Abstract
The goal of this work was to study the efficacy of the positron emission tomography (PET) tracers 2-deoxy-2-[18F]fluoro-D-glucose ([18F]FDG) and 64Cu-diacetyl-bis(N4-methylthiosemicarbazone) ([64Cu]ATSM) and in monitoring placental and fetal functional response to acute hyperoxia in late-term pregnant mice subjected to experimentally induced chronic hypoxia. E15 mice were maintained at 12% inspired oxygen for 72 hours and then imaged during oxygen inhalation with either [18F]FDG to monitor nutrient transport or 64Cu-ATSM to establish the presence of hypoxia. Computed tomography (CT) with contrast allowed clear visualization of both placentas and fetuses. The average ratio of fetal to placental [18F]FDG uptake was 0.45 ± 0.1 for the hypoxic animals and 0.55 ± 0.1 for the normoxic animals, demonstrating a significant decrease (p = .0002) in placental function in dams exposed to chronic hypoxic conditions. Hypoxic placentas and fetuses retained more 64Cu-ATSM compared to normoxic placentas and fetuses. Herein we report first-in-mouse PET imaging of fetuses employing both tracers [18F]FDG (metabolism) and 64Cu-ATSM (hypoxia). [18F]FDG PET/CT imaging allowed clear visualization of placental-fetal structures and supported quantification of tracer uptake, making this a sensitive tool for monitoring placental function in preclinical rodent models. These measurements illustrate the potentially irreversible damage generated by chronic exposure to hypoxia, which cannot be corrected by acute exposure to hyperoxia.
THE PLACENTA MAINTAINS dynamic fetal homeostasis by performing a wide range of physiologic functions, which, after birth, are carried out by multiple organs, such as the kidney, liver, gastrointestinal tract, lungs, and endocrine glands. The primary functions of the placenta are to mediate the transfer of respiratory gases, water, ions, and nutrients; provide an immunologic barrier between fetus and mother; and produce and secrete a vast array of hormones, cytokines, and signaling molecules. Malformations in the structure or function of the placenta, the uterus, or the connection between them can lead to life-threatening sequelae for the mother and progeny.1,2 Maternal and fetal blood flow within the placenta and exchange of nutrients and oxygen between the two vascular systems are essential aspects of placenta-fetal units. Insufficiencies in these functions may result in a variety of severe prenatal and postnatal syndromes, including intrauterine growth restriction (IUGR), preeclampsia, and fetal death in utero, as well as a predisposition to systemic cardiovascular and pulmonary vascular dysfunction.3–6 Furthermore, increasing epidemiologic evidence has demonstrated that aberrant placental function during fetal development can have long-term negative impacts on human adult health, including increased risk of ischemic heart disease.7,8 Acute administration of oxygen is frequently used to reduce fetal hypoxic stress in pregnant women.
Characterizing placental function is vital for understanding fetal physiology, growth patterns, and complications during pregnancy. Thus, understanding the factors that affect transport across the placenta can impact both broad areas of basic research and clinical practice. Noninvasive imaging methods can play a key role in assessing the efficiency and integrity of this transport function. A wide variety of preclinical magnetic resonance imaging (MRI) studies of placenta structure and function have been described.9–13 By contrast, relatively few studies employing radionuclides to study placenta have been reported. Although such experiments may not readily translate to the clinic, they can provide valuable functional and metabolic information in the preclinical setting. Recently, several groups have developed nonhuman-primate animal models for investigation of drug transfer from mother to fetus.14–16 However, radionuclide-based studies employing nonhuman primates are prohibitively expensive, and the development of positron emission tomography (PET) in small animal models of pregnancy could provide important insights into placental function; however, relatively few of these studies have been conducted.17,18 Herein we report first-in-mouse PET studies employing the tracers 2-deoxy-2-[18F]fluoro-D-glucose ([18F]FDG) (metabolism) and 64Cu-diacetyl-bis(N4-methylthiosemicarbazone) (64Cu-ATSM) (hypoxia).
