Abstract
Affibody molecules, small (7 kDa) scaffold proteins, are a promising class of probes for radionuclide molecular imaging. Radiolabeling of Affibody molecules with the positron-emitting nuclide 68Ga would permit the use of positron emission tomography (PET), providing better resolution, sensitivity, and quantification accuracy than single-photon emission computed tomography (SPECT). The synthetic anti-HER2 ZHER2:S1 Affibody molecule was conjugated with DOTA at the N-terminus, in the middle of helix 3, or at the C-terminus. The biodistribution of 68Ga- and 111In-labeled Affibody molecules was directly compared in NMRI nu/nu mice bearing SKOV3 xenografts. The position of the chelator strongly influenced the biodistribution of the tracers, and the influence was more pronounced for 68Ga-labeled Affibody molecules than for the 111In-labeled counterparts. The best 68Ga-labeled variant was 68Ga-[DOTA-A1]-ZHER2:S1 which provided a tumor uptake of 13 ± 1 %ID/g and a tumor to blood ratio of 39 ± 12 at 2 hours after injection. 111In-[DOTA-A1]-ZHER2:S1 and 111In-[DOTA-K58]-ZHER2:S1 were equally good at this time point, providing a tumor uptake of 15 to 16 %ID/g and a tumor to blood ratio in the range of 60 to 80. In conclusion, the selection of the best position for a chelator in Affibody molecules can be used for optimization of their imaging properties. This may be important for the development of Affibody-based and other protein-based imaging probes.
TARGETED CANCER THERAPIES use molecular recognition of cancer-associated abnormalities to exert their antitumor action. The specificity of targeted therapy is dependent on careful patient stratification as only a fraction of patients would have tumors expressing a particular target, and only this fraction would benefit from a particular targeted therapy. The use of molecular imaging for visualization of molecular therapeutic targets is a promising approach in the development of personalized cancer treatment. 1 In contrast to biopsy-based methods, noninvasive radionuclide molecular imaging methods are less prone to sampling errors and are repeatable, permitting monitoring of expression changes in response to therapy. 2 Thus, there is a need for imaging agents that provide high sensitivity and specificity in the detection of molecular targets in malignant tumors. 3
One straightforward approach for the development of imaging agents for visualization of molecular targets is to label the therapeutic antibodies specific to these targets with radionuclides suitable for imaging. 4 However, the intact antibodies demonstrate slow extravasation and tumor penetration as well as slow clearance of unbound antibody from blood. These factors result in low imaging contrast. 5 Moreover, imaging agents with a molecular weight of more than 45 kDa have a tendency to nonspecific accumulation in tumors. 6 Development of smaller targeting proteins may provide imaging of higher sensitivity and specificity. 7
Several alternative nonimmunoglobulin-based scaffold proteins, such as Affibody molecules, DARPins, and knottins, 7 have demonstrated an apparent potential for providing sensitive molecular imaging. The three-helix affinity proteins, Affibody molecules, are an example of alternative tracers with favorable properties for radionuclide molecular imaging.8,9 Randomization of 13 amino acids in Affibody molecules creates a large library from which high-affinity binders to different targets can be selected. 10 Affinity maturation has provided the development of Affibody molecules with subnanomolar affinity to several receptors overexpressed in malignant tumors.11–14 The small size (7 kDa) of Affibody molecules provides rapid extravasation, tumor penetration, and clearance of unbound tracer from healthy organs and tissues, which creates high-contrast images a few hours after injection.15,16
Affibody molecules targeting the human epidermal growth factor receptor type 2 (HER2) are the most studied.15,16 HER2 is a transmembrane tyrosine kinase receptor involved in signaling pathways that regulate cell proliferation, differentiation, motility, and suppression of apoptosis. 17 HER2 is overexpressed in a considerable fraction of breast, urinary bladder, ovarian, colorectal, and prostate cancers.17,18 Treatment of HER2-expressing breast and gastrointestinal cancers with the anti-HER2 antibody trastuzumab,19,20 the antibody-drug conjugate trastuzumab-DM1, 21 or the tyrosine kinase inhibitor lapatinib 19 significantly improves the survival of patients with disseminated cancer. Determination of HER2 expression in all breast cancers is recommended by the American Association of Clinical Oncology to stratify patients for HER2-targeting therapy. 22 Radiolabeled HER2-binding Affibody molecules have provided high-contrast images of HER2-expressing tumors in both preclinical 16 and clinical23,24 studies. HER2-binding Affibody molecules have been labeled with several radionuclides for singlephoton emission computed tomography (SPECT) and positron emission tomography (PET).25–29 This makes the HER2-binding Affibody molecule a convenient model for investigation of how the labeling chemistry influences the biodistribution properties of Affibody-based tracers.
