Abstract
A new approach in the treatment of clear cell renal carcinoma (ccRCC) is radioimmunotherapy (RIT) using anti-carbonic anhydrase IX (CAIX) antibody G250. To investigate the potential of RIT with lutetium 177 (177Lu)-labeled G250, we conducted a protein dose escalation study and subsequently an RIT study in mice with intraperitoneally growing ccRCC lesions. Mice with intraperitoneal xenografts were injected with 1, 3, 10, 30, or 100 μg of G250 labeled with 10 MBq indium 111 (111In) to determine the optimal protein dose. The optimal protein dose determined with imaging and biodistribution studies was used in a subsequent RIT experiment in three groups of 10 mice with intraperitoneal SK-RC-52 tumors. One group received 13 MBq 177LU-DOTA-G250, a control group received 13 MBq nonspecific 177LU-MOPC21, and the second control group was not treated and received 20 MBq 111In-DOTA-G250. The optimal G250 protein dose to target ccRCC in this model was 10 μg G250. Treatment with 13 MBq 177LU-DOTA-G250 was well tolerated and resulted in significantly prolonged median survival (139 days) compared to controls (49-53 days, p = .015), indicating that RIT has potential in this metastatic ccRCC model.
THE APPROVAL OF targeted therapies such as tyrosine kinase inhibitors (TKIs) and inhibitors of mammalian target of rapamycin has revolutionized the treatment of advanced clear cell renal cell carcinoma (ccRCC) during the last decade. Sunitinib, pazopanib, temsirolimus, and bevacizumab in combination with interferon-α have improved progression-free survival and are now approved as first-line treatments for metastatic ccRCC.1–5 Unfortunately, these agents have several disadvantages. First, they are associated with sometimes severe side effects, such as diarrhea, hypertension, fatigue, and hand-foot syndrome. 6 Moreover, treatment with these drugs is not curative, implicating chronic treatment. In addition, discontinuation of therapy may lead to flare-up of disease activity in some patients. 7 Continued treatment despite disease progression or a switch to another angiogenesis inhibitor with a relatively short interval is therefore necessary.
The need for a less toxic therapeutic option with good and durable responses led to studies targeting the carbonic anhydrase IX (CAIX) using monoclonal antibody (mAb) G250 (chimeric mAb also known as girentuximab). G250 specifically binds to CAIX, which is expressed in approximately 94% of ccRCCs, and expression in normal tissues is limited. 8 This preferential high expression in ccRCC has been used in many preclinical and clinical studies for imaging and therapy of ccRCC. 9
Due to the limited efficacy observed in the first clinical trials using 131I-girentuximab, the search for more suitable radionuclides was initiated. A preclinical study by Brouwers and colleagues revealed the superior therapeutic efficacy of lutetium 177 (177Lu)-, yttrium 90-, or rhenium 186-labeled girentuximab compared to iodine 131 (131I)- labeled girentuximab. As tumor growth in mice with subcutaneous xenografts was delayed most effectively by the 177Lu-labeled antibody, 10 a phase I dose escalation trial with 177Lu-girentuximab in patients with advanced ccRCC was initiated. The results of this trial were very promising as 177Lu-girentuximab radioimmunotherapy (RIT) was generally well tolerated and resulted in disease stabilization in the majority of patients. 11 However, now the question arises as to how to combine RIT with other treatment modalities, such as TKIs, as we recently found that the uptake of girentuximab is markedly reduced during treatment with these agents. 12 To design the optimal treatment strategy, additional preclinical studies regarding the sequencing of therapies are of the utmost importance.
In preclinical studies to date, tumor growth was induced by subcutaneous injection of tumor cells or grafting of harvested xenograft tissue, resulting in a single, palpable, visible tumor.10,13 Although the growth of subcutaneous tumors can be monitored easily with caliper measurements, subcutaneously growing xenografts differ substantially from tumor lesions in patients, particularly with respect to blood supply and physiology.14,15 In this study, we evaluated the potential of combined radioimmunodetection and RIT with murine G250 in an intraperitoneal ccRCC xenograft model that may more closely mimic human metastatic ccRCC.