[18F]FDG PET has long been used to measure glucose transport in many disease states and thus is an ideal tracer for our proof-of-principle studies.19–23 The overarching goal of our work is to enhance our knowledge of placental-fetal transport of nutrients, in this case glucose, to provide a better understanding of the unique properties of mammalian reproduction. To this end, studies to assess the ability of FDG PET to measure changes in placental function were conducted in normal pregnant animals and pregnant mice exposed to chronic hypoxic conditions. All mice were subject to acute hyperoxia during FDG-PET imaging.
Materials and Methods
All animal experiments were conducted according to the guidelines of the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health under a protocol approved by the Washington University Animal Studies Committee. Timed pregnant CD-1 (Crl:CD1(ICR)) mice were obtained from Charles River Laboratories (Wilmington, MA).
Hypoxia studies were conducted using a hypoxia chamber (BioSpherix, Lacona, NY), as previously reported. 24 Briefly, mice were fed a standard rodent chow and housed in a 12:12-hour light-dark cycle in room air. The hypoxic atmosphere was maintained at fraction of inspired oxygen (FIO2) = 12% with a ProOx 110 gas oxygen controller (BioSpherix). The hypoxic cohort was exposed to an atmosphere with FIO2 = 12% beginning on embryonic day 15 (E15) and maintained in a hypoxic environment for 72 hours. Mice were imaged immediately following hypoxic exposure.
Radiochemistry
[18F]FDG was produced on a GE Fastlab and 64Cu was produced on a CS-15 biomedical cyclotron, both in the Washington University Cyclotron Facility.25,26 64Cu-ATSM was prepared according to previously published methods.26,27
Small Animal PET/Computed Tomography
Prior to imaging, animals were anesthetized with 2% isoflurane in either oxygen (FDG study) or air (64Cu-ATSM study) and the tail vein was catheterized using 0.025 OD × 0.012 ID Micro-Renathane tubing (Braintree Scientific, Braintree, MA) attached to a heparinized 0.3 cc, 29-gauge, half-inch insulin syringe filled with saline. One hundred microcuries of either [18F]FDG (hypoxic n = 6, normoxic n = 3) or 64Cu-ATSM (hypoxic n = 4, normoxic n = 4) was then injected coincidentally with the start of the PET scanning. Dynamic PET images were acquired for 60 minutes. The PET study was followed by the injection of 3.5 μgI/g of body weight Omnipaque (iohexol, 350 mgI/mL, GE Healthcare, Princeton, NJ) equivalent to a dose of 0.028 mmol/kg inorganic iodine through the tail-vein catheter, coincidental with the initiation of 6-minute computed tomography (CT). PET data were reconstructed using standard methods with the maximum a posteriori probability (MAP) algorithm. CT images were overlaid on the PET images, and volumes of interest (VOI) were drawn using CT anatomic guidelines. Additional details can be found in the supplementary information (Figure S1 and Figure S2, online version only).
Statistical Analysis
Data are expressed as mean ± SD unless noted otherwise. Statistical significance was determined with an unpaired, two-tailed Student t-test and 95% confidence level, and p values less than .05 were considered significant. All data were analyzed and plotted using GraphPad Prism version 6.0 (GraphPad Software, La Jolla, CA).
Results and Discussion
To evaluate [18F]FDG as an imaging modality for studying placental function, we investigated the effect of chronic hypoxia on maternal-fetal glucose transport. E15.5 mice were exposed to a hypoxic environment for 72 hours and imaged at E18.5 with [18F]FDG as described in the Methods section. To achieve the contrast needed for the anatomically guided structure determination, Omnipaque, an iodine-based CT contrast agent, was used, as described in Methods. Imaging with CT contrast allowed clear visualization of placentas and fetuses, as demonstrated by coregistered PET/CT axial, coronal, and sagittal projections (Figure 1, A and B). Time-activity curves (TACs) obtained from the VOI analysis showed a rapid uptake of [18F]FDG and slow washout in normoxic and hypoxic animals (Figure 1, C and D). To investigate the efficiency of the nutrient transfer from placenta to fetus, the ratios of fetal-to-placental dynamic [18F]FDG uptake were calculated from the TAC data collected between 10 and 60 minutes following tracer injection. A comparison of the ratios of fetal to placental FDG uptake showed a significant decrease (p = .0002) in placental function in dams exposed to hypoxic conditions (Figure 2A). The average ratio between fetuses and placentas for the hypoxic animals was 0.44 ± 0.1 (n = 42), whereas the normoxic animals demonstrated a ratio of 0.55 ± 0.1 (n = 25). The mean [18F]FDG uptake by hypoxic and normoxic placentas and fetuses is compared in Figure 2B. [18F]FDG uptake was significantly higher in the fetuses and placentas of normoxic animals (p = .0001 for both pairs), suggesting a transfer defect in hypoxic animals due to the chronic hypoxic injury, which is not corrected by acute hyperoxia (Table 1). Interestingly, there was no significant difference in mean tracer uptake between the hypoxic placentas and fetuses (p = .059). Overall, [18F]FDG PET demonstrated the robustness required to detect small differences in placental-fetal nutrient transfer.