Incorporation of chelators during peptide synthesis of Affibody molecule provides homogeneous conjugates appropriate for site-specific labeling.30–33 Previous studies have demonstrated that conjugation of chelation moieties can modify the physicochemical properties of an imaging agent, such as local charge or lipophilicity. These modifications can considerably alter the fate of radiolabeled short peptides.34–36 This is also true for Affibody molecules. In previous work, we showed that HER2-binding Affibody molecules labeled with 99mTc via different peptide-based chelators positioned at the N-terminus exhibit different biodistribution and targeting properties.32,33,37 Moreover, the in vivo properties of the 111In-labeled synthetic Affibody molecule ZHER2:s1 with a DOTA, NOTA, or NODAGA chelator at the N-terminus were dependent on the chelator type. 38 The results of that study demonstrated that even subtle differences in chelator structure have the potential to further increase the sensitivity of molecular imaging using Affibody molecules. 38 In an earlier study, we also conjugated the DOTA chelator to three different positions of a synthetic variant of the anti-HER2 ZHER2:342 Affibody molecule—more specifically to position A1 (the N-terminus), position K50 (located in the middle of helix 3), and position K58 (the C-terminus) via amide bond formation using an orthogonal protection strategy. 39 Direct in vivo comparison of the biodistribution properties of the 111In-labeled conjugates showed that different positioning of DOTA could be used to modify the targeting properties of Affibody molecules. For example, 111In-[DOTA-K50]-ZHER2:S1 showed suboptimal biodistribution properties in comparison with the two other variants. 39 Other studies have shown that coupling of different nuclides using the same chelator considerably affects organ uptake and the blood clearance rate of Affibody molecules.40–43
In this study, Affibody molecules conjugated with DOTA at A1 (the N-terminus), K50 (located in the middle of helix 3), and K58 (the C-terminus) were labeled with 68Ga. The influence of the DOTA chelator position on the biodistribution of 68Ga-labeled Affibody molecules was evaluated and compared to the biodistribution properties of their 111In-labeled counterparts.
Material and Methods
Materials
Buffers, such as 0.1 M phosphate-buffered saline (PBS), pH 7.5, 0.2 M ammonium acetate, pH 5.5, and 1.25 M sodium acetate, pH 3.6, were prepared using common methods from chemicals supplied by Merck (Whitehouse Station, NJ). Ethylenediaminetetraacetic acid (EDTA) was acquired from Sigma (St. Louis, MO). High-quality Milli-Q water (resistance higher than 18 MΩ cm) was used for preparing solutions. Metal contamination was removed from the buffers used for conjugation and labeling by purification using 2 to 3 g/L Chelex 100 resin (Bio-Rad Laboratories, Hercules, CA). [111In] chloride was purchased from Covidien. The 68Ge/68Ga generator (Eckert and Ziegler AG, Berlin, Germany) was eluted with 0.1 M hydrochloric acid (prepared from 30% ultrapure HCl from Merck).
The human ovarian carcinoma cell line SKOV3, 1.6 × 106 HER2 receptors per cell (ATCC, purchased via LGC Promochem, Boras, Sweden), was used in the experiments. The cells were cultured in RPMI medium (Flow Irvine, Lake Forest, CA) supplemented with 10% fetal calf serum (Sigma), 2 mM L-glutamine, and PEST (penicillin 100 IU/mL and 100 mg/mL streptomycin) (all from Biokrom AG, Berlin, Germany).
The yields of the labeled Affibody constructs were determined using 150 to 771 DARK GREEN Tec-Control Chromatography strips from Biodex Medical Systems (Shirley, NY), eluted with 0.2 M citric acid, pH 2.0. Sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE), 200 V constant, was performed using NuPAGE 4 to 12% Bis-Tris Gel (Invitrogen AB, Stockholm, Sweden) in MES buffer (Invitrogen AB) to confirm the identity of 68Ga-DOTA-A1-ZHER2:S1, 68Ga-DOTA-K50-ZHER2:S1, and 68Ga-DOTA-K58-ZHER2:S1. Cells in the in vitro experiments were detached using trypsin-EDTA solution (0.25% trypsin, 0.02% EDTA in buffer; Biochrom AG). Ketamine (50 mg/mL; Ketalar, Pfizer, New York, NY), xylazine (20 mg/mL; Rompun, Bayer, Leverkusen, Germany), and heparin (5,000 IE/mL; Leo Pharma, Ballerup, Denmark) were obtained commercially. The radioactivity was measured using an automated gamma-counter with a 3-inch NaI (Tl) detector (1480 Wizard, Wallac Oy, Turku, Finland). The distribution of radioactivity along the thin-layer chromatography strips and SDS gels was measured on a Cyclone storage phosphor system (PerkinElmer, Waltham, MA) and analyzed using OptiQuant image analysis software (PerkinElmer).
Statistical Analysis
Data on cellular uptake and biodistribution were analyzed by the two-tailed t-test using GraphPad Prism version 4.00 for Windows (GraphPad Software, La Jolla, CA) to determine the significance of differences (p < .05). The unpaired t-test was used for comparison of bio distribution of different conjugates. For analysis of the data, from a dual label in vivo study, the paired t-test was used.