Materials and Methods
Antibodies
G250 is a murine IgG1 mAb that is directed against the CAIX antigen expressed in ccRCC. The G250 antibody was affinity purified on a Protein-A column from the supernatant of G250 hybridoma cell cultures. MOPC21 (Sigma-Aldrich, Zwijndrecht, The Netherlands) is a murine IgG1 mAb not directed against any known antigen and was used as a negative control in the RIT experiment.
Conjugation, Radiolabeling, and Quality Control
For the antibody dose escalation study, both G250 and MOPC21 were conjugated with isothiocyanato-benzyl-diethylenetriaminepentaacetic acid (ITC-DTPA), as described previously. 10
For biodistribution studies, DTPA-conjugated G250 was incubated with 111In (Covidien BV, Petten, The Netherlands) in 0.1 M MES buffer, pH 5.5, at room temperature, under strict metal-free conditions for 20 minutes as described previously. 16 The specific activity of the antibody preparation was 1.07 MBq/μg (yield 98%). The protein dose was adjusted to 3, 10, 30, or 100 μg by adding unlabeled G250. For single-photon emission computed tomography (SPECT)/computed tomography (CT) studies, 67.2 μg DTPA-conjugated G250 was incubated with 403 MBq 111In. This preparation was purified on a PD10 column. The specific activity of 111In-DTPA-G250 used for imaging studies was 3.4 MBq/μg (overall yield 19%). Again, the protein dose for the different groups was adjusted by adding unlabeled G250. Ten megabecquerels of 111In-DTPA-G250 was administered intravenously via the tail vein (0.2 mL).
For the therapy experiment, isothiocyanato-benzyl- 1,4,7,10-tetraazacyclododecanetetraacetic acid (ITC-DOTA) was used as a bifunctional chelator because of the slightly better stability with 177Lu. 10 DOTA conjugation was performed essentially as described by Lewis and colleagues. 17 No-carrier-added 177Lu was acquired from ITG (Garching, Germany). For RIT, antibody-DOTA conjugates were radiolabeled with 177Lu (G250: 46% yield, MOPC21 53% yield) or with 111In (74% yield). The radiochemical purity of the radiolabeled antibody preparations in the RIT experiment all exceeded 95%. The specific activity of 177Lu-DOTA-G250, 177Lu-DOTA-MOPC21, and 111In-DOTA-G250 was 1.3, 1.3, and 1.6 MBq/μg, respectively.
Cell Culture
Tumor growth was induced by an intraperitoneal injection of 0.2 mL of a suspension of 3 × 10 6 SK-RC-52 cells, a CAIX-expressing human ccRCC cell line, 18 resulting in tumor nodules (submillimeter to 3 mm in diameter) in the peritoneal cavity after 2 to 4 weeks.
Animals
All experiments were conducted in accordance with the principles laid out by the revised Dutch Act on Animal Experimentation (1997) and approved by the institutional Animal Welfare Committee of the Radboud University Nijmegen. Animals were housed and fed according to the Dutch animal welfare regulations. The experiments were performed in female nude BALB/c nu/nu mice (8-10 weeks old) weighing 20 to 25 g (Janvier, le Genest-Saint-Isle, France). Mice were accustomed to laboratory conditions for at least 1 week before experimental use and housed under nonsterile standard conditions in filter-topped cages with free access to animal chow and water.
Protein Dose Escalation Study
Twenty-five female athymic BALB/c nu/nu mice were injected with SK-RC-52 cells as described above and divided in five groups. Three weeks after inoculation, the mice were injected with 3, 10, 30, or 100 μg G250-DTPA labeled with 10 MBq 111In. At the 1 μg dose level, the protein dose was too low for labeling with activity doses required for SPECT imaging. Therefore, the conjugate in this group was labeled with 1 MBq only, which is sufficient to determine the biodistribution of the radiolabeled antibody.
Imaging with the U-SPECT II scanner (MILabs, Utrecht, The Netherlands) was performed in the 3, 10, 30, or 100 μg groups 48 hours postinjection of 111In-DTPA-G250. A 1.0 mm diameter pinhole collimator tube was used. Mice were anesthetized with a mixture of oxygen, N2O, and isoflurane and placed in a supine position in the scanner. Body temperature was maintained at 38°C during the scan. The total scan time was approximately 60 minutes per animal for SPECT acquisition and 3 minutes for CT imaging. Images were reconstructed using the MILabs software. Immediately after completion of image acquisition, mice were euthanized, and the intraperitoneal cavity was inspected meticulously for the presence of intraperitoneal tumor depositions. Tumor nodules and tissues were dissected, and their concentration of the radiolabel (%ID/g) was determined. If no tumor lesions were detected, animals were excluded from the ex vivo biodistribution studies.
