Abstract
For intraoperative imaging, antibodies labeled with both a radionuclide and a fluorophore may be used to tag the tumor lesion with a radiolabel and a fluorescent signal at high tumor to background ratios. However, labeling antibodies with fluorescent moieties may affect the in vivo behavior of the antibody depending on the dye to antibody substitution ratio. To investigate the optimal substitution ratio for use in dual-modality image-guided surgery, we conjugated three different antibodies, MN-14 (anti-CEACAM5), girentuximab (anti-CAIX), and cetuximab (anti-EGFR), with both diethylene triamine pentaacetic acid (DTPA, for labeling with 111In) and IRdye 800CW at dye to antibody ratios of 0, 1, 1.5, 2, and 3 and assessed in vivo behavior. Biodistribution studies showed that at high dye to antibody ratios, liver uptake of the dual-labeled antibodies increased, whereas tumor uptake decreased. Conversely, very low ratios may not be optimal either because in that case, only a few antibody molecules will be dual-labeled (i.e., contain both a DTPA and an IRDye 800CW moiety), which may complicate interpretation of dual-modality data. The present study shows that, provided that the chelator to antibody ratio is high enough, a dye to antibody ratio in the range of 1 to 1.5 is optimal for antibody-targeted dual-modality imaging applications. However, the optimal configuration is antibody dependent and should be determined for each dual-labeled antibody individually.
FOR MANY TYPES OF CANCER, radical surgical resection of tumor tissue is the cornerstone of effective treatment. 1 To identify tumor lesions, surgeons mostly rely on palpation and visual inspection, together with any preoperatively acquired data. Intraoperative tumor localization may be improved by image-guided surgery. For example, for sentinel lymph node mapping, peritumoral injection of dyes, radioisotopes, or nanocolloids containing a combination of these may be used to guide lymph node dissection.2,3 However, for assessment of tumor borders and resection margins, a tumor-targeted approach is needed. 4 For example, antibodies seem particularly suitable for this purpose because they specifically accumulate in tumor tissue and can be relatively easily conjugated to the signaling molecules of interest.
Antibodies bound to various radionuclides have been used for radioguided surgery of tumors using a gamma probe. 5 However, gamma-probe tumor detection does not provide an accurate delineation of the tumor and resection margins. Tumor-targeted fluorescence imaging could allow real-time delineation of tumor lesions intraoperatively, but the penetration depth of emitted light in biological tissue is limited to a few millimeters. 6 Therefore, a combination of these techniques may be most appropriate for image-guided surgery. Combining a radiotracer for the detection of tumor tissue and an optical tracer for subsequent accurate tumor delineation enables dual-modality image-guided surgery, as has been shown in preclinical studies.7–17
For this approach, antibodies directed against a tumor-associated antigen have to be conjugated with either a multifunctional single attachment point 18 or sequentially with both a chelator for radiolabeling and a fluorophore. Most chelating agents can be conjugated to antibodies in a ratio of up to approximately 5:1 without having significant detrimental effects on biodistribution and immunoreactivity of the antibody.10,19–22 This is not the case for fluorophores. For conjugated fluorescent dyes, the dye to antibody ratio (average number of fluorophores conjugated to one antibody molecule, also known as the degree of labeling [DOL] or the fluorophore/protein [F:P] labeling ratio) may influence the in vivo behavior of the labeled antibody, showing increased liver accumulation at higher ratios. 10 At low ratios, however, a substantial amount of the injected antibody will remain unlabeled. Since the actual number of fluorophores conjugated to an antibody follows a Poisson distribution,23–25 at a molar substitution ratio of 1, 37% of the injected antibody molecules are unlabeled, 37% will contain one fluorophore moiety, 18% will contain two fluorophores, and 8% will contain three or more fluorophores (Table 1). These differently labeled antibodies may show different biodistributions and different in vivo contributions from the overall fluorescent signal in the tumor.
Percentage of Antibody Molecules with 0, 1, 2, 3, 4, and > 4 Fluorophores at Different Dye to Antibody Ratios, According to the Poisson Distribution*

For example, at a dye to antibody ratio of 1, on average, 18% of all antibody molecules will contain 2 fluorophore moieties.
