Abstract
The purpose of this study was to evaluate the time course of contrast enhancement of spleen, liver, and blood using eXIA 160 XL in healthy mice. eXIA 160 XL was intravenously injected in C57bl/6 mice (n = 12) at a dose of 0.1 mL/20 g (16 mg iodine [I]/20 g) (n = 6) or 0.2 mL/20 g (32 mg I/20 g) (n = 6). The distribution was analyzed by repeated micro–computed tomographic scans up to 48 hours after contrast administration. Images were analyzed using Amide software. Regions of interest were drawn in the spleen, liver, and left ventricle. Contrast enhancement was measured and expressed as a function of time. Peak contrast enhancement of the spleen was reached at 30 minutes, and peak contrast enhancement of the liver occurred 45 minutes after 16 mg I/20 g. Given that this contrast was found to be rather low in the spleen in comparison with former eXIA 160 products, experiments were done at a higher dose. However, the 32 mg I/20 g dose was lethal for mice. Enhancement inside the heart lasts for 1 hour. Administration of eXIA 160 XL results in long-lasting blood pool contrast with higher contrast enhancement in heart and liver in comparison with eXIA 160; however, the administered dose should be limited to 16 mg I/20 g.
HIGH-RESOLUTION x-ray micro–computed tomography (micro-CT) is a noninvasive longitudinal imaging modality that decreases experimental variability and generates detailed three-dimensional images. 1 It is a rapidly evolving preclinical imaging method that allows structural and functional visualization of the inner anatomy of small animals.2,3 Compared to clinical CT, the spatial resolution of micro-CT is significantly better, from 1 to 100 μm. 4 When planning small-animal imaging experiments, optimization of the CT protocol is strongly advised to minimize the biologic consequences of the additional dose associated with the CT while maintaining sufficient resolution. 5
However, because micro-CT uses x-rays for imaging different materials, it suffers from very poor contrast between soft tissues, which makes it impossible to distinguish different organs. 6 To improve soft tissue imaging, exogenous contrast agents can be injected in laboratory animals.7,8
X-ray contrast agents developed for clinical purposes are hydrophilic iodinated molecules with a low molecular weight that are very quickly eliminated by glomerular filtration. 9 However, the administration of clinically approved iodinated extracellular water-soluble contrast agents is not useful because of the high heart rate of the test animals and the fast excretion of these products.
Over the past decade, micro-CT-compatible contrast agents have been developed for x-ray imaging that have a long residence time in the bloodstream.9–12 Adequate micro-CT contrast agents are characterized by their formulation as nanoparticulate systems such as liposomes to avoid renal filtration, 13 their functionalization of the surface by grafting on hydrophilic polymers such as polyethylene glycol (PEG), 14 and their content of x-ray contrasting materials such as iodine. 13 Furthermore, the agents should be nontoxic to the animals. Commercially available micro-CT contrast agents such as Fenestra and eXIA 160 have been described in the literature. 15
Recently, a new blood pool contrast agent, eXIA 160 XL (eXIA 160 eXtra Lasting, Binitio Biomedical, Inc., Ottawa, ON), became commercially available. It has been claimed that the agent remains longer in the circulation in comparison with eXIA 160 due to its size and surface modifications. However, to our knowledge, there are no published experimental imaging data regarding eXIA 160 XL. The purpose of our study was to evaluate the time course of contrast enhancement of spleen, liver, and blood in mice following administration of eXIA 160 XL and to compare this agent to other contrast agents, described in Willekens and colleagues. 15 Also, the optimal experimental concentration for adequate enhancement was determined.
Materials and Methods
Animal Preparation
Our study was approved by the ethical committee for small animal studies of our university.
