Abstract
The aim of this study was to evaluate the impact of different anesthetics on 3′-[18F]fluoro-3′-deoxythymidine ([18F]FLT) uptake in carcinomas and arthritic ankles. To determine the amount of [18F]FLT uptake in subcutaneous CT26 colon carcinomas or arthritic ankles, spontaneously room air/medical air–breathing mice were anesthetized with isoflurane, a combination of medetomidine/midazolam, or ketamine/xylazine. Mice were kept conscious or anesthetized during [18F]FLT uptake before the 10-minute static positron emission tomographic (PET) investigations. [18F]FLT uptake in CT26 colon carcinomas and arthritic ankles was calculated by drawing regions of interest. We detected a significantly reduced (4.4 ± 0.9 %ID/cm3) [18F]FLT uptake in the carcinomas of ketamine/xylazine-anesthetized mice compared to the [18F]FLT-uptake in carcinomas of medetomidine/midazolam- (7.0 ± 1.5 %ID/cm3) or isoflurane-anesthetized mice (6.4 ± 1.5 %ID/cm3), whereas no significant differences were observed in arthritic ankles regardless of whether mice were anesthetized or conscious during tracer uptake. The time-activity curves of carcinomas and arthritic ankles yielded diverse [18F]FLT accumulation related to the used anesthetics. [18F]FLT uptake dynamics are different in arthritic ankles and carcinoma, and the magnitude and pharmacokinetics of [18F]FLT uptake are sensitive to anesthetics. Thus, for preclinical in vivo [18F]FLT PET studies in experimental tumor or inflammation models, we recommend the use of isoflurane anesthesia as it yields a stable tracer uptake and is easy to handle.
POSITRON EMISSION TOMOGRAPHY (PET) is an established noninvasive in vivo method for longitudinal studies, for example, for monitoring therapy responses or disease progression in preclinical research and clinical diagnosis. 3′-[18F]Fluoro-3′-deoxythymidine ([18F]FLT), a proliferation tracer, is used primarily for the detection of cell proliferation in carcinomas and is not yet well established for detecting cell proliferation in inflamed tissues.1,2 [18F]FLT is transported into the cell, phosphorylated by thymidine kinase 1 (tk1), and trapped within the cell. This process allows for the noninvasive in vivo measurements of cellular tk1 activity, which is linked to cellular proliferation. 3
However, to enable noninvasive in vivo preclinical measurements, animals need to be anesthetized during the PET scans, which might last between 10 minutes for static data acquisition and 2 hours for dynamic data acquisition. The most commonly used anesthetics in rodent research studies are isoflurane, ketamine/xylazine, and medetomidine/midazolam. 4 Anesthetics such as isoflurane, sevoflurane, ketamine, pentobarbital, and midazolam can impair heart function, suppress breathing frequency, change blood pressure and glucose metabolism, reduce tumor blood flow and oxygenation, and counteract inflammatory processes.5–9 Isoflurane, a γ-aminobutyric acid (GABAA) receptor agonist, is used as an inhaled anesthetic well known for its rapid anesthesia induction and recovery. 10 Ketamine is an Nmethyl-D-aspartate (NMDA) receptor antagonist that also binds to μ-opioid and σ-opioid receptors and is well suited for anesthesia induction. 11 Medetomidine and xylazine are α2-adrenoreceptor agonists that confer skeletal muscle relaxation and hypnotic effects enforcing anesthesia. 12 The benzodiazepine midazolam, a GABAA receptor agonist, has potent hypnotic and skeletal muscle relaxant properties 13 and is thus best qualified for anesthesia induction (Table S1, online version only).
In previous studies, we observed respiratory acidosis and, consequently, a reduced [18F]FLT uptake in carcinomas and inflamed ankles of ketamine/xylazine-anesthetized mice that were conscious and breathed oxygen during tracer uptake but not in mice that breathed air. 14 The aim of this study was to identify the best suited protocol for anesthesia induction for noninvasive in vivo examination of [18F]FLT-PET studies in an experimental carcinoma and inflammation model. Consequently, we analyzed the differential effects on the pharmacokinetics of [18F]FLT uptake in subcutaneous CT26 colon carcinomas,15,16 muscle tissue, and glucose-6-phosphate isomerase (GPI)-arthritic or healthy ankles 17 of short- or long-duration ketamine/xylazine-, medetomidine/midazolam-, or isoflurane-anesthetized mice (Figure 1).

In vivo [18F]FLT-PET scan protocols that were used for the measurement of arthritic and healthy ankles and CT26 colon carcinomas under air breathing. A, Dynamic PET protocol to identify differences in the time-activity curves under different anesthetics. B, PET protocol when mice were conscious during tracer uptake followed by an anesthetized 10-minute static PET scan. C, PET protocol when mice were anesthetized during [18F]FLT tracer uptake followed by a 10-minute static PET scan.
Material and Methods
GPI-Arthritis Model
The K/BxN arthritis mouse model needs two different mouse strains: heterozygous KRN T-cell receptor transgenic B10.BR C57Bl/6 mice and homozygous NOD/Lt mice carrying the Ag7 allele. 18 K/BxN mice express the transgenic KRN T-cell receptor and the major histocompatibility complex (MHC) class II molecule Ag7 and develop severe inflammatory arthritis beginning at 3 weeks of age. Serum obtained from K/BxN mice can be injected into different mouse strains and causes arthritis within 1 day after injection due to GPI-specific autoantibodies. 18 GPI serum was obtained retrobulbarly from male and female K/BxN transgenic mice at the age of 1 to 3 months that were bred under specific pathogen-free (SPF) conditions. Control serum was obtained retrobulbarly from healthy male and female C57BL/6 mice at the age of 1 to 6 months.
