Abstract
Folate receptor β (FR-β) is overexpressed on activated, but not resting, macrophages involved in a variety of inflammatory and autoimmune diseases. A pivotal step in atherogenesis is the subendothelial accumulation of macrophages. In nascent lesions, they coordinate the scavenging of lipids and cellular debris to define the likelihood of plaque inflammation and eventually rupture. In this study, we determined the presence of FR-β-expressing macrophages in atherosclerotic lesions by the use of a fluorine-18-labeled folate-based radiotracer. Human endarterectomized specimens were used to measure gene expression levels of FR-β and CD68. Increased FR-β and CD68 levels were found in atherosclerotic plaques compared to normal artery walls by quantitative real-time polymerase chain reaction. Western blotting and immunohistochemistry demonstrated prominent FR-β protein levels in plaques. FR- β-positive cells colocalized with activated macrophages (CD68) in plaque tissue. Carotid sections incubated with 3′-aza-2′- [18F]fluorofolic acid displayed increased accumulation in atherosclerotic plaques through in vitro autoradiography. Specific binding of the radiotracer correlated with FR-β-expressing macrophages. These results demonstrate high FR-β expression in atherosclerotic lesions of human carotid tissue correlating with CD68-positive macrophages. Areas of high 3′-aza-2′-[18F]fluorofolic acid binding within the lesions represented FR-β-expressing macrophages. Selectively targeting FR-β-positive macrophages through folate-based radiopharmaceuticals may be useful for noninvasive imaging of plaque inflammation.
ATHEROSCLEROSIS, a progressive inflammatory disease, plays a key role in the pathogenesis of cardiovascular diseases. 1 It is characterized by thickening of the artery walls due to accumulation of lipids and fibrous elements in the tunica intima leading to the formation of atherosclerotic plaques. Activated macrophages accumulate at the site of inflammation and take up large quantities of lipoprotein particles, become foam cells, and discharge inflammatory cytokines and growth factors that promote the recruitment and proliferation of other cells. 2 Matrix metalloproteinases (MMP), reactive oxygen species, and proinflammatory cytokines such as tumor necrosis factor a (TNF-α) provoke plaque destabilization, which makes it prone to rupture. These advanced plaques, so-called vulnerable plaques, can lead to serious thrombotic events, comprising the risk of initiating myocardial infarction or stroke.3–5
The influx of macrophages is recognized as an important feature of atherosclerotic plaque formation. In general, the macrophage population is of high phenotypic heterogeneity, and cells can express a wide spectrum of activation programs. Exploiting the fact that activated macrophages but not resting macrophages or other immune cells overexpress the folate receptor (FR) could be of clinical significance with regard to the diagnosis and therapy of inflammatory diseases. 6
Several isoforms of the FR have been reported with distinct expression patterns in normal and pathologic tissues. The two most relevant FR isoforms are FR-α (FOLR1 gene), overexpressed by cancer cells, and FR-β (FOLR2 gene), present on activated macrophages. Both isoforms possess a glycosylphosphatidylinositol anchor and both bind folic acid (FA) with high affinity (KD < 10−9 M).7,8 As a molecular probe, FA offers several advantages, such as its small size (molecular weight = 441 g/mol), stability over a broad range of temperature and pH values, and the possibility of site-specific chemical modifications without losing its high affinity for the FR.9,10 Preclinical studies demonstrated that folate-based targeting agents were taken up by activated macrophages involved in inflammatory processes and that the uptake was mediated by FR-β.6,11–15
In the field of nuclear imaging, a number of folate-based radiopharmaceuticals have been developed and preclinically evaluated for the diagnosis of FR-positive cancer and inflammatory diseases by single-photon emission computed tomography (SPECT) with the isotopes 99mTc, 111In, and 67Ga and by positron emission tomography (PET) with 18F and 68Ga.13,16–19
99mTc-EC20 was successfully employed for the imaging of atherosclerotic plaques in apolipoprotein E (ApoE)- deficient mice, a model of atherosclerosis. 20 Ex vivo autoradiography and subsequent histology of excised aortas demonstrated a correlation between 99mTc-EC20 uptake and macrophage accumulation. Winkel and colleagues demonstrated that the folate-based SPECT radiotracer 111In-EC0800 is not a clear-cut marker for the detection of vulnerable plaques but detects both stable and vulnerable atherosclerotic plaques in a shear stress-induced atherosclerosis mouse model. 21 Another group showed ex vivo accumulation of a folate–fluorescein isothiocyanate (FITC) conjugate in excised human atherosclerotic plaque samples. 22 Regional accumulation correlated with the density of CD68, a marker for activated macrophages, and with hypoxia as visualized by staining for hypoxia-inducible factor 1a (HIF-1α). 22
In this work, we addressed the questions whether (1) expression levels of human FR-β differ between plaques of stable and vulnerable appearance and (2) whether our recently developed 18F-labeled folate-based radiotracer, 3′- aza-2′-[18F]fluorofolic acid, 23 binds to human atherosclerotic plaques. Excised artery wall segments from carotid endarterectomy (CEA) were classified into stable plaques, vulnerable plaques, and healthy artery walls, respectively, by a vascular surgeon, and classification was confirmed by histochemistry methods according to published guidelines. 24 Expression of CD68, FR-α and FR-β was quantified on the messenger ribonucleic acid (mRNA) level by quantitative real-time polymerase chain reaction (qPCR) and on the protein level by Western blotting and immunohistochemistry. Binding of 3′-aza-2′-[18F]fluorofolic acid to human atherosclerotic plaques was evaluated by in vitro autoradiography.
