Abstract
The purpose of this study was to develop a clinically relevant orthotopic xenotransplantation model of pancreatic cancer and to perform a preclinical evaluation of a new positron emission tomography (PET) imaging probe, 64Cu-labeled cyclam-RAFT-c(-RGDfK-)4 peptide (64Cu-RAFT-RGD), using this model. Varying degrees of αvβ3 integrin expression in several human pancreatic cancer cell lines were examined by flow cytometry and Western blotting. The cell line BxPC-3, which is stably transfected with a red fluorescence protein (RFP), was used for surgical orthotopic implantation. Orthotopic xenograft was established in the pancreas of recipient nude mice. An in vivo probe biodistribution and receptor blocking study, preclinical PET imaging coregistered with contrast-enhanced computed tomography (CECT) comparing 64Cu-RAFT-RGD and 18F-fluoro-2-deoxy-D-glucose (18F-FDG) accumulation in tumor, postimaging autoradiography, and histologic and immunohistochemical examinations were done. Biodistribution evaluation with a blocking study confirmed that efficient binding of probe to tumor is highly αvβ3 integrin specific. 64Cu-RAFT-RGD PET combined with CECT provided for precise and easy detection of cancer lesions. Autoradiography, histologic, and immunohistochemical examinations confirmed the accumulation of 64Cu-RAFT-RGD in tumor versus nontumor tissues. In comparative PET studies, 64Cu-RAFT-RGD accumulation provided better tumor contrast to background than 18F-FDG. Our results suggest that 64Cu-RAFT-RGD PET imaging is potentially applicable for the diagnosis of αvβ3 integrin–expressing pancreatic tumors.
PANCREATIC CANCER has the worst prognosis among malignant solid tumors. It is one of the leading causes of cancer-related mortality across the world, with an incidence almost equaling mortality. 1 The 5-year survival rate is still as low as 6% according to the American Cancer Society 2012 statistics. 2 Providing early diagnosis and efficient therapeutic measures remains a major challenge, and suitable animal models that mimic the clinical situation as closely as possible are essential for developing and comparing new diagnostic and therapeutic strategies. 3 There are presently two main types of human xenograft mouse models used for pancreatic cancer research, subcutaneous and orthotopic, depending on the location of the implanted xenograft. Orthotopic tumor xenografts are increasingly being recognized for increased clinical relevance and explored accordingly.
An ideal imaging modality and reagent for diagnosis of pancreatic tumors remains elusive. In recent years, positron emission tomography (PET) using 18F-fluoro-2-deoxy-D-glucose (18F-FDG) has been most commonly used for the evaluation of pancreatic cancer, as in the other clinical settings of various cancers. 4 However, certain pitfalls, including false-positive findings due to pancreatitis or false-negative diagnosis in hyperglycemic patients, limit the accuracy of detection. 5 The availability of a more disease-specific imaging agent targeting tissue biomarkers of pancreatic carcinoma would overcome the shortcomings of metabolic probes.
Integrins are a family of adhesion molecules that mediate cell–cell and cell–extracellular matrix interactions and contribute to angiogenesis. 6 Integrins regulate diverse functions in tumor cells, including adhesion, migration, invasion, proliferation, survival, and metastasis. 7 The αvβ3 integrin, one of the most widely studied and well-characterized integrins, is highly expressed on proliferating endothelial cells during angiogenesis, 8 as well as on cancer cells of various origins. 9 Hosotani and colleagues examined the expression of αvβ3 integrin in human pancreatic cancer samples by immunohistochemical (IHC) analysis and reported that αvβ3 integrin was expressed in 58% of invasive pancreatic ductal carcinomas. 10 Hence, αvβ3 integrin would be a good target, and PET imaging using a radiolabeled αvβ3 integrin tracer has the possibility to be a measure for the diagnosis of pancreatic cancer.
Radiolabeled cyclic peptides containing the Arg-Gly-Asp (RGD) amino acid sequence are commonly used as αvβ3 integrin imaging probes.11,12 Our group has developed a PET imaging probe, 64Cu-labeled cyclam-RAFT-c(-RGDfK-)4, encompassing a cluster of four RGD peptides (designated 64Cu-RAFT-RGD), for visualization and quantification of αvβ3 integrin expression in some animal tumor models. A tetrameric cRGD-containing peptide, known as RAFT-c(-RGDfK-)4, was synthesized by separately grafting four cyclo(-RGDfK-) monomers onto the upper side of the cyclic regioselectively addressable functionalized template (RAFT) decapeptide platform, 13 conjugated with a bifunctional chelator, 1,4,8,11-tetraazacyclotetradecane (cyclam), and then radiolabeled with the positron emitter 64Cu14–16 (Figure 1).

Molecular structure of 64Cu-RAFT-RGD.
