Abstract
Biomarker-specific imaging probes offer ways to improve molecular diagnosis, intraoperative margin assessment, and tumor resection. Fluorescence and photoacoustic imaging probes are of particular interest for clinical applications because the combination enables deeper tissue penetration for tumor detection while maintaining imaging sensitivity compared to a single optical imaging modality. Here we describe the development of a human epidermal growth factor receptor 2 (HER2)-targeting imaging probe to visualize differential levels of HER2 expression in a breast cancer model. Specifically, we labeled trastuzumab with Black Hole Quencher 3 (BHQ3) and fluorescein for photoacoustic and fluorescence imaging of HER2 overexpression, respectively. The dual-labeled trastuzumab was tested for its ability to detect HER2 overexpression in vitro and in vivo. We demonstrated an over twofold increase in the signal intensity for HER2-overexpressing tumors in vivo, compared to low–HER2-expressing tumors, using photoacoustic imaging. Furthermore, we demonstrated the feasibility of detecting tumors and positive surgical margins by fluorescence imaging. These results suggest that multimodal HER2-specific imaging of breast cancer using the BHQ3-fluorescein trastuzumab enables molecular-level detection and surgical margin assessment of breast tumors in vivo. This technique may have future clinical impact for primary lesion detection, as well as intraoperative molecular-level surgical guidance in breast cancer.
MULTIMODAL MOLECULAR IMAGING has the potential to improve diagnosis and intraoperative margin assessment of breast cancer. A promising strategy for improving cancer imaging specificity is the use of imaging probes that are targeted to particular biological markers. One such marker is human epidermal growth factor receptor 2 (HER2), which is overexpressed in 25 to 30% of breast cancer patients. 1 HER2 detection in breast tumors can guide therapeutic decisions and may serve as a predictive biomarker for disease outcome. 2
Recent reports describe the use of dual-labeled radionuclide and fluorescence imaging probes targeted against HER2 for noninvasive diagnosis and surgical guidance, respectively.3,4 Such imaging-based diagnostic approaches are advantageous compared to conventional techniques, such as immunohistochemical staining of ex vivo biopsies, which often lack reproducibility and accuracy. 5 In fact, interobserver variability and deficient standardized preanalytical methodology can contribute to a misclassification rate as high as 40%.6,7 Among various imaging techniques for diagnostic applications, optical and photoacoustic imaging techniques may be best suited for routine clinical use as they are easily accessible, minimally invasive, and inexpensive approaches that do not rely on receptor-targeted radiopharmaceuticals.8,9 Although fluorescence imaging has been previously used for HER2 visualization of breast tumors in vivo, photoacoustic imaging may be optimal for noninvasive diagnosis because it provides better spatial resolution beyond the depth penetration limit of fluorescence imaging.10,11 Various photoacoustic imaging agents, such as HER2 antibody–conjugated fluorophores, 12 dye-encapsulating nanoparticles, 13 and gold nanorods, 14 have been tested in vitro. Of those, HER2 antibody–conjugated gold nanorods have been tested in vivo, demonstrating contrast enhancement in an HER2-overexpressing oral squamous carcinoma model. 15
In addition to diagnostic applications, HER2-targeted fluorescence probes have been tested in preclinical breast cancer models for real-time intraoperative tumor imaging and surgical margin assessment for surgical guidance. 16 Such techniques potentially overcome the limitations of common histology-based surgical margin assessment methodologies, such as frozen-section analysis, which significantly prolong the operating time (up to 30 minutes) and have a high variance in sensitivity (65–78%) due to the variability in pathologists' experience. 17 Recently, clinical trials on intraoperative breast tumor visualization using targeted and nontargeted fluorescent dyes have been approved, and patient accrual is ongoing.18,19
In the current study, we sought to develop and validate a new dual-labeled photoacoustic-fluorescent HER2 imaging probe that could be used to examine differential HER2 expression in breast cancer in vivo. We used the commercially available Black Hole Quencher 3 (BHQ3) as a photoacoustic contrast agent because we and others have observed that BHQ3 yields a high photoacoustic signal in the near-infrared range compared to conventional fluorescent dyes, such as Alexa Fluor 750. 20 BHQ3 is also advantageous compared to gold nanorods, one of the common contrast agents for photoacoustic imaging, because the latter are thermally unstable and thus require surface coating to avoid optical absorption spectral changes during photoacoustic imaging. 14 N-Hydroxysuccinimide (NHS)-fluorescein was chosen for fluorescence imaging because NHS esters react efficiently with primary amino groups to form stable amide bonds. Overall, we demonstrate noninvasive in vivo photoacoustic imaging of HER2 expression and real-time intraoperative surgical margin assessment using fluorescence imaging of breast tumors in a murine breast tumor xenograft model.
