Abstract
The aim of this study was to determine whether the severity of contact hypersensitivity reactions (CHSRs) can be observed by noninvasive in vivo optical imaging of matrix metalloproteinase (MMP) activity and whether this is an appropriate tool for monitoring an antiinflammatory effect. Acute and chronic CHSRs were elicited by application of a 1% trinitrochlorobenzene (TNCB) solution for up to five times on the right ear of TNCB-sensitized mice. N-Acetylcysteine (NAC)-treated and sham-treated mice were monitored by measuring ear swelling and optical imaging of MMP activity. In addition, we performed hematoxylin-eosin staining and CD31 immunohistochemistry for histopathologic analysis of the antiinflammatory effects of NAC. The ear thickness and the MMP activity increased in line with the increasing severity of the CHSR. MMP activity was enhanced 2.5- to 2.7-fold during acute CHSR and 3.1- to 4.1-fold during chronic CHSR. NAC suppressed ear swelling and MMP signal intensity in mice with acute and chronic CHSR. During chronic CHSR, the vessel density was significantly reduced in ear sections derived from NAC-treated compared to sham-treated mice. In vivo optical imaging of MMP activity measures acute and chronic CHSR and is useful to monitor antiinflammatory effects.
T CELL–DEPENDENT autoimmune diseases such as rheumatoid arthritis and psoriasis vulgaris are classified as delayed-type hypersensitivity reactions (DTHRs), orchestrated by interferon-γ-producing CD8+ (Tc1) and CD4+ (Th1) T cells.1,2 Most DTHRs, such as cutaneous contact hypersensitivity reactions (CHSRs), are accompanied by accumulations of polymorphonuclear neutrophils (PMNs), which elicit an oxidative burst and are critically involved in the pathogenesis of several autoimmune diseases. 3 The oxidative burst is characterized by a rapid release of reactive oxygen species (ROS) generated by PMNs and macrophages.4,5 ROS regulate inflammatory immune responses through interactions with nuclear factor (NF)- κB or p38 mitogen-activated protein (MAP) kinase signaling pathways 6 and display either tissue-destructive or -protective effects. 7
Our previous work focused on studying the effector phase in a murine model of acute and chronic trinitrochlorobenzene (TNCB)-induced CHSR.3,8 We observed a strongly reduced PMN accumulation and oxidative burst in inflamed ear tissue of TNFR1−/− and mast cell–deficient mice with acute TNCB-induced CHSR.3,8 Using in vivo positron emission tomography (PET) and 18F-RGD-peptide, which binds to activated αVβ3-integrin, we distinguished acute from chronic TNCB-induced CHSR and followed angiogenesis during the progression of local inflammation. 9
As chronic inflammation is highly associated with angiogenesis, 9 antiinflammatory or antiangiogenic drugs can suppress chronic cutaneous DTHR. 10 The underlying mechanisms of inflammation-induced angiogenesis are driven by hypoxia 11 and proinflammatory mediators such as tumor necrosis factor (TNF), macrophage inflammatory protein (MIP)-2, nitric oxide (NO), angiopoietin, and matrix metalloproteinases (MMPs).3,12 MMPs are a family of enzymes that degrade components of the extracellular matrix by cleaving molecules such as collagen that are essential for tissue conversion, cell migration, and angiogenesis. 13 MMP activity has been investigated in recent years in several experimental disease models, such as cardiovascular diseases, 14 rheumatoid arthritis, 15 chronic obstructive pulmonary disease, 16 and tumor progression and metastasis. 17 MMP activity can be examined noninvasively in vivo by an activatable optical imaging fluorescent probe with an MMP-specific cleavage site. The activatable probe is designed in a way that without MMPs, a peptide sequence fixes two fluorochromes closely together, causing a quenching of the fluorescence signal by fluorescent resonance energy transfer (FRET). 18 Cleaving of the MMP-specific peptide sequence activates the fluorochromes, enabling the measurement of the fluorescence signal by optical imaging. 19
Noninvasive in vivo detection of MMP activity by MMP-specific activatable fluorescent probes has been used in models of experimental rheumatoid arthritis, 20 allergic airway inflammation, 21 cerebral ischemia, 22 aortic aneurysms, 23 myocardial infarction, 24 atherosclerotic plaques, 25 and various tumors, 26 but to our knowledge, not in T cell–mediated cutaneous DTHR. Thus, the aim of our study was to analyze whether in vivo measurements of MMP activity can determine the severity of acute and chronic CHSR and to monitor antiinflammatory effect. Our experimental CHSR model is a unique tool to study the mechanisms of DTHR and includes an internal naïve control on the left ear as we elicit TNCB-specific CHSR exclusively on the right ear.