The purpose of the present study was to develop a clinically relevant orthotopic xenotransplantation model of pancreatic cancer that reflects the clinical situation and to perform preclinical evaluation of the diagnostic PET imaging probe 64Cu-RAFT-RGD, allowed in this model. Previously, we used the subcutaneous models derived from stable transfectants of human embryonic kidney HEK293 cell lines (HEK293(β1), HEK293(β3)), overexpressing integrin β1 and β3 subunits, respectively, and human glioblastoma cell line U87 MG. Evaluation of 64Cu-RAFT-RGD in an orthotopic tumor model derived from pancreatic cancer cell lines that have endogenous integrin expression has not been examined yet. We investigated integrin (αvβ3 and αv) expression in pancreatic cancer cell lines, establishment of orthotopic pancreatic xenograft tumors, αvβ3 integrin–specific accumulation of 64Cu-RAFT-RGD in tumors, lesion visualization by microPET/contrast-enhanced computed tomography (CECT) with comparison to 18F-FDG PET, and the validation of 64Cu-RAFT-RGD uptake in tumor by autoradiography (ARG), histologic, and IHC examinations.
Materials and Methods
Cell Culture and Reagents
The human pancreatic cancer cell lines BxPC-3, MIAPaCa-2, AsPC-1, and PANC-1 and the human glioblastoma cell line U87MG were purchased from American Type Culture Collection (Manassas, VA). BxPC-3-RFP expressing red fluorescent protein (RFP; DsRed2) was purchased from AntiCancer, Inc. (San Diego, CA). All cell lines except MIAPaCa-2 and PANC-1 were cultivated at 37°C in a humidified atmosphere containing 5% CO2 in RPMI 1640 medium (Sigma, St. Louis, MO) supplemented with 10% fetal bovine serum (Nichirei Biosciences, Tokyo, Japan), 100 U/mL penicillin-G sodium, and 100 mg/mL streptomycin sulfate (Invitrogen, Carlsbad, CA). Dulbecco's Modified Eagle's Medium was used for cultivation of MIAPaCa-2 and PANC-1 cell lines.
Flow Cytometric Analysis of Cellular αvβ3 Expression Level
Cellular αvβ3 integrin expression levels were measured using R-phycoerythrin-conjugated antihuman integrin αvβ3 monoclonal antibody (MAB1976H) (Chemicon, Temecula, CA). Incubation of cells with conjugated antibody was performed for 30 minutes at 4°C on cell suspensions in phosphate-buffered saline (PBS) containing 1% bovine serum albumin, 1 mM CaCl2, and 1 mM MgCl2. Cells were rinsed with PBS for flow cytometric analysis using Guava EasyCyte Plus Systems (Guava Technologies-Millipore, Hayward, CA). Cells treated with PBS without antibody were used as the negative control.
Western Blot Analysis
Western blotting (WB) was performed to analyze αv integrin expression in both cultured cells and tissues samples. Whole-cell lysates were prepared using RIPA buffer (Wako Pure Chemical Industries, Osaka, Japan) with protease inhibitor cocktail (PIC) (Sigma-Aldrich, St. Louis, MO). Tumor tissue and normal pancreatic tissue were homogenized using tissue protein lysis reagent (CelLytic MT) with PIC. The amount of total protein was measured using the Nanodrop spectrophotometer (NanoDrop Technologies, Inc., Wilmington, DE). Protein (50 μg) was separated on a 10%polyacrylamide gel (ATTO Corporation, Tokyo, Japan) and transferred to Immobilon-P membrane (Millipore, Billerica, MA). The membrane was blocked with Block-Ace reagent (Dainippon Pharmaceutical, Osaka, Japan) at room temperature for 1 hour and incubated with primary antibody at room temperature for 1 hour. Rabbit antihuman integrin αv (CD51) polyclonal antibody (Millipore, Temecula, CA) and goat antihuman actin (C-11) antibody (Santa Cruz Biotechnology, Santa Cruz, CA) were used. The membrane was washed with Tris-buffered saline (TBS) and incubated with the appropriate secondary antibody, which was a horseradish peroxidase (HRP)-linked donkey antirabbit or antigoat IgG (GE Healthcare, Little Chalfont, UK; Santa Cruz Biotechnology). Immunoreactive bands were visualized using the Enhanced Chemiluminescence Plus Western blotting detection system (GE Healthcare).
Development of an Orthotopic Pancreatic Cancer Xenograft Model in Mouse
All animal experiments were carried out in accordance with the guidelines for animal experimentation determined by the Animal Care and Use Committee at our institution. At first, BxPC-3-RFP cells (5 × 106) were mixed with BD Matrigel Matrix (BD Biosciences, Bedford, MA) and subcutaneously inoculated into the flank of nude mice (8-week-old female BALB/cA Jcl-nu/nu mice, CLEA, Japan). When subcutaneous tumors had reached a size of 10 mm in longest diameter, the tumors were harvested and minced with scissors into 2 mm3 pieces in cold normal saline. We performed the surgical orthotopic implantation (SOI) under a 7× magnification LG-PS2 microscope (Olympus Corp., Tokyo, Japan). Under anesthesia, the recipient mouse abdomens were opened by left upper abdominal paramedian incision, and the pancreatic tail with the spleen was gently exteriorized. One donor tumor fragment was stitched into the pancreatic tail using 5-0 absorbable surgical suture (Akiyama Medical, Tokyo, Japan). The organs were then returned to the peritoneal cavity, and the abdominal wall and skin were closed in two layers with surgical suture. The SOI recipient mice were monitored and weighed daily to check for any signs of stress, trauma, or surgical complication.