Material and Methods
Subjects' Characteristics, Clinical Data, and Tissue Banking
This study was conducted with excised tissue from 37 patients referred to the Clinic for Cardiovascular Surgery of the University Hospital Zurich (Zurich, Switzerland) for CEA between 2010 and 2012. Written informed consent was obtained from all patients. The atherosclerotic plaques were excised from the internal, external, and common carotid artery by bifurcation advancement.25,26 All excised specimens were photographed for macroscopic reevaluation before transfer within 5 minutes into RNAlater solution (Sigma, St. Louis, MO) and stored at 4°C until further use. According to the classification of the American Heart Association, plaques were characterized as stable if they macroscopically presented a lipid core encapsulated between a fibrous cap and the media without endothelial disruption. Plaques were considered vulnerable if there was intraplaque hemorrhage evolving in the necrotic core, impaired endothelial surface, or thrombosis.24,26 These macroscopic features of vulnerable plaques and the presence of the intact fibrous cap in stable plaques were confirmed by histologic analysis with hematoxylin-eosin (HE) (Sigma) and Masson-Goldner's trichrome staining (Morphisto, Frankfurt/Main, Germany) according to the manufacturer's protocols (Figure S1, online version only). Furthermore, plaques with a general fibrous cap thickness < 500 μm and a minimum cap thickness < 200 μm were classified as vulnerable according to the Oxford Plaque Study. 27 The microscopic classification was in agreement with the macroscopic classification. In this study, human tissue samples of 6 normal artery walls and 25 stable and 15 specimens of vulnerable plaques were used. The normal artery walls are redundant segments, mostly from Arteria thyroidea or Arteria sternocleidomastoidea, which had to be removed during surgeries.
Mouse Tissue
FR-positive KB xenografts, generated by subcutaneous inoculation of human KB cells (cervical carcinoma cell line, subclone of HeLa cells) into the shoulder region of athymic nude mice (CD-1 Foxn-1/nu, Charles River, Sulzfeld, Germany) kept on a folate-deficient diet (Harlan Laboratories, Indianapolis, IN), and kidneys were excised and used as positive controls for high FR expression.7,28,29
RNA Isolation, Reverse-Transcription Reaction, and qPCR
A small segment of plaque was used for total ribonucleic acid (RNA) isolation according to the protocols of the Isol-RNA Lysis Reagent (5 PRIME, Gaithersburg, MD) and the bead-milling TissueLyser system (Qiagen, Hilden, Germany). A QuantiTect Reverse Transcription Kit (Qiagen) was used to generate complementary deoxyribonucleic acid (cDNA). Human β-actin (ACTB) primer, forward 5′-CATGTACGTTGCTATCCAGGC-3′, reverse 5′-CTCCTTAATGTCACGCACGAT-3′, was synthesized by Microsynth (Balgach, Switzerland). All other primers were purchased from Qiagen: human FOLR1 (QT01674316); human FOLR2 (QT00062608), and human CD68 (QT00037184).
qPCR of β-actin, FOLR1, FOLR2, and CD68 gene expression was performed with the DyNAmoTM Flash SYBR Green qPCR Kit (Thermo Scientific, Runcorn, UK) using a 7900 HT Fast Real-Time PCR System (Applied Biosystems, Carlsbad, CA). All reactions were performed in duplicate and within three independent runs. Quantitative analysis was performed using SDS Software v2.4 (Applied Biosystems, Carlsbad, CA) and a previously described 2−ΔΔCt quantification method with β-actin as reference for normalization. 30 The specificity of the PCR products of each run was determined and verified with the SDS dissociation curve analysis feature.