Synthesis of DOTA-Conjugated Affibody Molecules
A synthetic variant of the ZHER2:342 Affibody molecule, denoted ZHER2:S1, with the sequence AEAKYAKEMRNAY-WEIALLPNLNNQQKRAFIRSLYDDPSQSANLLAEAKKLN-DAQAPK-amide was assembled on a 433A Peptide Synthesizer (Applied Biosystems, Foster City, CA) using Fmoc/tBu chemistry in 0.1 mmol scale and Fmoc-rink amide resin with a substitution level of 0.39 mmol/g. The synthesis, purification, and characterization of the DOTA-conjugated Affibody molecules were previously described. 39
Radiolabeling Chemistry and In Vitro Studies
For 68Ga labeling, the generator was eluted with 0.1 M HCl and one fraction of 1,000 μL (void volume), and two fractions of 500 μL were collected. The second fraction of 500 μL, containing the maximum radioactivity, was used for labeling. An aliquot of a conjugate (50 mg), reconstituted in 10 μL of 0.2 M ammonium acetate, pH 5.5, was mixed with 25 MBq 68Ga-containing eluate. The pH of the reaction mixture was adjusted to between 3.0 and 3.5 by the addition of 1.25 M NaOAc, pH 3.6. Then the reaction mixture was incubated at 95°C for 15 minutes. Thereafter, small aliquots (1.4 μL) of the reaction mixtures were taken and analyzed by radio-instant thin-layer chromatography (ITLC).
To evaluate the stability of labeling, 68Ga-DOTA-K50-ZHER2:S1 and 68Ga-DOTA-K58-ZHER2:S1 were incubated with 500-fold excess of EDTA for 1 hour and then analyzed using ITLC. As a control, the conjugates were incubated with PBS. The experiments were performed in duplicate. The stability of 68Ga-DOTA-A1-ZHER2:S1 was evaluated earlier by Strand and colleagues. 43 Labeling of variants by 111In was performed as previously described. 39
An in vitro specificity test and a comparison of 68Ga-[DOTA-A1]-ZHER2:S1, 68Ga-[DOTA-K50]-ZHER2:S1, and 68Ga-[DOTA-K58]-ZHER2:S1 processing by SKOV3 cells were performed according to the method developed and validated by Wållberg and Orlova. 44 Briefly, the cells (≈ 1 × 106 per dish) were incubated with the labeled compounds (19.9 ng/dish, 270 pM) at 37°C, 5% CO2. At predetermined time points (0.5, 1, and 2 hours after incubation), the medium from a set of three dishes was removed and the cells were washed with ice-cold serum-free medium. The cells were then treated with 0.5 mL 0.2 M glycine buffer containing 4 M urea, pH 2.5, for 5 minutes on ice. The acidic solution was collected, and the cells were also washed with 0.5 mL glycine buffer, which was collected and pooled with the first fraction. The radioactivity in the acid wash fractions was considered membrane-bound radioactivity. After the addition of 0.5 mL 1 M NaOH, cells were incubated at 37°C for 0.5 hours, and thereafter, the alkaline solution containing cell debris was collected. The cell dishes were washed with an additional 0.5 mL NaOH, and the alkaline fractions were pooled. The radioactivity in the alkaline fractions was considered internalized. The percentage of internalized radioactivity was calculated for each fraction at each time point. The specificity and cellular processing of 111In-labeled variants were tested in earlier studies. 39
Comparative In Vivo Studies
Animal experiments were performed according to national legislation on laboratory animal protection and were approved by the Ethic Committee for Animal Research of Uppsala University (Permit Number: 48/11). Euthanasia was performed under xylazine/ketamine anesthesia, and all efforts were made to minimize animals' suffering. 68Ga- and 111In-labeled conjugates were compared using a dual label study to improve the statistical power and to minimize the number of tumor-bearing animals, as described by Honarvar and colleagues. 42 The goal of the study was a comparative evaluation of the influence of 68Ga and 111In on the biodistribution and targeting properties of DOTA-A1-ZHER2:S1, DOTA-K50-ZHER2:S1, and DOTA-K58-ZHER2:S1.
The targeting properties of the different conjugates were compared in female NMRI nu/nu mice (weight 24.25 ± 1.53 g) bearing SKOV3 xenografts 2 hours after injection. The cells (107 cells per mouse) were subcutaneously implanted in the right hind leg about 5 weeks before the experiment. At the time of the experiment, the average tumor weight was 0.2 ± 0.12 g. A mixture of 10 kBq 111In-labeled and 380 kBq 68Ga-labeled variants of each conjugate in 100 μL PBS was injected intravenously (tail vein). The injected peptide dose was adjusted to 1 mg (0.14 nmol) per mouse by adding unlabeled peptide. To check the specificity of xenograft targeting, another group of four mice was subcutaneously preinjected with 500 mg (3,800 nmol) nonlabeled ZHER2:342 Affibody molecule 40 minutes before injection of each radiolabeled conjugate. Blood and organ samples—lung, liver, spleen, kidneys, tumor, muscle, bone, gastrointestinal tract (with content)—and the remaining carcass were collected and weighed and the radioactivity was measured.