In Vivo Fluorescence Imaging to Confirm Establishment of the Implanted Tumor
To identify the development of BxPC-3-RFP orthotopic tumors, in vivo fluorescence imaging of recipient mice was conducted every 3 to 4 days by using the IVIS Lumina optical imaging system (Xenogen Corp., Alameda, CA). A filter set for DsRed and constant imaging parameters (exposure time: 1 second; binning: medium; lens aperture [f/stop]: 1; field of view [FOV]: 12.5 cm; floor lamp level: high) were used. Gross dissected specimen of orthotopic tumor was reconfirmed by an MZ16F stereo fluorescence microscope (Leica Microsystem, Wetzlar, Germany).
Peptide Synthesis and Labeling
Cyclam-RAFT-c(-RGDfK-)4 peptide and radiolabeling with 64Cu were performed according to our previously described method.14–16 The radiochemical purity and specific radioactivity of 64Cu-RAFTRGD used in the present study were > 98% and ≈ 18.5 MBq/nmol, respectively.
In Vivo Radiolabeled Peptide Biodistribution and Receptor-Blocking Studies
Mice bearing orthotopic pancreatic tumors were divided into two groups, blocking and nonblocking (n = 3 for each group). Mice of the nonblocking group were intravenously injected with 64Cu-RAFT-RGD (0.37 MBq) via the tail vein, and the blocking group received coinjection with an excessive amount of unlabeled peptide (150 nmol/200 μL) according to the previously reported technique. 15 Two hours after injection, the mice were sacrificed, each tissue of interest was excised, and the radioactivities were measured by a gamma-counter. Radioactivity uptake was expressed as a percentage of injected dose per gram of tissue (%ID/g) normalized to a 20 g mouse body weight.
PET Imaging Coregistered with CECT
Mice bearing orthotopic pancreatic tumors were intravenously injected with 7.4 MBq of 64Cu-RAFT-RGD. Two hours after injection, static PET data acquisition was conducted for 30 minutes using a small-animal PET system (Inveon, Siemens Medical Solutions, Malvern, PA) under 1.5% isoflurane anesthesia. Images were reconstructed using a maximum a posteriori reconstruction method with attenuation correction and Inveon Acquisition Workplace software (Siemens Medical Solutions). To obtain the relevant anatomic information, mice fixed in the same bed position were moved to the in vivo micro x-ray computed tomographic (CT) system R_mCT2 (Rigaku Co., Tokyo) scanner immediately after PET scanning. There the mice were intravenously injected with 200 μL of Iopamiron 370 contrast medium (Nihon Schering, Osaka, Japan), and CECT imaging was conducted 20 minutes after injection. The whole-body image was taken with an exposure time of 2 minutes, tube voltage of 90 kV, a current of 200 μA, and an FOV of 60 mm in diameter. Images were obtained with the image reconstruction, visualization, and analysis program provided by the manufacturer. For comparison of 18F-FDG and 64Cu-RAFT-RGD, mice underwent a continuous 2-day imaging protocol with 18F-FDG PET on day 1 and 64Cu-RAFT-RGD PET on day 2. After overnight fasting, mice were intravenously injected with 18F-FDG (3.7 MBq). Fifty minutes after injection, static PET data acquisition was conducted for 10 minutes and CECT was also performed for all mice. Both the PET and CECT data sets were coregistered to confirm the anatomic location of tumors, and fused images were obtained using PMOD PET data analysis software (PMOD Technologies, Zurich, Switzerland). Using ASIPro VM software (CTI Concorde Microsystems, Knoxville, TN), regions of interest (ROI) were manually drawn on three slices in three planes (coronal, transverse, sagittal) of each tumor, and the mean standardized uptake value (SUVmean) was measured in each ROI for quantitative analysis.
Postimaging Ex Vivo Autoradiography, Histologic Examination, and IHC and Immunofluorescence Staining
After 64Cu-RAFT-RGD PET imaging, the mouse was sacrificed, and the tumor with surrounding normal pancreatic tissue was removed, embedded in Tissue-Tek OCT compound (Sakura Finetek, Torrance, CA), and frozen by immersion in n-hexane precooled at −80°C. Frozen sections were serially cut into 10 μm thickness for ARG and hematoxylin-eosin (HE), IHC, and immunofluorescence (IF) staining. Tracer ARG was acquired by exposing the frozen sections to an imaging plate (BAS-MS 2040, Fujifilms Co. Ltd., Tokyo, Japan) in a freezer overnight. The imaging plate was scanned with an FLA-7000 bioimaging analyzer (Fujifilms Co. Ltd.), and the serial sections were stained with HE for histologic examination. For IHC staining, sections were fixed in acetone and incubated with mouse antihuman integrin αvβ3 monoclonal primary antibody (MAB1976) (Millipore, Temecula, CA). EnVivion+ HRP-labeled polymer antimouse antibody (Dako, Glostrup, Denmark) was used as a secondary antibody. Then the sections were stained with diaminobenzidine (Dako) and nuclei were counterstained with hematoxylin. For IF staining of microvessels, rat antimouse CD31 monoclonal antibody (BD Biosciences) and Alexa Fluor 594–conjugated goat antirat secondary antibody (Invitrogen) were used. Images were acquired with an epifluorescence microscope (Olympus X61) equipped with an imaging tiling system (e-Tiling, Mitani Corporation, Fukui, Japan).
