Abstract
Viewing individual cells and ambient microvasculature simultaneously is crucial for understanding tumor angiogenesis and microenvironments. We developed a confocal fluorescence microscopy (CFM) and photoacoustic microscopy (PAM) dual-modality imaging system that can assess fluorescent contrast and optical absorption contrast in biologic samples simultaneously. After staining tissues with fluorescent dye at an appropriate concentration, each laser pulse can generate not only sufficient fluorescent signals from cells for CFM but also sufficient photoacoustic signals from microvessels for PAM. To explore the potential of this system for diagnosis of bladder cancer, experiments were conducted on a rat bladder model. The CFM image depicts the morphology of individual cells, showing not only large polygonal umbrella cells but also intracellular components. The PAM image acquired at the same time provides complementary information on the microvascular distribution in the bladder wall, ranging from large vessels to capillaries. This device provides an opportunity to realize both histologic assay and microvascular characterization simultaneously. The combination of the information of individual cells and local microvasculature in the bladder offers the capability of envisioning the viability and activeness of these cells and holds promise for more comprehensive study of bladder cancer in vivo.
CFM, proven to be one of the most important advances ever achieved in optical microscopy, shows outstanding sensitivity and ability to specifically target structural components and dynamic processes in living cells and tissues. PAM has been explored for imaging of live cells too but has been successful for only limited types of cells, such as red blood cells and melanoma cells, both with strong optical absorption. CFM, powered by a huge variety of fluorescent probes, can image not only any type of cells but also specific regions, such as the cytoskeleton, mitochondria, Golgi apparatus, endoplasmic reticulum, and nucleus. CFM can also achieve the evaluation of dynamic processes and microenvironment variables, including inorganic metallic ions, pH, and oxygen, as well as the monitoring of intra- and extracellular activities, such as endocytosis, exocytosis, membrane fluidity, protein trafficking, signal transduction, enzymatic activity, and cellular integrity (live versus dead and apoptosis).
In previous work, PAM has been integrated with other imaging modalities, including ultrasonic microscopy 4 and optical coherence tomography,5,6 to obtain complementary diagnostic information. In comparison with these modalities, CFM could be a better candidate for integration with the newer PAM technique, considering that both CFM and PAM are generated by light absorption, although the signal of PAM is from nonradiative relaxation. When the two modalities are fully integrated, the fluorescent and photothermal events in the tissue are induced by the same laser pulses and, therefore, happen at the same time. In this case, the cellular and molecular information presented by CFM and the microvascular and hemodynamic information presented by PAM, both necessary for understanding the tumor microenvironment, are synchronized exactly, which is crucial for observing the transient phenomena in live tissues, for example, the fast exchange between individual cells and capillaries. Facilitating the imaging of microvessels and cells simultaneously, a PAM-CFM dual-modality device could enable in vivo investigation of the interaction of cancer cells with ambient microenvironments and help better understand the mechanism of disease onset, progression, and responses to therapy.
Recent work on photoacoustic and fluorescence dual-modality imaging realized mapping of blood vessels and draining lymphatic vessels simultaneously by filling the lymphatic vessels with 20% rhodamine B isothiocyanatedextran.7,8 In other research, the melanin and lipofuscin in retinal pigment epithelium were visualized by using photoacoustic and fluorescence imaging, respectively, taking advantage of lipofuscin autofluorescence. 9 So far, presenting cellular morphology and vascular structures simultaneously by photoacoustic and fluorescence imaging still has not been achieved successfully. In this work, imaging of individual cells and local microvasculature simultaneously with a fully integrated PAM and CFM system has been demonstrated for the first time on a rat bladder model. Presenting more comprehensive diagnostic information, such a dual-modality device, when conducted in an endoscopic manner in the future, holds promise for improved detection and guided treatment of nonmuscle invasive bladder cancer.