As an antiinflammatory approach, we used N-acetylcysteine (NAC), a sulfur-containing cysteine amino acid derivative, which is found naturally in food and is a powerful antioxidant that can decrease ROS levels in vitro 27 and in vivo. 28 Furthermore, NAC suppresses the activation of NF-κB 29 and p38 MAP kinase, 30 both essential for the activation of endothelial cells and angiogenesis.31,32 Data about the antiinflammatory effect of NAC on acute CHSR are controversial.33–35 Thus, we elucidated the antiinflammatory potential of NAC in chronic CHSR.
Materials and Methods
Animals
In our experiments, we used 8- to 12-week-old female C57BL/6 mice from Charles River Laboratories (Sulzfeld, Germany). Animal experiments were approved by the Regierungspräsidium Tübingen.
In Vivo Experiments
Mice were sensitized on the shaved abdomen (size approximately 2 cm × 2 cm) by the application of 80 μL 5% TNCB (dissolved in a 4:1 mixture of acetone/Miglyol 812; SASOL, Witten, Germany). One week later, mice were challenged with 20 or 80 μL of 1% TNCB (dissolved in a 9:1 mixture of acetone/Miglyol 812) on both sides of the right ear to elicit acute CHSR. To induce chronic CHSR, mice were recurrently challenged on the right ear every 2 to 3 days for a total of up to five ear challenges. The application of 80 μL 1% TNCB solution coated a larger ear skin area than 20 μL and thus was expected to cause a more severe inflammatory reaction. Ear swelling was quantified by measuring the ear thickness with a micrometer (Kroeplin, Schlüchtern, Germany) before the TNCB ear challenge and 12 to 24 hours afterwards. All measurements of the challenged right ears were compared to the nonchallenged left ears of the mice. The ear swelling response data were obtained from different experiments (20 μL TNCB challenge solution: two experiments (n = 10 and n = 8); 80 μL TNCB challenge solution: three experiments (n = 10, n = 6, n = 5). The experimental mice examined by optical imaging in Figure 1 (n = 3; two groups) are included in the ear swelling data set in Figure 2, A to D. Similarly, the mice from the optical imaging study in Figure 3 (first challenge: n = 3, two groups; fifth challenge: n = 3, two groups) were included in the ear swelling data set of Figure 4B.
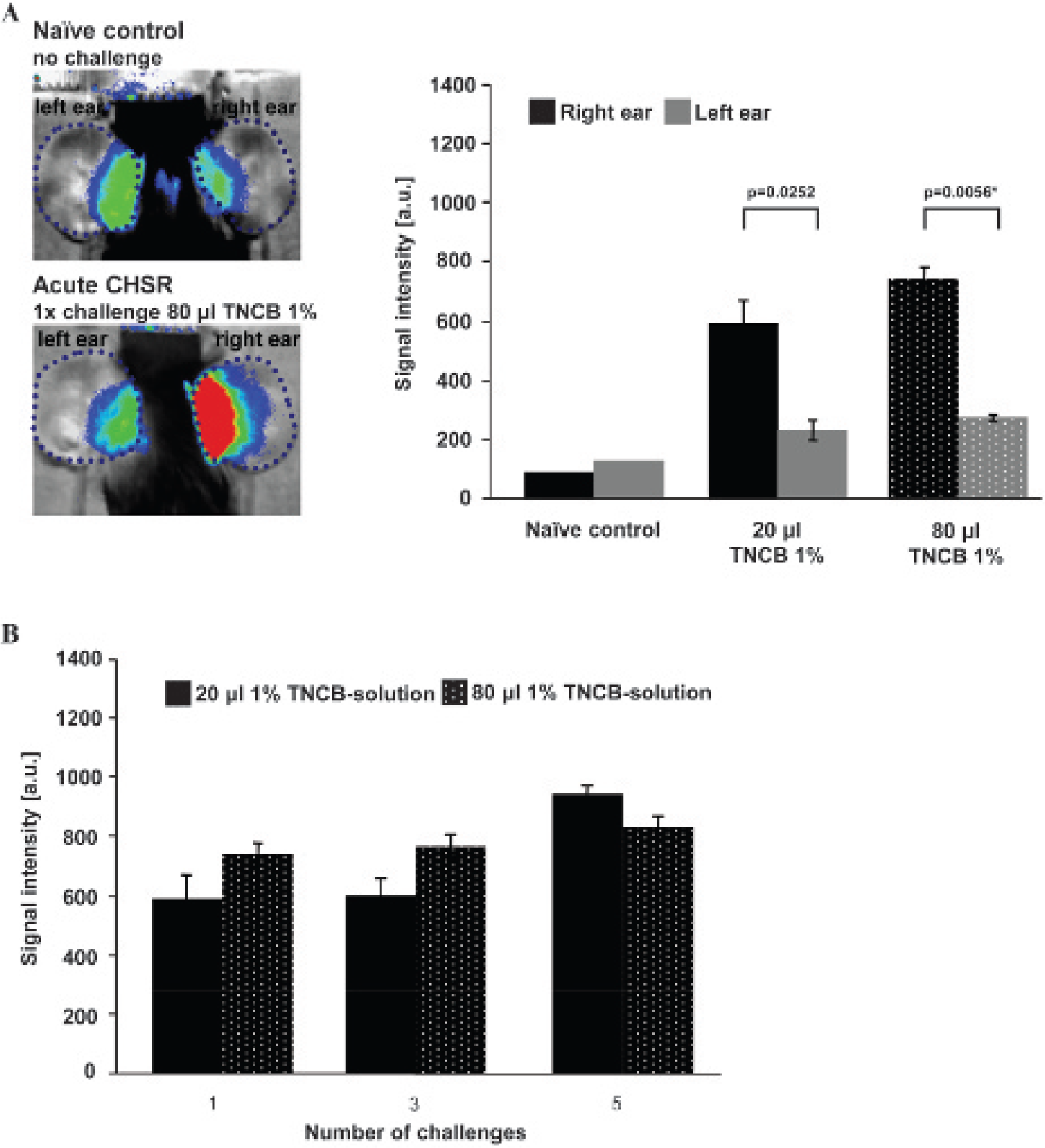
Noninvasive in vivo measurement of MMP activity. A, TNCB-sensitized mice were challenged once with 20 or 80 μL TNCB solution to elicit acute contact hypersensitivity reaction (CHSR). The signal intensity of the MMP-activatable probe was measured by optical imaging and indicates highly increased MMP activity in the challenged right ear compared to the untreated left ear (20 μL, n = 3; 80 μL, n = 3; naïve control, n = 1). The upper-left image is a representative fluorescent image of the signal intensity in the ears of a naïve mouse. The lower-left image shows the signal intensity of a mouse that was challenged once on the right ear with 80 μL TNCB solution. The p values without asterisks were not significant after correcting for multiple testing. B, The MMP signal intensity in inflamed ears slightly increased in chronic cutaneous CHSR (n = 3). The data are presented as the mean ± SEM.