Twelve healthy 6-week-oldmale C57bl/6 mice were used in this study. The mice ranged in weight from 21 to 34 g (average 25 g). A single tail vein injection was performed to administer an iodinated blood pool contrast agent, eXIA 160 XL (160 mg iodine [I]/mL). The injected dose was 0.1 mL/20 g body weight (n = 6), as indicated in the manufacturer's manual, and 0.2 mL/20 g body weight, an injection volume corresponding to 16.6% of the blood volume (n = 6) (16mg I/20 g and 32mg I/20 g). The decision to inject a double dose was made after analysis of the scans of 16 mg I/20 g, which showed lower contrast enhancement in the spleen in comparison with eXIA 160. The bolus injection took a few seconds, between 3 and 6. Anesthesia was induced with 5% isoflurane (Abbott, North Chicago, IL) and maintained at 2% for the duration of the imaging, which was done with the mice breathing spontaneously through a mask.
The scans were performed in similar environmental conditions. In our experimental setup, all injections were performed by the same person.
Micro-CT Imaging
Imaging was performed using an in vivo micro-CT system (SkyScan 1178 micro-CT system, SkyScan, Kontich, Belgium) with a spatial resolution of 83 μm. The voxel size was 83 μm 3 . The system is made up of two metalloceramic tubes with 0.5 mm Al filtration and two 1,280 × 1,024 pixel digital x-ray cameras. Images were acquired at 50 kV, 615 μA, with a 121-second acquisition time and a rotation step of 1.08°. The mice were placed on their left side on an animal bed and positioned in the isocenter of the scanner. Each animal underwent micro-CT scanning before contrast injection (baseline) and immediately following contrast administration, postinjection. Subsequent images were obtained 15, 30, and 45 minutes and 1, 2, 3, 4, 24, and 48 hours after contrast administration. Between the scans, the mice were put in their cage again. An experienced micro-CT user placed the mice manually in the scanner, taking into account the anatomic references. All CT data were acquired without the use of respiratory or cardiac gating. The radiation dose was 0.507 Gy per scan.
Image Analysis
Images were reconstructed using filtered backprojection (NRecon, SkyScan) and analyzed using Amide software (http://amide.sourceforge.net/). 16 Square regions of interest (ROI) measuring 1 mm2 were drawn manually in the liver, spleen, muscle, bladder, and left ventricle. The left ventricle density was used as a blood reference value. The ROI of the muscle was drawn in the upper leg. The average pixel value and its standard deviation were measured within every ROI for each mouse. The ROI was placed manually on the same anatomic reference to compensate small displacements. Contrast enhancement within tissues calculated for the different tissues and the different time points was defined as follows:
where PVorgan ti is the average pixel value of the organ at time ti (ranging from 0 to 48 hours) and PVorgan t0 is the average pixel value of the organ at baseline (before contrast administration). 13
For all data points, the median and corresponding first and third percentiles were calculated. Organ enhancement values obtained with eXIA 160 XL were compared to previously published similar data in 6-week-old male C57bl/6 mice for eXIA 160. 15 Mice survival was monitored up to 48 hours after injection of eXIA 160 XL. The half-life was calculated in GraphPad (GraphPad Inc., San Diego, CA).
Results
All test animals (6 of 6) injected with a dose of 16 mg I/20 g survived. Five mice injected with a dose of 32 mg I/20 g died between 24 and 48 hours after contrast administration (16.7% survival rate).
Contrast enhancement of blood, liver, and spleen was observed in all animals. Contrast enhancement of these organs is shown in Table 1 for the low-dose (16 mg I/20 g) and in Table 2 for the high-dose (32 mg I/20 g) injection. No uptake of contrast (< 1% contrast) was observed in the muscle or in the bladder within the time interval of the experiment (48 hours). With the low dose, peak contrast enhancement of the spleen was 22% at 30 minutes after injection (Figure 1A). With the high dose, the peak contrast enhancement was 60% at 45 minutes after injection (Figure 1B and Figure 2). Peak liver contrast enhancement was 14% at a dose of 16 mg I/20 g at 45 minutes after injection (see Figure 1A). For the high dose, peak contrast enhancement was 28% at 4 hours after injection (see Figure 1B and Figure 3).