A tumor sample and samples of normal tissues (blood, muscle, heart, lung, spleen, pancreas, stomach, duodenum, kidney, liver) were dissected, weighed, and counted in a gamma counter (1480 Wizard 3, LKB/Wallace, Perkin-Elmer, Boston, MA). Injection standards were also counted to correct for radioactive decay. Tissue uptake of the radiolabeled antibody was expressed as a percentage of the injected dose per gram (%ID/g).
RIT Experiment
For the RIT experiment, 30 female BALB/c nu/nu mice were injected with SK-RC-52 cells as described and divided into three groups. Two weeks after tumor cell inoculation, mice were injected with 10 μg 13 MBq 177Lu-DOTA-G250 (n = 10), 13 MBq 177Lu-DOTA-MOPC21 (control group, n = 10), or 20 MBq 111In-DOTA-G250 (control group, n = 10).
The weight of the mice was measured weekly, and mice were euthanized in case of excessive palpable tumor growth, substantial weight loss, or other signs of significant disease progression, as determined by trained biotechnicians who were blinded for the treatment given to the mice. As in the protein dose escalation study, the presence of intraperitoneal tumor depositions was scored macroscopically. Animals were excluded from survival analyses if no tumor lesions were detected after inspection of the abdomen. SPECT/CT imaging to monitor disease progression was performed at 3-week intervals as described above. Primary end points were overall survival and toxicity.
Statistical Analysis
Statistical analyses were performed using IBM SPSS Statistics version 20.0 (IBM, Armonk, NY) and GraphPad Prism version 5.03 (GraphPad Software, San Diego, CA). Differences in uptake of radiolabeled antibodies were tested for significance using the nonparametric Kruskal-Wallis test and were considered significant at p < .05, two-sided. Tumor to blood (T/B) ratios were calculated by averaging the T/B ratios of the individual animals (tumor uptake [%ID/g] divided by blood activity [%ID/g]). Values are expressed as mean ± standard deviation. Median survival differences were tested for significance using a Mantel-Cox test and were considered significant at p < .05.
Results
Protein Dose Escalation Study
Mice did not show clinical signs of discomfort 3 weeks after inoculation of the SK-RC-52 tumor cells. At that time, the intraperitoneal tumor nodules were not palpable. On macroscopic inspection of the opened abdomen, multiple solid tumors were found, predominantly located at the subhepatic, -splenic, and -phrenic spaces and within the mesentery (Figure 1). The number of tumor lesions per mouse was typically more than 10, ranging in diameter from less than a millimeter to 3 mm. Eighteen of 25 mice (72%) had macroscopically visible tumors at dissection. Mice without macroscopically visible tumor growth were excluded from analyses.

Intraperitoneal tumor depositions close to the pancreas and spleen 21 days after tumor cell inoculation. Note the extensive neovascularization surrounding the tumors (arrow).
SPECT/CT imaging showed high mAb uptake in the abdomen of mice at all protein dose levels. No clear differences in image quality were observed between the different groups. After the animals were sacrificed, all imaged areas coincided with intraperitoneal tumors, which ranged from 1 to 2 mm, demonstrating preferential mAb uptake in the tumors. The biodistribution of 111In-DTPA- G250 was determined 48 hours postinjection (Figure 2). Tumor uptake of the radiolabeled antibody was very high at all protein dose levels, with the highest mean uptake observed in the 10 μg group (54.9 ± 3.5), but the difference between the groups was not significant.