Imaging
The conjugate with the best tumor to organ ratio, in this case 68Ga-[DOTA-A1] -ZHER2:S1, was imaged to confirm the capacity for HER2 visualization. Two SKOV3 tumor-bearing mice were injected with 2.5 MBq of 68Ga-[DOTA-A1]-ZHER2:S1 (5 μg peptide). Immediately before imaging (1 and 2 hours postinjection), the animals were sacrificed, and the urine bladder was dissected to avoid interfering activity in urine close to tumor xenograft and kidneys. PET/computed tomography (CT) was performed using the Triumph TM Trimodality system (Gamma Medica, Inc), which is a fully integrated SPECT/PET/CT hardware and software platform optimized for small animals. The acquisition time was 60 and 120 minutes. The PET data were reconstructed into a static image using a MLEM 2D algorithm (10 iterations). The CT raw file was reconstructed using filtered backprojection. PET and CT DICOM files were analyzed using PMOD version 3.12 software (PMOD Technologies Ltd.).
Results
Synthesis of DOTA-Conjugated Affibody Molecules
Data concerning the chelator-conjugated Affibody molecules are summarized in Table 1. All conjugates had a purity of ≥ 97%. The melting point of DOTA-K50-ZHER2:S1, 57°C, was slightly lower than the melting points for the two other variants, [DOTA-A1]-ZHER2:S1 and [DOTA-K58]-ZHER2:S1, which were 64°C and 62°C, respectively. The affinities (equilibrium dissociation constants) of all three variants toward the HER2 receptor were in the same range, with KD around 100 pM, with slightly higher affinity for [DOTA-K58]-ZHER2:S1 (94 pM) than for [DOTA-A1]-ZHER2:S1 and [DOTA-K50]-ZHER2:S1 (133 and 107 pM, respectively) (see Table 1).
Biophysical Characteristics of the Affibody Variants 39

Radiolabeling Chemistry and In Vitro Studies
A labeling yield of about 99% was obtained for 111In-[DOTA-A1]-ZHER2:S1, 111In-[DOTA-K50]-ZHER2:S1, and 111In-[DOTA-K58]-ZHER2:S1. No additional purification was required.
Reasonable radiochemical yield was obtained for [DOTA-A1]-ZHER2:S1, [DOTA-K50]-ZHER2:S1, and [DOTA-K58]-ZHER2:S1 after incubation with 68Ga at 95°C for 15 minutes (Table 2). Further purification with NAP-5 size-exclusion chromatography provided a radiochemical purity of more than 96%. The SDS-PAGE analysis confirmed the identity of 68Ga-[DOTA-A1]-ZHER2:S1, 68Ga-[DOTA-K50]-ZHER2:S1, and 68Ga-[DOTA-K58]-ZHER2:S1 (Figure 1). No measurable release of radioactivity from the conjugates after 1 hour of incubation with 500-fold excess EDTA was detected (see Table 2), demonstrating high stability of all 68Ga-labeled conjugates.
Labeling Yield (Decay Corrected) and Radiochemical Purity of 68Ga-Labeled Affibody Molecules

EDTA = ethylenediaminetetraacetic acid.

Representative SDS-PAGE analysis of the identity of 68Ga-[DOTA-A1]-ZHER2:S1 (A), 68Ga-[DOTA-K50]-ZHER2:S1 and 68Ga-[DOTA-K58]-ZHER2;S1 (C). 1 = 68Ga-labeled Affibody molecule; 2 = 68Ga citrate was used as a marker for low-molecular-weight compounds. The signal was measured as a digital light unit and is proportional to the radioactivity in a given point of a lane in the SDS-PAGE gel.
A significant (p < .01) decrease in HER2 binding for 68Ga-[DOTA-A1]ZHER:S1, 68Ga-[DOTA-K50]-ZHER2:S1, and 68Ga-[DOTA-K58]-ZHER2:S1 to HER2-expressing SKOV3 cells after saturation of the receptors using nonlabeled ZHER2:342 indicated that the binding of radiolabeled Affibody molecules was receptor mediated. This experiment demonstrated retained binding specificity of the radioconjugates after labeling (Figure 2).