Material and Methods
The schematic of the integrated system for PAM and CFM dual-modality imaging is shown in Figure 1. These two imaging modalities showing different optical contrasts share the same laser source and scanning optical path. A neodymium:yttrium-aluminum-garnet (Nd:YAG) laser (Spot-10-200-532, Elforlight Ltd, Daventry, Northants, UK) serves as the light source. The Nd:YAG laser working at a 532 nm wavelength has a pulse duration of 2 nanoseconds and a maximal repetition rate up to 50 kHz. The fluctuation of the laser intensity is monitored by a photodiode (DET10A, Thorlabs, Newton, NJ). The laser beam passes through an excitation pinhole (50 μm) and is then reflected by a dichroic mirror (DMLP567, Thorlabs). A two-dimensional (2D) raster scan is realized by a 2D galvanometer. When the laser is pulsing at a 1 kHz repetition rate, a 2D raster scan over 256 × 256 points takes 2 minutes. An achromatic lens with a focal length of 30 mm is used as the objective lens. The focused laser spot is scanned over the tissue sample to excite both photoacoustic signal and fluorescence emission. The induced photoacoustic signal is detected by a highly sensitive custom-built needle hydrophone with a center frequency of 35 MHz and a −6 dB bandwidth of 100%. The back-traveling fluorescence light emitted from the tissue sample passes through the dichroic mirror and a longpass filter (FEL0550, Thorlabs). This design is intended to eliminate the reflected light at the excitation wavelength, allowing only the reflected fluorescent light at wavelengths above 550 nm to be transmitted for detection. Constricted by a detection pinhole (100 μm), which is used for the implementation of confocal microscopy, the fluorescence light is finally collected by an avalanche photodetector (APD110A, Thorlabs). The amplified photoacoustic and fluorescence signals are digitized by an A/D card (Razor CS14×2, GaGe, IL). The lateral resolution of this system on both PAM and CFM modes is determined by the size of the optical focal spot. By imaging a United States Air Force resolution template (T-20-P-TM, Applied Image Inc., Rochester, NY), this lateral resolution is quantified as 5 μm. The axial resolution of this system on the PAM mode is quantified as 33 μm, which is limited by the central frequency and the bandwidth of the hydrophone. The validation of the lateral and axial resolution for PAM has been demonstrated in our previous work on the same system.10–12

Schematic of a PAM and a CFM dual-modality imaging system.
To explore the potential of this imaging system for improved diagnosis of bladder cancer in the future, fresh rat bladders harvested immediately after the rats were euthanized were employed as the samples for this study. 12 All animal procedures were approved by the University of Michigan Committee on the Use and Care of Animals. Before the imaging procedure, the bladder specimens were treated with rhodamine 6G fluorophore. Rhodamine 6G has peak absorption at a wavelength of 530 nm, which is close to the excitation wavelength of 532 nm, and peak emission at a wavelength of 566 nm. A stock solution of rhodamine 6G solution was diluted in ethanol at a concentration of 10 mM and then was further diluted in phosphate-buffered saline to a concentration of 100 μM. The whole bladders were immersed in the solution for 30 minutes and then were rinsed with saline. Because the laser pulses for PAM are short (2 nanoseconds in our case), the