Materials and Methods
Conjugation, Purification, and Characterization of BHQ3-Fluorescein Trastuzumab
One milligram of amine-reactive NHS-fluorescein dye (ThermoFisher Scientific, Waltham, MA) and 1 mg BHQ3 of carboxylic acid succinimidyl ester (Biosearch Technologies, Novato, CA) were each dissolved in 200 μL of anhydrous dimethyl sulfoxide (DMSO; Sigma-Aldrich, Oakville, ON), separately under reduced lighting conditions. Concentrations of these two stock solutions were determined by the absorption spectra with extinction coefficients as follows: εFluorescein = 70,000 M−1 cm−1 at 493 nm and εBHQ3 = 40,700 M−1 cm−1 at 615 nm. The absorption spectra were measured using a Varian Cary 50 ultraviolet (UV)-visible spectrophotometer with a UV quartz cuvette. Reagents were aliquoted to the designated molar amount, dried by a centrifugal evaporator, and stored at −20°C for future use. Trastuzumab (Genentech, San Francisco, CA) was diluted in phosphate-buffered saline (PBS) at a concentration of 1 mg/mL. One hundred microliters of trastuzumab solution was first mixed with 10 μL of 0.1 M sodium bicarbonate buffer to adjust the pH value to 8. Fluorescein and BHQ3 reagents were each dissolved in 5 mL of anhydrous DMSO separately, and covalent conjugation reactions with trastuzumab were performed by agitation under reduced lighting conditions for 2 hours at room temperature. Labeled trastuzumab was passed through a Bio-Spin 30 column (Bio-Rad, Mississauga, ON) to remove the major portion of free fluorescein and BHQ3 and was further purified by reverse-phase analytical high-performance liquid chromatography (HPLC) on a Waters Model 2695 controller with a TSKgel G4000SWxL column (Tosoh, G4000SWxL 8 μm, 7.8 mm × 30 cm) and a Model 2996 photodiode array detector. The conditions were as follows: solvent 100% water without gradient, flow rate = 0.5 mL/min. The degree of labeling (DOL) and the concentration of the labeled trastuzumab were then measured as follows:
where extinction coefficient ε280nm = 225,000 M−1 cm−1 for trastuzumab, ε494nm = 69,000 M−1 cm−1 for fluorescein, and ε615nm = 407,000 M−1 cm−1 for BHQ3. The 0.234*Abs494nm refers to the fluorescein absorption contribution at 280 nm as the constant ratio of εFluorescein(280nm)/εFluorescein(494nm) is 0.234. The 0.7*Abs615nm refers to the BHQ3 absorption contribution at 280 nm [εBHQ3(280nm)/εBHQ3(615nm) = 0.7].