Statistical Analysis
Significant differences between groups were determined by Student t-test, and p values < .05 were considered significant.
Results
αvβ3 Integrin Expression in Human Pancreatic Cancer Cell Lines
Four human pancreatic cancer cell lines were examined by flow cytometry for αvβ3 expression (Figure 2, A and B). For reference, the human glioblastoma cell line U87MG, reported for its high endogenous αvβ3 expression, was used as a positive control. BxPC-3 and PANC-1 showed high αvβ3 expression levels comparable to those of the positive control. MIAPaCa-2 and AsPC-1 showed relatively low expression of αvβ3. The αv expression levels in respective cell lysates in WB analysis were concordant with αvβ3 expression measured with flow cytometry (Figure 2C). With WB analysis, the BxPC-3-RFP cell line demonstrated an αv expression level equivalent to that of BxPC-3, and BxPC-3-RFP tumor tissue lysate also demonstrated αv expression.

A, Flow cytometric analysis of αvβ3 integrin expression in four human pancreatic cancer cell lines. Human glioblastoma cell line U87MG was used as the positive reference. The representative results of two experiments are shown. B, Numbers in the upper-right quadrant indicate the percentage of cells that were αvβ3 positive, shown with a bar graph. C, With Western blotting, αv integrin expression in cell lysates of U87MG (lane 1), MIAPaCa-2 (lane 2), AsPC-1 (lane 3), PANC-1 (lane 4), BxPC-3 (lane 5), and BxPC-3-RFP (lane 6) were detected. Tissue lysates of BxPC-3-RFP tumor (lanes 7 and 9) showed αv integrin expression. Mouse normal pancreatic tissues were used as a negative control (lanes 8 and 10).
Establishment of an Orthotopic Pancreatic Cancer Xenograft Model Using SOI
As shown in Figure 3A, we performed the SOI to establish transplanted tumor. At 3 to 6 days after SOI, most of the mice showed enhanced fluorescence activity in the tumor-implanted site, indicating the existence of tumor. After 18 days, all mice had orthotopic xenografts observed by in vivo fluorescence imaging (Figure 3B). The tumor take rate of SOI was 100% in our model.

A, Intraoperative photographs of the surgical orthotopic implantation (SOI) procedure. B, In vivo fluorescence image confirmed the establishment of implanted tumor 6 days after SOI. peri = peritoneum.
Biodistribution and Receptor-Blocking Studies
The biodistribution of 64Cu-RAFT-RGD was examined in mice bearing BxPC-3-RFP orthotopic tumors 2 hours after intravenous injection (Figure 4), and the blood radioactivity was as low as 0.14 ± 0.04 %ID/g, indicating rapid blood clearance. Radioactivity in orthotopic pancreatic tumor was 6.01 ± 0.75 %ID/g, and surrounding normal pancreatic tissue uptake was 0.88 ± 0.17 %ID/g. Radioactivity accumulation in the kidney was as high as 15.79 ± 2.43 %ID/g, indicating prominent renal excretion of the probe. Moderate liver uptake of 5.38 ± 0.04 %ID/g was seen. Radioactivity accumulation in other tissues was low or negligible. Heart, lung, spleen, stomach, small intestine, muscle, and bone showed uptake of 0.95 ± 0.13, 2.70 ± 1.13, 3.41 ± 0.38, 2.40 ± 0.64, 2.50 ± 0.19, 0.65 ± 0.08, and 1.28 ± 0.20 %ID/g, respectively (Table S1, available online only). The tumor to blood, tumor to muscle, tumor to pancreas, and tumor to liver ratios were 46.64 ± 9.93, 9.3 ± 0.25, 7.11 ± 2.17, and 1.12 ± 0.15, respectively (see Figure 4B). The binding specificity of 64Cu-RAFTRGD was validated by coinjection with an excessive amount of unlabeled peptide. As demonstrated in Figure 4, a significant decrease, 95.5%, in radioactivity was achieved in tumors after blocking (see Figure 4A). All other examined tissues (except kidney, lung, and spleen) showed significant reduction as well.

A, Biodistribution of 64Cu-RAFT-RGD in nude mice bearing a BxPC-3-RFP orthotopic tumor 2 hours after receiving 64Cu-RAFT-RGD injection alone (gray) or coinjection with an excessive amount of unlabeled probe (black). B, 0.1 X Tumor to blood, tumor to muscle, tumor to pancreas, and tumor to liver ratios were compared. Data are represented as mean ± SD (n = 3 for each group) (*p < .05).