The aim of this study was to assess the optimal dye to antibody ratio for dual-labeled antibodies, considering the theoretical Poisson distribution of conjugated fluorophores, and in vivo biodistribution data of three different dual-labeled antibodies. For this purpose, MN-14 (antibody against carcinoembryonic antigen [anti-CEACAM5]), girentuximab (antibody against carbonic anhydrase IX [anti-CAIX]), and cetuximab (antibody against epidermal growth factor receptor [anti-EGFR]) were labeled with 111In–diethylene triamine pentaacetic acid (DTPA) and IRDye 800CW at dye to antibody ratios of 0, 1, 1.5, 2, and 3, and tumor and normal tissue uptake was determined in corresponding tumor mouse models.
Materials and Methods
Materials
Cetuximab (Erbitux) was purchased from Merck (Darmstadt, Germany). mMN-14 was a gift from Immunomedics, Inc. (Morris Plains, NJ). Girentuximab was a gift from Wilex AG (Munich, Germany). IRDye 800CW-NHS was purchased from Li-COR Biosciences (Lincoln, NE). 111In was purchased from Mallinckrodt (Petten, the Netherlands). Isothiocyanate–diethylene triamine pentaacetic acid (ITC-DTPA) (p-SCN-Bz-DTPA) was purchased from Macrocyclics (Dallas, TX). Human cell lines FaDu-hSCC, LS174T, and SK-RC-52 were purchased from the American Type Culture Collection (ATCC, Manassas, VA).
Dual-Labeled Antibody Synthesis: Conjugation of Fluorophore to the Antibodies
First, the antibodies were dialyzed against phosphate-buffered saline (PBS) using a 20,000 molecular-weight cutoff (MWCO) Slide-a-Lyzer Dialysis Cassette (Life Technologies, Inc.) to remove any additives. Dialysis was performed at 4°C for 5 to 7 days with buffer replacements every 24 hours. To the conjugation reaction mixture containing the antibody, 1.0 M pH 8.5 metal-free phosphate buffer was added at one-tenth of the calculated final volume to adjust the pH to 8.5. Finally, the IRDye 800CW was dissolved in dry dimethyl sulfoxide (DMSO) (3–10 mg/mL) and added to the conjugation reaction mixture. The DMSO volume never exceeded 10% of the total volume. To obtain the appropriate dye to antibody molar substitution ratio, the correlation between the molar conjugation reaction ratio and molar substitution ratio was assessed and plotted. According to these data, the necessary conjugation reaction ratio for the dye and the antibody was calculated. The final antibody concentrations were 3.5 to 4 mg/mL. The reaction mixtures were allowed to react at room temperature for 75 minutes on an orbital shaker (200 rpm).
Dual-Labeled Antibody Synthesis: Conjugation of Chelating Agent to Antibody
Subsequently, ITC-DTPA was conjugated to the dye-labeled antibodies (the labeled antibodies containing only a fluorescent dye, not a radiolabel). For this conjugation, the procedure was followed as per the dye conjugation, except now a phosphate buffer of pH 9.5 was used to optimize the yield of the conjugation reaction. A conjugation reaction ratio (ITC-DTPA:antibody) of 20:1 was used. ITC-DTPA was dissolved in dry DMSO (8–15 mg/mL) and added to the reaction mixture in the same manner as the IRDye 800CW. The mixtures were again allowed to react at room temperature for 75 minutes on a stirring plate (200 rpm).
Purification of Dual-Labeled Antibody
The synthesized dual-labeled antibodies were purified using 20,000 MWCO Slide-a-Lyzer Dialysis Cassettes against 0.25 M ammonium acetate (pH adjusted to 5.5). Dialysis was performed at 4°C for 5 to 7 days with buffer replacements every 24 hours.
Radiolabeling
The dual-labeled antibodies were labeled with 111In at 0.67 MBq/μg. First, indium chloride solution was buffered using a double amount of 0.5 M 2-(N-morpholino)ethanesulfonic acid (MES). After addition of the antibody, the solutions were left to react for 20 minutes at room temperature. Reaction volumes were typically around 50 μL, although dependent on the 111In activity. Subsequently, 50 mM ethylene diamine tetraacetic acid (EDTA) solution was added at 10% of the total reaction volume to complex unincorporated 111In.