Cell Culture
The MDA-MB-231 human breast cancer cell line was obtained from the American Type Culture Collection (Manassas, VA), and the MDA-MB-231-H2N human breast cancer cell line was kindly provided by Dr. Robert S. Kerbel (Sunnybrook Health Sciences Centre, Toronto, ON). The MDA-MB-231 cell line expresses minimal levels of the ERBB2 gene, which encodes for HER2 in humans. The MDA-MB-231-H2N human breast cancer cell line was previously transfected to stably overexpress the ERBB2 gene in the range of endogenous ERBB2-overexpressing breast cancer cell lines such as SKBR3. 21 Both cell lines were maintained in Dulbecco's Modified Eagle's Medium (DMEM) with 10% fetal bovine serum at 37°C and 5% CO2. Cytospun cells were fixed and stained for HER2 using anti-HER2 monoclonal antibody, and the slides were scanned using the Aperio slide scanner and processed using an Aperio Image Scope (Aperio Technologies, CA).
In Vitro Cytotoxicity Assay
In vitro cytotoxicity of BHQ3-fluorescein trastuzumab was determined by colorimetric (3-[4,5-dimethylthiazol-2-yl]-2,5 diphenyl tetrazolium bromide) (MTT) assay. For 2 days, 2.0 × 104 MDA-MB-231 cells, grown in a 96-multiwell plate, were incubated with 50 μg/mL and 100 μg/mL of the dual-labeled trastuzumab prepared in DMEM for 3 and 24 hours each. The cells were then incubated with 0.5 mg/mL of MTT solution (Invitrogen, Burlington, ON) dissolved in DMEM for 2 hours. The MTT solution was removed after the incubation period and replaced with 200 μL of 1:1 DMSO/70% isopropanol in 0.1 M HCl. The resulting optical density (OD) at 570 nm was measured on a Bio-Tek ELx800 microplate reader (MTX Lab System, Vienna, VA). Untreated MDA-MB-231 cells were used as a control. Percent viability was calculated as follows: [1 – (OD sample – OD blank control)/(OD control – OD blank control)] × 100.
Tissue Phantom and Cell-Encapsulating Gelatin Bead Preparation
Cell-encapsulating gelatin beads were prepared by fixing 107 MDA-MB-231 and MDA-MB-231 H2N cells in 3% formaldehyde for 2 hours, followed by blocking with 1% bovine serum albumin for 1 hour. The cells were then incubated with BHQ3-fluorescein trastuzumab for 1 hour and washed with PBS. Following washing, a 5 to 8% weight gelatin solution was added to form gelatin beads. The beads were embedded in holes (2–3 mm diameter, 5 mm deep) created in a piece of chicken breast tissue, which was covered with another piece of 10 mm thick chicken breast tissue for imaging.
Mouse Xenograft Model
Eight-week-old female athymic NCRNU mice (Taconic, Hudson, NY) were injected with 2.0 × 106 MDA-MB-231 (n = 4) or H2N (n = 8) cells subcutaneously into the flanks. Tumors were left to grow until they reached approximately 100 mm3. All animal procedures were conducted in accordance with appropriate regulatory standards under Animal Use Protocol 2577 approved by the University Health Network Institutional Animal Care and Use Committee.
Photoacoustic Imaging and Image Analysis
All photoacoustic imaging was performed using the Vevo LAZR system (VisualSonics Inc., Toronto, ON) with a 21 MHz center frequency. In vivo photoacoustic imaging was performed while the mouse was under gas anesthesia consisting of 100% oxygen mixed with 2% isoflurane. The mice were imaged before and 1, 4, 8, 24, and 48 hours after tail vein administration of BHQ3-fluorescein trastuzumab (50 μg per mouse, dissolved in 200 μL PBS). To position the tumor parallel to the platform, anesthetized mice were held with their hindleg extended and a piece of gauze placed underneath. Three-dimensional (3D) photoacoustic and ultrasound images were acquired simultaneously with a single excitation wavelength of 700 nm. This wavelength was chosen based on the in vitro characterization of the BHQ3-fluorescein trastuzumab. The imaging could not be performed at a wavelength lower than 680 nm because the imaging system had a wavelength range of 680 to 970 nm. The 3D tumor volume was defined by manual contouring of the tumor boundary using built-in software, and the amount of photoacoustic intensity (defined as the percentage of the photoacoustic signal within the tumor volume) was quantified. During serial photoacoustic imaging of tumors, the following experimental parameters were kept constant: photoacoustic gain, transmitted laser power, focus depth, signal intensity at each depth, frame averaging, and frame rate.