Contrast Enhancement in Blood, Liver and Spleen after Low-Dose Contrast Administration (0.1 mL/20 g)

PI = postinjection.
Data points represent the median value of six measurements, with the 1st and 3rd percentiles.
Contrast Enhancement in Blood, Liver, and Spleen after High-Dose Contrast Administration (0.2 mL/20 g)
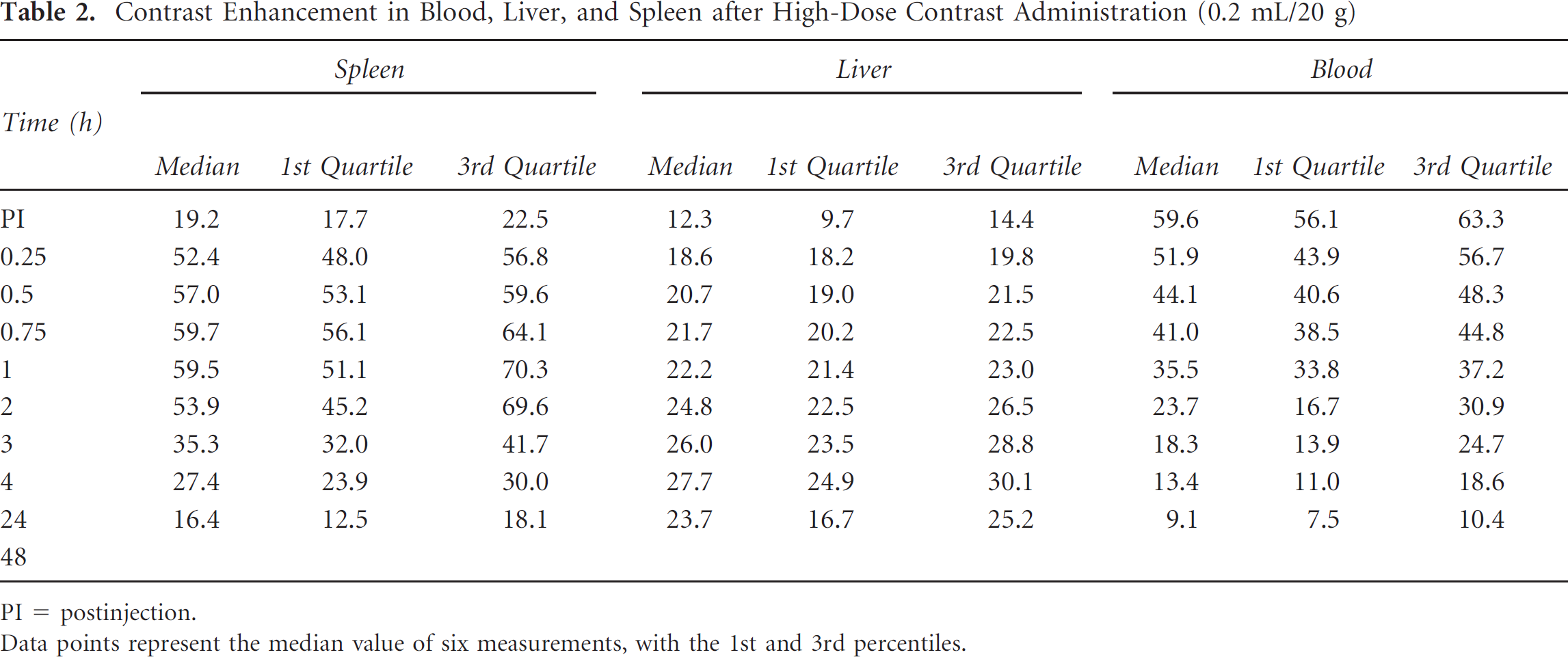
PI = postinjection.
Data points represent the median value of six measurements, with the 1st and 3rd percentiles.