Substitution Ratio
The dye to antibody substitution ratios were determined by measurement of absorbance at 280 and 774 nm with a 3% correction due to IRDye 800CW absorption at 280 nm (according to the manufacturer's protocol) using an Ultrospec 2000 spectrophotometer (Pharmacia Biotech, Amersham, UK). Quantification of the IRDye 800CW concentration was done using a calibration curve of IRDye 800CW in PBS.
Quality Control
Radiochemical purity was analyzed with instant thin-layer chromatography (ITLC) using a 0.1 M sodium citrate buffer as the mobile phase. The ITLC was read using a Fuji Bas-1800 II phosphor imager and analyzed using Aida software v.4.21.033 (Raytest, Straubenhardt, Germany). The integrity of the dual-labeled antibodies was assessed by sodium dodecyl phosphate polyacrylamide gel electrophoresis (SDS-PAGE) using an 8% gel. A sample of 10 μg was prepared in a 20 μL volume under nonreducing conditions and denatured by heating to 95°C for 15 minutes. The results were interpreted by visual inspection. The immunoreactive fraction (IRF) of the dual-labeled antibodies was estimated using an assay as described by Lindmo and colleagues. 26 In short, a small amount of labeled antibody (±200 Bq) was added to a serial dilution of cells. The IRF was then calculated by plotting a double inverse plot of the bound fraction against the cell concentration and taking the inverse of the y-axis intercept.
Animals
All experiments were approved by the Animal Welfare Committee of the Radboud University Medical Center and were conducted in accordance with the Revised Dutch Act on Animal Experimentation (Wet op de Dierproeven). Female BALB/c nude mice (6–8 weeks) were purchased from Janvier (le Genest-Saint-Isle, France). The animals were marked by a tail tattoo and were housed in enriched filter-top cages (five animals per cage). They were allowed to acclimatize to their new environment for a week before starting the experiment. The welfare of the animals was assessed daily by biotechnicians, and mice were weighed during the experiment to monitor possible weight loss. The mice were given ad libitum access to food and water. Three days before the imaging experiments, the animals were put on a chlorophyll-free diet to reduce autofluorescence.
Biodistribution Studies
For each antibody, 25 mice were subcutaneously injected in their right flank with a 200 μL cell suspension in RPMI (3 × 105, 2 × 106, 5 × 106 cells for LS174T, SK-RC-52, and FaDu-hSCC, respectively). The tumor cell lines used in the MN-14, girentuximab, and cetuximab experiments were LS174T, SK-RC-52, and FaDu-hSCC, respectively. After 2 weeks of tumor growth (tumor size ≈ 0.2 mL), the 25 animals were randomly divided into 5 groups of 5 animals, each being injected with 30 μg of dual-labeled antibody with a dye to antibody substitution ratio of 0, 1, 1.5, 2, or 3. In two animals of each group, single-photon emission computed tomographic/computed tomographic (SPECT/CT) images were acquired (see below). These animals were injected with 30 μg of dual-labeled antibody (20 MBq). The rest of the animals were injected with 2 MBq of dual-labeled antibody. After 48 hours, the animals were anesthetized (isoflurane), scanned, euthanized, and dissected. The tumor, organs of interest, and blood were weighed and activity was measured using a Wizard automatic gamma counter (PerkinElmer, Waltham, MA). Data were analyzed using Prism v.5 (GraphPad Software, La Jolla, CA).
SPECT/CT
The animals selected for SPECT/CT imaging were scanned using a MILabs USPECT-II/CT system (MILabs, Utrecht, the Netherlands) with a 1.0 mm mouse collimator and 48 bed positions. The total scan time was approximately 35 minutes. The dynamic scanning setting was used. Reconstructions and three-dimensional images were made using the MILabs reconstruction software and an ordered-subset expectation maximalization algorithm.
Optical Imaging
Fluorescence images were acquired of all animals using the Xenogen - IVIS Lumina II system (Caliper Life Sciences, Hopkinton, MA). The excitation wavelength used was 745 nm, and the filter used was the indocyanine green filter set (810–885 nm). The field of view was set to ‘C’ and F/stop to 2. The animals were scanned for 1 to 5 minutes. Images were corrected for autofluorescence using an autofluorescence background image recorded at an excitation wavelength of 675 nm.