Fluorescence Imaging and Image Analysis
All fluorescence imaging was performed using the Maestro hyperspectral fluorescence imaging system (CRI, Woburn, MA) in the 500 to 720 nm range, using a 445 to 490 nm excitation filter and a 515 nm longpass emission filter. To demonstrate surgical margin assessment based on fluorescence, mice were imaged 48 hours after injection of BHQ3-fluorescein trastuzumab following euthanasia. The surrounding skin was removed to reduce autofluorescence, and the tumor bed was imaged prior to and after surgical resection of the tumor. In addition, resected tumors were imaged to quantify intratumoral fluorescence signal intensity using built-in software.
Statistical Analysis
All statistical analyses were performed using the two-tailed Student t-test. A p value < .05 was considered significant, and the data were reported as mean ± SD.
Results
Synthesis and Characterization of BHQ3-Fluorescein Trastuzumab
BHQ3-fluorescein trastuzumab was analyzed using HPLC and spectrophotometry to confirm the dual labeling. As seen in Figure 1A, the absorption spectra of the dual-labeled trastuzumab indicated distinct optical absorption peaks, with fluorescein peaking at 495 nm and BHQ3 peaking at 607 nm. The latter observation is similar to a previous report, which measured an absorption peak of 612 nm for BHQ3 in water. 22 BHQ3 is described as having peak absorption of < 700 nm when measured in polymerase chain reaction buffer, as reported by the manufacturer. The observed difference in peak absorbance can be described by a change in the refractive index of the dissolving media. 23 The DOL for BHQ3-fluorescein trastuzumab was calculated to be 4.8, with an approximate 1:1 ratio of BHQ3 and fluorescein concentrations. A standard MTT assay was performed for the MDA-MB-231 cells that minimally express HER2 to determine the cytotoxicity of BHQ3-fluorescein trastuzumab. The assay revealed little cytotoxicity of the BHQ3-fluorescein trastuzumab (50 μg/mL) compared to untreated cells, with an average cell viability of 91.03% versus 100%, respectively (Figure 1B). Increased toxicity was observed with a higher concentration (100 μg/ML) and longer incubation (24 hours). Given that trastuzumab alone is minimally toxic to MDA-MB-231 cells (surviving fraction of 90% when incubated with 50 μg/mL for 1 hour 24 ), the observed cytotoxicity may have been due to the dyes. Based on these results, further in vitro and in vivo experiments were performed using 50 μg/mL of the BHQ3-fluorescein trastuzumab.

Characterization of BHQ3-fluorescein trastuzumab. A, Absorption spectra for BHQ3 alone (green), fluorescein alone (blue), and the BHQ3-fluorescein trastuzumab (red) in water. B, MTT assay for cells that were untreated (cells alone) or treated with 50 μg/mL and 100 μg/mL of the BHQ3-fluorescein trastuzumab and assessed at 3 and 24 hours posttreatment. Experiments performed in triplicate; error bars = SD.