Protein Isolation, Western Blotting, and Densitometry Analysis
For preparation of protein lysate, frozen plaque tissue was grinded in a mortar under liquid nitrogen. The powder was collected in 300 to 500 μL CellLytic MT buffer (Sigma) containing Halt protease inhibitor (Promega, Madison, WI). Mixtures were homogenized by sonification and centrifuged at 12,000g for 10 minutes at 4°C. The supernatants were collected and analyzed by Western blot. Separation of proteins was performed by 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE), according to standard protocols (Bio-Rad, Hercules, MA). For detection of FR-α, protein lysates were mixed with nonreducing sample buffer (64.3 mM Tris/HCl, pH 6.8; 10% glycerol; 0.02% bromophenol blue), whereas reducing conditions (including 2% SDS, 5% β-mercaptoethanol) were applied for detection of FR-β and β-actin. After SDS-PAGE, proteins were transferred to nitrocellulose membranes (GE Healthcare, Little Chalfont, UK). The blots were blocked with 5% dried milk powder in Tris-buffered saline-Tween 20 (pH 7.4). They were then processed for immunostaining with either a mouse monoclonal antibody against human FR-α (1:500, MAB5646, R&D Systems, Abingdon, UK), rabbit polyclonal to human FR-β (1:800, ab103998, Abcam, Cambridge, UK), or an antibody against β-actin (1:8,000, A2066, Sigma) at 4°C overnight. Bound antibodies were visualized with anti-rabbit or anti-mouse immunoglobulin G (IgG) secondary antibodies conjugated with horseradish peroxidase diluted to 1:80,000 (A0545 and A4416, respectively, Sigma). The antigen-antibody complexes were detected by enhanced chemiluminescence. Western blots were repeated at least twice to confirm the results. The stained bands were scanned and quantified with ImageJ 1.45 software (National Institutes of Health, Bethesda, MD). The integrated optical density of the FR-β band was normalized to the integrated density of the corresponding β-actin band. This ratio was further normalized to the corresponding ratio of a normal artery defined as a reference sample. The results are therefore presented as relative expression.
Histology and Immunohistochemistry
For histology, immunohistochemistry, and autoradiography, human plaque segments were embedded in Neg-50 Frozen Section Medium (Thermo Scientific, Runcorn, UK) and cut in 7 and 20 μm slices, respectively, on a cryostat (Cryo-Star HM 560 MV, Microm International, Walldorf, Germany), thaw-mounted onto coated glass slides (Superfrost Plus, Thermo Scientific, Runcorn, UK), and stored at −80°C until further use. Frozen sections (≈ 10 μm thick) from mouse kidney and KB xenografts were generated as described previously. 29 For microscopic characterization, HE (Sigma) staining was applied according to the manufacturer's protocols.
Immunohistochemistry was performed according to the protocol of Dako REAL EnVision Detection System (peroxidase/DAB, rabbit/mouse) (Dako, Carpinteria, CA) using mouse anti-human FR-α (1:100, MAB5646, R&D Systems), rabbit anti-human FR-β (1:100, ab103998, Abcam), or mouse anti-human CD68 (1:50, ab955, Abcam) antibodies to stain consecutive plaque sections. Nuclei were counterstained with hematoxylin solution, Gill No. 1 (Sigma). All tissue sections were mounted with Eukitt quick-hardening mounting medium (Sigma). For every immunohistochemical experiment, a negative control was performed by omitting the primary antibody. Sections were evaluated by microscopy (Zeiss AxioImager A1, Jena, Germany, or digital slide scanner Pannoramic 250, Sysmex, Horgen, Switzerland). Eight independent investigators, not familiar with the project, blindly scored the stained sections as weak, moderate, and strong staining intensities with a training table as reference (Figure S2, online version only). The results were compiled as a percentaged average of the eight scores. 26
In Vitro Autoradiography