Course of ear swelling during acute (A–B) and chronic (C–D) TNCB-induced contact hypersensitivity reaction (CHSR). A and B, 20 μL: TNCB sensitized: n = 19; 80 μL: TNCB sensitized: n = 16. C and D, Chronic CHSR was induced by repetitive ear challenges up to five times with 20 or 80 μLTNCB solution and yielded a continuous increase in ear thickness. The unchallenged left ear served as a control (20 μL, n = 21; 80 μL, first and second challenge n = 16, third and fourth challenge n = 12, fifth challenge n = 18). The ear swelling of the right ear of each mouse was always higher than on the left ear. The data are presed as the mean; error bars show ± 1 SEM. E, Hematoxylin-eosin histology of ear tissue derived from a naïve, non–TNCB-challenged mouse shows the physiologic state (left). Ears of mice with acute CHSR induced by 20 μL TNCB solution show strong edema and leukocyte infiltration, predominantly polymorphonuclear neutrophils (center, 12 hours after challenge). Ears with chronic CHSR show tissue remodeling characterized by acanthosis, hyperkeratosis, new blood vessel formation, and a dense leukocyte infiltrate (right, 12 hours after challenge, magnification 100×).

NAC protects against tissue conversion by inhibiting MMP activity. A, Noninvasive in vivo imaging of the MMP signal intensity in the right TNCB-challenged ear of a representative sham- (upper) or NAC-treated (lower) mouse with acute and chronic TNCB-induced contact hypersensitivity reaction (one or five 80 μL TNCB ear challenges). B, Quantitative analysis of the MMP signal intensity in the ears of NAC- or sham-treated mice shows an almost 50% reduction in MMP signal intensity values in NAC-treated littermates (n = 3; 80 μL TNCB ear challenge). The data are presented as the mean; error bars show ± 1 SEM. The p values without asterisks were not significant after correcting for multiple testing.

NAC suppresses acute and chronic TNCB-induced contact hypersensitivity reaction (CHSR). A and B, NAC reduced the TNCB-induced ear swelling response during acute and chronic CHSRs. The graphs represent the ear swelling in NAC- or sham-treated mice during acute (one ear challenge) chronic CHSR (five ear challenges) when 20 μL (A) or 80 μL (B) TNCB solution was applied to the right ear (20 μL: sham, n = 6; NAC, n = 7; 80 μL: sham, n = 6; NAC, n = 6). The data are presented as the mean; error bars show ± 1 SEM. The p values without asterisks were not significant (after correcting for multiple testing).