BHQ3-Fluorescein Trastuzumab Enables Detection of HER2-Overexpressing Breast Tumor Cells In Vitro
MDA-MB-231 wild-type (WT) and MDA-MB-231 H2N tumor cells (Figure 2A) were used to demonstrate the signal enhancement for HER2-overexpressing H2N cells by BHQ3-fluorescein trastuzumab in vitro.21,25 Tumor cell–encapsulating gelatin beads were placed in the chicken breast phantom to mimic the subsurface tissue-scattering properties observed in human tissues. 26 As expected, imaging of the fluorescein fluorescence signal demonstrated a strong binding of the BHQ3-fluorescein trastuzumab to the MDA-MB-231 H2N tumor cells compared to the WT cells (Figure 2B). Similarly, photoacoustic imaging of BHQ3 demonstrated strong binding of the BHQ3-fluorescein trastuzumab to the H2N cells compared to the WT cells (Figure 2, C and D). Moreover, the H2N cells demonstrated a broad photoacoustic absorption spectrum ranging from 690 to 790 nm with a maximum absorption peak at 700 nm, which is characteristic of BHQ3 (Figure 2E). However, we anticipate that higher photoacoustic intensity could be obtained if the available photoacoustic system had an excitation laser wavelength range that started from 600 nm because we measured the peak absorption to be 607 nm during initial probe characterization. The in vitro spectrum information of BHQ3 was used to select the optimal excitation wavelength for in vivo photoacoustic imaging of the BHQ3-fluorescein trastuzumab.
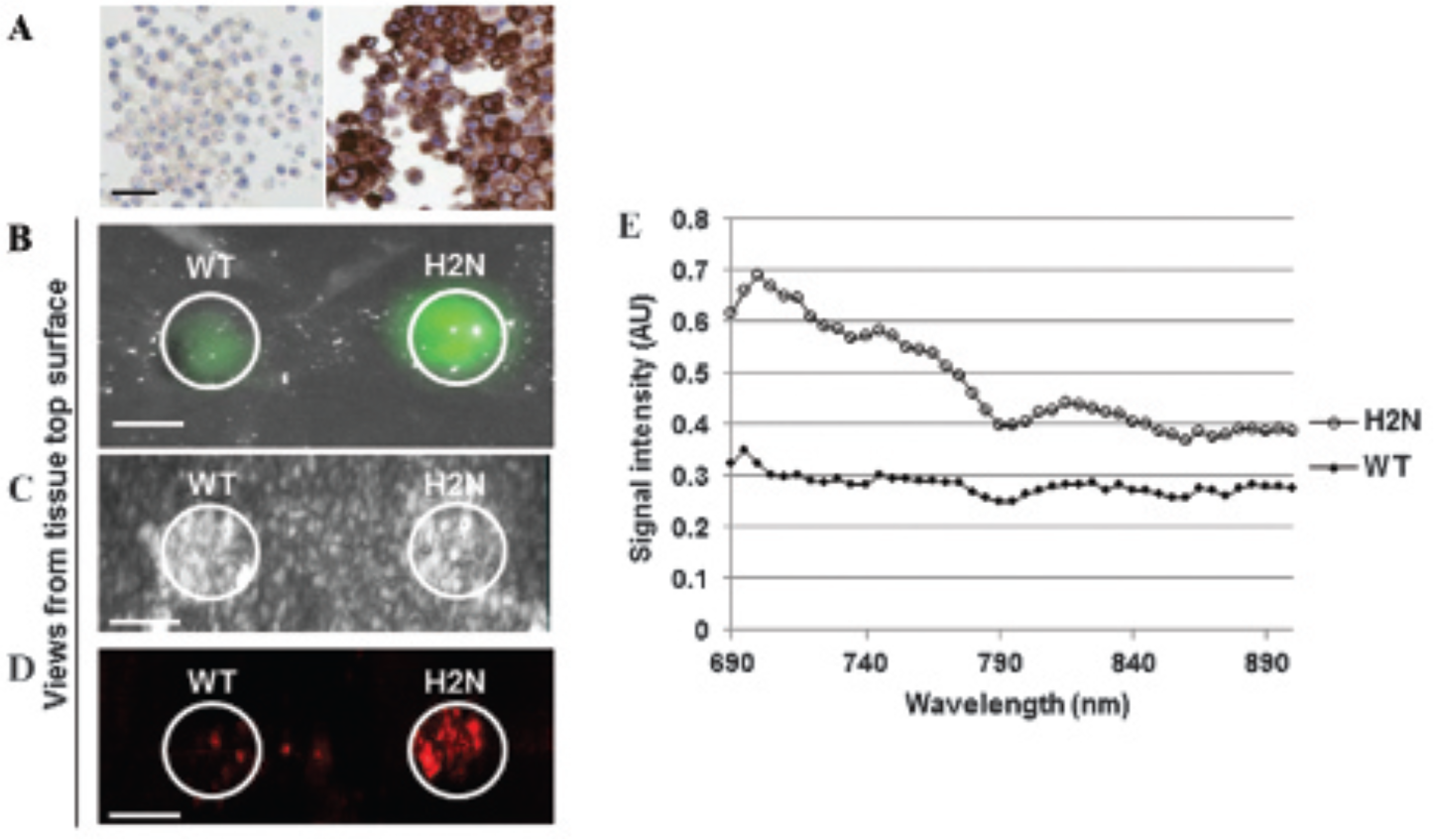
In vitro imaging of BHQ3-fluorescein trastuzumab. A, MDA-MB-231 wild-type (WT) (left) and H2N (right) cells stained with the anti-HER2 monoclonal antibody. B, “Top-down” fluorescence image of the MDA-MB-231 WT (left) and H2N (right) cell-encapsulating gelatin beads embedded in the chicken breast tissue phantom. “Top-down” three-dimensionally rendered ultrasound (C) and corresponding (D) photoacoustic images of the MDA-MB-231 WT and H2N cell–encapsulating gelatin beads, respectively. The ultrasound image shows the size and location of the beads within the chicken breast phantom, whereas the corresponding photoacoustic image demonstrates the difference in photoacoustic signal intensity between the WT and H2N cells in tissue. E, Spectral analysis of wavelength-dependent photoacoustic absorption for the WT and H2N cell–encapsulating gelatin beads within the tissue phantom. Scale bars: A, 30 μm; B–D, 2 mm.