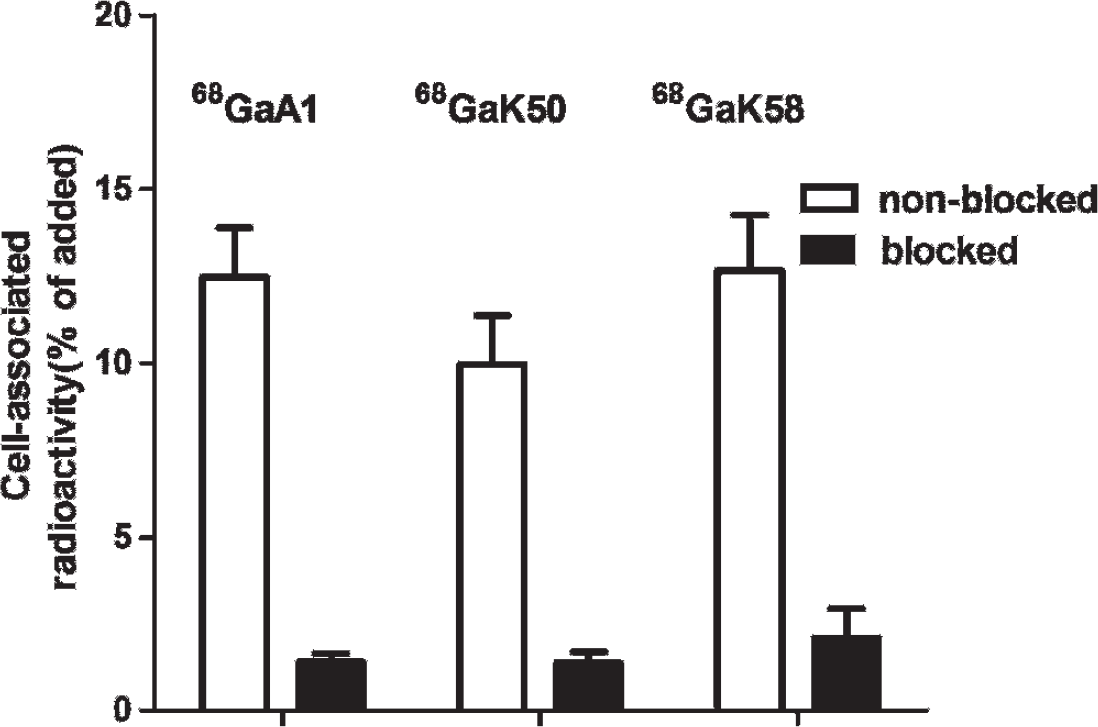
In vitro specificity of 68Ga-[DOTA-A1]-ZHER2;S1, 68Ga-[DOTA-K50]-ZHER2:S1, and 68Ga-[DOTA-K58]-ZHER2:S1 binding to the SKOV3 cell line. Radioactivity uptake in HER2-overexpressing SKOV3 cells presaturated with nonlabeled ZHER2:342 was significantly (p < .01) lower than in nonsaturated HER2-overexpressing SKOV3 cells.
Cellular processing of 68Ga-[DOTA-A1]-ZHER2:S1, 68Ga-[DOTA-K50]-ZHER2:S1, and 68Ga-[DOTA-K58]-ZHER2:S1 by HER2-expressing SKOV3 cells is presented in Figure 3. Both binding and internalization profiles were very similar for all conjugates. The difference between the amounts of cell-associated radioactivity for the radioconjugates was within the accuracy of the experimental method. After rapid binding within the first 30 minutes of incubation, the cell-associated radioactivity increased with a slower rate. The internalization of the compounds was relatively slow and increased slightly throughout the experiment. After 2 hours of incubation, approximately 10% of the radioactivity was internalized by SKOV3 cells. Data were in a good agreement with data for other ZHER2:342-based radiolabeled conjugates.40,41,44

In vitro cellular processing of 68Ga-[DOTA-A1]-ZHER2:S1, 68Ga-[D0TA-K50]-ZHER2:S1, and 68Ga-[DOTA-K58]-ZHER2:S1 by SKOV3 cells. Cells were incubated with the labeled compounds at 37uC. Acid wash was used to determine the membrane-bound radioactivity. Cell-bound activity is normalized to the maximum uptake. Error bars might not be seen when they are smaller than the symbols.
Comparative In Vivo Studies
Data concerning the head-to-head comparison of targeting of HER2-expressing SKOV3 xenografts using [DOTA-A1]-ZHER2:S1, [DOTA-K50]-ZHER2:S1, and [DOTA-K58]-ZHER2:S1 labeled with 68Ga and 111In are presented in Figure 4, Table 3, and Table 4.
The results of the in vivo specificity tests are presented in Figure 4. The tumor uptake of all 68Ga conjugates was significantly (p < .0001) reduced by presaturation of HER2 receptors in tumors using nonlabeled ZHER2:342 Affibody molecule. This demonstrates the saturable character of their uptake and suggests that targeting was specific. The tumor uptake of 111In-labeled conjugates was also specific and in agreement with previous data. 39
Biodistribution Comparison of 68Ga- and mIn-Labeled Affibody Molecules in NMRI nu/nu Mice Bearing SKOV3 Xenografts 2 Hours after Intravenous Injection

GI = gastrointestinal.
Data are presented as an average %ID/g value for four animals ± standard deviation. (Data for the GI tract with content and carcass are presented as %ID/sample.)

In vivo specificity of targeting of HER2-expressing SKOV3 xenografts by 68Ga-(A) and 111In-(B) labeled Affibody molecules 2 hours after intravenous injection. The tumor uptake is expressed as %ID/g. Radioactivity uptake in HER2-overexpressing SKOV3 xenografts presaturated with nonlabeled ZHER2:342 was significantly (p < .05) lower.
All the 111In- and 68Ga-labeled constructs demonstrated rapid blood clearance via kidneys with subsequent tubular reabsorption. At 2 hours after injection, 65 to 75% of injected radioactivity was found in kidneys, resulting in uptake over 200%ID/g. The radioactivity accumulation in the gastrointestinal tract (a measure of hepatobiliary excretion) was low for all the radioconjugates.
The rapid blood clearance demonstrated the absence of transchelation of 68Ga to blood plasma proteins. Moreover, the low uptake of radioconjugates in liver and spleen indicates minor probability of radiocolloid formation.