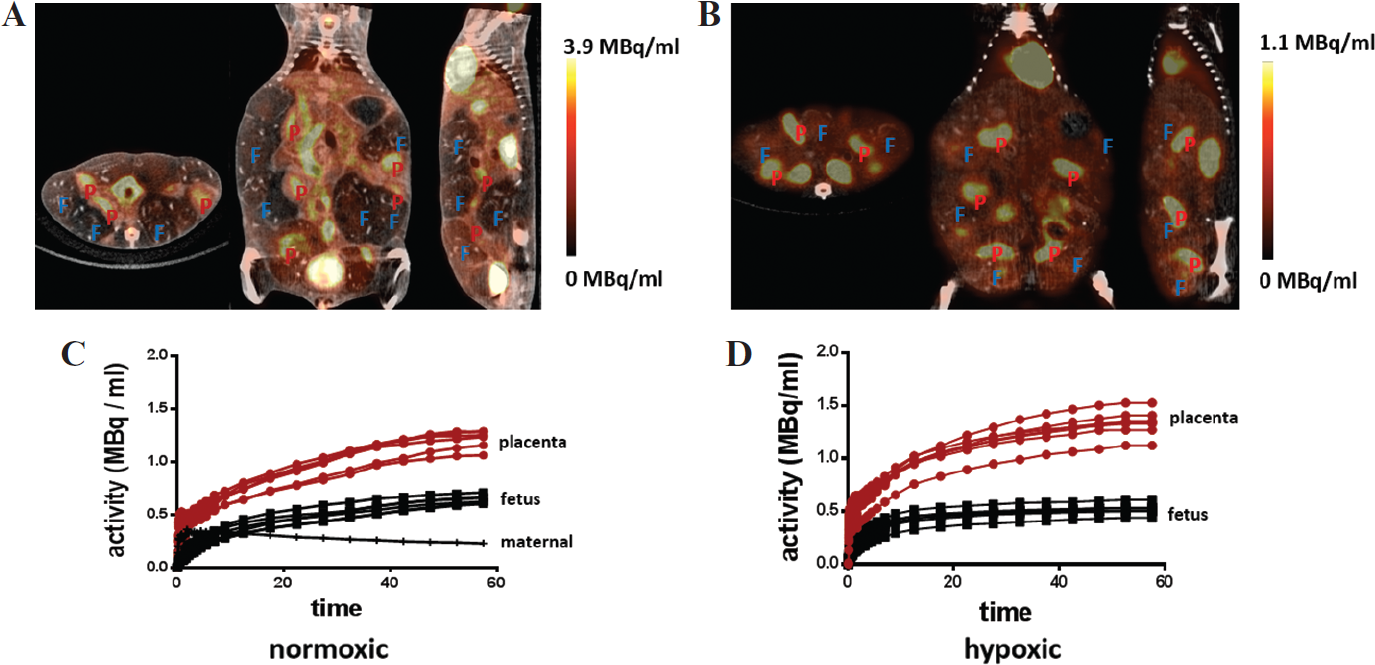
[18F]FDG PET/CT imaging of glucose transport. [18F]FDG PET/CT images (A and B) and time-activity curves (C and D) for E18.5 normoxic (A and C) and hypoxic (B and D) mice. F = fetus; P = placenta.

[18F]FDG fetal placental ratios as a measurement of placental function. A, Average ratio of fetal-to-placental uptake of [18F]FDG in hypoxic and normoxic animals. B, Mean [18F]FDG uptake in fetuses and placentas of hypoxic and normoxic animals.