BHQ3-Fluorescein Trastuzumab Accumulates in HER2-Overexpressing Tumors In Vivo
Athymic mice bearing subcutaneous MDA-MB-231 WT and H2N tumors were imaged with the photoacoustic system before tail vein injection of the BHQ3-fluorescein trastuzumab and for 48 hours after injection. We expected that BHQ3-fluorescein trastuzumab would absorb most optimally at 700 nm in vivo based on the previously described in vitro data; therefore, we imaged the WT and the H2N tumors at 700 nm. As seen in Figure 3A, the photoacoustic signal intensity, measured at 700 nm as a percentage of 3D tumor volume, increased maximally by over twofold at 8 hours postinjection in the H2N tumors compared to the baseline (p = .0148). On the other hand, the photoacoustic signal intensity measured in the WT tumors did not change significantly after the administration of BHQ3-fluorescein trastuzumab, demonstrating the ability of BHQ3-fluorescein trastuzumab to detect HER2 overexpression in vivo. Figure 3B further demonstrates that the photoacoustic signal was visible in the core of the tumor following administration of the BHQ3-fluorescein trastuzumab compared to the baseline. Endogenous absorbance by deoxyhemoglobin is also present at 700 nm11,27 and is represented as the baseline measurement of percent photoacoustic signal in Figure 3A. Notably, the signal was detected within the tumor with a penetration depth of 5 mm, which surpasses the tissue penetration depth of fluorescence imaging.

In vivo photoacoustic imaging of BHQ3-fluorescein trastuzumab. A, Photoacoustic signal of MDA-MB-231 wild-type (WT) (n = 4) and H2N (n = 8) tumors grown subcutaneously in nude mice. Tumors were imaged 1 day before (baseline) and 1, 4, 8, 24, and 48 hours after intravenous injection of the dual-labeled trastuzumab. Percent photoacoustic signal was calculated based on the contoured 3D tumor volume. Error bars = SD; *p = .0148. B, Representative images of photoacoustic signal within H2N tumor volume, obtained at 700 nm. The images were 3D rendered to represent the 3D volume. Scale bar: 2 mm.