The influence of radionuclide on biodistribution and targeting was clear for all three DOTA-conjugated Affibody molecules. The tumor uptake was 20 to 30% higher for the 111In-labeled variants in comparison with the 68Ga-labeled counterparts (p < .05 in a paired t-test). The radionuclide had a clear effect on the uptake in normal organs as well. However, this effect was dependent on the position of the chelators. It was most pronounced for [DOTA-K50]-ZHER2:S1, where the 68Ga-labeled variant had significantly (p < .05) higher uptake in blood, liver, spleen, muscle, and bone, but lower in kidneys. For [DOTA-A1]-ZHER2:S1, uptake of the 68Ga-labeled variant was also significantly (p < .05) higher in blood and lower in kidneys. 68Ga-[DOTA-K58]-ZHER2:S1 had lower uptake than 111In-[DOTA-K58]-ZHER2:S1 in lung, liver, tumor, and bone.
At 2 hours after injection, the influence of the position of the DOTA chelator had a moderate influence on the biodistribution of 111In-labeled Affibody molecules. There was no significant difference between 111In-[DOTA-A1]-ZHER2:S1 and 111In-[DOTA-K58]-ZHER2:S1 or between 111In-[DOTA-K58]-ZHER2:S1 and 111In-[DOTA-K50]-ZHER2:S1 at this time point. However, the uptake of 111In-[DOTA-K58]-ZHER2:S1 was significantly (p < .05) higher than the uptake of 111In-[DOTA-K50]-ZHER2:S1 in liver and lung.
In the case of 68Ga, the influence of the chelator position was more pronounced. Apparently, positioning of DOTA at the N-terminal alanine caused the most favorable biodistribution profile. The uptake of 68Ga-[DOTA-A1]-ZHER2:S1 was significantly (p < .05) lower than the uptake of 68Ga-[DOTA-K50]-Z HER2:S1 in blood, lung, liver, spleen, and bone and uptake of 68Ga-[DOTA-K58]-ZHER2:S1 in lung.
Tumor to blood, tumor to liver, and tumor to spleen ratios were significantly (p < .05) higher for 68Ga-[DOTA-A1]-ZHER2:S1 and 68Ga-[DOTA-K58]-ZHER2:S1 than for 68Ga-[DOTA-K50]-ZHER2:S1 at 2 hours after injection (see Table 4).
Comparison of Tumor to Organ Ratios of 68Ga- and 111In-Labeled Affibody Molecules in NMRI nu/nu Mice Bearing SKOV3 Xenografts 2 Hours after Intravenous Injection

Data are presented as an average %ID/g value for four animals ± standard deviation.
Significant difference (p < .05) between 68Ga-[DOTA-A1]-ZHER2:S1 and 68Ga-[DOTA-K50]-ZHER2:S1.
Significant difference (p < .05) between 111In-[DOTA-A1]-ZHER2:S1 and 68Ga-[DOTA-A1]-ZHER2:S1.
Significant difference (p < .05) between 111In-[DOTA-K50]-ZHER2:S1 and 68Ga-[DOTA-K50]-ZHER2:S1.
Significant difference (p < .05) between 111In-[DOTA-K58]-ZHER2:S1 and 111In-[DOTA-K50]-ZHER2:S1.
Significant difference (p < .05) between 68Ga-[DOTA-A1]-ZHER2:S1 and 68Ga-[DOTA-K58]-ZHER2:S1.
Significant difference (p < .05) between 111In-[DOTA-A1]-ZHER2:S1 and 111In-[DOTA-K58]-ZHER2:S1.
Significant difference (p < .05) between 111In-[DOTA-K58]-ZHER2:S1 and 68Ga-[DOTA-K58]-ZHER2:S1.
Significant difference (p < .05) between 68Ga-[DOTA-K50]-ZHER2:S1 and 68Ga-[DOTA-K58]-ZHER2:S1.
There was no significant difference between 111In-[DOTA-A1]-ZHER2:S1 and 111In-[DOTA-K50]-ZHER2:S1.
In general, all tumor to organ ratios were considerably lower for 68Ga-[DOTA-K50]-ZHER2:S1 than for 68Ga-[DOTA-A1]-ZHER2:S1 and 68Ga-[DOTA-K58]-ZHER2:S1 (see Table 4). Apparently, there was a strong influence of the radionuclide on tumor to organ ratios. This influence was most clearly seen for blood, where the 111In label provided appreciably (two- to fourfold) higher tumor to blood ratios for [DOTA-A1]-ZHER2:S1 and [DOTA-K58]-ZHER2:S1 than for 68Ga. Furthermore, the effect of the label nuclide was dependent on the position of the chelator. For example, the tumor to organ ratios of 68Ga-[DOTA-K50]-ZHER2:S1 were lower than those of 111In-[DOTA-K50]-ZHER2:S1 for all organs, which was not the case for [DOTA-K58]-ZHER2:S1 and 68Ga-[DOTA-KA1]-ZHER2:S1.
Imaging
High-contrast images of HER2-expressing xenografts in nude mice were acquired at 1 and 2 hours after intravenous injection of 68Ga-[DOTA-A1]-ZHER2:S1. In agreement with the biodistribution data, the only organ with high uptake was kidneys (Figure 5). Coronal images are presented as maximum intensity projections.

