold WC model has been widely used in studies of the tumor microenvironment,17,18 tumor–stroma interactions and metastasis,19,20 drug delivery, 21 and response to treatments such as antiangiogenic therapy,22,23 nanoparticles,24,25 chemo- 26 28 and/or radiation, and photodynamic therapy 29 . Although useful, questions about this model's biological relevance have been raised because subcutaneous tumors that grow flat in vivo may not accurately represent the more clinically relevant orthotopic tumor microenvironment that is required to promote tumor development.30,31 Furthermore, some tumors fail to grow when injected at the time of the WC surgery due to spontaneous apoptosis. 32 To overcome these limitations, “pseudo-orthotopic” 33 and “ectopic-orthotopic” 34 tumor models have been investigated by Borgstrom and colleagues. Both models involve implantation of tumor cells or tumor spheroids with their respective orthotopic stromal tissues in the murine dorsal skinfold WC, resulting in faster and well-vascularized tumor growth characteristics. Borgstrom and colleagues demonstrated the ability to implant stromal tissues from a variety of organs, making the dorsal skinfold WC model suitable for studying tumor and stromal cells of different anatomical origins in vivo. 34 Alternatively, tumor cells can be injected and left to grow in the dorsal skinfold prior to performing WC implantation surgery. This method has been shown to improve tumor growth in the dorsal skinfold WC by affording an initial period of stable tumor growth and vascularization before the WC installation. 35 Another method for making the WC models suitable for studying orthotopic tumors is to use organ-specificWCs. 36 Such a system allows for serial imaging of primary tumors in situ in the orthotopic setting. Moreover, tumor metastases can be investigated in metastasis-prone organs in vivo. Here we summarize some of these models established in various organs.

Examples of window chamber (WC)-based animal models used in optical intravital imaging. (A) Dorsal skinfold WCs (top left, metal; bottom left, plastic) surgically implanted on a mouse (right). The metal WCs were purchased from Research Instruments Inc. (Durham, NC). Plastic WCs are compatible for imaging with photoacoustic and magnetic resonance imaging. Scale bar = 5 mm. (B) Vasculature imaged through the dorsal skinfold WC. Fluorescence microscopy (left) requires contrast agent, such as labeled dextran, seen in green, whereas speckle-variance optical coherence tomography (svOCT, right) enables visualization of vasculature based on blood cell motion. Scale bar = 500 μm. (C) Plastic abdominal WC (left) surgically implanted over the exteriorized pancreas of a mouse (right). Scale bar = 5 mm. (D) Fluorescence image of the vasculature (green) and DsRed-BxPC3 human pancreatic tumor cells (red) in the pancreas. Scale bar = 500 μm. (E) Metal femur WC (left) surgically implanted on a mouse (right). The metal bar is placed under the femur to secure the WC in place. Scale bar = 5 mm. (F) Fluorescence image of the femoral vasculature (green). Scale bar = 100 μm. (G) Plastic spinal cord WC (left) surgically implanted on a mouse (right). Scale bar = 1 cm. (H) Fluorescence (left) and svOCT (right) images showing corresponding vascular structures in the spinal cord. Scale bar = 1 mm. (G) to (H) adapted with permission from Figley SA et al. 32
Cranial WC Model
A chronic cranial WC implant was first described in 1975. 37 Since then, the cranial WC model has been widely used to study cortical microcirculation and structures. 38 It has also been used as an orthotopic model of glioma to study vascular structure and permeability, 39 as well as to evaluate optical contrast agents for tumor margin detection. 40 Furthermore, a recent study demonstrated the use of triple transgenic mice with fluorescent astrocytes, neurons, microglia, and dendritic cells to image the dynamic microenvironment of glioblastoma. 41 The study developed a multiphoton imaging protocol that enables imaging of six different fluorescent proteins within the cranial WC of reporter mice, which is applicable to other intravital animal models. Imaging within the brain can be performed with a chronic transparent window or a thinned skull window, although a chronic window is superior for long-term imaging. 31
Pancreatic/Abdominal WC Model
A pancreatic WC was first used to study the effect of antivascular endothelial growth factor (VEGF) antibody treatment on angiogenesis, vascular permeability, and tumor growth in an orthotopic model of pancreatic cancer. 42 A follow-up study demonstrated the effect of anti-VEGF treatment on pancreatic tumors by imaging and quantifying changes to vascular density and diameter. 43 Using a similar abdominal WC model, Ritsma and colleagues imaged colorectal tumor cells in the hepatic microvasculature and tracked T cells in the spleen. 44 They demonstrated that their abdominal WC model enables imaging of abdominal organs, such as the spleen, pancreas, liver, and small intestine, for up to 28 days. Recently, our own group developed a version of the pancreatic WC model adapted from the model developed by Tsuzuki and colleagues 42 to investigate radiation response of orthotopic human pancreatic tumors and their vasculature in vivo (Figure 2, C and D). The model was developed to permit longitudinal microscopic imaging of tumors and vessels using white light reflectance, fluorescently labeled vascular imaging agents, ultrasonography, and photoacoustic imaging.
Mammary Fat Pad WC Model
A mammary WC was first described in 2003 in rats and mice to obtain images of angiogenic tumor vasculature. 45
The model has since been used to track metastatic cell migration using photoswitching of Dendra-2-labeled cells. 46 The photoswitching technique enabled tracking of a distinct group of cells in relation to blood vessels and collagen matrix. A recent study used the Confetti mouse model, which contains a stochastic multicolor reporter, to identify the presence of mammary CSCs by lineage tracing for up to 3 weeks. 47 A study by Schafer and colleagues demonstrated compatibility of the mammary WC model with MRI to study anatomic features of tumor, in addition to fluorescence-based visualization of tumor cell growth and death. 48
Other WC Models
In addition to the models listed above, WC models have been developed for other anatomic sites. For example, a femur WC model was described for the study of bone microcirculation, leukocyte–endothelium interactions, and tumor growth in mice femora. 49 Our own group recently developed a femur WC murine model to study tumors and their vasculature in vivo in response to radiation and other treatments (Figure 2, E and F). A lymph node WC model was developed to study mechanisms of non-Hodgkin lymphoma progression and dissemination for over 2 weeks. 50 In addition to tumor models established in WCs, it is also possible to develop organ-specific WC systems for studying micrometastasis. For example, a lung WC model can be used to visualize metastasis to the lungs. Although the lung WC has not been developed as a chronic implantation model, it has been used to image lung microvasculature changes in response to hypoxic pulmonary vasoconstriction for up to 2 hours using a fluorescent vessel tracking dye. 51 In addition, response to inflammation, based on blood perfusion and neutrophil motion, was studied using a thoracic chamber with a vacuum system, which enabled the lung to enter the field of view of the window while sustaining its function. 52 The vacuum model allowed for loose adherence of the WC to the lung surface to maintain perfusion and ventilation for up to 3 hours. Recently, we reported the development of a spinal cord WC model for imaging micrometastasis of medulloblastoma tumor cells to the spinal cord using multimodal intravital imaging of the spinal cord and surrounding tissues. In this work, we demonstrated the use of fluorescence microscopy, power Doppler ultrasonography, photoacoustic imaging, and OCT imaging of the spinal cord, including the use of small animal x-ray microirradiation (Figure 2, G and H). 32
Collectively, application of these animal WC models opens up possibilities for dynamic microscopic imaging in various organs. To our knowledge, dorsal, cranial, and spinal cord WC models have been used in the context of radiobiology research, mostly to investigate cellular and vascular response to radiation.27,32,53,54 Other orthotopic tumor models, as described above, can also be considered when investigating normal and tumor tissue response to radiation, as well as the effect of radiation treatment on tumor metastasis in vivo.