A, Contrast enhancement in blood, liver, and spleen after 16 mg I/20 g of eXIA 160 XL. B, Contrast enhancement in blood, liver, and spleen after 32 mg I/20 g of eXIA 160 XL.
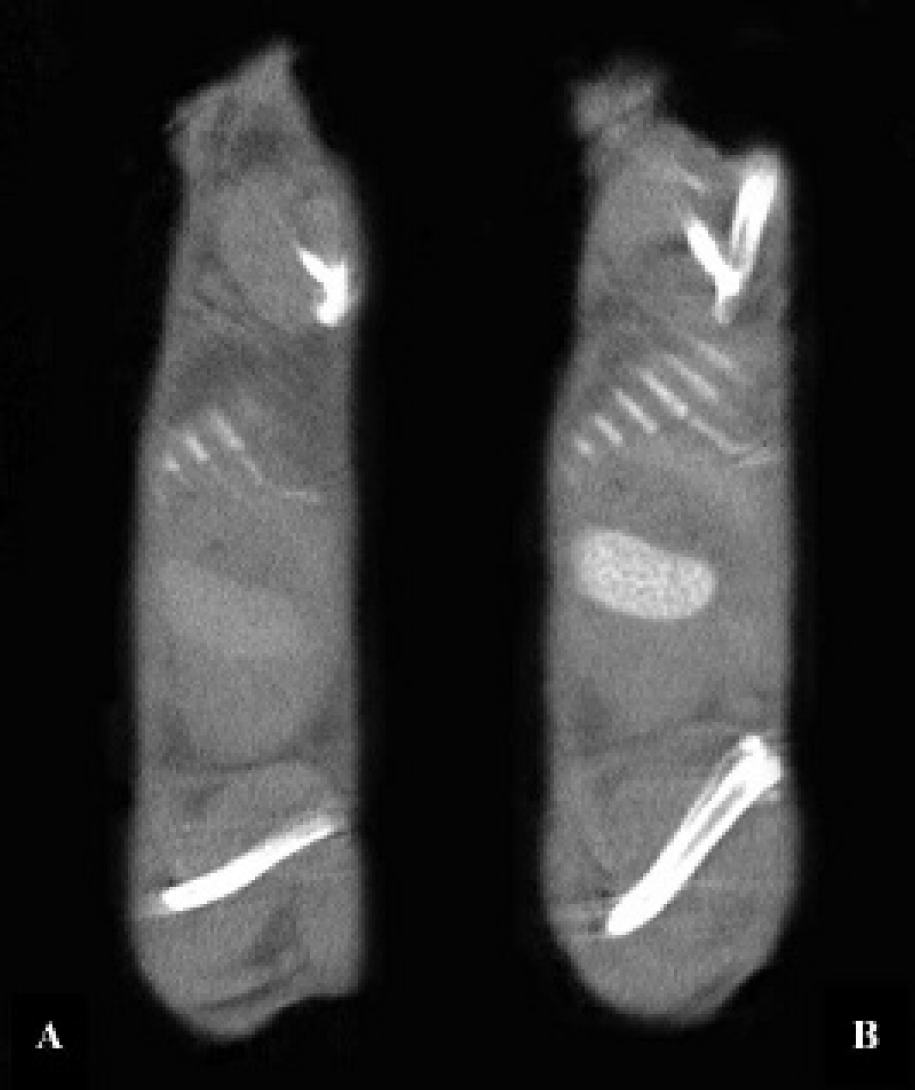
Contrast-enhanced images with eXIA 160 XL (from sagittal slices). Peak enhancement in the spleen (A) 30 minutes after administration of eXIA 160 XL at a dose of 16 mg I/20 g and (B) 45 minutes after administration of eXIA 160 XL at a dose of 32 mg I/20 g.

Contrast-enhanced images with eXIA 160 XL (from coronal slices). Peak enhancement in the liver (A) 45 minutes after administration of eXIA 160 XL at a dose of 16 mg I/20 g and (B) 4 hours after administration of eXIA 160 XL at a dose of 32 mg I/20 g.