MicroPET/CECT
Static PET scans followed by CECT were undertaken. CECT images of mice delineated the tumor and depicted the surrounding nontumor tissues, such as liver, stomach, intestine, and kidneys (Figure 5, B and E). Images showing tumor and whole-body uptake of radioactivity 2 hours after 64Cu-RAFT-RGD injection (Figure 5A) and 50 minutes after 18F-FDG injection (Figure 5D) were compared. Coregistered and superimposed PET images on CECT images facilitated precise and easy detection of pancreatic cancer lesions (Figure 5, C and F). In addition, tracer uptake by nontumor tissues could be distinctly identified on fusion images. Tumor was clearly visualized by high 64Cu-RAFT-RGD accumulation, giving good contrast. 18F-FDG uptake in tumor seemed to be low, with poor contrast. By quantifying the tumor uptake, the SUVmean for 64Cu-RAFT-RGD tended to be higher than that for 18F-FDG (0.48 ± 0.19 vs 0.34 ± 0.06), although not statistically significant (Figure 5G), with a p value of .081.
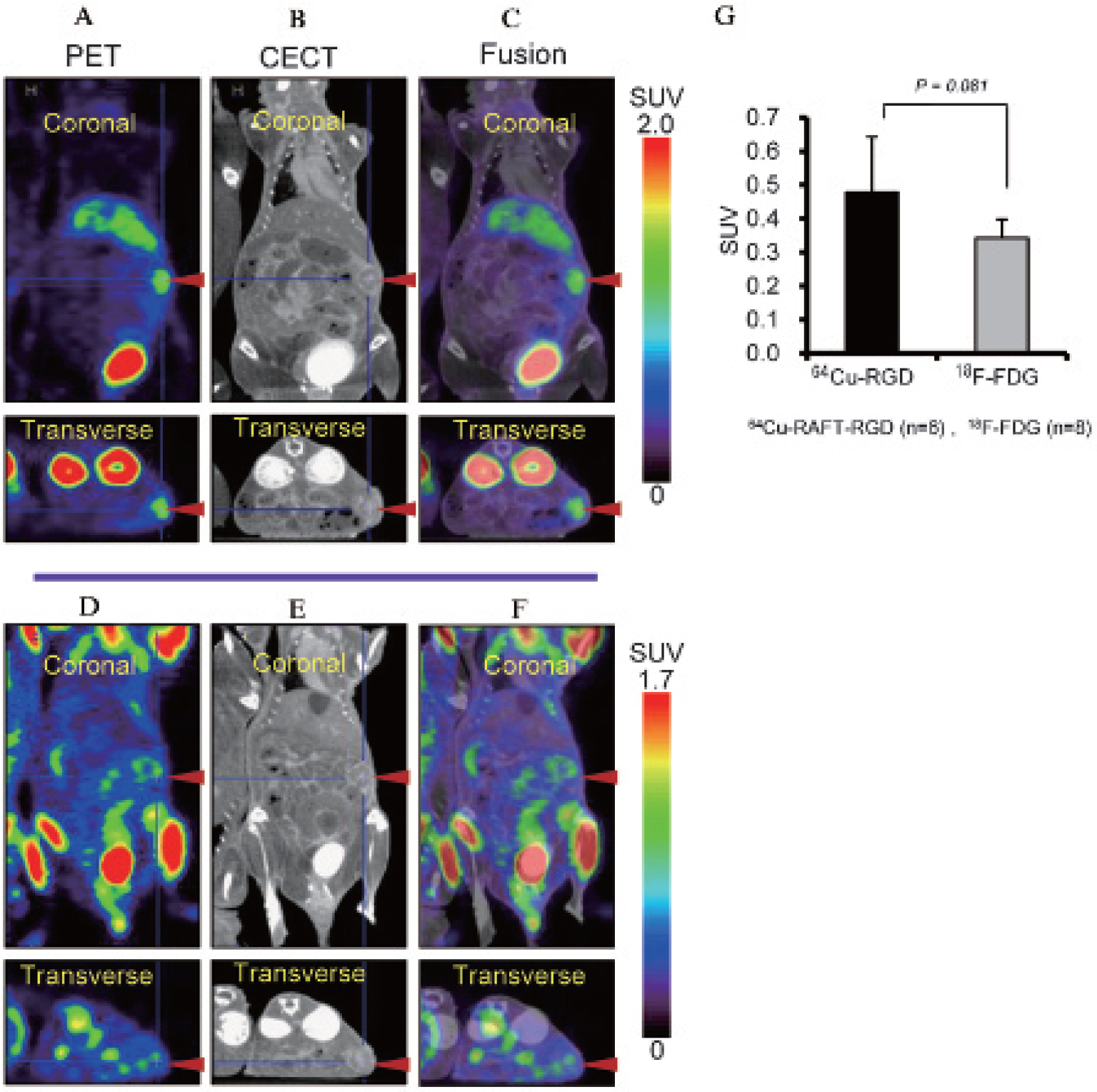
MicroPET/CECT images (coronal and transverse) of a representative mouse bearing an orthotopic pancreatic tumor. Images of the upper panel (A, B, and C) were obtained 2 hours after injection of 64Cu-RAFT-RGD. Images of the lower panel (D, E, and F) were obtained 50 minutes after injection of 18F-FDG in the same mouse 1 day before 64Cu-RAFT-RGD PET. The fusion images (C and F) obtained by superimposing of PET images (A and D) on corresponding CECT images (B and E) indicated marked tumor to background contrast and slightly higher uptake of 64Cu-RAFT-RGD compared to 18F-FDG in tumor lesions (arrowhead). G, Tumor 64Cu-RAFT-RGD and 18F-FDG uptake (SUVmean) were compared. Data are represented as mean ± SD (n = 6 for 64Cu-RAFT-RGD and n = 8 for 18F-FDG) (p = .081).