Small Animal X-Ray Irradiation Techniques
Intravital imaging of radiation response can benefit greatly from a capability to perform image-guided focal irradiation within animal WC setups. Precise targeting with a wide variety of irradiation beam sizes and energies is well suited for irradiation of tumors within a WC setting (typically 3 to 5 mm in size). A major advantage of small animal irradiation systems is that they allow investigators to recapitulate specific clinical radiation treatment schema that deliver complex dose patterns based on radiologic image guidance (e.g., intensity-modulated radiation therapy [IMRT]), 55 improving the relevance of preclinical studies to clinical radiation treatment regimens. Here we briefly summarize recent developments in small animal irradiation platforms and their relevant applications.
A system named Small Animal Radiation Research Platform (SARRP) was developed at Johns Hopkins University. 56 The system is equipped with an x-ray source (energy ranging from 50 to 225 kVp), cone-beam computed tomography (CT) image guidance, and a robotic stage for positioning animals during treatment. Using the system, a deposited radiation dose can be confined to within 1 mm. 56 In addition to CT and planar x-ray imaging for target localization, the platform also enables bioluminescence image-guided irradiation. This image-guidance technique was successfully used to deliver spatially confined x-ray to orthotopic pancreatic tumors, which are otherwise difficult to target using CT alone due to the lack of intrinsic soft tissue contrast in the abdomen. 57
Another commercialized system, named X-RAD 225Cx, was developed at the Princess Margaret Cancer Centre. 58 It consists of an x-ray tube with a cone-beam CT panel mounted on a rotating gantry arm for image-guided radiotherapy (Figure 3A). Similar to the SARRP system, the X-RAD system enables irradiation with a focal spot size as small as 1 mm at a dose rate ranging from 1 to 3 Gy/min. 58 The system has been used to irradiate two-dimensional (2D) complex dose distribution geometry at the millimeter scale, suggesting its potential application for “dose-painting” in experimental models such as the dorsal skinfold WC model. 59 In addition, a dedicated treatment planning system named SmART-Plan was developed to enable high-throughput animal experiments for complex dose delivery in three dimensions (3D). 60 Recently, the X-RAD system was also equipped with 3D bioluminescence imaging capability, 61 which could enable better tumor to normal contrast than the onboard CT imager for improved treatment planning and longitudinal imaging of tumor response to irradiation.
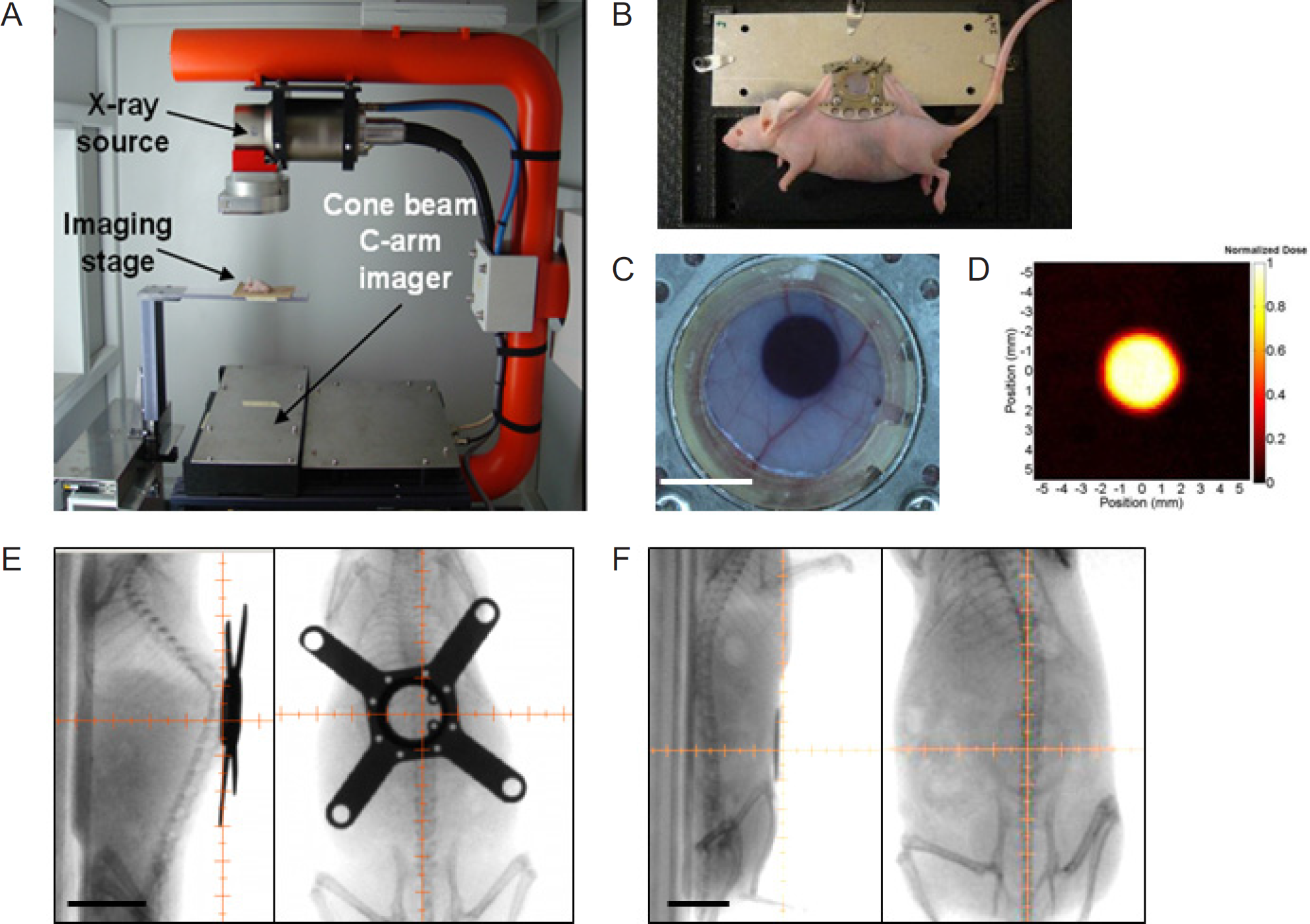
Small animal irradiator and its application in window chamber (WC) animal models. (A) The X-RAD 225Cx system, equipped with a CT imaging panel and an x-ray source mounted on a rotating gantry. (B) A photograph of an anesthetized dorsal skinfold WC-bearing mouse restrained on a custom-made mount for irradiation. (C) A radiochromic film enables visualization of the delivered dose. An image of a fluorescent tumor is used to move the stage to a desired location to target tumors. Scale bar = 5 mm. (D) The irradiated radiochromic film can be scanned to perform dosimetry. The colored bar represents doses delivered. Using a custom-made collimator, an approximate 4 mm diameter dose was deposited. (B) to (D) images adapted with permission from Maeda A et al. 27 X-ray fluoroscopy images of (E) spinal cord WC-bearing mouse and (F) pancreatic WC-bearing mouse. Scale bar = 1 cm. The small animal irradiator can be used in various WC animal models. X-ray fluoroscopy can be used to target tumors or structures within the WCs. The motorized stage enables precise targeting with collimators of different sizes corresponding to varying sizes of radiation dose delivery. The beams can also be delivered from multiple angles to spare healthy organs. (E) images adapted with permission from Figley SA et al. 32
Both SARRP and X-RAD systems can be used with WC models, allowing for focal x-ray irradiation based on CT, x-ray, and/or bioluminescence imaging. For example, the X-RAD system has been used with intravital optical imaging techniques to study bone marrow cell recruitment and changes to the tumor microenvironment in response to radiation.27,53 Figure 3, B to F, demonstrates its application with dorsal skinfold, 27 pancreas, and spinal cord 32 WC models. Other potential applications of focal irradiation with intravital imaging may include delivery of varying doses to different populations of cells defined by imaging or monitoring of cellular action in and out of the treatment field.