NAC Administration
NAC (5 mg/mL; Sigma-Aldrich, Steinheim, Germany) was added continuously to the drinking water, starting 2 days prior to the first TNCB ear challenge. Because mice drink approximately 6 mL per day, 36 the daily NAC incorporation was expected to be approximately 30 mg/mouse. The oral bioavailability ranges from 6 to 10% in humans.37,38 Sham-treated mice received normal drinking water. Ear swelling of sensitized mice was measured 12 and 24 hours after each TNCB ear challenge. Seven mice received 20 μL of 1% TNCB on the right ear, and six mice received 80 μL of 1% TNCB. We analyzed the ear swelling response after each challenge, with the exception of the third and fourth challenges with 80 μL of 1% TNCB, when only two mice were measured (see Figure 4).
Optical Imaging
We measured MMP activity using MMPsense680 (PerkinElmer, Waltham, MA), an MMP-activatable fluorescent optical imaging probe. According to the manufacturer, MMPsense680 is cleavable preferentially by MMP-9 and -13 but also by MMP-2 and MMP-3. 39 MMPsense680 was injected intravenously 12 hours after TNCB ear challenge, and in vivo optical imaging measurements were conducted 24 hours later. For in vivo investigations, we used a Hamamatsu Aequoria Dark Box and a C4880 Hamamatsu Dual Mode cooled charge-coupled device (CCD) camera (Hamamatsu Photonics Deutschland GmbH, Herrsching, Germany). The exposure time was 30 seconds using a 680 nm excitation filter and a 700 nm emission filter. To locate the ears, a bright-field image without the emission filter was recorded with an exposure time of 0.01 seconds. During optical imaging, mice were anesthetized by inhalation of isoflurane-O2 (1.5% Forane, Abbott GmbH, Wiesbaden, Germany) and placed on a heating pad to maintain body temperature between 36°C and 37°C. To measure the whole upper surface area of the mouse ears, the ears were carefully fixed on a flat black plate by a nylon thread (Figure S1, online version only). Regions of interest (ROI) were drawn on the right and left ears (as indicated by the dotted blue line in Figure 1A and 3A), enabling a semiquantitative analysis of the detected fluorescence light intensity in arbitrary units. The background signal was subtracted from the ROI mean values; however, we did not use an area-normalized unit but reported all photons counted in the specific ROI. Image analysis was performed with Wasabi Imaging Software (Hamamatsu).
Histology and Immunohistochemistry
Mice were sacrificed 12 hours after the last TNCB ear challenge. Ear tissue was fixed in 4% formalin. After embedding in paraffin, 5 μm sections were cut using a microtome (Leica, Wetzlar, Germany) and stained with hematoxylin and eosin (H&E) according to standard procedures. 3 An experienced dermatologist examined the H&E ear sections and analyzed them qualitatively.
For immunohistochemistry, mouse ears were embedded in Tissue-Tek (Sakura Finetek, Torrance, CA) and frozen in liquid nitrogen. Seven-micrometer sections were cut by a cryotome (Leica, Wetzlar, Germany) and placed on Superfrost/Plus slides (Menzel, Braunschweig, Germany). To detect blood vessels, ear sections were stained with a CD31 (PECAM-1) antibody (BD Pharmingen, Franklin Lakes, NJ) using a Vectastain ABC Kit (Vector Laboratories, Burlingame, CA). As positive controls, we used ear sections with vessels displaying a clear lumen and intraluminal erythrocytes (Figure S2, online version only). To determine unspecific staining, ear tissue was stained without using the primary antibody. Counterstaining was performed with hemalaun. CD31 slides were scanned using the Hamamatsu NanoZoomer (Hamamatsu) and analyzed in NDP.view2 software (Hamamatsu). One or two representative areas per sample were chosen in the middle section of the ear, where appropriate staining quality was visible. The stained vessels were counted and divided by the surface area (mean 0.7 ± 0.09 mm2 ± 1 SEM) to calculate the vessel density. As the surface areas in the ear sections varied due to the different ear swelling responses (the cross-section diameter) in the sham-treated and NAC-treated experimental groups, the vessel density represents the antiinflammatory effects of NAC much better than an absolute quantification of the vessels. Analysis of CD31 immunohistochemistry was not performed blinded as this issue is hard to achieve because the differences in CD31-stained ear slices between the sham-treated and the NAC-treated experimental groups were easily viewable.