The 18F-labeled FA derivatives 3′-aza-2′-[18F]fluorofolic acid and [18F]fluoro-deoxy-glucose folate were synthesized according to the procedures previously published by our group.23,31 Frozen slices from human atherosclerotic plaques (20 μm), mouse kidney (10 μm), and KB xenograft (10 μm) were thawed at room temperature for 10 minutes and washed in Tris buffer (50 mM Tris, 150 mM NaCl, pH 7.4) containing 0.1% bovine serum albumin (BSA) for 10 minutes on ice. The slices were incubated with 100 μL 3′-aza-2′-[18F]fluorofolic acid or [18F]fluoro-deoxy-glucose folate solution (1 nM in Tris buffer/0.1% BSA) for 40 minutes in a humidified chamber at room temperature. For blockade conditions, both radiotracer solutions contained 10 mM FA (Sigma). After decanting, the slices were washed for 5 minutes in Tris buffer/0.1% BSA, 3 × 3 minutes in Tris buffer and 2 × 5 seconds with distilled water at 4°C. The slices were dried and exposed for 10 to 30 minutes to a BAS-MS 2025 phosphor imaging plate (Fuji Film, Dielsdorf, Switzerland), which was subsequently scanned in a BAS- 5000 bioimaging analyzer (Fuji Film). Data were analyzed and processed with AIDA 4.5 software (Raytest, Sprockhövel, Germany). The amount of radioactivity bound to the tissue samples was quantified with a calibration curve derived from a series of standards deposited on Whatman paper (Whatman, Bottmingen, Switzerland) and exposed to the same plate. Radioactivity signals were within the linear range of the calibration curve. Tissue radioactivity, corrected for plaque size, was subtracted from the accumulated radioactivity under blocking conditions to calculate the specific binding. The specific binding was normalized to one of the vulnerable plaques (value of specific binding equals 1) serving as reference in all experiments and, therefore, expressed as relative binding. In the end, all sections were HE stained as described above. Experiments were repeated twice (for each radiofolate).
Statistics
The significance of intergroup differences was evaluated with SigmaPlot 12.5 (Systat Software, San Jose, CA) using nonparametric statistical analyses (Mann-Whitney U test), and p < .05 was considered significant.
Results
Subjects' Characteristics
Patient characteristics are summarized in Table 1. The asymptomatic patients group contained five men and three women, with a mean age of 69.7 ± 5.5 years (range 64-81 years). The symptomatic patients group consisted of eight men and four women, with a mean age of 74.5 ± 7.7 years (59-83 years). Six normal artery segments and 25 stable and 15 vulnerable plaque specimens were obtained.
Characteristics of the Patients Included in This Study

CEA = carotid endarterectomy.
Increased Expression of FR-β and CD68 in Human Carotid Lesions
FOLR1, FOLR2, and CD68 gene expression was analyzed in human carotid plaques and in normal artery samples by qPCR (Figure 1). FOLR1 mRNA expression was very low in all groups (see Figure 1A). FOLR2 expression was significantly increased in atherosclerotic plaques (Stab+Vuln) compared to normal arteries (7.6-fold; p = .047) (see Figure 1B). However, no difference was found between the stable and vulnerable plaque groups. The findings were similar for CD68, with a significant increase in atherosclerotic plaques compared to normal tissue (4.7-fold; p = .011) (see Figure 1C). Relative FOLR2 expression of each specimen plotted against the respective CD68 expression is shown in Figure 1D. FOLR2 and CD68 gene expression profile showed a positive correlation with R = .32.
Western blot analyses were performed to assess protein levels of the FR isoforms (Figure 2A). As found by qPCR, FR-α protein expression was negligible in normal and plaque tissue, but the protein was abundantly present in KB xenograft lysates, which was used as a positive control. FR-β was detected on the protein level at a size of 40 kDa and in significantly higher amounts in plaques than in normal artery walls (8.2-fold; p = .048; see Figure 2, A and B). However, FR- β expression in stable and vulnerable plaques did not differ.

Quantitative real-time PCR was used to determine relative mRNA levels of FOLR1 (A), FOLR2 (B), and CD68 (C) in normal arteries (open circles), stable plaques (gray circles), and vulnerable plaques (black circles). Stab+Vuln (semifilled circles) combines values obtained from stable and vulnerable plaques. Expression levels were calculated relative to β-actin (n = 3). Box plots represent statistical values. The boundaries of the box indicate the 25th and the 75th percentile, respectively. A dashed line graphs the mean and a solid line the median. Whiskers (error bars) above and below the box indicate the 10th and 90th percentiles. Outliers are indicated by black dots outside the whiskers. D, Correlation between relative mRNA levels of FOLR2 and CD68. Open circles = normal arteries; gray circles = stable plaques; black circles = vulnerable plaques.