Role of Imaging in Current Topics in Radiobiology
Intravital imaging has been used to answer relevant radiobiology questions. These reports, which are discussed in this section, have mainly focused on the effect of radiation on normal and tumor vasculature, tumor hypoxia, and immune cell recruitments. Emerging areas of interest in radiobiology, such as the role of CSCs and tumor invasion, can also be assessed using intravital imaging. Here we review recent intravital imaging techniques for assessing these biological aspects in vivo and discuss their current and potential applications for novel discoveries in radiobiology.
Radiation-Induced Vascular Effects
Growing evidence suggests that ionizing radiation affects normal and tumor vasculature. Radiation may cause serious complications, such as acute and late thrombosis and vascular occlusion,62,63 thereby contributing to radiation-induced toxicity in normal tissue. More recently, experimental studies have demonstrated that radiation-induced vascular alterations can affect tumor response to radiation. For example, the direct effect of radiation-induced vascular damage on tumor response is thought to be significant when tumors are irradiated with a single dose high enough to cause substantial endothelial cell death, leading to tumor cell death.64,65 However, the observed outcome of radiation-induced changes to tumor vasculature varies between different studies, depending on radiation dose as well as tumor type, size, and stage. 65 For example, recent studies using a primary sarcoma cancer model in mice have shown that, contrary to the observations made in transplanted tumor models, endothelial cells undergo delayed mitotic cell death after radiation 66 and that endothelial cell death does not affect tumor response to radiation. 67 Radiation-induced effects on vasculature may also vary depending on the fractionation scheme. For example, a recent study demonstrated that ablative hypofractionated radiotherapy (36 Gy in three fractions) decreases vascular density while increasing perivascular coverage, suggesting that ablative hypofractionated radiotherapy normalizes tumor vasculature, leading to a decrease in tumor hypoxia. 68 Moreover, Potiron and colleagues demonstrated that conventional fractionated radiotherapy (2 Gy daily for 2 weeks) causes vascular remodeling, based on vascular wall maturation and increased perivascular coverage. 69 This study further showed that vascular remodeling increases perfusion and decreases hypoxia while maintaining vascular density. Collectively, these studies propose that radiation-induced vascular changes impact both normal tissue toxicity and tumor cell response to radiation.
Imaging Vasculature
Radiation is thought to cause vascular damage and remodeling depending on the dose and fraction. Intravital microscopy is suited to monitor such vascular changes over time, at both structural and functional levels. Here we review recent studies that use in vivo optical imaging to study vascular structure and function and their applications in monitoring vascular changes induced by irradiation (Table 1 and Figure 4).

Examples of in vivo imaging modalities for studying vascular structure and function. (A) Fluorescence imaging of tumor vasculature. The human pancreatic cancer cell line BxPC3 was grown in the dorsal skinfold window chamber model, and 2 MDa fluorescein isothiocyanate (FITC)- dextran was injected intravenously to provide contrast to perfused vessels. Scale bar = 500 μm. (B) Speckle-variance optical coherence tomography (svOCT) imaging of the BxPC3 tumor vasculature, providing structural information. Scale bar = 2 mm. (C) Photoacoustic imaging of tumor vasculature. Human colorectal adenocarcinoma LS174T cells were grown subcutaneously in flanks of mice, and the image was obtained 12 days postinoculation. The dashed line represents the tumor margin, and the arrows represent landmarks that can be tracked over time. Image adapted with permission from Laufer J et al. 80 (D) Fluorescence imaging of vascular permeability using 158 kDa tetramethylrhodamine isothiocyanate (TRITC)- dextran following VEGF treatment in an ex ovo chicken embryo model. Extravasation of the dextran molecules can be seen by the increased fluorescence intensity in the extravascular space. Images adapted with permission from Pink DB et al. 87 (E) Fluorescence imaging and velocimetry technique to study blood flow. Imaging of mouse brain was performed using two-photon fluorescence microscopy with 2 MDa Texas red-dextran. Velocity (in bold) and diameter measurements were made at various locations by velocimetry as indicated. Images adapted with permission from Kim TN et al. 95 (F) Cross-sectional Doppler and structural OCT images of a mouse spinal cord. The Doppler image demonstrated blood flow in the posterior spinal vein, whereas the structural image highlighted anatomic features: (1) glass coverslip of the window chamber, (2) anterior spinal vein, (3) white matter, and (4) gray matter. Images adapted with permission from Figley SA et al. 32 (G) Photoacoustic imaging of blood flow in a mouse ear. Total flow speed was measured by combining axial and transverse flow components. Image adapted with permission by Yao J et al. 97 Copyright © 2012 by SAGE Publications, Inc. Reprinted with permission of SAGE Publications, Inc.
Imaging vascular structure. The structure of functional vasculature can be visualized and quantified using single- or multiphoton fluorescence microscopy (see Figure 4A). Typically, a large-molecular-weight dextran (2 MDa) is injected prior to imaging to label functional vasculature.27,70 Image analysis tools, such as freeware ImageJ (National Institutes of Health, Bethesda, MD), commercially available image analysis packages, and custom-made software, 71 73 can be used to measure various features of vasculature, such as fractal dimension, diameter, vascular density, branch length and branching points, vascular surface area, and tortuosity based on 2D and 3D fluorescence images.35,70,74 These features are important indicators of vascular status. For example, reduced vascular diameter and tortuosity are thought to be indicators of vascular normalization, which can lead to improvement in radiation therapy efficacy. 22 A study by Winkler and colleagues used intravital fluorescence microscopy to characterize the time course of functional and structural changes to tumor vasculature in response to vascular endothelial growth factor receptor 2 (VEGFR2) blockade. 75 By using fluorescence imaging to measure vascular length, diameter, volume, and permeability, they demonstrated that VEGFR2 blockade temporarily induces vascular normalization, which is characterized by improved vascular function. Together with histology, they identified a “normalization window” in which radiation therapy is most effective when combined with VEGFR2 blockade. The same group later studied nitric oxide (NO) distribution in addition to vascular function and structure using intravital fluorescence microscopy. 76 An NO-sensitive fluorescent probe enabled identification of NO gradient in perivascular areas, which induced vascular normalization. They further demonstrated that perivascular production of NO suppressed tumor growth after fractionated radiation, suggesting that vascular normalization may improve tumor response to radiation.
Short Summary of Different Imaging Techniques for Visualizing Vasculature

pO2 = partial pressure of oxygen; OFDI = optical frequency domain imaging; sO2 = oxygen saturation; svOCT = speckle-variance optical coherence tomography.
Various optical imaging modalities can be used to image vascular structure, function, and oxygenation. Notably, OCT, PAM, and hyperspectral imaging do not require contrast agents for vascular imaging, whereas fluorescence and phosphorescence require contrast agents.
Together these studies exploited intravital imaging to visualize and quantify changes in tumor vasculature over time to help investigate the effect of combining vascular-targeting strategies with radiation therapy.