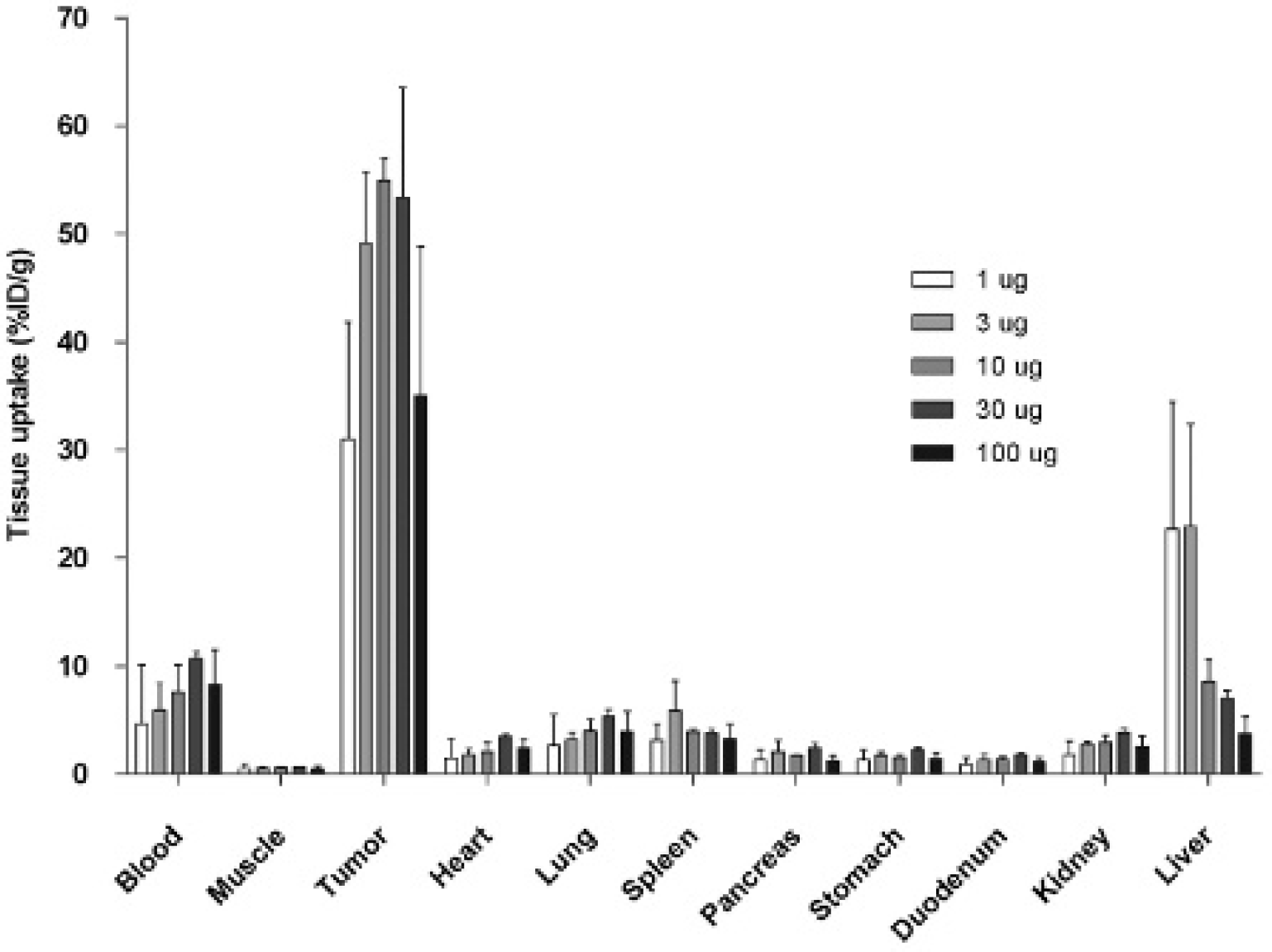
Biodistribution and specific tumor targeting of escalating doses of 111In-DTPA-G250 48 hours after injection. Tissue uptake is expressed as %ID/g. Values represent mean ± standard deviation. Note the high liver uptake in the 1 and 3 μg groups.
The highest T/B ratio was found in the 1 μg group (14.7 ± 11.6), mainly due to lower blood values, but the ratio was not statistically significant different from that of the other groups (p = .297). Significantly higher liver uptake was observed in the 3 μg group (23.0 ± 9.5) compared to the 10, 30, and 100 μg dose level groups (p = .036). At protein dose levels ≥ 10 μg, favorable T/B ratios (8.5 ± 3.3, 5.1 ± 1.,8 and 4.4 ± 1.1, respectively) and lower liver uptake was observed, especially in the 10 μg group (T/B ratio 8.5 ± 3.3). Therefore, it was concluded that the optimal G250 dose to target ccRCC in this model was 10 μg.