Statistics
All statistical analyses were performed using SPSS 20 (IBM Corp., Armonk, NY). Comparisons of the uptake of the dual-labeled antibodies in tumor, liver, and tumor to liver and tumor to blood ratios were analyzed for each antibody with univariate analyses of variance with post hoc Bonferroni corrected tests. Statistical significance was set at a p level of less than .05.
Results
Synthesis of Dual-Labeled Antibodies
To enable synthesis of antibodies with accurate predefined dye to antibody ratios, the molar substitution ratio was plotted against the molar conjugation ratio for the three different antibodies (Figure 1). Although these relationships followed a descriptive logarithmic curve, this pattern was different for the three antibodies tested. For this study, all dual-labeled antibodies were within a 0.2 margin from their target dye to antibody ratio, except for cetuximab target ratio 3, which was 2.7. The DTPA to antibody ratio was estimated from the ITLC analyses as described. These ratios were 2.8, 3.9, 4.1, 3.6, and 4.2 for girentuximab; 2.1, 2.3, 1.9, 2.3, and 2.6 for MN-14; and 2.1, 2.6, 2.8, 3.4, and 2.2 for cetuximab for the dye to antibody ratios of 0, 1, 1.5, 2, and 3, respectively. All preparations were labeled with 111In with a radiochemical purity of at least 95%, except for the dual-labeled cetuximab with a dye to antibody ratio of 3, which had to be purified on a PD-10 column after labeling. The resulting radiochemical purity of this preparation after purification was > 98%.

Conjugation ratio (molar ratio between the number of dye molecules per antibody molecule in the conjugation reaction mixture) versus the dye to antibody substitution ratio for the three antibodies.
Quality Control