Statistics
A paired, two-tailed Student t-test was used to examine the individual ear swellings from 12 to 24 hours after TNCB challenge (see Figure 2, A and B), whereas an unpaired two-sample, two-tailed Student t-test was used to make group comparisons of ear swelling between mice challenged with 20 or 80 μL TNCB (see Figure 2, A and B). Due to four comparisons, we adjusted the significance level by Bonferroni correction to 1.25%.
The course of ear swelling responses from the first to the fifth ear challenge (see Figure 2, C and D) was analyzed by analysis of covariance (ANCOVA) using a combination of “challenge” (as continuous variable) and “ear side” (TNCB-challenged right ear or untreated left ear as a nominal variable), nested under “mouse ID” (nominal variable with random effect).
A paired, two-tailed Student t-test was used to make group comparisons of optical imaging signal intensities of the right (TNCB challenged) versus the left (untreated) ears of the identical TNCB-sensitized mice (see Figure 1A). We adjusted the significance level to 2.5% according to Bonferroni correction for two comparisons. The course of optical imaging signal intensities was analyzed by ANCOVA using a combination of challenge (as continuous variable) and mouse ID (as a nominal variable with random effect; see Figure 1B).
Differences in both ear swelling responses (see Figure 4, A and B) and optical imaging signal intensities (see Figure 3B) between NAC- and sham-treated mice were examined by two-sample, two-tailed Student t-test. The significance level was adjusted in Figure 4, A and B, by Bonferroni correction to 1.67% due to multiple testing (three comparisons: 1× TNCB challenge: 12 and 24 hours; 5× TNCB challenge 12 hours). In Figure 3B, the significance level was adjusted to 2.5%, making a Bonferroni correction for two comparisons (1× TNCB challenge; 5× TNCB challenge).
A Wilcoxon test was used to compare the vessel density of NAC- and sham-treated mice (Figure 5C). The p values below .05 were considered statistically significant. Quantitative data are reported as means and standard errors of the mean (± 1 SEM).

NAC protects against angiogenesis during chronic contact hypersensitivity reaction (CHSR). A, Hematoxylin-eosin (H&E)-stained ear sections from NAC-treated mice with chronic CHSR show noticeably reduced ear thickness, edema, hyperkeratosis, acanthosis, infiltration of inflammatory cells, and number of blood vessels compared to sham-treated littermates (magnification: 100×; five 80 μL TNCB ear challenges). B and C, Quantitative analysis of the vessel density of CD31-positive cells in ear samples from NAC- or sham-treated mice with chronic CHSR (NAC treated, n = 8; sham treated, n = 10; five 80 μL TNCB ear challenges). The data are presented as the mean; error bars show ± 1 SEM.
Results
Our study focused on quantifying the severity of skin inflammation, enabling us to differentiate between acute and chronic CHSR by noninvasive in vivo optical imaging of MMP activity.
Course of Ear Swelling during Acute and Chronic TNCB-Induced CHSR
The initial ear challenge with 20 μL TNCB caused an increase in ear thickness in TNCB-sensitized C57BL/6 mice of 151 ± 14 μm (± 1 SEM) after 12 hours and 174 ± 16 μm after 24 hours (see Figure 2A).
Application of 80 μL TNCB solution caused no significant difference in ear swelling responses at 12 hours (175 ± 11 μm) and 24 hours (142 ± 14 μm) compared to ears challenged with 20 μL TNCB solution. Twenty-four hours after TNCB ear challenge with 80 μL TNCB solution, the ear swelling decreased significantly compared to 12 hours after challenge (see Figure 2, A and B).
Repetitive TNCB ear challenges, every 2 to 3 days for up to five exposures to 20 or 80 μL of 1% TNCB solution caused chronic CHSR. In chronic TNCB-specific CHSR provoked by 20 or 80 μL of 1% TNCB solution, we observed a continuous increase in ear thickness that reached 315 ± 31 μm 12 hours after the fifth 20 μL TNCB ear challenge and 310 ± 27 μm after the fifth 80 μL TNCB ear challenge (see Figure 2, C and D). Statistical data analysis using ANCOVA revealed that ear swelling was significantly higher in the TNCB-treated right ears than in untreated left ears 12 hours after every TNCB challenge. Ear swelling responses consistently increased over the course from the first to the fifth challenge regardless of the amount of TNCB solution used.
H&E histology of inflamed ears 12 hours after the first TNCB ear challenge showed strong edema, slight leukocyte infiltration dominated by PMNs, dilated vessels, and an unchanged epidermis during acute CHSR. In contrast, in ears with chronic CHSR, we observed massive hyperkeratosis, acanthosis, an increase in the number of blood vessels, and a dense infiltration of PMNs and lymphocytes (see Figure 2E).