RIT Study
Twenty-nine of 30 mice (97%) developed visible tumors during the experiment as assessed by meticulous macroscopic inspection of the peritoneal cavity. All mice survived the treatment, and apart from mild transient weight loss (< 15%) associated with the 177Lu-labeled antibody preparations, treatment was generally well tolerated. Three mice died during the imaging procedures (injections or anesthesia) and were excluded from the analyses. One of these animals did not have macroscopically visible tumor growth.
Survival curves of the three groups are shown in Figure 3. Mice reached the humane end point when signs of significant disease progression or clinical deterioration (mainly severe weight loss) occurred, as judged by blinded biotechnicians. Lesion size ranged from < 1.0 to 12 mm. The response to treatment was also monitored with 111In-DTPA-G250 SPECT/CT. As shown in Figure 4, the progression of tumor lesions could easily be monitored in vivo, and when animals were sacrificed, a close correlation between SPECT images and macroscopic tumors was observed.
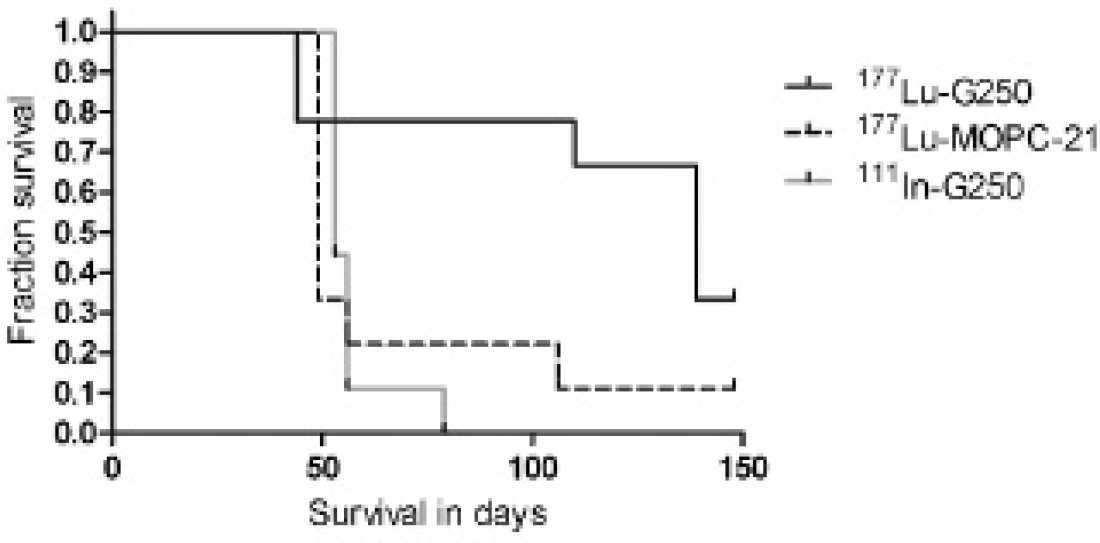
Kaplan-Meier survival estimates for the three groups in the radioimmunotherapy experiment. Treatment with 177Lu-DOTA-G250 resulted in significantly prolonged median survival of 139 days, in comparison with 49 days (177Lu-DOTA-MOPC21) and 53 days of 111In-DOTA-G250 (Mantel-Cox p 5 .015).

In vivo SPECT/CT imaging of mice with intraperitoneal clear cell renal cell carcinoma tumors. Rapid disease progression occurred in an animal in the 111In-DOTA-G250 group, which reached a humane end point after 5 weeks (A). Another animal was treated with 177Lu-DOTA-G250 immediately after baseline imaging (B). Note the presence and progression of a tumor in the left side of the abdomen after 3 and 6 weeks (arrows). The tumor was not detected with SPECT/CT after 9 weeks, was not found during dissection, and possibly regressed due to radioimmunotherapy.