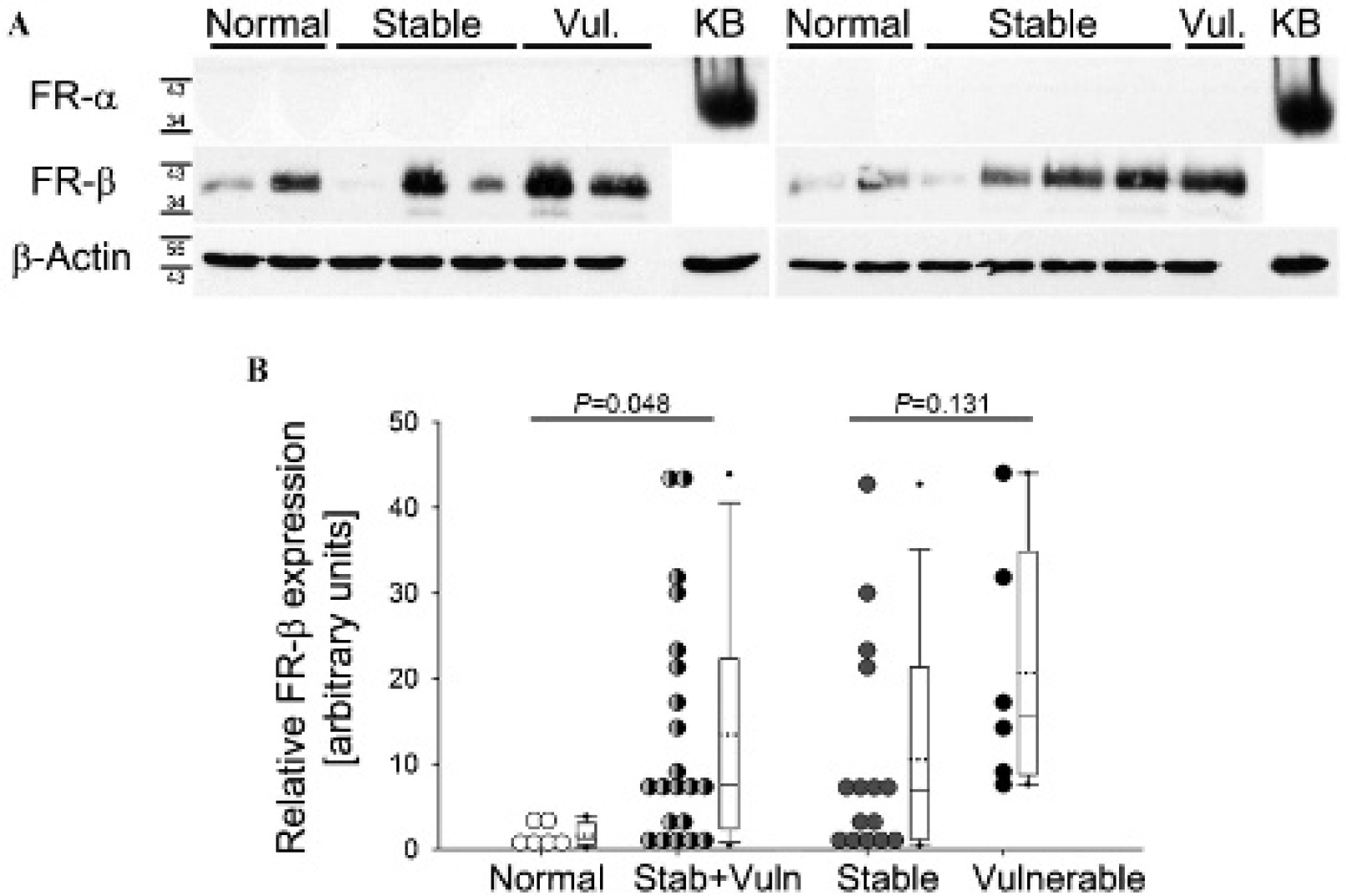
A, Representative Western blots of protein expression from human normal arteries, stable plaques, vulnerable (Vul.) plaques, and KB xenografts (KB) using human-specific antibody against FR-α or FR-β. β- Actin served as a loading control. B, Semiquantification of FR-β-band intensities from three independent experiments, normalized to β-actin. Stab+Vuln (semifilled circles) combines values obtained from stable and vulnerable plaques.