Compared to fluorescence imaging, which visualizes functional vasculature using an injected fluorescent dye, OCT can be used to image vasculature (see Figure 4B) without contrast agents and independent of blood flow. 77 Vakoc and colleagues developed a technique termed optical frequency domain imaging (OFDI), a “next generation” of OCT, with rapid image acquisition speed and high sensitivity. 23 Using this system, they monitored tumor and vascular response to cellular- and vascular-targeted therapies to evaluate the use of OFDI for therapeutic response monitoring. A similar technique termed speckle-variance optical coherence tomography (svOCT) was used to monitor vascular changes in response to adenoviral infection 78 and radiation therapy. 27 In addition, an image analysis tool was developed for svOCT to quantify vascular changes in the 3D volume. 77 A major advantage of OCT imaging for intravital models compared to single- or multiphoton fluorescence imaging of vasculature is its depth of penetration in tissues. Using OCT, blood vessels beyond 1 mm in depth can be observed, whereas the depth of penetration for multiphoton microscopy is limited to 250 to 400 μm. 23 Our group studied the effect of a single dose of radiation on tumor vasculature using both fluorescence and svOCT imaging to distinguish functional and structural damage to vasculature in vivo over several weeks following treatment. 27 We demonstrated that some tumor vasculature is functionally impaired following irradiation yet maintains its structural integrity over time, demonstrating the complementary role of the two imaging techniques. The ability to distinguish between structural and functional changes in the tumor vasculature in vivo and serially following radiation treatment may improve our understanding of how some tumor cells are able to survive and regrow following treatment. 79
Lastly, photoacoustic imaging has been used in intravital models to analyze vascular structure owing to strong optical absorption of hemoglobin. 80 The first description of optical resolution photoacoustic microscopy (PAM) demonstrated simultaneous imaging of vascular structure as well as intravascular oxygen saturation (sO2) under physiologic hyperoxic and hypoxic conditions. 81 Using a subcutaneous model, Laufer and colleagues monitored the development of tumor vasculature and its response to a vascular-disrupting agent, achieving depths of almost 10 mm with sub-100-micron spatial resolution 80 (see Figure 4C). PAM has also been combined with svOCT to image vascular structure 82 and with fluorescence confocal microscopy for in vivo angiography and lymphangiography, both in the mouse ear model. 83 Photoacoustic imaging can be used in a similar manner to fluorescence or OCT imaging for measuring radiation-induced vascular structural and functional changes in situ but with improved imaging depth. As a result, photoacoustic imaging may be more suitable for applications where the organs of interest (in organ-specific WC models) are not superficial. For example, our group recently demonstrated the combination of photoacoustic imaging with intravital fluorescence microscopy to visualize vascular structure and function, as well as metastatic tumor foci in a murine spinal cord WC model. 32
Imaging vascular function. Vascular function can be determined by measuring vascular permeability and blood flow. In animal models, vascular permeability is typically measured using the Miles vascular permeability assay, 84 which involves injection of Evans blue dye into an animal to examine visible blue stains around the injected site after euthanasia. This assay requires animal sacrifice and does not allow dynamic tracking of vascular permeability. Alternatively, intravital fluorescence microscopy has recently been used to study both normal and tumor vascular permeability in vivo. In contrast to imaging functional vascular structure using fluorescence microscopy, low-molecular-weight dextran molecules are typically used to assess vascular permeability by allowing the dextran molecules to extravasate from the vasculature. Images can be acquired at various time points after injection of the dextran, and the images are registered in order to segment intravascular and extravascular space. 85 Low-molecular-weight dextrans (< 40 kDa) rapidly accumulate in the extravascular compartment of tumors, whereas high-molecular-weight dextrans have reduced tumor permeability. 86 Dextrans of various sizes have been used to study changes to vascular permeability in a chicken embryo model and the dorsal skinfold WC model in response to treatments such as vascular endothelial growth factor (VEGF) 87 (see Figure 4D), vascular-disrupting agent,85,88 doxorubicin, 28 and bevacizumab, 89 as well as radiation therapy. For example, assessment of vascular permeability and leukocyte adhesion by intravital imaging demonstrated that a high dose of radiation (20 Gy) induces an increase in vascular permeability and leukocyte adhesion in the blood-brain barrier.90,91 These studies further demonstrated that targeting intracellular adhesion molecule 1 (ICAM-1) reduces such an effect, making it a potentially effective way to manage radiation-induced vascular injury in the central nervous system. Similarly, Mouthon and colleagues used fluorescence microscopy to study platelet interaction with the endothelium following irradiation. 92 The study demonstrated that radiation increases platelet adhesion mostly in postcapillary venules, likely by upregulation of adhesion molecules in the endothelium rather than a direct activation of platelets, thus identifying a potential target for reducing radiation-induced thrombosis. Thus, the use of intravital imaging for assessing vascular function has enabled the study of dynamic processes (vascular permeability, cell–cell interaction) at microscopic resolution over time in response to radiation. This in turn has helped uncover potential avenues for reducing normal tissue vascular injury following irradiation, which has translational value.
Fluorescence microscopy can also be used to measure blood flow. Fluorescence imaging of labeled red blood cells can be used to track their velocity within tumor vasculature using an algorithm that predicts the track of red blood cells based on a previous history of their location. 93 More recently, Kamoun and colleagues developed two techniques, termed “residence timeline scanning” and “relative velocity field scanning,” to analyze flow velocity in tumor vasculature using fluorescently labeled red blood cells. 94 However, to measure high blood velocity in vessels and arterioles, the former method was limited to a measurement at a single pixel, and the latter method was limited by the angle of measurement. To enable analysis of high blood velocity, Kim and colleagues developed a line-scanning particle imaging velocimetry technique, which determines the displacement of red blood cells between pairs of line scans 95 (see Figure 4E). Using the technique, they identified changes in red blood cell velocity within arteriovenous shunts in a mouse cranial WC model of arteriovenous malformation. Doppler OCT imaging32,96 (see Figure 4F) and PAM 97 (see Figure 4G) have also been used to study tumor blood flow. Typically, blood flow measurements are combined with measurements of other vascular characteristics, such as vessel diameter and saturated oxygenation, to characterize tumor hypoxia and metabolism. These imaging techniques can be used to investigate how radiation-induced changes alter blood flow.
Radiation-induced vascular damage, whether structural or functional, affects tumor microenvironment and overall tumor response to radiation therapy. The various imaging techniques described above enable differentiation of vascular structure and function, as well as visualization of processes within vasculature in response to radiation. Intravital imaging enables investigation of the direct effects of radiation on vascular structure and function, as well as the ability of vascular-targeting agents to augment tumor response to radiation. Together these imaging techniques can be used to assess radiation-induced vascular damage and the consequent tumor response, as well as novel ways to mitigate vascular damage and the effect of combination therapies.
Tumor Hypoxia
Tumor hypoxia is an important indicator of tumor aggressiveness. Measuring tumor hypoxia before clinical treatment has a prognostic value as tumor hypoxia is associated with decreased local tumor control and lower rates of disease-free and overall survival. 98 Tumor hypoxia is also an indicator of reduced tumor sensitivity to radiation. Experimental studies have shown that hypoxic cells are two to three times more resistant to a single fraction of radiation than those with normal levels of oxygen due to decreased fixation of DNA double-strand breaks produced by free radicals. 99 Furthermore, under hypoxic conditions, expression of various genes, such as those involved in angiogenesis and metastasis, are induced by upregulation of hypoxia-inducible factor 1 (HIF-1). Hypoxia-induced genomic changes may also impact cellular radioresistance, for example, by altering DNA repair pathways and increasing proliferative potential.100,101 HIF-1 expression is also upregulated by radiation, leading to endothelial cell survival and consequent radioresistance. 54
Hypoxia occurs as a consequence of imbalance between availability and metabolism of oxygen; therefore, strategies for increasing oxygen availability and/or decreasing oxygen metabolism have been investigated as ways to overcome hypoxia and radioresistance. For example, methods to increase delivery of oxygen through the bloodstream using hyperbaric oxygen, carbogen, and nicotinamide 102 and to decrease oxygen consumption by cells using NO donors 103 have been studied. More recently, classes of hypoxia sensitizers, hypoxia-activated cytotoxins, and direct inhibitors of hypoxia-activated signaling pathways have been developed. One such example is tirapazamine, a highly toxic hypoxia-activated cytotoxin that effectively enhances radiation therapy in experimental settings. 102 In summary, tumor hypoxia is an important target for radiation therapy as it limits therapeutic efficacy. It will continue to be aggressively studied in the context of emerging combination treatment strategies in both preclinical and clinical settings to overcome the biological mechanism of resistance.