Treatment with 177Lu-DOTA-G250 resulted in significantly prolonged median survival of 139 days, which is significantly longer than the survival of the mice in both control groups. The median survival of the mice that received 177Lu-DOTA-MOPC21 was 49 days and 53 days in the 111In-DOTA-G250-treated group (Mantel-Cox test, p = .015). The study was terminated after 150 days, the preset end point for assessment of survival.
Discussion
In this study, the potential of radioimmunodetection and RIT with radiolabeled G250 in an intraperitoneal ccRCC xenograft model was evaluated. The intraperitoneal model with the SK-RC-52 cell line proved reliable and highly reproducible as substantial tumor growth was established in the vast majority of the inoculated mice (in approximately 85%).
Several studies indicate that substantial differences in blood supply and physiology exist within different tumor models.14,15 In the current study, we opted for intraperitoneally growing tumor nodules to more closely mimic metastatic disease with smaller tumor depositions in comparison with subcutaneous xenografts. The drawbacks of intraperitoneal tumor growth are that the lesions are not palpable the first weeks after induction and that straightforward visual monitoring of the tumor growth is not possible. This makes in vivo imaging techniques indispensable if longitudinal quantification of tumor burden is required.
In the protein dose escalation study to determine the optimal antibody dose for in vivo targeting of the intraperitoneal ccRCC xenografts, mice did not show clinical signs of discomfort 3 weeks after inoculation of the SK-RC-52 tumor cells, nor were the intraperitoneally growing tumors palpable. Despite the small size of the intraperitoneal tumors, small depositions of approximately 1 to 2 mm and larger were well visualized with SPECT/CT 48 hours postinjection of 111In-DTPA-G250, emphasizing the specific and high accumulation of mAb G250. No clear differences in image quality between the different dose levels were observed. Due to the low protein and radiation dose administered, it is highly unlikely that multiple 111In-DTPA-G250 injections used for disease monitoring will have any effect on tumor progression.
The protein dose escalation experiment demonstrated that optimal T/B ratios and low liver uptake levels were reached at a protein dose of 10 μg. Very high liver and low blood levels were observed in the animals with normal tumor load after 3 weeks in the 1 and 3 μg groups. The mAb G250 biodistribution in the animals without any tumor depositions in these low-protein dose groups was similar to that in the 10, 30, and 100 μg groups (data not shown). This suggests that the antibody distribution at low protein doses is heavily influenced by the presence of tumor. An inverse correlation was observed between blood levels and liver uptake at the lower protein doses. This correlation could be the result of binding of 111In-DTPA-G250 to circulating antigen in the blood due to antigen shedding by the tumor, 19 which will lead to rapid clearance of the antibody-antigen immune complex via the liver, although we have no formal evidence of circulating antigen.
An additional advantage of an antibody dose of 10 μg or more is that it is easier to label the antibody with higher doses of radioactivity, which are required for RIT. In the subsequent RIT study, we found a significantly prolonged median survival in the group treated with 177Lu-DOTA- G250. The difference in median survival in this study (139 days) and our previous results in a subcutaneous model (300 days) 10 is likely due to the location of the tumor depositions as the intraperitoneal tumors often result in obstruction of the gastrointestinal tract, which subsequently leads to rapid weight loss and clinical deterioration of the animals. In both models, 177Lu-DOTA-G250 clearly shows therapeutic potential.
Recently, several clinical studies reported severely hampered antibody targeting when radiolabeled antibodies were administered during TKI treatment.12,20 To successfully combine TKI treatment and RIT in the future, additional preclinical studies are warranted to investigate the optimal sequence and timing of both treatment modalities. The intraperitoneal ccRCC xenograft model used in current study mimics metastatic ccRCC and is therefore suitable for future experiments, for instance, to determine for how long antibody targeting to the tumor is reduced after cessation of the TKI treatment. Better understanding of underlying mechanisms could be an important step in the development of successful RIT strategies for ccRCC.
Footnotes
Acknowledgments
We thank Lieke Claessens-Joosten, Annemarie Eek, Hanneke Peeters, Bianca Lemmers-van de Weem, and Henk Arnts for their technical assistance.
Financial disclosure of authors: P.F.A. Mulders, W.J.G. Oyen, O.C. Boerman, and E. Oosterwijk serve(d) on an advisory board for Wilex AG, Munich, Germany.
Financial disclosure of reviewers: None reported.