Correlating FR-β and CD68 Expression in Immunostained Atherosclerotic Plaques
We stained normal arteries and stable and vulnerable plaques to evaluate the presence and localization of FR-β- expressing cells. Macrophage-rich areas were identified by CD68-positive immunohistostaining. We compared the distribution of CD68 staining to that of FR-β-positive staining in serial sections. As shown in Figure 3 and Figure S3 (online version only), normal artery walls showed no staining for CD68 and FR-β. In both stable and vulnerable plaques, distinct FR-β-positive spots were observed. The majority of FR-β-expressing cells colocalized with CD68-positive macrophages at sites within the plaque. Immunohistostaining for FR-α was overall weak in normal arteries and both plaque types but positive in KB xenograft sections (see below and Figure S2).

Representative immunohistochemical stainings of a normal artery (A, B) and stable (C, D) and vulnerable (E, F) plaques using anti-CD68, anti-FR-β, and anti-FR-α antibodies or control IgG (omitting primary antibody). Boxes in A, C, and E indicate high-magnification areas shown below each plaque in B, D, and F. Boxes apply for all four stainings. Sections were counterstained with hematoxylin, resulting in blue nuclei. Lu = blood vessel lumen. Scale bar: 2.5 mm in low-magnification (A, C, E) and 1 mm in high-magnification (B, D, F) images.
Figure 4 shows the scoring of the staining intensities by eight independent investigators. The CD68-staining intensity was estimated as strong in 47% of the stable and 64% of the vulnerable plaques. The antibody anti-FR-β showed a strong staining in 64% of the stable and 77% of the vulnerable plaques.

Estimation of the staining intensities obtained with antibodies anti-CD68, anti-FR-β, and anti-FR-α. Six normal arteries, 22 stable plaques, and 12 vulnerable plaques were analyzed.
Consistent with the Western blotting and qPCR, immunostaining results revealed increased expression of FR-β and CD68 in atherosclerotic plaque samples, and absence of FR-α. FR-β expression correlated with the presence of the activated macrophage marker CD68.
Accumulation of 3′-Aza-2′-[18F]Fluorofolic Acid in Inflammatory Loci of Human Carotid Plaques
For in vitro autoradiography studies, an 18F-labeled FA derivative, designated as 3′-aza-2′-[18F]fluorofolic acid, was used. Our group previously published the radiochemistry and in vitro/in vivo characteristics of 3′-aza-2′- [18F]fluorofolic acid, demonstrating high binding affinity in the range of FA with about 1.4 ± 0.5 nM and high in vivo specificity for FR-positive tissue as determined by PET. 23 3′-Aza-2′-[18F]fluorofolic acid bound preferentially to atherosclerotic plaques (Figure 5, A and B, and Figure S4, online version only). Nonspecific binding was determined in the presence of excess unlabeled FA, showing a complete blockade in all sections. The pattern of radioactivity was similar to the immunohistochemical staining patterns for FR-β and CD68 on adjacent sections (see Figure 5A). FR-β- and CD68-positive cells were located in areas of high binding of 3′-aza-2′- [18F]fluorofolic acid. Mouse kidney sections showed strong binding of 3′-aza-2′-[18F]fluorofolic acid to the renal cortex, which was completely blocked in the presence of FA. In addition, 3′-aza-2′-[18F]fluorofolic acid specifically bound to sections of KB xenografts. KB xenograft sections show positive FR-α but negative FR-β immunohistostaining.

A, Representative immunohistochemistry, hematoxylin-eosin staining, and autoradiography images of a human normal artery, stable plaque, vulnerable plaque, mouse kidney, and KB xenograft. For immunohistochemical staining of human artery samples and KB xenograft, antibodies anti-CD68 and anti-FR-β, and for KB xenograft an anti-FR-α antibody, were used. All antibodies used are human specific and not applicable (n/a) on mouse kidney sections. Adjacent sections were used for in vitro autoradiography with 3′-aza-2′-[18F]fluorofolic acid under baseline conditions or blocking conditions with excess of folic acid (FA). Autoradiograms (baseline and blocked) are overlapped with the corresponding hematoxylin-eosin-stained section. Scale bar for human artery tissue: 2.5 mm; mouse kidney and KB xenograft: 5 mm. Lu = blood vessel lumen. B and C, Semiquantitative autoradiography-derived measurement for the specific binding of 3′-aza-2′-[18F]fluorofolic acid (B) and [18F]fluoro-deoxy-glucose folate (C) in normal arteries and stable and vulnerable plaques. Stab+Vuln (semifilled circles) combines values obtained from stable and vulnerable plaques.