Imaging Tumor Hypoxia
Tumor hypoxia is influenced by oxygen availability and metabolism, both of which can be measured within vessels and extravascularly in the interstitial space using intravital imaging to assess radiation response. In addition, alteration of molecular expression due to hypoxia can be visualized. Most studies have used more than one optical imaging modality to better understand tumor hypoxia and metabolism (Figure 5). Here we review hypoxia-imaging techniques that have been used to assess the response to radiation (phosphorescence and fluorescence imaging), as well as those that have potential unique applications to answer radiobiological questions.

Examples of imaging modalities for studying vascular oxygenation and hypoxia. (A) Phosphorescence imaging of vascular partial pressure of oxygen (pO2) using Oxyphor R2 in a rat cortex. Selected locations and corresponding pO2 measurements are highlighted in green. Arterioles are indicated in red, and venules are indicated blue. Image adapted with permission from Yaseen MA et al. 105 (B) Phosphorescence imaging of tumor hypoxia using iridium complex for different tumors at 0 and 60 minutes following tail-vein injection of the iridium complex. The iridium complex (red) was found to be localized within tumors, indicating their extravasation and local accumulation. The system has not been used in a window chamber–based animal model. Images reprinted with permission from the American Association for Cancer Research: Zhang S et al. 110 Scale bar = 1 cm. (C) Fluorescence imaging of 4T1 mammary carcinoma cells (red) and its HIF-1 expression (green) in a dorsal skinfold window chamber model. Images adapted with permission from Palmer GM et al. 111 Scale bar = 1 mm. (D) Photoacoustic imaging of vasculature in the dorsal skinfold window chamber. A commercial photoacoustic imaging system (Vevo LAZR, VisualSonics Fujifilm) was used. The color bar indicates the relative level of vascular oxygen saturation (sO2). Scale bar = 5 mm. (E) Hyperspectral imaging of sO2 in a Caki-2 tumor–bearing dorsal skinfold window chamber 4 days following tumor implantation. Images adapted with permission from Lee JA et al. 117 (F) Hemoglobin saturation (top) and flavin adenine dinucleotide (FAD)/reduced nicotinamide adenine dinucleotide (NADH) ratiometric redox (bottom) imaging in a non–tumor-bearing window chamber mouse during oxygen and nitrogen inhalation. Hemoglobin saturation was measured by absorption between the wavelength range of 500 to 620 nm, whereas redox was calculated based on NADH and FAD autofluorescence. Images adapted with permission from Skala MC et al. 118 Scale bar = 1 mm.
Phosphorescence imaging enables quantitative measurement of partial pressure of oxygen (pO2) based on oxygen-dependent quenching of phosphorescence. 104 Oxygen-sensitive probes can be injected intravenously to measure pO2 in the vessels or interstitially to measure pO2 in the tissue. Palladium (Pd) and platinum (Pt) porphyrin-based probes have been used to measure intravascular pO2. 104 For example, the Pd porphyrin–based dendritic oxygen probes Oxyphor R2 and G2 were used in combination with confocal microscopy to monitor changes in vascular pO2 within a cranial WC model while altering the fraction of inspired oxygen and performing functional brain stimulation 105 (see Figure 5A). More recently, a “next-generation” Pt-based probe (PtP-C343) was developed to measure intravascular pO2 in mouse cortical arterioles before and after photothrombotic occlusion. 106 In this study, an additional measurement of blood flow was obtained using the laser speckle contrast imaging technique. A recent study by Spencer and colleagues also used PtP-C343 to measure pO2 in the bone marrow with two-photon phosphorescence lifetime microscopy. 107 They compared pO2 at different locations from the bone, as well as differences in pO2 as a function of vascular diameter. Interestingly, they also demonstrated elevation of pO2 after radiation therapy in normal vasculature. The increase in oxygenation was due to reduced oxygen consumption because bone marrow cells were reduced after irradiation while blood flow was maintained. The ability to image multiple components simultaneously was necessary to reach this conclusion. The study demonstrates the unique ability of imaging to assess changes in oxygen consumption and demand in response to irradiation. Phosphorescence imaging has also been used with OCT to measure cerebral pO2 and blood flow, 108 as well as with PAM to image changes to the intravascular sO2 and pO2 levels in response to hyperoxia and normoxia. 109 Oxygen-sensitive phosphorescence probes can also be injected into the interstitial space of animals to measure tissue pO2 levels. For example, Zhang and colleagues used a red-emitting iridium complex for phosphorescence imaging of tumor hypoxia 110 (see Figure 5B). As an alternative to metalloporphyrins, which are relatively large, the iridium complex could be chemically modified to improve water solubility as well as absorption and emission properties for improved imaging depth. Unlike phosphorescence imaging, which requires specialized instrumentation, boron nanoparticles have been developed to enable ratiometric imaging of tissue oxygen tension. 111 Boron nanoparticles have been used in the dorsal WC model to measure dynamic tissue oxygenation in combination with hyperspectral imaging for vascular sO2 and fluorescence imaging for HIF-1 expression (see Figure 5C). Fluorescence-based imaging of HIF-1 expression has been used in a study by Moeller and colleagues to directly assess HIF-1 activation following irradiation. 54 The ability to visualize distribution of HIF-1-activated tumor cells in time and space provided important information that led to linking HIF-1 levels to reoxygenation after irradiation. Imaging also revealed that an increased amount of free radicals was related to radiation-induced HIF-1 activation. Given these observations, they demonstrated that HIF-1 regulates endothelial cell radioresistance, which has implications in diminishing tumor response to radiation. Harada and colleagues also used a similar system, a hypoxia-inducible luciferase expression vector, 112 to study cell fate after irradiation in a tumor xenograft model. The authors demonstrated elevated cellular levels of HIF-1 activity in areas of radiation-induced reoxygenation and that these surviving tumor cells translocate toward blood vessels, facilitating local tumor recurrence. Overall, the study used imaging to observe molecular changes related to hypoxia after irradiation in a time- and space-dependent manner, providing useful information on the effect of radiation on tumor cells.