SDS-PAGE showed that all dual-labeled antibodies were intact and remained intact for at least 1 month when stored at 4°C in the dark. Lindmo assays were carried out to assess the IRF of all dual-labeled antibody preparations. The IRF of MN-14 was more than 60% for each conjugate. For girentuximab, the IRF was more than 85% for each conjugate. For cetuximab, the IRF was more than 75% for each conjugate. No correlation between the dye to antibody ratio and the IRF could be found for any of the dual-labeled antibodies.
Biodistribution Studies
To obtain quantitative information on the biodistribution of the dual-labeled antibodies following intravenous injection, mice were euthanized, tissues were dissected and weighed, and activity was measured. The results are shown in Figure 2. The antibodies without a fluorophore (dye to antibody ratio 0) served as a control. For all three antibodies, the same trend was observed, with lower tumor uptake and higher liver accumulation at higher dye to antibody ratios. However, the extent of this effect was different for the three different antibodies tested. For example, the biodistribution of dual-labeled girentuximab seems only to be affected significantly at dye to antibody substitution ratios exceeding 2. This can also be observed from the tumor to liver ratios (Figure 3), where all dual-labeled antibodies show the same trend, although, unexpectedly, the tumor to liver ratio of MN-14 at a dye to antibody ratio of 1 is lower than ratio 1.5 (p = .056). Tumor to blood ratios were not significantly different.
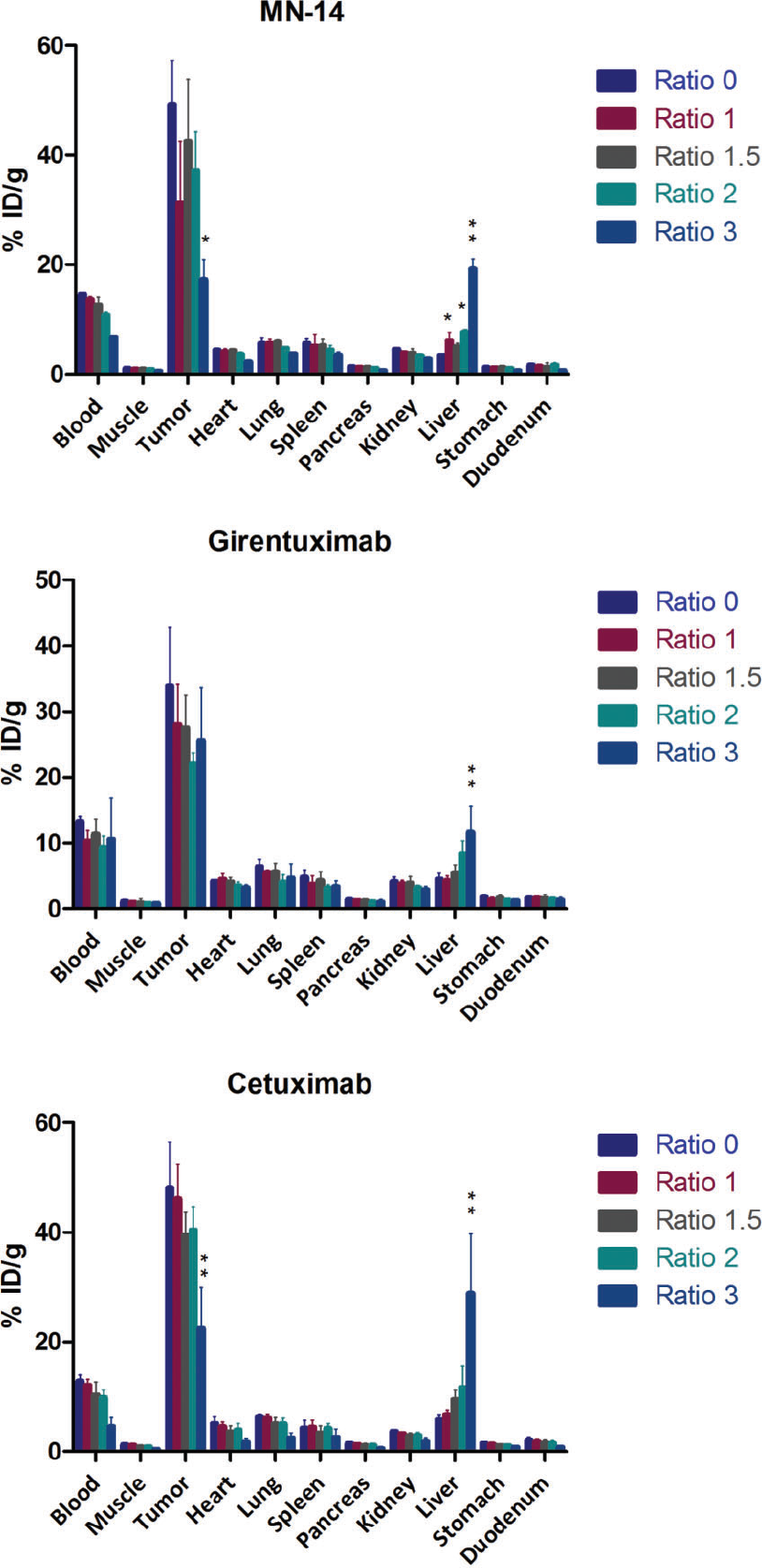
Biodistribution of dual-labeled MN-14, girentuximab, and cetuximab at different dye to antibody substitution ratios. With increasing substitution ratios, accumulation of the dual-labeled antibody in the tumor decreased, whereas accumulation in the liver increased for all antibodies. *p < .05, **p < .001 compared to the control dye to antibody ratio 0.
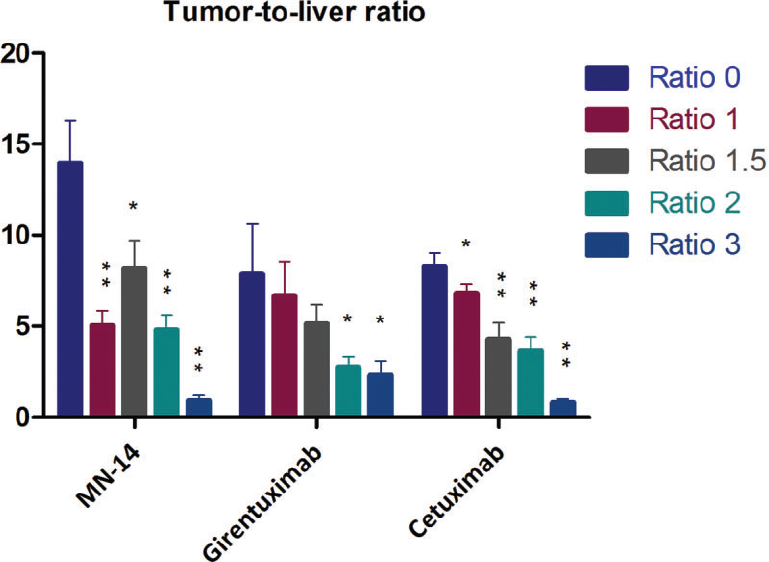
Tumor to liver uptake ratios of the biodistribution data presented in Figure 2. *p < .05, **p < .001 compared to the control dye to antibody ratio 0.