BHQ3-Fluorescein Trastuzumab Enables Fluorescence-Based Surgical Margin Detection
At the experimental end point of 48 hours following probe injection, mice were sacrificed and the skin around the tumors was surgically removed to acquire fluorescence images of the intact tumor. Immediately following imaging, tumors were resected under white light, and fluorescence imaging of the ex vivo tumor and surgical bed was performed to visualize the surgical margin. Fluorescence imaging distinguished the H2N tumors from the adjacent tissue in vivo (Figure 4A) and was sensitive to the detection of residual tumor mass postoperatively (Figure 4B). The average fluorescence intensity of resected H2N tumors was over twofold higher than that of the WT tumors, further validating the ability to detect signal enhancement due to HER2 overexpression in an intraoperative setting (Figure 4C).

Fluorescence-based surgical margin assessment. White light (left) and corresponding fluorescence (right) images of the tumor (MDA-MB-231 H2N) (A) before and (B) after surgical resection. The yellow arrow highlights an area that contained residual tumor cells. Scale bar: 0.5 mm. C, Average fluorescence signal intensity from resected wild-type (WT) and H2N tumors (n = 4 each). Error bars = SD; *p = .002.
Discussion
Molecularly specific photoacoustic and fluorescence imaging probes have the potential to improve breast cancer management by improving the accuracy of diagnosis and intraoperative surgical margin assessment. 28 Here we designed the dual-labeled trastuzumab probe to detect different HER2 expression levels in vitro and in vivo using photoacoustic and fluorescence imaging. Identifying the expression levels of breast tumor receptors, such as HER2, could have a predictive value by enabling the identification of patients who are most likely to benefit from receptor-targeted therapies.
Notably, in vivo photoacoustic imaging demonstrated a twofold increase in the signal intensity for HER2-overexpressing tumors 8 hours following the BHQ3-fluorescein trastuzumab administration compared to tumors with a lower expression of HER2. However, longitudinal fluorescence imaging data demonstrated that the HER2-overexpressing tumors retained fluorescence for as long as 48 hours after intravenous injection due to the prolonged presence of the BHQ3-fluorescein trastuzumab, in accordance with a previous report on trastuzumab pharmacokinetics. 29 The observed reduction in the photoacoustic signal of BHQ3-fluorescein trastuzumab in the tumor may be due to the metabolization of BHQ3 following administration in vivo. 22 In addition, further studies are needed to correlate fluorescence and photoacoustic imaging data to known receptor expression levels to quantitatively analyze the relative percentage of HER2-overexpressing cells within a tumor, which may add prognostic value to the assay. 30 Such an assay may also help differentiate in vivo HER2 expression in HER2-overexpressing tumors from normal tissues such as breast duct epithelium, which expresses low levels of HER2. 5
Our results also demonstrated that the BHQ3-fluorescein trastuzumab enabled detection of HER2-overexpressing tumors at the surgical margin, which is consistent with a recent report using a subcutaneous breast tumor model. 16 Thus, our dual-labeled trastuzumab probe may provide a way to guide intraoperative margin assessment. Fluorescence-based breast cancer surgical margin detection is more promising than conventional histologic techniques or emerging point-sampling tumor detection technologies such as MarginProbe or SpectroPen,31,32 mainly because the latter cannot examine large surgical fields quickly. Additional investigation to test our probe using an orthotopic breast cancer model is planned.