A high-resolution photoacoustic imaging system similar to the PAM system has been used in combination with fluorescence and pulse echo imaging to track tumor and vascular development, including measurement of vascular sO2, in a dorsal skinfold WC model. 113 Similarly, a commercial photoacoustic imaging system was used with the dorsal skinfold WC model to measure vascular sO2 (see Figure 5D). PAM was also combined with spectral domain OCT to measure the metabolic rate of oxygen in a flow phantom and in a mouse ear model. 114 In this study, sO2 and artery and vein diameters were measured by PAM, whereas the mean blood flow velocities in the artery and vein were measured by spectral domain Doppler OCT to calculate the metabolic rate of oxygen. Vascular sO2 can also be measured using hyperspectral imaging, which is based on spectral analysis of reflected absorbance collected at different wavelengths. 115 Hyperspectral imaging has been used in combination with video recording of blood flow to study the course of wound-induced vascular abnormalities in the dorsal WC model. 116 Similarly, hyperspectral imaging and fluorescence imaging of blood transit time have been used in combination to analyze vascular oxygenation and blood flow 117 (see Figure 5E). Both PAM and hyperspectral imaging can be used in a similar manner to phosphorescence imaging for studying alterations in vascular oxygenation following radiation, thereby obtaining simultaneous information on blood flow when integrated with other imaging systems. For example, tumor metabolic demand can be quantified in vivo using a combination of intravital imaging techniques. In a study by Skala and colleagues, redox ratiometric fluorescence, hyperspectral and Doppler OCT were combined to study tumor metabolic demand, sO2, vessel morphology, and blood flow 118 (see Figure 5F). This study enabled imaging of oxygen supply and demand in tumors, which is an important indicator of tumor hypoxia.
Imaging-based assessment of hypoxia can be challenging because hypoxia results from the combined effect of oxygen availability and metabolism. The combined use of imaging methods described above can help delineate multiple factors regulating hypoxia, such as blood flow, vascular oxygenation, and upregulation of hypoxia-related genes. These assays complement conventional assays, which are typically invasive or require histologic analysis, to provide dynamic information of hypoxia in the context of radiobiology. Moreover, they enable assessment of therapeutic strategies to overcome the radioresistant nature of hypoxic tumors.
Radiation-Induced Immunomodulation
Radiation-induced vascular damage can lead to HIF-1- dependent stimulation of cytokine signaling and subsequent recruitment of a heterogeneous population of tumor-associated macrophages and immune cells. 119 Kioi and colleagues demonstrated that bone marrow–derived cells (BMDCs) restore irradiated vasculature by vasculogenesis, thus promoting tumor regrowth. 120 Kozin and colleagues confirmed that tumor recurrence after irradiation is supported by infiltrating CD11b+ monocytes. 121 Thus, recruitment of BMDCs to the tumor could favor local relapse after irradiation due to vascular remodeling. 122 In addition to BMDC-mediated local vascular remodeling, increasing clinical evidence suggests that the effect of radiation therapy extends beyond the treatment field, described by the term “abscopal effect,” an event occurring at a distance from the irradiated volume but within the same organism. 123 The abscopal effect is thought to be mediated by the immune system, involving dying cells, their microenvironment, and the host immunologic niche. 124 Experimental studies suggest multiple effects of radiation on the immune system, which may help us understand the abscopal effect in patients. For example, radiation induces immunogenic cell death, a type of cell death that activates the immune response by various molecular mechanisms. 125 Immunogenic cell death promotes presentation of tumor-associated antigens by dendritic cells (DCs), which may be activated and migrate from the treatment field to trigger a cytotoxic T-cell response against tumor cells.122,126 Radiation can also induce chemokine expression, attracting cytotoxic T cells to the irradiated site. 127 In addition, surviving cancer cells display expression of adhesion molecules, death receptor, and major histocompatibility complex (MHC) class I, enhancing recognition by cytotoxic T cells. 127 At present, it is not known which of these radiation outcomes directly account for the abscopal effect. 128 Further experimental studies are needed to elucidate the underlying inductive mechanisms of the abscopal effect, as well as the role of BMDCs to promote tumor recurrence following irradiation. 127
Imaging Immunomodulation
Fluorescence imaging can be applied in intravital imaging models to visualize cell–cell interactions and cellular movements in the living system in response to treatments. For example, BMDCs are recruited in response to radiation as well as chemotherapy and may influence vascular remodeling and tumor cell kill. 129 Nakasone and colleagues demonstrated recruitment of myeloid cells after doxorubicin treatment through a mammary WC, showing that myeloid cell recruitment correlates with tumor relapse. 28 However, the authors suggest that myeloid cells can also induce the direct killing of cancer cells, concluding that the functional differences of recruited myeloid cells are likely due to different subpopulations. Burrell and colleagues demonstrated in a cranial WC model that BMDCs, isolated from green or red fluorescent transgenic donor mice and reconstituted into recipient mice, are recruited following cranial irradiation in a dose- and time-dependent manner. 53 Intravital imaging was used in this study to demonstrate that despite their initial expectations, BMDCs do not differentiate into CD31-positive endothelial cells. Based on these observations, they investigated the fate of BMDCs recruited by irradiation and showed that these BMDCs can differentiate into inflammatory cells and microglia in the cortex. Similar studies can be performed in tumor models to study BMDC engraftment into irradiated tumor vasculature, which may influence tumor recurrence. 120
In addition to BMDCs, recruitment of other immune cells in response to treatment can be visualized. For example, Cummings and colleagues directly visualized cutaneous DCs using fluorescently labeled MHCII antibody to monitor changes in DC density on irradiation. 130 In agreement with ex vivo results, they demonstrated that irradiation causes migration of DCs outside the irradiated field, which triggered a cytotoxic T-cell response (Figure 6A). DC-triggered T-cell response has been investigated in other therapeutic contexts. For example, T-cell interaction with CD11c+ DCs has been studied in a spontaneous breast cancer model, 131 as well as in subcutaneous and orthotopic kidney tumor models. 132 In the latter study, the authors showed that the proportion of T cells interacting with DCs increases after chemotherapy, suggesting that this interaction limits the antitumor effect of chemotherapy. Lastly, Ritsma and colleagues used the abdominal WC model and fluorescence microscopy to study CD8+ T-cell localization in the spleen, tracking acute T-cell infiltration by antigen challenge. 133 In tumor models, CD8+ T-cell infiltration into the tumor site has been studied by intravenous injection of T cells into tumor-bearing mice in the dorsal skinfold WC model. 134 The authors studied T-cell interaction with fibrosarcoma cells longitudinally, as well as the effect of T cells on tumor vasculature. They observed increased vessel leakiness as the T cells entered the tumor. This observed phenomenon may explain why adaptive T-cell transfer causes solid tumors to regress from the lesions' core and suggests that adaptive transfer of T cells may synergize with radiation therapy by targeting hypoxic areas of a solid tumor. 135

Examples of imagingmodalities for immunomodulation, cancer stemcells (CSCs), and invasion. (A) Major histocompatibility complex (MHC) class II+ cell population in the mouse ear before (left) and after (right) irradiation. Anti-MHCII was used to label cutaneous dendritic cells, and images were obtained at 55 and 64 mm in depth from the ventral surface of the ear. Images adapted with permission from Cummings RJ et al. 130 Scale bar = 50 mm. (B) Tumor propagation by CSCs (green) and non-CSCs (cyan) with respect to FITC-dextran–labeled vasculature (pink) 35 (top) and 38 (bottom) days following injection. CSCs and non-CSCs were enriched from human glioma cells based on CD133 expression and were transduced to express yellow fluorescent protein (YFP) and cyan fluorescent protein (CFP), respectively. The cells were coinjected into the cortex of a mouse and imaged through a cranial window chamber. Following injection, CSCs outgrow non-CSCs to forma tumor. Images adapted with permission fromLathia JD et al. 143 Scale bar = 100 μm. (C) Extravasation of PC14-PE6 lung carcinoma cells (red) with respect to FITC-dextran–labeled vasculature (green), imaged through a cranial window. The tumor cells were injected through the carotid artery. The images demonstrate development of metastasis in the brain over time. Images adapted with permission from Macmillan Publishers Ltd: Nature Medicine, Kienast Y et al. 158 , copyright 2010. Scale bar = 100 μm.