fluorescence emission from the laser pulses for PAM is not efficient. Extending the staining period or treating the sample with fluorescent dye at a higher concentration could help enhance the fluorescence emission only within a certain range because quenching may occur when the dye concentration in the sample is too high. Therefore, producing satisfactory CFM images of cells with the same laser for PAM of vasculature is challenging and can be achieved only by treating the tissues with suitable fluorophores at appropriate concentrations. For in vivo transurethral imaging of bladder in the future, a similar preparation procedure can be realized by instilling the phosphate solutions into the bladder noninvasively through urethrocystoscopy. 13
Results
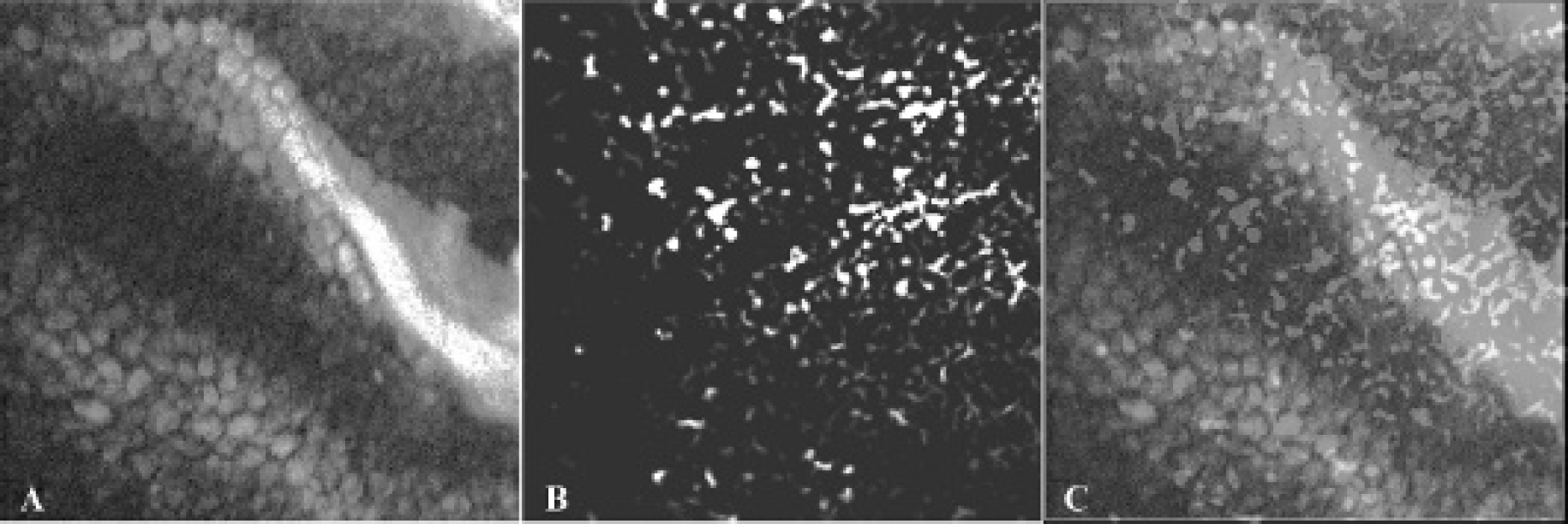
All PAM images were acquired in three dimensions (3D) and are presented here as 2D maximum amplitude projection (MAP) images for better comparison with 2D CFM images. The slice thickness of a 3D PAM image used to generate the corresponding 2D MAP image is 54 μm, which matches with the depth coverage of the 2D CFM image from the same tissue. In other words, the vasculature observed by PAM is in the same plane as the cells visible with CFM. Before imaging with PAM and CFM simultaneously, we examined and optimized the performance of this system on each modality. A rat bladder was first imaged in the PAM mode. Then, after being treated with fluorophore, the same sample was imaged in the CFM mode. Figure 2A represents a CFM image (1.125 mm × 1.125 mm) of the bladder wall, indicating individual cells and adventitia. Figure 2B shows an enlarged CFM image (0.35 mm × 0.35 mm) depicting the shape and size of the individual cells. Besides the large polygonal umbrella cells, some intracellular components, such as nuclei in some cells, can also be discerned. With the CFM image showing the morphologic cellular information in the bladder wall, the PAM image, as shown in Figure 2C, provides complementary information, based on the endogenous optical absorption contrast, of vascular distribution inside the bladder wall. Vascular structures from large vessels down to the distributed capillary network can all be recognized, proving the excellent sensitivity and the high spatial resolution of this imaging system.

The CFM image and the PAM image shown in Figure 3, A and B, were acquired simultaneously from a rat bladder that had been treated with rhodamine 6G, demonstrating the capability of this system to map two different molecular contrasts in tissues through a single scan. Based on the fluorescent contrast and the optical absorption contrast, one can visualize individual cells and blood vessels, respectively. The imaging result from another rat bladder sample is shown in Figure 4. In comparison with the results in Figure 3, the CFM image in Figure 4A shows the cellular structures better, whereas the PAM image in Figure 4B shows richer capillaries, although no large vessels, such as those in Figure 3B, were found. Because the PAM image and the CFM image of each sample were scanned using the same device at exactly the same time, they are coregistered naturally. Figure 3C and Figure 4C give the coregistered CFM and PAM images of the two bladder samples, respectively, showing both cells and vessels in the bladder wall using different colors.

Simultaneous CFM and PAM imaging of a rat bladder.
Simultaneous CFM and PAM imaging of another rat bladder.