Validation by Ex Vivo ARG, Histologic, IHC, and IF Examinations
Immediately after PET imaging, mice were euthanized and the abdominal wall was opened to inspect the pancreatic cancer lesion macroscopically. The position of gross tumor (Figure 6D) was identical to that of the region of high tracer uptake seen in 64Cu-RAFT-RGD PET (Figure 6C) and enhanced fluorescence activity seen on in vivo fluorescence imaging (Figure 6B). Gross specimens of tumor with surrounding normal pancreatic tissue (Figure 6E) were removed and processed for ARG and HE, IHC, and IF staining. On ARG images, high 64Cu-RAFT-RGD accumulation was observed in tumor but not in normal pancreas (Figure 6F). HE staining of adjacent sections showed clear a histopathologic difference between tumor and normal pancreatic acinar cells (Figure 6G). The tumor area exhibited neoplastic cells with high-grade nuclear features (pleomorphic nuclei, prominent nuclei) and some regions of desmoplasia, which are characteristic of human pancreatic cancer. With the IHC staining, αvβ3 integrin expression in tumor cells and their inhomogeneous distribution in tumor area, compared to normal pancreatic acinar cells, were observed (Figure 6I). CD31 IF staining revealed that that there was no abundant tumor microvascular network over the normal area, indicating that our pancreatic tumor had less vascularity compared to the common feature of other vascular-rich tumors (Figure 6H).

A, White light image and fluorescence image of gross dissected specimen. Optical (B), PET (C), and dissected (D) whole-body specimen images from a representative mouse bearing an orthotopic pancreatic tumor (arrowhead). E, Gross specimen of tumor and normal pancreatic tissue excised from this mouse. F, Tumor area showed high radioactivity on ex vivo autoradiographic (ARG) image. G, Adjacent section was stained with hematoxylin and eosin (HE), and histopathology was observed with low- (X4) and high-power magnification (X20). HE staining demonstrated neoplastic cells with high-grade nuclear features and some regions of desmoplasia in the tumor area and normal acinar cells in pancreatic parenchyma. H, CD31 immunostaining of the serial section indicated unabundant vascularity in the tumor area. I, Enhanced immunohistochemical (IHC) staining of αvβ3 integrin in tumor cells but not in normal acinar cells was visualized with low- (X4) and high-power magnification (X20). I = intestine; IF = immunofluorescence; N = normal pancreatic tissue; S = spleen; T = tumor.
Discussion
Compared to some limitations of the subcutaneous model, orthotopic pancreatic tumor more closely mimics the natural biologic behavior and characteristics of human pancreatic cancer because it grows in its native micro-enviroment and reliably replicates certain important characteristics of human malignancy, such as local invasion and metastasis. 17 Therefore, the orthotopic tumor model is more suitable for examination of the tumor–host interactions directing disease-relevant metastases and loco-regional growth, the tumor site–dependent therapeutic response, and specific gene expression in target organs and tumor. On the other hand, some drawbacks exist in the generation of an orthotopic tumor model, which is time and labor consuming and demands skilled work, high cost, longer healing and recovery time after surgery, and limited throughput because of imaging methods that are necessary to monitor the tumor. Nevertheless, orthotopic tumor models are emerging as the preference for pancreatic cancer research due to the greater clinical relevance. 18 There are a variety of techniques to generate an orthotopic pancreatic cancer model in immunodeficient mice, such as direct injection of tumor cells into the pancreas with or without Matrigel,19,20 and surgical transplantation of a small tumor fragment derived from subcutaneous tumors in donor animals 21 or directly from patient specimens 22 has also been reported by several groups. In this study, we established orthotopic pancreatic cancer models in recipient nude mice by the SOI method and investigated the possibility of accurate detection of this cancer using this model and the PET imaging agent 64Cu-RAFT-RGD. We used the human pancreatic cancer cell line BxPC-3-RFP with stable and high expression of RFP, which could easily be monitored with an optical imaging system and facilitated real-time visualization of tumor development inside the abdomen. Tumor growth at the implanted site was visible a few days following operation, and tumor became visible in all recipient mice within 18 days, exhibiting a 100% tumor take rate in our experiment (see Figure 3). The main beneficial characters of SOI were rapid, and predictable tumor growth and feasibility to avoid undesired peritoneal spread and carcinomatosis occurred by leakage of cell suspension in a direct injection method, which was used to form a traditional orthotopic model. Moreover, with the difference from the subcutaneous model, orthotopic tumor invasion to surrounding organs such as the small intestine, stomach, liver, spleen, and peritoneum was noticed in some cases. Metastases to distant organs were not evaluated in this work.