Multiple immune cells are modulated on irradiation, thereby affecting tumor response. The imaging methods discussed above demonstrate the imaging of both myeloid and lymphoid-lineage cells, suggesting a wider application of these methods. Studies have demonstrated in vivo visualization of immune cells within and outside the radiation field in a longitudinal manner, as well as cell-to-cell crosstalk, which directly influence tumor response to radiation. When such techniques are combined with imaging of tumor cells, complex interactions and timescales thereof between immune and tumor cells may be visualized, thereby elucidating the postirradiation effect of immunomodulation on tumor response.
CSCs and Radioresistance
CSCs are a population of cells that self-renew and generate the heterogeneous cell lineages that comprise tumors. 136 In general, CSCs are thought to be intrinsically resistant to radiation compared to non-CSC populations as experimental studies suggest that a high proportion of CSCs in the tumor correlates with greater radioresistance. 137 Some mechanisms for intrinsic radiosensitivity of CSCs have been elucidated. For example, CSCs contain increased levels of free radical scavenging substances, resulting in low levels of reactive oxygen species that mediate cell killing after radiation exposure. 138 In addition, DNA checkpoint activation in CSCs leads to efficient repair of radiation-induced DNA strand breaks, contributing to CSC survival. 139 Furthermore, radiation may induce CSC cycle redistribution and consequent repopulation, negatively affecting therapeutic outcome of conventional fractionated radiation therapy regimens. 136 Importantly, the genotypic and phenotypic characteristics of CSCs may vary within the tumor due to the influence of the heterogeneous tumor microenvironment on CSC maintenance. In fact, there are three potential niches for maintenance of stemness: hypoxic, perivascular, and invasive niches. Such variable niches are composed of heterogeneous cell types, as well as matrix and signaling proteins that contribute to CSC maintenance and activity. 137 Thus, identification of the spatial distribution of CSCs, for example by imaging techniques, is critical to understand the mechanisms of CSC maintenance and the subsequent influence of CSCs on therapeutic outcome. In addition, predictive markers of CSCs and/or identification of a CSC niche may enable spatially localized delivery of higher doses of radiation to the CSC population, in a manner that is similar to dose-painting for hypoxic regions, which is expected to improve local tumor control.136,137
Imaging CSCs
To our knowledge, intravital microscopic imaging of CSCs in vivo has yet to be applied in the context of radiobiology. Rather, various imaging methods have been used to identify CSCs in heterogeneous cell populations. Specifically, whole-body imaging techniques, such as radionuclide imaging, MRI, bioluminescence, and whole-body fluorescence imaging, have been used to image CSCs. 140 For example, CD44 was used as a marker to identify patient-derived orthotopic breast CSCs using a dual-function reporter for bioluminescence and fluorescence to detect spontaneous lung and lymph node metastasis. 141 In addition to directly labeling CSCs based on their surface marker expression, proteasome activity has been used as a marker of CSCs for whole-body imaging. 142 In a study by Vlashi and colleagues, glioma cells were engineered to express fluorescence for 26S proteasome, which has reduced activity in CSCs. 142 They showed that the CSCs localized around blood vessels, indicating that glioma stem cells reside in a perivascular niche. Importantly, they also showed that there was reduced proteosomal activity in cells that survive fractionated radiation, indicating that radiation may accelerate repopulation of those CSCs. Although this study did not use intravital microscopy techniques, their model could easily be applied to study the microscopic CSC niche and the response of CSCs to radiation in vivo over time.
Other studies have used intravital microscopy for imaging CSCs. A study by Lathia and colleagues used a fluorescent labeling approach for CSCs in primary glioblastoma cells grown inside a cranial WC 143 (Figure 6B). In this study, CSCs were enriched based on CD133 expression, fluorescently labeled, and mixed with non-CSCs for transplantation into the cortex. Using multiphoton microscopy, they imaged CSCs during the course of tumor progression and determined that a small fraction of tumor cells can propagate a heterogeneous tumor. They also showed that CSCs can generate cells that no longer express the stem cell marker, demonstrating the transition between stem cell states. This study identified the need to study CSC plasticity, defined as the gain or loss of function. To enable imaging of CSC plasticity, Zomer and colleagues developed a mouse model to perform lineage tracing in a primary mammary tumor model. 47 In this study, a mammary WC model was used with a spontaneous breast cancer model modified to express the Cre-inducible Confetti construct for lineage tracing. This lineage tracing approach successfully identified a population of cells with stem cell capacities, including those that do not express established CSC markers. The CSC population identified using the lineage tracing approach induced tumor regrowth by dividing into differentiated tumor cells, as well as giving rise to new tumor cells with stem cell capacities that can be clonally expanded.
Techniques using whole-body and intravital microscopy imaging of CSCs, mostly fluorescence based, are emerging. However, CSC imaging can be challenging because there is no single universal marker for CSCs. Moreover, as described above, the function of CSCs and their expression of biomarkers at various stages of differentiation must be carefully considered to fully appreciate their role in radiobiological processes. An appropriate cell labeling method to identify CSC populations in vivo is needed before intravital optical imaging can be used to visualize the effect of radiation on CSCs. Such CSC imaging technique can be combined with other imaging techniques described earlier, such as those for vasculature and hypoxia, to investigate the effect of CSC niche on radioresistance by combining imaging for tumor cells and CSCs.
Radiation-Induced Tumor Invasion
The effect of radiation on promoting metastasis has been well documented based on clinical observation. 144 Importantly, metastasis can be found in both previously irradiated normal tissues and areas of local tumor irradiation, 144 suggesting that both stromal and tumor cells alter their phenotype to promote metastasis.
Experimental studies suggest that stimulation of stromal cells by irradiation contributes to tumor invasiveness. 145 For example, HIF-1 expression, which can be upregulated by irradiation, induces expression of various proteases that can promote collagen degradation. 146 This effect contributes to extracellular matrix (ECM) remodeling, characterized by degradation of basement membranes, which can in turn promote tumor invasion. 147 Similarly, radiation-induced inflammation has been correlated with increased expression of multiple factors such as cytokines146,148 and promigratory molecules, 149 thereby promoting formation of metastases. Bouchard and colleagues suggest that antiinflammatory agents may improve radiation therapy outcomes by preventing radiation-induced cancer cell migration. 149 Furthermore, fibroblasts and myofibroblasts are the stromal components that can promote tumor cell invasion. Early in vitro data suggest that irradiated fibroblasts accelerate invasion and motility of cancer cells through enhanced tumor– stroma interactions mediated by transforming growth factor P (TGF-P) signaling pathways. 145 Fibroblasts are also transformed into myofibroblasts through the same pathway; myofibroblasts deposit collagen and promote desmoplasia, which is characterized by disruption of tissue structure, to facilitate tumor invasion.150,151 Myofibroblasts may also produce cytokines that initiate vascular growth and promote tumor invasiveness. 150 In addition to radiation-induced changes to the stromal components, some irradiated solid tumors may themselves alter their phenotype to promote invasion. For example, radiation induces epithelial-mesenchymal transition (EMT), also through activation of TGF-P signaling pathways. 152 EMT is characterized by a decrease in epithelial markers such as E-cadherin and cytokeratin and an increase in mesenchymal markers such as N-cadherin and vimentin. EMT has been linked to the aggressiveness of metastatic disease, which may be due to increased migratory capacity, stemness, and intrinsic resistance to therapies. 153 EMT may be induced by other stimuli, such as hypoxia, promoting increased invasion and metastasis, increased CSC renewal, and radioresistance. 154 These experimental studies suggest a complex effect of radiation on tumor stromal and tumor cells to promote invasion and metastasis.