Binding of 3′-aza-2′-[18F]fluorofolic acid was 7.7-fold higher on plaque tissue than on normal arteries (p = .049) (see Figure 5B). Specific binding was slightly but not significantly higher in vulnerable than in stable plaques (2.3-fold; p = .052). These results were confirmed with another highly specific folate derivative, [18F]fluorodeoxy-glucose folate (Figure 5C).
Discussion
The molecular differentiation of plaques at risk from stable plaques remains an important yet elusive challenge of cardiovascular research. Macrophages are decisive in the chronic inflammatory processes that provoke atherogenesis. 2 It is tempting to expect that the accumulation of activated macrophages in atherosclerotic lesions correlates with plaque vulnerability. However, macrophage infiltration has both anti- and proatherogenic effects, and the involvement of macrophage subtypes in processes leading to vulnerability or stabilization is not completely resolved yet. 2 According to a recent study with > 200 patients, occurrence of a cardiovascular event did not correlate with the total number of CD68-positive foam cell macrophages but rather with the proportion of MMP12-positive foam cells within a representative plaque. MMP12 was upregulated in foam cells undergoing apoptosis, a beneficial event in early but a detrimental event in advanced plaques.2,32
It is currently not known whether plaque burden of FR-β-positive macrophages and imaging of FR, respectively, are predictive of plaque rupture or occurrence of a life-threatening event. There are some indications that FR- β expression on macrophages may correlate with plaque vulnerability. 22 FR-β-expressing macrophages produce TNF-α and reactive oxygen species, indicative of proinflammatory M1-type macrophages. 6 TNF-α and reactive oxygen species are both involved in plaque destabilization.
FR-β/CD68-positive foam cells expressing TNF-α were present at high levels in plaques of ApoE-deficient mice. 33 Moreover, FR-β-targeted immunotoxin treatment not only resulted in a significant reduction in macrophages expressing CD68, FR-β, and TNF-α but also reduced progression of atherosclerotic lesions in this model. 33
In our immunohistochemical study, FR-β and CD68 were observed mainly on the medial side of the intima, where macrophages tend to be larger than those on the luminal side. 34 As observed by others, not all FR-β-positive regions stained positive for CD68. 33 This could result from the presence of FR-β-containing cell remnants. Such cell remnants may origin from apoptosis of foam cell macrophages. Due to high apoptotic activity in vulnerable plaques, we would assume increased FR-β levels. However, this was not the case, and higher sample numbers are needed to elucidate that.
To date, [18F]fluorodeoxyglucose ([18F]FDG) has been most commonly used as a PET agent in vascular biology.35–38 The rationale for using [18F]FDG to identify plaques is the metabolic activity of macrophages. However, issues of specificity of [18F]FDG uptake in inflammatory macrophages remain, as well as how this inflammatory signal can be employed for predicting plaque rupture. [18F]FDG-PET is limited by the abundance of imaging target throughout various stages of atherosclerosis development. Proximity to the highly metabolically active myocardium, and consequent signal spillover, limits the use of [18F]FDG for coronary imaging. The choice of targets for imaging atherosclerotic plaques is clearly important. Promising targets found in the process of inflammation are radiolabeled cytokines, chemokines, or their receptors. Several studies evaluated the feasibility of noninvasive detection of MMP activity, using, for example, 99mTc- or 18F-labeled MMP inhibitors.39,40 The tripeptide arginine-glycine-aspartic acid (RGD) is a high-affinity ligand of αVβ3-integrin that is expressed on macrophages and endothelial cells and is upregulated in angiogenesis. Various radiolabeled RGD derivatives, among them [18F]galacto-RGD, were suggested for the imaging of vulnerable plaques. 41 [99mTc]annexin V, with high affinity to phosphatidylserine, targets the surface of cells undergoing apoptosis. 42 However, apoptosis occurs relatively late when plaque rupture is already ongoing. [99mTc]apcitide (also known as [99mTc]P280) is a radiolabeled peptide that binds to glycoprotein IIb/IIIa receptors expressed on activated platelets in thrombi. 43 As is the case for apoptosis imaging, thrombus imaging is only successful at late stages of vulnerability, that is, when rupture is already acute.