On the clinical front, the best option for pancreatic cancer treatment is curative surgery, and timely detection of lesions is critically important. In current practice, 18F-FDG-PET imaging has been widely applied as a noninvasive, functional imaging technique using 18F-FDG to characterize the enhanced glucose use of cancer cells for detection, staging, monitoring treatment response, and finding recurrence of various malignant diseases, including pancreatic cancer. 23 Still, no conclusive consensus has been reached with regard to whether the role of 18F-FDG-PET is crucial in pancreatic cancer management. 24 According to the literature, despite the finding that 18F-FDG-PET is useful in the management of pancreatic cancer, it is not regarded as an ideal method because 18F-FDG uptake can overlap in benign and malignant diseases and not provide high enough diagnostic accuracy for malignant pancreatic tumors in some cases, 25 and it has a limited role in the evaluation of metastasis. 26 False-positive findings due to nonspecific 18F-FDG uptake by several benign conditions, such as inflammatory disease, granulomatous disease, and gastrointestinal, muscle, and brown fat uptake, and false negative results due to high blood glucose levels in patients or low glucose transporter protein and glucose phosphorylation enzyme expression in indolent tumors are major flaws of 18F-FDG. In light of this fact, a more disease-specific imaging probe that targets specific molecular or genetic abnormalities of disease, rather than depending only on metabolic changes, would be a desirable probe for tumor detection.
To visualize the tumor with a targeted imaging approach, an adequate level of accessible tumor-specific target is important to detect and delineate lesions from background. αvβ3 Integrin, the most actively studied of the integrins, serves as an excellent molecular target for tumor imaging because it is not readily detectable in quiescent vessels but becomes highly expressed in proliferating tumor vasculature 6 and in tumor cells themselves. High αvβ3 integrin expression in many types of carcinoma, such as glioma 27 ; melanoma 28 ; breast, 29 ovarian, 30 lung, 31 and colon 32 cancer; and squamous cell carcinoma of head and neck 33 has been reported, whereas its expression in corresponding normal tissue is low or absent. The ability to noninvasively visualize and quantify αvβ3 integrin expression levels can provide an outlook to verify the presence of tumor cells themselves along with sprouting surrounding tumor vasculature. As with certain other malignant tumors, αvβ3 integrin is often highly expressed in pancreatic cancer cells, 10 whereas it is hardly expressed in normal pancreatic cells. Concordantly, our flow cytometric analysis exhibited high expression of αvβ3 integrin in BxPC-3 and PANC-1 pancreatic cancer cell lines and relatively low levels in MIAPaCa-2 and AsPC-1 lines (see Figure 2, A and B). The observation of αvβ3 integrin expression in other varieties of pancreatic cancer cell lines in future studies should improve the consensus. The αv integrin subunit expression in corresponding cell lysates seen in our WB analysis was also consistent with αvβ3 integrin expression measured with flow cytometry (see Figure 2C). The BxPC-3-RFP that we used as a cell line for the orthotopic model and PET experiments had almost identical αv integrin expression level as BxPC-3. In addition, BxPC-3-RFP tumor tissue lysate revealed av integrin expression on protein blot. Our IHC examination also described the increased αvβ3 integrin expression in cells of tumor frozen section, and it was concordant with the WB result. Thus, visualization of pancreatic cancer using αvβ3-targeted radiolabeled RGD peptides can be expected. We previously compared the cell binding of 64Cu-RAFT-RGD peptide using monolayer cultured of HEK293(β1) and HEK293(β3) cell lines. The binding activity that was expressed as the cell-bound radioactivity in percentage of totally added radioactivity normalized to 100 μg of protein were 0.34 ± 0.04% and 15.67 ± 0.74%, which indicated good binding of 64Cu-RAFT-RGD for αvβ3. Moreover, we also studied the feasibility of selectively imaging αvβ3 integrin–expressing tumors in vivo, 16 as well as tumor angiogenesis integrated by αvβ3-expressing activated endothelial cells during angiogenesis 34 using the 64Cu-RAFT-RGD PET probe. Furthermore, we reported high 64Cu labeling efficiency for cyclam conjugate 15 and achieved a 64Cu-RAFT-RGD probe with favorable pharmacokinetics in terms of biostability, quick blood clearance with prevailing renal excretion, high specificity, and strong affinity to αvβ3 integrin expression.16,34 As an extension to these achievements, in the present study, we sought to evaluate the potential use of 64Cu-RAFT-RGD for pancreatic cancer detection. The αvβ3 integrin–targeted specificity of 64Cu-RAFT-RGD was assessed by means of an in vivo radiolabeled probe biodistribution with receptor blocking study, and, in good agreement with the previous report, in vivo αvβ3-binding specificity of 64Cu-RAFT-RGD was validated by coinjection with an excessive amount of unlabeled probe. 64Cu-RAFT-RGD accumulation was significantly diminished by blocking probe, which produced a 95.5% reduction in 64Cu-RAFT-RGD accumulation in tumor and certain reductions in other tissues and demonstrated the αvβ3-specific binding character of RGD targeted to inherent high and basal αvβ3 integrin expression of tumor and nontumor tissues (see Figure 4, A and B).