The use of photoacoustic imaging for visualizing tumor biomarkers must consider the potential for background photoacoustic signals caused by endogenous absorption, such as that of hemoglobin and melanin. 33 One way to minimize interference by endogenous optical absorbers is to ensure that the optical dye used for the imaging probe has a significantly higher absorption coefficient than the intrinsic tissue or blood absorbers. In the present study, we achieved this by using BHQ3 for its inherently high optical absorption in the near-infrared range. To further differentiate the imaging probe signal from that of endogenous absorbers, a more robust multispectral analysis could be used to classify each image pixel as a given absorber, based on known absorption spectra. However, this capability was not available in the commercial preclinical photoacoustic imaging system used for the present study.
We also demonstrated that the BHQ3-containing probe had little cell toxicity, which is consistent with similar studies using BHQ3-containing targeted molecular beacons. 34 However, a recent report suggests the need for caution when using BHQ3 in vivo due to its unstable azo bond, which could result in production of metabolites with unknown toxicity. 22 In future experiments, we plan to examine methods to reduce the risk of potential toxicity, for example, using a polyethylene glycol moiety as a backbone. 35 Alternatively, improving the detection sensitivity of the photoacoustic system by using a more sensitive transducer could enhance detection of the imaging probe at a lower concentration. 36
HER2 expression plays an important role in primary breast cancer diagnosis, treatment planning, and prediction of disease outcome. 2 Indeed, HER2 expression has also been identified in lymph nodes and distant metastases.37,38 The detection of distant metastases is of significant clinical importance, and several clinical studies have demonstrated the potential use of radiolabeled trastuzumab for noninvasive detection of HER2-positive metastases using scintigraphy and positron emission tomography.39,40 As an alternative to such nuclear imaging techniques, which have limited spatial resolution and are complicated by cost and radioactivity, our hybridized photoacoustic fluorescence probe approach enables the noninvasive detection of HER2-positive primary tumors and lymph nodes or distant metastases using emerging clinical prototype dual-mode imaging systems. 41 In fact, the use of photoacoustic imaging to detect distant lymph node metastases with endogenous and exogenous contrast has been previously demonstrated in animal models.42,43 As we demonstrated, our dual-modality imaging approach improves lesion detection compared to fluorescence imaging alone by achieving tissue depth penetration of 5 mm using a preclinical photoacoustic imaging system. Studies suggest that clinical ultrasound-photoacoustic imaging systems can achieve several centimeters of tissue depth sampling, which can simultaneously provide important complementary anatomic and morphologic information about the tumor in situ.41,44 Thus, with deeper tissue penetration and high resolution, our dual imaging modality probe has the potential to be translated to clinical use for detection of primary lesions and distant metastasis. We anticipate that our approach could also be modified to facilitate multiplexed imaging by incorporating other antibodies against breast tumor biomarkers such as human epidermal growth factor receptor (EGFR), which would further augment detection capabilities and improve biological characterization of breast tumors. 45
Overall, our current approach demonstrates the feasibility and future potential of breast tumor–specific hybrid photoacoustic fluorescence imaging for achieving noninvasive detection of primary lesions, as well as molecular-level margin assessment and surgical guidance in breast cancer. As new multimodal imaging systems emerge for clinical applications in breast cancer, there will be a need for complementary multimodal imaging agents. Our results help form the basis of such studies that will accelerate the translation of new molecular imaging diagnostic and intraoperative imaging procedures into the clinical domain.
Footnotes
Acknowledgments
Thanks are extended to Cheng Jin for help with animal handling, Dr. Iris Kulbatski and Dr. Phil Medeiros for manuscript editing, Dr. Alexandra Easson for providing the HER2 antibody, and Dr. Robert Kerbel for providing the MDA-MB-231-H2N human breast cancer cell line.
Financial disclosure of authors: This work was supported by funding provided to R. DaCosta by the Canadian Institutes of Health Research and by the International Collaboration R&D Program, South Korean Ministry of Knowledge Economy. We also acknowledge imaging support provided by STTARR Innovation Facility and the Advanced Optical Microscopy Facility of University Health Network.
Financial disclosure of reviewers: None reported.