Peak vascular enhancement was observed immediately following intravenous administration of contrast (30% at a dose of 16 mg I/20 g and 60% at a dose of 32 mg I/20 g) (see Figure 1A and Figure 4).

Contrast-enhanced images with eXIA 160 XL (from sagittal slices). Peak enhancement in the vasculature (A) immediately following administration of eXIA 160 XL at a dose of 16 mg I/20 g and (B) immediately following administration of eXIA 160 XL at a dose of 32 mg I/20 g.
Discussion
eXIA 160 XL was well tolerated by the mice receiving a dose of 16 mg I/20 g. The survival rate of mice after administration of eXIA 160 XL at a dose of 16 mg I/20 g was 100%, which is similar to the survival rate of eXIA 160 at an equal load. 14 However, we observed that at the dose of 32 mg I/20 g of eXIA 160 XL, survival was only 16.6%. Most of the mice receiving the double dose died between 24 and 48 hours after contrast injection. We assume that the high dose became toxic. Unfortunately, we did not perform toxicity tests on our mice, so we have no physiologic data to explain the death. Mortality due to contrast has already been described in the literature. In their experiments, Vandeghinste and colleagues favored the use of eXIA 160 in comparison with Fenestra VC because of the high mortality related to the latter. 17 The decreased death rate of eXIA 160 may be explained by the possibility of administering only half of the volume of contrast agent for the same image quality.
Detombe and colleagues suggested that there might be an upper tolerance limit on the total amount of eXIA 160 that a mouse can adequately process and eliminate. 18 These conclusions are probably also applicable to the newer contrast agent, eXIA 160 XL.
Clearance and metabolic degradation of contrast agents are of great importance. The physiologic changes related to nonbiodegradable nanoparticulate contrast can prove to be lethal for the test animals. These nonbiodegradable agents are predominantly removed by the Kupffer cells of the liver, where they will remain permanently. This will lead to phagocytic overload of Kupffer cells and functional disturbances. First, permanent mechanical obstruction and impairment of sinusoidal blood flow in the liver occur, which may lead to ischemia and necrosis. This was previously described by Detombe and colleagues, who performed postmortem studies on the affected animals. 18 These studies showed necrotic areas in the kidneys and liver. Second, activated Kupffer cells will release toxic mediators, contributing to endothelial cell damage and leading to endotoxemia, sepsis, and, finally, death.
Due to the lethality of the small animals after injection of a dose of 32 mg I/20 g, we decided to further discuss only the results of the scans obtained after injection of a dose of 16 mg I/20 g of eXIA 160 XL.
Contrast enhancement of 30% was measured in the blood pool immediately after injection of only 16 mg I/20 g of eXIA 160 XL. Only half of the blood pool contrast enhancement (15%) could be reached using the same dose of eXIA 160 15 (Figure 5A). However, both contrast agents contain the same amount of iodine, namely 16 mg I/20 g (500 mg I/kg). Even a double dose of Fenestra LC and VC was required to obtain an enhancement of 19% versus 23%. 15 This difference is related to the iodine loading: eXIA 160 and eXIA 160 XL contain 3.2 times more iodine per milliliter than Fenestra LC and VC.

A, Contrast enhancement in blood after 16 mg I/20 g of eXIA 160 XL in comparison with eXIA 160. The data on eXIA 160 were obtained from earlier experiments. 15 B, Contrast enhancement in spleen after 16 mg I/20 g of eXIA 160 XL in comparison with eXIA 160. The data on eXIA 160 were obtained from earlier experiments. 15 C, Contrast enhancement in liver after 16 mg I/20 g of eXIA 160 XL in comparison with eXIA 160. The data on eXIA 160 were obtained from earlier experiments. 15
This study also demonstrates that the elimination of eXIA 160 XL from the blood pool was slower in comparison with that of eXIA 160 15 (see Figure 5A). Because of its colloidal nature, eXIA 160 and eXIA 160 XL can remain within the intravascular compartment for a longer time than conventional contrast agents. According to information provided by the manufacturer, a longer vascular residence time of eXIA 160 XL was achieved by size and surface modifications of the eXIA 160 contrast agent to make it less susceptible to clearance by the liver. Whereas most of the contrast is removed from the circulation by the liver, it should be completely metabolized and cleared within 24 hours and total mouse CT attenuation values should return to preinjection levels at around 48 hours following administration. We observed that the contrast returned to its background signal within 48 hours after administration (see Table 1). The median values of liver and spleen were below 1%.