As imaging probes become more specific regarding successful measurable uptake in only the tumor or other tissue, it is important to depict the exact anatomic location of the uptake. Many studies have shown that by combining anatomic and functional imaging techniques, PET/CT images surpass either PET, CT, or PET and CT images viewed side by side. 35 Generally, PET depends on biochemical or molecular characteristics of diseases and may yield unsatisfactory resolution of the image, whereas CT affords morphology and structure of diseased organs with very high spatial resolution. Thus, by precise coregistration of PET and CT, synergistic and complementary effects may be expected to increase the specificity, sensitivity, and interpretation of PET and indentification of organ underlying probe accumulation. In the present study, we performed PET and CECT and coregistered for a single set of fused images. Through PET/CECT fusion images of three planes, coronal, transverse, and sagittal, we could identify the location of tumor and surrounding organs, which were relatively clearly visualized on CECT. Even tumors that were closely located to kidney and liver and somewhat hard to delineate due to high renal or moderate liver uptake of 64Cu-RAFT-RGD in PET images alone were successfully recognized on fusion images. Together with CECT, 64Cu-RAFT-RGD PET apparently demonstrated 100% of all implanted orthotopic pancreatic tumors in the present study.
We also evaluated 64Cu-RAFT-RGD and 18F-FDG uptake 2 hours and 50 minutes after injection, respectively. These time points were known and suitable to obtain satisfactory contrast between tumor and nontumor tissues according to our preliminary experimental experience. When the PET images of the two radiotracers were compared, the 64Cu-RAFT-RGD PET gave a much better tumor to background contrast (see Figure 5, A–F) and slightly higher tumor uptake compared to 18F-FDG (see Figure 5G).
Additionally, our postimaging ARG, histopathologic, and IHC examinations supported the potential of 64Cu-RAFT-RGD PET for imaging of pancreatic cancer because a probe accumulation difference between tumor and surrounding normal pancreatic tissue was clearly found in ARG (see Figure 6F) and was confirmed by HE (see Figure 6G) and IHC (see Figure 6I) staining of adjacent sections. Taken together, the overall results of our model study have demonstrated that αvβ3 integrin–targeted imaging using 64Cu-RAFT-RGD can function as a highly specific imaging tool for pancreatic cancer, visibly distinguishing it from adjacent normal tissues. Generally, human pancreatic cancer is relatively hypovascular in nature compared to the other tumors known for rich vascular supply. 36 We also found a relatively poor microvessel network in tumor by IF detection method for panendothelial cell marker CD31 using antimouse monoclonal antibody (see Figure 6H). Hence, 64Cu-RAFT-RGD accumulation in our tumor model might be predominantly associated with αvβ3 integrin expression of tumor cells themselves rather than its expression on activated endothelial cells.
Recently, Yoshimoto and colleagues reported that single-photon emission computed tomography (SPECT) with 111In-DOTA-c(RGDfK) has great potential for the early diagnosis of pancreatic cancer in the hamster carcinogenesis model by showing specific uptake in tumor but not in inflammatory lesions and normal pancreas. 37 They used a monomeric RGD peptide that has been reported by other groups as causing high radioactivity accumulation in the liver and intestine due to its unfavorable hepatobiliary pathway. 38 Now, multimerization of RGD peptides is the trend in the development of RGD peptides to enhance binding affinity and specificity for αvβ3 integrin, leading to high tumor accumulation and retention in the tumor. 39 Our tetrameric RGD-based peptide fulfilled these properties, and its high renal accumulation could be reduced by pre- or coinjection with the plasma expander gelofusine. 40 With PET advantages in terms of detection sensitivity and image resolution over SPECT, we anticipate that 64Cu-RAFT-RGD will be a more preferable probe.
Although further translational studies in humans will be needed, the high-quality performance of present-day clinical PET in terms of spatial resolution and hybrid PET/CT technologies should provide clinical use of 64Cu-RAFT-RGD in the management of pancreatic cancer in the near future. According to the radiochemical properties of the labeled isotope 64Cu (half-life 12.7 hours; β+ 19%; Eβ+max 0.655 MeV; β− 40%; Eβ–max 0.573 MeV) and its feasibility of production via a small biomedical cyclotron, 41 along with the possibility of reduced risk of nephrotoxicity by coinjection of gelofusine, 64Cu-RAFT-RGD should be an attractive and favorable probe for diagnostic PET imaging and internal radiotherapy of pancreatic cancer, which is another interesting subject of the research that should be performed in the future.
Conclusion
The present study on an orthotopic xenotransplantation model in mouse and the comprehensive results suggest that the targeted molecular probe 64Cu-RAFT-RGD possesses specific binding to αvβ3 integrin of pancreatic tumor cells, may provide higher detectability over 18F-FDG-PET, and might be a potentially applicable candidate for the diagnosis of pancreatic cancer expressing αvβ3 integrin.
Footnotes
Acknowledgments
We thank Hisashi Suzuki and other members of the Molecular Probe Program for 64Cu production; the Cyclotron Operation Section for cyclotron operation; all members of the Diagnostic Imaging Program, Molecular Imaging Center, National Institute of Radiological Sciences, and Dr. Jean-Luc Coll of INSERM U823, Institut Albert Bonniot, Université Joseph Fourier, Grenoble, France, for helpful discussion.
Financial disclosure of authors: This study was partly supported by a Grant-in-Aid for Scientific Research (C) (24591804) from the Ministry of Education, Culture, Sports, Science and Technology, Japan.
Financial disclosure of reviewers: None reported.