Imaging
SPECT/CT images were consistent with the results of the biodistribution studies showing the same trend: decreased accumulation in the tumor and increased accumulation in the liver at increasing dye to antibody ratios. In Figure 4, the SPECT/CT images are shown of mice bearing SK-RC-52 tumors in the right flank after injection of dual-labeled girentuximab. Fluorescence images of the same mice are also shown in Figure 4. Scaling of all images was kept just above the noise threshold. These images were not interpreted quantitatively.

SPECT images (top) and fluorescence images (bottom) of mice bearing SK-RC-52 tumors in the right flank 48 hours after injection of 111In-DTPA-girentuximab-IRdye 800CW with dye to antibody substitution ratios of 0, 1, 1.5, 2, and 3 (from left to right), illustrating tumor accumulation of the dual-labeled antibody and increased liver uptake at higher substitution ratios.
Discussion
Intraoperative imaging is a promising tool to improve the outcome of oncologic surgery. Dual-modality approaches combining radionuclide detection and fluorescence imaging may overcome the limitations of the two separate techniques. Preclinical studies using dual-labeled antibodies have shown promising results, both in pre- and intraoperative imaging and image-guided surgery, although the success of this approach is dependent on the expression of the antigen on the tumor.7–17 One of the main challenges for clinical application of this technique, however, is to achieve sufficiently high signal intraoperatively for tumor detection and even more to visualize tumor resection margins. In this study, we investigated the optimal configuration of dual-labeled antibodies for dual-modality image-guided surgery.
In particular, the dye to antibody ratio of dual-labeled antibodies seems to be an important factor determining in vivo behavior. 10 At ratios of 2 or higher, the uptake of the dual-labeled antibody in the liver increases, whereas the uptake in the tumor decreases compared to a radiolabeled antibody (a labeled antibody containing only a radiolabel, not a fluorescent dye), possibly due to the reduced circulation time of the antibody.10,15 Although higher dye to antibody ratios may yield more fluorescent signal, too high ratios may be undesirable because quenching due to resonance energy transfer between fluorophores on the same protein molecule will reduce the fluorescent signal. This may occur particularly when two dye moieties are located closer together, although the size of this effect may be fluorophore dependent.27–29 On the other hand, dye to antibody ratios could also be too low. In this case, many antibody molecules remain unlabeled because labeled proteins follow a discrete (Poisson) distribution. This distribution pattern is supported theoretically for discrete, low-probability events (a protein may have tens of potential conjugation sites, of which only a few are labeled), and was confirmed experimentally using Cy3-labeled anti-EGFR antibodies.23,25
For dual-labeled antibodies, Poisson distributions for both labels may be assumed as long as enough potential binding sites remain and no interactions between the labels occur. This means that if the chelator to antibody ratio is 3 and the fluorophore to antibody ratio is 1.5, 74% of the antibody molecules are dual labeled (i.e., contain at least one chelator and at least one fluorophore moiety). For low ratios, this shifts to a suboptimal distribution; for example, at ratios of 1 and 0.5 for the two labels, only 25% of all antibody molecules are dual labeled, whereas more than half (53%) only contain either a chelator or a fluorophore. As these different monolabeled antibodies may show different in vivo behavior, this may complicate interpretation of the dual-modality imaging signals. In this case, the biodistributions of the two monolabeled antibodies may be determined to show that both antibodies have the same in vivo behavior. 30 In addition to labeling at random sites of the antibody, newer labeling strategies are currently under investigation, for example, site-specific techniques based on click chemistry. 31
As a consequence of both the theoretical considerations and the in vivo data, the optimal dye to antibody ratio of dual-labeled antibodies is around 1.5 as these ratios do not significantly impact on immunoreactivity or in vivo behavior of the labeled antibody. This ratio was found to be higher (approximately 2–5) when only immunoreactivity of the labeled antibody was considered. 25 In the current study, we conjugated antibodies at dye to antibody ratios of 0, 1, 1.5, 2, and 3. Since the Poisson distributions of these labeled antibodies are known, and the tumor and liver uptake has been measured, one could theoretically calculate the tumor to liver ratio for antibodies tagged with 0, 1, 2, 3, or > 3 separately by solving five equations with five unknowns. For example, for girentuximab, at a dye to antibody ratio of 1.5, 22% of the antibodies contain zero fluorophores, 33% contain one fluorophore, and 25% contain two fluorophores, etc. (see Table 1), and the resulting uptake in the tumor and liver is 27.6 %ID/g and 5.5 %ID/g, respectively. So, in the equation, 0.22*Ab0 + 0.33*Ab1 + 0.25*Ab2 + 0.13*Ab3 + 0.07*Ab>3 = 27.6% ID/g in the tumor (Abx denotes an antibody containing x number of fluorophores). Similar calculations can be made for liver uptake and for all five dye to antibody ratios, which can then be solved for Abx. However, solving these mathematical equations did not yield reliable results because small variations in the biodistribution data lead to large variations in the estimated tumor to liver ratios for the antibodies tagged with 0, 1, 2, 3, or > 3 labels separately. Still, the biodistribution data of the dual-labeled antibodies with different dye to antibody ratios and the corresponding Poisson distributions of the fluorophore strongly suggest that antibodies containing a higher number of fluorophores have lower tumor to liver ratios.