MicroPET/CT imaging of HER2 expression in SKOV3 xenografts in NMRI nu/nu mice using 68Ga-[DOTA-A1-ZHER2:S1]. The image was acquired 1 hour (A) and 2 hours (B) after administration of the tracer. The animals were euthanized immediately before imaging. Arrows point to tumor (T) and kidneys (K).
Discussion
Radionuclide imaging of molecular targets for therapy has great potential for rendering targeted therapy of cancer more personalized and, therefore, more successful. For this strategy to be useful, it is critical that the highest possible sensitivity and specificity of the molecular imaging are achieved. The small size of engineered scaffold proteins provides higher contrast and specificity in comparison with antibody-based imaging agents, which makes this type of imaging probes very promising. 7 However, the approaches for further enhancing imaging contrast and, hence, sensitivity, have to be identified and explored to improve detection of target expression in small metastases.
The imaging contrast depends on the ratio of radioactivity uptake in tumors and in normal tissues. Therefore, the decrease in the uptake of a radiotracer in normal tissues is as important as the increase in accumulation of radioactivity in tumors. Whereas factors determining accumulation of protein-based imaging agents in tumors (size, affinity, residualizing character of a label, and bioavailability) are relatively well understood, 45 factors determining accumulation in normal tissues are much less studied and understood. The most studied aspect is renal reabsorption, where several mechanisms have been identified for short peptides, 46 and some information is also available on hepatic uptake and hepatobiliary excretion. 47 This prompted us to systematically evaluate factors influencing the uptake of Affibody molecules in normal tissues, and one of the factors is the labeling chemistry.
The labeling chemistry (understood as the combination of the chemical properties of a radionuclide, the chelator or prosthetic group used for its attachment to a peptide, and the chemical nature of the linker between the chelator and the peptide) modifies the chemical and physicochemical properties of the solvent-exposed surface of the targeting peptide. Distribution of charge and lipophilic patches on the surface become altered, which should alter the interactions of the probe with blood proteins and constituents of normal tissue. This was previously shown for short (8–10 amino acids) radiolabeled peptides.34,36,48 Surprisingly, we found that the labeling chemistry also has a noticeable influence on the biodistribution profile of Affibody molecules, although their size (58 amino acids) is much larger than that of short peptides. 49 In principle, this opens a way to finding a labeling chemistry that will provide optimal biodistribution of Affibody molecules and, potentially, other engineered scaffold proteins of similar size.
Previously, we showed that the position of the DOTA chelator, which is conjugated to the synthetic anti-HER2 ZHER2:S1 Affibody molecule via an amide bond, influences the uptake of 111In-labeled Affibody molecules in normal organs and tissue and the rate of its clearance from blood. 39 Comparison of the biodistribution suggested that 111In-[DOTA-K58]-ZHER2:S1 provided the best tumor to blood and tumor to spleen ratios at 4 hours after injection and is therefore the best construct to be used as an imaging agent. Substitution of 111In with the short-lived positron-emitting nuclide 68Ga (T1/2 = 67.6 minutes) should enable the use of PET for imaging of HER2 expression with Affibody molecules. The use of PET would provide better spatial resolution, sensitivity, and quantification capacity of the imaging. 50 It would be attractive to use the same DOTA-conjugated ZHER2:S1 for both PET (where possible) and SPECT. However, it is not obvious that findings for 111In could be translated to the 68Ga label because the structures of gallium and indium complexes of DOTA are quite different. Indium has a coordination number of eight in its complex with monoamide-DOTA, and the complex has the geometry of a slightly distorted square antiprism. 51 Gallium in such a complex is hexacoordinated, and the complex has a cis-pseudo-octahedral geometry with one free carboxylate group. 51 Accordingly, the local distribution of charge in indium and gallium complexes is different, which may influence off-target interaction and uptake in normal tissues. Therefore, in this study, we performed an evaluation of the influence of the DOTA position (conjugation via formation of the amide bond at the N-terminal alanine, A1, lysine in the middle of helix 3, K50, or the C-terminal lysine, K58) on the biodistribution and targeting properties of the synthetic ZHER2:S1 Affibody molecule labeled with 68Ga. As expected, the use of DOTA in combination with labeling at high temperature permitted high labeling yield and high stability of the label (see Table 2). The conjugates demonstrated preserved saturable binding to HER2-expressing cells in vitro, which confirms the expected specificity of their binding (see Figure 2). There was no noticeable influence of chelator position on cellular processing of 68Ga-labeled ZHER2:S1 (see Figure 3).