eXIA 160 XL enhancement dropped 60% within the first hour (see Table 1). However, the enhancement remained above 15% during the first 30 minutes after contrast injection, providing good contrast enhancement for several consecutive micro-CT scans. The enhancement remained above 3% during 4 hours after administration of eXIA 160 XL (see Table 1), whereas after injection of eXIA 160, this enhancement remained only during the first hour. 15
Additionally, thanks to the injection of eXIA 160 XL, other organs, including the spleen and liver, can be well detected on the micro-CT images. This is related to the hepatobiliary clearance of the contrast agent, which is also seen with Fenestra LC and VC. 19 Peak contrast enhancement of eXIA 160 XL in the spleen occurred 30 minutes after injection, in contrast to eXIA 160, which had a peak contrast enhancement in the spleen already 15 minutes after contrast injection (22% versus 38%) (Figure 5B). The spleen plays a central role in the reticuloendothelial system (RES). The enhancement of the spleen might be explained by the elimination of eXIA 160 XL by the hepatobiliary system and the RES system. Peak contrast enhancement of the liver occurred 45 minutes after injection, similar to eXIA 160 (14% with eXIA 160 XL compared to 9.5% with eXIA 160) 15 (Figure 5C). Originally, eXIA 160 was developed as a hepatocyte-specific contrast agent to be used for detection of liver tumors as well as evaluation of diffuse liver disease. Hence, eXIA 160 XL results in higher contrast peak values in the liver but lower peak values in the spleen.
The peak contrast enhancement reached by eXIA 160 (9.5%) 14 or even higher values could be obtained by using eXIA 160 XL during the first 3 hours after contrast enhancement. This makes eXIA 160 XL more valuable in serial micro-CT scanning of the liver. In spleen imaging, eXIA 160 is more useful because the peak enhancement is 40% higher compared to eXIA 160 XL (see Table 1).
Renal filtration was not observed due to the absence of contrast enhancement in the bladder.
For experiments with eXIA 160 XL, a scan delay at the maximum enhancement will result in optimal image quality. However, depending on the purpose of the study and the desired image quality, the time interval to scan the mouse can be more flexible. For example, the contrast enhancement in the liver is more than 10% after the 15-minute scan and remains above this level until the 3-hour scan. This can be sufficient to distinguish the organs from the surrounding tissue.
The pharmacokinetic characteristics of eXIA 160 have been previously reported in the literature. The manufacturer performed size and surface modifications on eXIA 160 to create eXIA 160 XL. eXIA 160 is an aqueous colloidal polydispersed iodinated contrast medium without renal filtration and without diffusion from intravascular to interstitial compartments. As such, it is possible to use it as a blood pool agent. It remains in the circulation for a prolonged period of time and is suitable for characterization of blood vessels and certain types of pathologic abnormalities found in cancer. eXIA 160 has the characteristic feature of extravasating slowly out of the vessels. 20
Conclusion
Administration of eXIA 160 XL results in long-lasting blood pool contrast with higher contrast enhancement in heart and liver in comparison with eXIA 160; however, the administered dose should be limited to 16 mg I/20 g due to lethal effects at the studied higher dose. Its high iodine content allows small injection volumes in the range of 0.1 mL/20 g, which is preferred in serial small-animal in vivo studies.
Footnotes
Acknowledgment
Financial disclosure of authors and reviewers: None reported.