Cohen and colleagues found that at dye to antibody ratios of 2, tumor uptake decreased significantly. 10 In our study, we confirmed these results and comment that ratios lower than 1 are not optimal either. In addition, we show that the optimal ratio may be different for different antibodies. For example, tumor uptake decreased significantly at a dye to antibody ratio of 3 for dual-labeled MN-14 and cetuximab but not for girentuximab. The tumor to liver ratio, however, showed the same trend for all three antibodies at different dye to antibody ratios. Similar to the antibody-dependent correlation between the molar conjugation ratio and the molar substitution ratio (see Figure 1), the optimal dye to antibody ratio also seems to be different for different dual-labeled antibodies. Explanations for this might be found in the physicochemical properties of the antibody, which may change if a fluorophore such as IRDye 800CW is conjugated to it. Increased lipophilicity of the labeled antibody may lead to increased albumin binding 32 and subsequent liver uptake. Also, the negative charge of the fluorophore will result in an overall decrease in net positive charge of the antibody, which leads to decreased tissue retention and increased blood clearance. 33 This mechanism has been proposed for chelator-conjugated antibodies as well at high substitution ratios. 34 However, whether the overall charge of the antibody will change to a critical value, changing in vivo behavior when conjugated to a negatively charged dye moiety, may be dependent on the isoelectric point of the unlabeled antibody, which may be different for different antibodies.
Radiolabeled antibodies have been used clinically for oncologic imaging and therapy for years.35,36 More recently, the first clinical application of targeted fluorescence imaging has been shown in ovarian cancer patients using a folate receptor α–targeted fluorescent agent. 37 This technique shows the high potential for fluorescence intraoperative imaging. Targeted dual-modality imaging using antibodies labeled with both a radionuclide and a fluorophore may enable both preoperative PET or SPECT imaging to assess disease extent and subsequent intraoperative radiodetection and fluorescence imaging for image-guided surgery. Since the preclinical results of this approach are promising, clinical translation of dual-modality imaging will be the next step.
The success of oncologic surgery depends on the accurate identification and subsequent radical resection of tumor tissue. Dual-labeled antibodies can be used to tag the tumor lesion with a radiolabel and a fluorescent signal at high tumor to background ratios. In the current study, we showed that at high dye to antibody ratios, the liver uptake of the dual-labeled antibodies increased, whereas tumor uptake decreased. Conversely, very low ratios may not be optimal either because in that case, only a few antibody molecules will be dual-labeled (i.e., contain both a DTPA and an IRDye 800CW moiety), which may complicate interpretation of dual-modality data. Considering tumor uptake, a dye to antibody ratio of 1 to 1.5 is optimal for antibody-targeted dual-modality imaging applications provided that the chelator to antibody ratio is high enough. However, pharmacokinetic effects due to fluorophore conjugation may be antibody dependent and should be determined for each dual-labeled antibody individually.
Footnotes
Acknowledgments
Financial disclosure of authors: David M. Goldenberg is an employee, stock holder, and patentee of Immunomedics, Inc.
Financial disclosure of reviewers: None reported.