In vivo, all conjugates labeled with both 111In and 68Ga demonstrated specific (saturable) uptake in HER2-expressing SKOV3 xenografts (see Figure 4). The relative biodistribution of the 111In-labeled conjugates (e.g., higher uptake of 111In-[DOTA-K58]-Z HER2:S1 in lung and liver than for 111In-[DOTA-A1] -ZHER2:S1) were in agreement with the previously published data, 39 although the biodistribution was evaluated at an earlier time point (2 hours after injection instead of 4 hours) in this study due to the short half-life of 68Ga. Furthermore, the biodistribution of 111In-[DOTA-A1]-ZHER2:S1 and 68Ga-[DOTA-A1]-ZHER2:S1 was compared earlier in a study concerning the influence of macrocyclic chelators attached at the N-terminus of ZHER2:S1. 43 These data were also in good agreement with the data from the current study (more rapid clearance of 111In from blood; higher uptake of 111In in tumor, lung, and bone; and higher uptake of 68Ga in liver). Thus, the experimental results were cross-validated by the results from other studies. The data suggest that substitution of 111In by 68Ga has a substantial influence on the biodistribution of all variants (see Table 3), with the most pronounced difference for [DOTA-K50]-ZHER2:S1. The biodistribution data suggest that labeling with 68Ga had a stronger influence on the biodistribution than 111In. One common feature was the somewhat lower uptake of 68Ga in tumor in comparison with 111In, but otherwise, there was no common pattern in the influence of the radionuclide on the biodistribution. For example, substitution of 111In by 68Ga was associated with higher radioactivity concentration in blood for 68Ga-[DOTA-A1]-Zher2:S1 (p < .05) and 68Ga-[DOTA-K50]-ZHER2:S1 (p < .005) but not for 68Ga-[DOTA-K58]-ZHER2:S1. Comparison of the liver uptake for the 68Ga label with the 111In label showed that the uptake was significantly (p < .0005) higher in the case of [DOTA-K50]-ZHER2:S1 and significantly lower (p < .05) in the case of [DOTA-K58]-ZHER2:S1, but there was no difference in the case of [DOTA-A1]-ZHER2:S1. This indicates, most likely, that several different off-target interactions are important for the biodistribution of Affibody molecules, which are affected in different ways by the substitution of label, depending on the label position. Unfortunately, this means that the results of optimization studies with one radionuclide cannot be translated to another radionuclide, and optimization should be performed for each potentially useful label.
Tumor to organ ratios are important characteristics of an imaging agent as they determine the imaging contrast. The results of this study suggest that 68Ga-[DOTA-K50]-ZHER2:S1 provides the lowest tumor to organ ratios and should therefore be excluded from consideration. There was no significant (p > .05) difference between the tumor to organ ratios of 68Ga-[DOTA-A1]-ZHER2:S1 and 68Ga-[DOTA-K58]-ZHER2:S1 for nearly all organs, but lung. The tumor to lung ratio of 68Ga-[DOTA-A1]-ZHER2:S1 (45 ± 5.2) was 1.7-fold higher than the ratio of 68Ga-[DOTA-K58]-ZHER2:S1. This is important as pulmonary metastases are frequent for breast cancer, 52 and HER2 is a vital predictive biomarker for breast cancer.
We previously investigated the influence of nuclide and chelators on HER2 targeting using the recombinantly produced anti-HER2 ZHER2:2395 Affibody molecules. 53 The ZHER2:2395 Affibody molecule has the same binding site for HER2 as ZHER2:S1. To ensure site-specific labeling, a C-terminal cysteine was engineered into ZHER2:2395, and maleimido derivatives of DOTA and NODAGA were conjugated. Thus, the major difference between these conjugates and the conjugates used in the current study was the conjugation chemistry. In recombinant DOTA-conjugated Affibody molecules, 111In provided significantly (p < .05 in a paired t-test) higher tumor to blood, tumor to liver, tumor to spleen, and tumor to bone ratios. In this study, with DOTA conjugated to the C-terminus via an amide bond, the 111In-labeled conjugate also had a significantly (p < .05) higher tumor to blood ratio, but tumor to liver, tumor to spleen, and tumor to bone ratios were higher for 68Ga. This is a strong indication that different ways to conjugate the same chelator can influence the imaging properties of the targeting conjugates and should be optimized.
In this study, both tumor uptake and tumor to blood ratio were higher for 111In-[DOTA-A1]-ZHER2:S1 than for 68Ga-[DOTA-A1]-ZHER2:S1, which might raise the question of whether the use of PET instead of SPECT is still justified in this case. The issue is the high sensitivity required for detection of small metastases with the diameter of 10 mm or smaller. With such an object size, the partial volume effect results in an appreciable decrease of the SPECT signal and deterioration of contrast (≈ four-to fivefold for 10 mm54). Therefore, 68Ga-[DOTA-A1]-ZHER2:S1 would provide better sensitivity for imaging of small objects despite an 18% decrease in tumor uptake and a 35% decreased tumor to organ ratio.
In conclusion, positioning of the DOTA chelator at the N-terminus provides the best tumor to organ ratios for 68Ga-labeled synthetic Affibody molecules. The position of the chelator in Affibody molecules strongly influences the biodistribution of the 68Ga-labeled tracer, and this effect can be used for optimization of the imaging properties. These observations may be important for development of Affbody-based imaging agents for other molecular targets, as well as for development of other engineered scaffold protein-based imaging probes.
Footnotes
Acknowledgments
Financial disclosure of authors: This work was supported by grants from the Swedish Research Council (Vetenskapsrådet) and Swedish Cancer Society (Cancerfonden).
Financial disclosure of reviewers: None reported.