Noninvasive In Vivo Measurement of MMP Activity
To investigate the dynamics of tissue conversion during acute and chronic CHSR, we measured MMP activity in vivo using optical imaging and a fluorescent probe activatable by several MMPs. The optical imaging probe was injected intravenously 12 hours after each TNCB ear challenge with 20 or 80 μL of 1% TNCB solution. We performed noninvasive in vivo optical imaging measurements after another 24 hours. Only a faint fluorescent signal was detected in a naïve control mouse, but acute CHSR increased the MMP-specific signal intensity (representing mainly MMP-9 and -13 activity) dramatically (see Figure 1A). After the first 20 μL TNCB ear challenge, we observed a 2.5-fold greater MMP signal in the TNCB-challenged right ears (595 ± 8 a.u.) compared to the untreated left ears (234 ± 3 a.u.). After application of 80 μL TNCB solution, the signal intensity of the TNCB-challenged right ear (745 ± 7 a.u.) was 2.7-fold greater than that of the contralateral ear (274 ± 1 a.u.; see Figure 1A).
In line with the severity of the cutaneous CHSR, we measured an increase in MMP signal intensity between the third (604 ± 6 a.u.) and the fifth TNCB ear challenge (948 ± 2 a.u.) when applying 20 μL TNCB but only a slight increase when applying 80 μL TNCB solution (third challenge: 771 ± 4 a.u.; fifth challenge: 835 ± 4 a.u.; see Figure 1B). The signal intensity did not increase significantly with the number of challenges for either the 20 μL or the 80 μL group.
NAC Suppresses Acute and Chronic TNCB-Induced CHSR
Next, we focused on antiinflammatory NAC. Thus, 5 mg/mL of NAC was added to the drinking water of experimental mice, which was expected to amount to a daily NAC dose of approximately 30 mg/mouse. Sham-treated mice received normal drinking water. NAC medication started 2 days prior to the first TNCB ear challenge. Twelve hours after the first 20 μL TNCB ear challenge, we observed reduced ear swelling compared to sham-treated littermates (see Figure 4A). Only slightly reduced ear swelling in NAC-treated mice was observed 12 hours after applying 80 μL of 1% TNCB solution (acute CHSR; see Figure 4B). After 24 hours, NAC displayed a significant antiinflammatory effect in mice challenged with 20 and 80 μL of 1% TNCB solution. During chronic CHSR after five challenges of the right ears, we measured a significantly reduced ear swelling response in NAC-treated mice compared to sham-treated mice. The antiinflammatory effect of NAC was even stronger in mice challenged with 80 μL of 1% TNCB solution. Ear swelling response in NAC- or sham-treated mice during the increasing severity of CHSR, while applying either 20 or 80 μL TNCB up to five times, showed a generally increased antiinflammatory effect of NAC over time (see Figure 4, A and B).
NAC Protects from Angiogenesis
We performed histopathologic analysis of H&E-stained ear sections derived from NAC- and sham-treated littermates 12 hours after the fifth ear challenge with 80 μL of 1% TNCB solution to evaluate the impact of NAC on chronic cutaneous DTHR. In ears from NAC-treated mice, we observed reduced edema, hyperkeratosis, acanthosis, inflammatory cell infiltrate, and blood vessel number compared to sham-treated mice (see Figure 5A). To quantify the number of blood vessels, we performed platelet endothelial cell adhesion molecule (CD31/PECAM) immunohistochemical staining (see Figure 5, B and C). We counted a significantly reduced number of blood vessels in ear sections derived from NAC-treated compared to sham-treated mice. Thus, we conclude that NAC significantly reduces blood vessel formation during chronic cutaneous CHSR (see Figure 5, B and C; Table S1, online version only).
NAC Protects from Tissue Conversion by Inhibition of MMP Activity
To investigate the impact of NAC on tissue conversion, we detected MMP activity in vivo in NAC- or sham-treated mice with acute and chronic CHSR by optical imaging. The MMP signal intensities in NAC-treated mice that were challenged one or five times with 80 μL of 1% TNCB solution were reduced by approximately 50% 12 hours after challenge compared to the ears of sham-treated littermates. These differences were statistically significant (see Figure 3). Repetitive TNCB ear challenges at the right ear elicit and reinforce CHSR accompanied by itching. Thus, the mice start scratching and thereby transfer small amounts of the TNCB-challenging solution to the contralateral left ear, causing a slight CHSR detectable by highly sensitive MMPsense optical imaging investigations (see Figure 5 and Figure S3, online version only).
We correlated ear swelling responses and MMPsense signal intensity from TNCB-challenged right ears of all experimental groups after one, three, and five TNCB ear challenges; the Pearson correlation coefficient for all measured mice was r = .74; for 80 μL of 1% TNCB solution, r = .75; and r = .50 for 20 μL of 1% TNCB solution (Figure S4, online version only).