Imaging Tumor Invasion
Although the emerging evidence that radiation treatment can induce a more migratory and thus invasive tumor cell phenotype is intriguing, establishing the clinical importance of these findings will take time. Intravital imaging may play an important role in further elucidating the biological mechanisms at play. For example, it has been shown that the TGF-β signaling pathway can be induced by radiation, leading to a variety of downstream effects, including enhanced invasiveness. A study by Giampieri and colleagues visualized TGF-β signaling activity using breast cancer cells engineered to express the fluorescence reporter for TGF-β. 155 In this study, the authors showed that TGF-β is required for metastasis via blood vessels but not lymphatics, indicating that tumor cells use heterogeneous routes to metastasize. They also studied expression of vimentin, one of the hallmark biomarkers for EMT, and observed its heterogeneous expression in the tumors, although no correlation was found for vimentin expression levels with respect to TGF-β signaling. Stromal cells such as myofibroblasts may also be visualized, although expression of markers such as α-smooth muscle actin is not specific. 27
Intravital microscopy can also be used to study the dynamic in vivo processes of tumor cell invasion and metastasis in relation to stromal cell components. Tumor cell motility was studied using B16F10 melanoma cells grown in a dorsal skinfold WC, demonstrating invasion along collagen-rich dermal tissue. 156 In addition, metastasis of murine mammary and human fibrosarcoma cells to the lung following intravenous injection was studied using intravital imaging to track metastatic colonies over time. 157 Through a cranial window, melanoma and lung cancer cells were seen metastasizing to the brain, revealing the process of cancer cell extravasation and consequent micrometastasis formation 158 (Figure 6C). In the abdominal WC model for liver, imaging of metastasis of colorectal tumors into the liver identified distinct steps in metastasis formation and migratory behavior at different stages of metastasis. 133 This study also demonstrated the interaction between tumor cells and ECM based on collagen imaging to provide a better understanding of metastasis formation.
Experimental evidence suggests that a complex interplay of components within the tumor and stroma influences tumor invasion on irradiation. As described above, intravital microscopy enables visualization of cytokine signaling using engineered cells and of the invasive process. In addition, stromal components such as collagen, which is involved in invasion, can also be visualized using microscopy. 159 Together, intravital microscopy can elucidate radiation-induced dynamic changes of multiple components within the tumor and stroma, as well as visualize the consequent metastatic process.
Application of Intravital Microscopy to Experimental Radiobiology: Limitations and Future Potential for Translational Impact
Technical advances in intravital microscopy have broadened the field of modern preclinical radiobiology through investigations of radiation-induced effects on tumor vasculature, hypoxia, and the immune system in vivo. However, other critical components of the tumor microenvironment, such as CSCs and stromal cells, may also substantially influence the antitumor effectiveness of radiation therapy and therefore treatment outcome. Since the intravital microscopy techniques for studying these components are currently available in experimental models, a unique, novel opportunity exists for in-depth studies of radioresponse using intravital microscopy.
Until now, many experimental studies have used noninvasive volumetric imaging methodologies, such as MRI and PET, to visualize the dynamic state of the tumor microenvironment, including tumor hypoxia, perfusion, and metabolism.11,160,161 The preclinical use of such volumetric imaging strategies aligns well with the fact that they can also be used clinically for treatment planning, guidance, and response monitoring. For example, a metabolic imaging technique, [18F]fluorodeoxyglucose PET, has been introduced for use with cancer patients for staging and response monitoring purposes and is under clinical investigation as a predictive marker of radiation therapy outcome.162,163 However, the experimental use of these volumetric imaging techniques to study cellular and molecular changes in the heterogeneous tumor microenvironment is limited due to cost, complexity, and suboptimal image resolution. Optical imaging techniques not only overcome such limitations but also provide advantages, such as simultaneous assessment of anatomic, functional, and cellular responses to treatment and the ability to multiplex imaging signal while using multiple complementary modalities (e.g., fluorescence, OCT, photoacoustics, etc.) in vivo and longitudinally. Intravital microscopy techniques have evolved over the past few years, in particular, to become a common laboratory tool for studying multiple cellular and molecular dynamic biological systems. In fact, the durability of intravital imaging techniques is represented by an increasing number of publications on novel animal models for intravital imaging of various organs that enable investigation of orthotopic tumor progression, stromal effects, and distant metastasis. Thus, intravital microscopy can be applied in experimental radiobiological studies using orthotopic tumor models. However, there are a few technical issues with intravital imaging methods that need to be considered prior to their use, including (1) imaging depth in tissues, (2) duration of each imaging session and length of an experiment, and (3) cell labeling techniques. Depth of tissue penetration, a major limitation of optical imaging techniques, is particularly relevant for fluorescence microscopy imaging, although the use of multiphoton and near-infrared light can improve the depth ofpenetration compared to single-photon confocal imaging. OCT and photoacoustic imaging have a greater depth of penetration (up to several millimeters in depth), providing structural images of tumor and vasculature with high resolution. The duration of each imaging session is limited by anesthesia, whereas the length of the experiment is limited by the use of a WC model. Typically, the dorsal skinfold WC model can be used for up to 3 weeks, and other WC models can be used anywhere between hours and weeks. Therefore, investigation of long-term biological effects, such as chronic response to an intervention (e.g., fractionated radiation treatment regimens) or metastasis formation, may be challenging. Although some of the issues associated with duration of the experiment can be addressed by implanting tumor cells in animals and allowing the tumors to grow to a useful experimental size prior to WC surgery, 35 long-term changes to tumors can only be observed in a typical in vivo xenograft tumor model together with histopathologic analysis. Finally, labeling of target cells to be imaged, particularly with an antibody, requires consideration of interference with cellular function. For example, Kilarski and colleagues used a less specific surface marker to label DCs rather than the more common surface marker CD11c, which is required for cell migration, to minimize the influence of labeling on DC function. 164 Alternatively, transgenic mice expressing CD11c under vector control131,132 can be used to avoid labeling and its potential interference with biological function of the target cell.
Despite the technical challenges discussed above, intravital microscopy is becoming increasingly relevant and informative in a variety of fields. Many of the techniques described above, including novel imaging probes, such as those used for oxygen measurement, and engineered cells and animal models that enable imaging of particular cell lineages, enable visualization of multiple cellular and molecular changes to tumors and their microenvironment. Image analysis and quantification methods, such as those developed for vascular measurements, further make intravital microscopy a convenient experimental tool. Moreover, tumors can be harvested at any time to validate in vivo observations, including investigation of gene expression and signaling pathways, for example. Importantly, the emergence of many primary-derived tumor cell lines represents a new avenue for studying radioresponse in patient-derived samples where clinical outcome data are available. Such additional capabilities make intravital imaging a valuable tool for studying tumors and their microenvironment at a cellular and a molecular level in vivo. These imaging tools, combined with animal models and small animal irradiation technologies, enable new ways to study the effect of radiation on each component of the tumor microenvironment, as well as to evaluate the effect of emerging combination therapies, such as radiation with antiangiogenic agents, hypoxia-targeting agents, or CSC-targeting agents. Collectively, the available tools and oncology model systems contribute to the durability of the field of preclinical radiobiology and can be applied to study the various in vivo processes related to tumor and normal tissue radiobiology, thereby informing the design of effective therapies while accelerating the pace of research toward translation of new treatment strategies.
Footnotes
Acknowledgments
Financial disclosure of authors and reviewers: None reported.