One Way Anova Multiple Comparison

To evaluate the exposure to chronic tissue hypoxia, hypoxia-challenged animals and normoxic animals were imaged during normal air inhalation using 64Cu-ATSM, a tracer that is specifically retained in tissues with low O2 tension.28–30 Systemic chronic hypoxia was induced for 72 hours as described in Methods. After injecting 100 μCi of 64Cu-ATSM, dynamic disintegration data were collected for 30 minutes. A comparison of 64Cu-ATSM retention in the tissues of animals exposed to systemic chronic hypoxia or normoxia showed significantly more 64Cu-ATSM associated with hypoxic tissues (Figure 3A, p = .033). The mean uptake of 64Cu-ATSM in hypoxic maternal tissue (muscle) was 32,500 ± 6,300 Bq/mL, whereas the normoxic muscle tissue retained an average of 15,800 ± 6,600 Bq/mL. Similar to [18F]FDG uptake, there was no significant difference in 64Cu-ATSM uptake among maternal tissues, fetuses, or placentas of hypoxic animals (Figure 3B). As shown in Figure 3C, the mean 64Cu-ATSM uptake in hypoxic (45,400 ± 17,100 Bq/mL, n = 37) and normoxic (28,600 ± 16,200 Bq/mL, n = 26) placentas (Figure 3C) was significantly different (p = .0002). A similar difference was observed when comparing the fetuses of hypoxic (31,700 ± 24,600 Bq/mL, n = 37) and normoxic (15,800 ± 5,600 Bq/mL, n = 26) animals, where p = .002 (Figure 3D). These data indicate the persistence of chronic hypoxia and suggest that the observed [18F]FDG transfer defect may be due to tissue injury resulting from chronic hypoxia. Further studies are required to determine if this effect is entirely irreversible.

64Cu-ATSM imaging of hypoxia. A, Mean 64Cu-ATSM retention in maternal muscle tissue (thigh) of hypoxic and normoxic animals. B, Mean 64Cu-ATSM retention in maternal muscle, placentas, and fetuses of hypoxic animals. Mean 64Cu-ATSM retention in (C) placentas and (D) fetuses of hypoxic and normoxic animals.
PET was used in earlier studies of maternal-to-fetal transfer in large animals, such as sheep 31 and nonhuman primates.32,33 Benveniste and colleagues used [18F]FDG PET and MRI to monitor the movement of labeled compounds across placenta and their trafficking within the fetal tissues in healthy Macaca radiata. 15 In a similar study in sheep under conditions of acute hypoxia performed by Tchirikov and colleagues, the investigators observed a decrease in fetal organ perfusion, whereas glucose uptake by the fetus was unaffected. 31 Although expensive and logistically complicated, these studies were instrumental in motivating the use of PET for investigating placental-fetal trafficking in preclinical models. Moreover, a broad range of experimental disease states, both acute and chronic, are available in small rodents, enabling a variety of studies of gestational pathologies and their effect on placental-fetal transfer.
In this work, we showed that excellent coregistration of PET and contrast-enhanced CT data could be achieved. However, due to the limitations in spatial resolution of the small animal PET scanner, accumulation of the labeled compound in specific fetal organs could not be determined. Nonetheless, [18F]FDG PET could detect small differences in tracer transfer caused by an experimentally induced pathology that was corroborated by an independent PET tracer. Overall, the use of PET/CT in small animal systems to track placental-fetal transport of a wide range of labeled compounds in normal and/or disease states could lead to a better understanding of prenatal pathologies.
Conclusions
[18F]FDG PET imaging was used to monitor the effects of chronic systemic hypoxia in a late-term murine pregnancy model. [18F]FDG PET images provided the high image contrast and spatial resolution required for accurate quantification of tracer distribution between placenta and fetus, demonstrating that [18F]FDG PET is a sensitive tool for monitoring placental function in preclinical mouse models.
Footnotes
Acknowledgments
We thank the Washington University Preclinical PET/CT Imaging Facility for the imaging studies and the Washington University Cyclotron Facility for FDG and 64Cu production.
Financial disclosure of authors: This work was supported by the US/Israel Binational Science Foundation, grant 2011405, and pilot funding from Mallinckrodt Institute of Radiology, Washington University.
Financial disclosure of reviewers: None reported.