Discussion
It is interesting to note that the large vessels shown well in Figure 3B can be barely recognized in the CFM image and are presented as weak and fuzzy shadows, as marked by the red arrows in Figure 3A. This may be due to the strong light absorption by blood in the large vessels, which reduced the local fluorescent signals. Another possible reason is that the fluorescent stain itself may enable the visualization of vessels, although the fluorescent contrast is not comparable to the strong optical absorption contrast presented by PAM. The shadows of the large vessels, however, do not significantly influence the clarity and integrity of the cells in the CFM image. On the other hand, the fluorescent stained cells are not visible in the PAM image. This is due to the fact that rhodamine 6G comes with a high fluorescence quantum yield of 0.95; therefore, only a small portion of the absorbed light can turn into heat, which cannot generate detectable photoacoustic signals. This finding demonstrates the low interference between the two imaging modalities that is desirable in most situations, for example, if one wants to quantify microvascular density and evaluate tissue hemodynamic properties by PAM without being affected by the procedure of CFM.
One may also notice that the image quality in Figure 3 and Figure 4, PAM images that were acquired after the fluorescent stain, is not comparable to that in Figure 2, PAM images that were acquired before the fluorescent stain. The reason is that during the immersion of the ex vivo bladder samples in the dye solution for fluorescent staining, the majority of the blood in the vessels were lost, which directly affected the PAM imaging of vasculature. Due to the loss of blood, worse continuity in presenting vessels, especially small capillaries, can be noticed. However, we do not expect this to be a problem for future clinical or preclinical imaging in vivo because treatment with fluorescent dye will not lead to any loss of blood from live tissues.14–16 Before transurethral imaging of human bladder in vivo becomes possible, the probe for signal scanning needs to be miniaturized and housed in a catheter. The current cystoscopic catheter for clinical diagnosis of bladder cancer has a typical size of 23 French (7.6 mm). A possible limitation of PAM for future endomicroscopic imaging in clinical settings is that the ultrasonic hydrophone for photoacoustic signal detection must be submersed in water or other acoustic coupling materials, although the acoustic coupling will not be a problem when working in a full bladder. A potential solution is to use noncontacting methods based on the interferometric measurement of photoacoustic signals.17–19
The choice of fluorescent dye depends highly on the information to be acquired. For example, the use of SYTO@17 in the study by Koenig and colleagues provided information on the nucleus and cytoplasm because the SYTO@17 binds to deoxyribonucleic acid (DNA) and to ribonucleic acid (RNA). 20 Rhodamine 6G and rhodamine 123 are cell-permeant, cationic, fluorescent dyes that are sequestered by active mitochondria. In the study by D'Hallwein and colleagues, rhodamine 123 was instilled at a concentration of 100 μM for in vivo endoscopic CFM imaging of AY-27 tumor–bearing rat bladders. 13 Compared to rhodamine 6G, rhodamine 123 is less toxic and, therefore, is a better candidate for in vivo applications. In the future, by using a tunable laser, instead of the one with a fixed wavelength at 532 nm, as used in this study, this dual-modality imaging system could work with many other fluorescent dyes and assess different diagnostic information.
Summary
We have successfully validated the feasibility of viewing individual cells and ambient microvasculature simultaneously using an integrated PAM and CFM system. Unlike many previously reported multimodality imaging devices, the two imaging modes in this system are fully integrated so that a single laser pulse can induce two signals presenting two different molecular contrasts in tissues at the same time. The two image types acquired with such a system are not only accurately coregistered but also exactly synchronized, potentially facilitating the study of many transient events in vivo for the first time, for example, the interchange of oxygen from capillaries to cells. Therefore, besides potentially enabling better diagnosis of diseases, such as bladder cancer, this novel technique may benefit basic science research by enabling cancer study in vivo at cellular and molecular levels.
Footnotes
Financial disclosure of authors: This work was supported in part by the Samsung GRO 2012 Program, the National Science Foundation (grant number 1256001), the National Institutes of Health (grant numbers R01AR060350, R01CA91713, and P41-EB2182), and a University of Michigan–Shanghai Jiao Tong University (UM-SJTU) joint grant.
Financial disclosure of reviewers: None reported.