Discussion
TNCB-induced CHSR is a well-established model for acute and chronic cutaneous DTHR. We recently showed that there is a close link between chronic inflammation and angiogenesis during TNCB-induced chronic cutaneous DTHR by in vivo 18F-RGD-PET targeting activated αVβ3-integrin. 9 Angiogenesis in inflammatory processes is caused by hypoxia 11 and proangiogenic mediators. Keratinocytes, mast cells, macrophages, endothelial cells, and other cell types secrete mediators such as TNF, MIP-2, NO, angiopoietin, and MMPs, which can promote angiogenesis.3,12 The mechanism of angiogenesis starts with vasodilatation and increased vessel permeability. Proteases such as MMPs degrade the basal lamina and the extracellular matrix as well as loosening the cell–cell contacts between endothelial cells, which subsequently allows endothelial cells to migrate and proliferate. 40 Because MMP activation is an essential step in the mechanism leading to angiogenesis, in vivo detection of active MMP is a highly promising tool to monitor the severity of inflammatory processes and the impact of antiinflammatory approaches that could prevent inflammation-induced tissue destruction.
Our noninvasive in vivo investigation of MMP activity by optical imaging revealed increased MMP activity during acute and chronic cutaneous TNCB-specific DTHR (see Figure 1). According to the manufacturer, MMPsense680 is cleavable predominantly by MMP-9 and -13. 39 Bremer and colleagues experimentally showed in vitro that the measured signal intensity from an activatable MMP probe directly correlates with MMP activity. 19 Unfortunately, for MMPsense, no control agent is available. However, the activatable optical imaging probe ProSense680 (PerkinElmer) has the identical mechanism of action and the same fluorochromes but is activated by several proteases. We evaluated ProSense and its uncleavable control substance, ProSense-control, in the model of TNCB-induced CHSR and detected a strong signal using ProSense680 and almost no signal using the uncleavable control probe ProSense-control (unpublished data, 2011). This substantiates our conclusion that activation of the optical imaging probe MMPsense is due to the specific cleavage of proteases at the cleavage site and is therefore independent of other effects, such as the increased perfusion.
In recent years, measuring MMP activity by in vivo optical imaging of MMP-activatable probes has been evaluated in various models of disease, such as allergic airway inflammation 21 and experimental arthritis. 20
Other MMP-targeting probes are often based on MMP inhibitors that bind to the active site of MMPs. These MMP inhibitors can be labeled with fluorochromes for optical imaging or radioactive isotopes for single-photon emission computed tomography (SPECT) or PET imaging and are often used for preclinical imaging of atherosclerosis 41 and cancer. 42 In a recent study by Waschkau and colleagues, a fluorescence-labeled MMP inhibitor was compared to an MMP-activatable probe. 43 They showed that imaging with a fluorescence-labeled MMP inhibitor requires a shorter uptake time but yields a lower signal to noise ratio compared to the activatable probe.
MMPs are important for tissue conversion, cell migration, activation of mediators, and angiogenesis.13,44 Thus, they play various roles in the establishment of inflammatory processes. Wang and colleagues reported reduced cutaneous DTHR in MMP-3−/− mice and, surprisingly, prolonged cutaneous DTHR in MMP-9−/− mice. 45 The investigators found reduced interleukin-10 protein expression in MMP-9−/− mice and therefore suggested a role for MMP-9 in cutaneous DTHR. 45 Many MMPs, such as MMP-9, can be both pro- and antiangiogenic. 13 Inhibition of MMPs causes reduced angiogenesis in different tumor models and thus can inhibit tumor progression and metastases. 17
Optical imaging is an emerging tool for noninvasive in vivo assessment of inflammation. Activatable probes provide a high specificity and a superior signal to noise ratio and have been created for the detection of proteases such as MMPs and cathepsins.39,46 Cathepsins represent a class of proteases that are critically involved in inflammatory immune processes. 47 Other optical imaging targets to measure ongoing inflammation are cellular and vascular adhesion molecules as well as integrins, such as the αVβ3 integrin.9,48 Inflammatory processes are accompanied by a strongly increased glucose metabolism, which could be determined by a fluorescence-labeled glucose analogue.49,50 In addition, transgenic mice with specific luciferase-linked reporter genes enable bioluminescence optical imaging–based detection of the activation of proinflammatory signaling pathways such as NF-κB. 51 Optical imaging is a fast, easy to use, and very versatile method, but its limitations are the lack of absolute quantification, no reliable three-dimensional reconstructions, and limited penetration depth. PET is a well-established approach to address these downsides; however, it is dependent on the availability of short-lived radiopharmaceuticals. In various studies, PET was used to examine inflammatory immune diseases focusing on glucose metabolism, cyclooxygenase expression, proteases, membrane antigens of immune cells, cellular receptors, and endothelial activation, among others. 52