Our recently developed 18F-labeled folate derivative, 3′- aza-2′-[18F]fluorofolic acid, 23 with high binding to both FR- α and FR-β, demonstrated ideal in vitro properties for the imaging of inflammatory processes by targeting activated macrophages in atherosclerosis. It showed high and specific accumulation in CD68- and FR-β-rich plaque regions at negligible unspecific retention in healthy artery walls. Specificity to FR-positive normal (kidney), inflammatory (vulnerable plaques), and malignant (KB xenograft) tissue has been validated. High specific but low unspecific accumulation is a prerequisite for the noninvasive imaging of atherosclerotic plaques by PET, in particular considering the small volume of atherosclerotic lesions in relation to the spatial resolution in PET. The latter is a few millimeters for clinical whole-body cameras. 44 Thus, partial volume effects need to be compensated by high signal to noise ratios. Since the binding of 3′-aza-2′-[18F]fluorofolic acid is limited to sites enriched in activated macrophages, image misinterpretation can be significantly reduced. The lack of anatomic details from PET images can be addressed by coregistration with computed tomography (CT) or magnetic resonance imaging (MRI). Our 18F-labeled folate derivative offers the opportunity to quantify intraplaque FR-β-positive macrophages with greater specificity than [18F]FDG. Although 3′- aza-2′-[18F]fluorofolic acid will admittedly image any site of active inflammation, its ease of synthesis and radiolabeling, excellent tissue penetration, rapid clearance from FR-negative tissue, lack of toxicity, and specificity for an abundant cell type in atherosclerosis (macrophages) render it a possible nuclear probe for further evaluation.
Applying the most recent guidelines for classification of plaque vulnerability,24,27 we did not find significant differences in FR-β or CD68 levels between plaques that were identified as vulnerable and stable, respectively. In addition, accumulation of our 18F-labeled FA did not allow distinguishing between the two plaque types. According to our findings, infiltration of CD68- and FR-β-positive macrophages occurs independently of cap thinning in atherosclerotic lesions or other signs of plaque vulnerability. Comparing FR-β and CD68 gene expression, we found a slightly but not significantly higher ratio of FR-β/CD68 in plaques classified as vulnerable rather than in those classified as stable. In addition, all methods applied in this study revealed a tendency without significance toward higher FR-β expression in vulnerable than in stable plaques.
Accumulation of folate derivatives in atherosclerotic plaques may not correlate with the immediately life-threatening potential of a lesion (the classification criteria in this study) but could be an indication of the potential of a plaque to become vulnerable and eventually rupture. There is currently no way to predict whether and in which time period a stable plaque will become vulnerable. Whether FR-β could serve as a marker in this respect, as suggested by several research groups,20,22,33 requires prospective clinical studies, as in the above example with MMP12. Despite its predictive value, expression of MMP12 did not correlate with intraplaque hemorrhage or the presence of thrombus, two indicators of plaque vulnerability. 32 In this work, we show in vitro that our novel 3′-aza-2′-[18F]fluorofolic acid is perfectly suited to address the above question in a prospective clinical study with PET. The translation of the radiosynthesis of 3′-aza-2′-[18F]fluorofolic acid to an automated synthesis module is completed, and clinical studies are planned. In a further step, we plan to evaluate 3′-aza-2′-[18F]fluorofolic acid, due to its fast and easy radiosynthesis, in a preclinical model of atherosclerosis based on a scavenger receptor BI KO/ApoE hypomorphic mouse and in shear stress-induced plaques.
Conclusion
This study demonstrated high FR-β expression in human endarterectomized plaque samples, as elucidated by qPCR, Western blotting, and immunohistochemistry and visualized by in vitro autoradiography using a novel 18F-labeled folate derivative. The positive correlation between FR-β- and CD68-expressing macrophages represents the cell membrane-associated FR as a suitable imaging marker to estimate intraplaque inflammation. We have shown that 3′-aza-2′-[18F]fluorofolic acid not only detects FR-α-positive tumors but is also a potential useful tool to image inflammatory diseases.
Footnotes
Acknowledgments
We are grateful to Claudia Keller, Petra Wirth, and Josefine Reber for technical assistance. We thank Jelena-Rima Ghadri, Michael Fiechter, Tobias Fuchs, and Julia Stehli for coordinating the constant collection of human tissue samples and Markus Schmid for taking care of the standardized process of tissue collection in the operating room of the University Hospital Zurich, Switzerland.
Financial disclosure of authors and reviewers: None reported.