The mechanisms in inflammatory diseases leading to chronic inflammation are not yet fully understood. As a large variety of molecular processes are involved, multimodal in vivo imaging approaches are promising to gain deeper insights into the pathophysiology of inflammatory diseases. As inflammation is a dynamic process, the ability to perform multiple noninvasive measurements during the progression of the inflammatory process is an enormous advantage of in vivo imaging methods. Although many imaging studies have been performed in inflammatory diseases, only limited data about the monitoring of the progression of an inflammatory disease from the acute to a chronic state are available. It has been shown in two different experimental arthritis models that 18F-fluorodeoxyglucose–PET (detecting glucose metabolism) and 18F-3′-deoxy-3′-18F-fluorothymidine (FLT)-PET (detecting cell proliferation) combined with magnetic resonance imaging (MRI) and computed tomography (CT) are promising tools to monitor disease progression.53,54
In the development of new treatment approaches, in vivo imaging allows the noninvasive monitoring of the antiinflammatory action. Efficient treatment of inflammatory diseases is still a challenge in modern individualized medicine due to unsatisfying treatment responses or severe side effects. 55 Because cutaneous DTHR is accompanied by dense PMN infiltrates, 3 which are a source of ROS produced by nicotinamide adenine dinucleotide phosphate (NADPH) oxidase and myeloperoxidase, 56 NAC, a powerful and widely used antioxidant, could be a promising treatment for DTHR. In our experiments, NAC displayed a strong antiinflammatory effect in acute and chronic TNCB-specific cutaneous DTHR. This effect is in line with data published by other groups using TNCB or other haptens to elicit acute cutaneous DTHR.33,35 To our knowledge, no other group has investigated the effect of NAC on chronic cutaneous DTHR. Because chronic DTHR causes tissue conversion and tissue destruction, it more closely reflects the clinical reality of autoimmune disease than acute DTHR. Our experiments on chronic TNCB-induced cutaneous DTHR revealed reduced edema, hyperkeratosis, acanthosis, inflammatory cell infiltrate, and blood vessel density in NAC-treated compared to sham-treated mice (see Figure 5).
Successful antiinflammatory NAC treatment has been reported for other DTHRs, such as experimental arthritis 57 and atherosclerosis. 58 In contrast, Wagberg and colleagues and Sarnstrand and colleagues reported enhanced ear swelling in NAC-treated mice in a model of oxazolone-induced acute cutaneous DTHR.33,58
NAC is commonly used in patients as a mucolytic agent, 59 but its efficacy is controversial.60,61 NAC is commonly accepted as an antidote for acetaminophen intoxication. 62 Interestingly, the antiinflammatory effects of NAC treatment have been shown in patients with cystic fibrosis 63 and septic shock. 64
NAC reduces ROS in vitro 27 and in vivo 28 and is a source of the antioxidant glutathione. NAC is able to suppress both the activation of NF-κB 29 and p38 MAP kinase 30 (see Supplementary Discussion, online version only).
In our noninvasive in vivo experiments, optical imaging using MMP-activatable probes allowed us to monitor the protective effects of antiinflammatory NAC medication on MMP activity in acute and chronic cutaneous TNCB-induced DTHRs (see Figure 3). We detected a decrease in MMP activity in the ears of NAC-treated mice during acute and chronic inflammation. Interestingly, we found a good correlation between the TNCB-induced ear swelling responses and the MMPsense signal intensities (see Figure S4, online version only).
In chronic diseases, the actual clinical end points are delayed and often accompanied by severe tissue destruction. Therefore, noninvasive in vivo imaging is an important tool to study the antiinflammatory effects of experimental therapies. 55 Noninvasive in vivo detection of MMP activity during antiinflammatory treatment has been documented in different models of inflammatory diseases, such as experimental allergic airway inflammation 21 and experimental arthritis. 65
Conclusion
Noninvasive in vivo optical imaging of MMP activity by specific activatable probes is a powerful diagnostic tool to identify sites of inflammation, to measure acute and chronic cutaneous DTHRs, and to monitor antiinflammatory effects. The protective power of NAC reduces the detrimental effects of acute and chronic cutaneous DTHRs and suppresses MMP activity and, consequently, angiogenesis.
Footnotes
Acknowledgments
We thank Maren Harant and Funda Cay for excellent technical support.
Financial disclosure of authors: This work was supported by the Medical Faculty of the Eberhard Karls University Tübingen (“Promotionskolleg”) and the Werner Siemens-Foundation.
Financial disclosure of reviewers: None reported.