Female BALB/c mice at the age of 6 to 10 weeks and female/male C57BL/6 mice were obtained from Charles River (Sulzbach, Germany). Mice were bred under SPF conditions and were kept in individually ventilated cages (IVCs) in our animal housing. The mean weight of the mice was 15 to 20 g.
A total of 200 μL of diluted GPI serum (1:1 serum/saline) was injected intraperitoneally into healthy BALB/c mice on days 0 and 2 to induce joint inflammation.17–19 Control mice were injected with diluted control serum. [18F]FLT-PET investigations were performed at days 5 to 7 after GPI or control serum injection.
Spontaneously breathing animals (group 1, n = 9) were scanned under isoflurane (n = 9), medetomidine/midazolam (n = 9), or ketamine/xylazine (n = 9) anesthesia with room air/medical air breathing on 3 consecutive days. A second group of 23 animals was scanned using ketamine/xylazine and room air/medical air (n = 8) or 100% oxygen (n = 15), isoflurane/room air/medical air (n = 14), and medetomidine/midazolam/room air/medical air (n = 7) on 2 consecutive days. A third group of animals (n = 9) was scanned under isoflurane/room air/medical air (Table S2, online version only).
CT26 Mouse Colon Carcinoma Model
Female 6- to 8-week-old BALB/c mice (Charles River) were injected subcutaneously with 5 × 105 CT26 colon carcinoma cells in 200 μL NaCl below the right upper flank (dorsal).15,16 The mean weight of the mice was 17.81 ± 1.13 g. CT26 mouse colon carcinoma cells were cultured as recently described.14,16 Ten to 14 days after tumor cell injection, PET measurements were performed.
Group 1 (n = 12) and group 2 (n = 11) animals were scanned using isoflurane/100% oxygen, isoflurane/room air/medical air, medetomidine/midazolam/room air/medical air, and ketamine/xylazine/room air/medical air on consecutive days. A third group of 20 mice was scanned with ketamine/xylazine/100% oxygen and room air/medical air breathing on 2 consecutive days (Table S3, online version only).
Anesthesia
The mice were anesthetized using isoflurane, ketamine/xylazine, or medetomidine/midazolam as follows:
• 1.5 vol.% isoflurane (Forene, Abbott Labs, Baar, Switzerland) was delivered in medical air at a flow rate of 0.8 L/min using a dedicated vaporizer (Vetland, Louisville, KY).
• A mixture of ketamine (100 mg/kg, Ratiopharm, Ulm, Germany) and xylazine (5 mg/kg, Rompun, Bayer HealthCare, Leverkusen, Germany) was administered intraperitoneally in a volume of 0.1 mL/10 g body weight.
• A mixture of medetomidine (0.5 mg/kg, Domitor, Pfizer, Karlsruhe, Germany) and midazolam (5 mg/kg, Dormicum, Ratiopharm, Ulm, Germany) was administered intraperitoneally. To reverse the anesthesia, a mixture of atipamezole (2.5 mg/kg, Antisedan, Pfizer, Karlsruhe, Germany) and flumazenil (0.5 mg/kg, Anexate, Fresenius, Bad Homburg, Germany) was injected subcutaneously in a volume of 0.1 mL/10 g body weight.
If necessary, redosing of ketamine/xylazine or medetomidine/midazolam anesthesia was applied by a subcutaneous catheter connected to an infusion pump (PHD 22/2000, Harvard Apparatus) with a continuous flow of 8 μL/min for 10 minutes.
Animal Preparation
To avoid a decrease in body temperature, mice were heat supported over the entire scan time and the [18F]FLT tracer uptake time by using a heating mat. The heating mat (37°C) was placed beneath the anesthesia box during the entire [18F]FLT tracer uptake time as well as beneath the mice on the PET bed during the scanning time. Mice were kept in IVCs in our animal housing facility. For the [18F]FLT PET studies, the IVCs were taken into our imaging laboratory. Afterward, at least 10 minutes before the [18F]FLT tracer injection or anesthesia induction, the capping of the IVC was removed. After [18F]FLT injection, mice that were anesthetized during the tracer uptake time were placed into an anesthesia box. Mice breathed medical air in the anesthesia box and on the PET bed during the entire PET investigation. Mice that were conscious during the tracer uptake time were placed back into the cage after [18F]FLT injection and breathed room air until anesthesia induction.
For dynamic PET investigations, anesthetized mice were injected intravenously into the tail vein with [18F]FLT on the PET scanner bed and investigated for 90 minutes (arthritis) or 120 minutes (CT26) (see Figure 1A).
Conscious mice were injected intravenously with [18F]FLT and allowed to move freely for 30 minutes (arthritis) or 115 minutes (CT26) in IVCs with open lids breathing room air prior to the induction of anesthesia and a 10-minute static PET scan (see Figure 1B).
The anesthetized mice were injected intravenously with [18F]FLT and kept continuously anesthetized during tracer uptake in a temperature-controlled anesthesia box before the 10-minute static PET scans were performed (see Figure 1C).
The [18F]FLT tracer was administered by intravenous injection using a 30-gauge needle (Becton Dickinson, Franklin Lakes, NJ) attached to a polyethylene catheter (High-Med-PE, 0.28 mm inner diameter, Reichelt Chemietechnik GmbH+Co, Heidelberg Germany) into the tail vein. The mice were injected with 9.5 ± 1.2 MBq (arthritis) or 5.8 ± 0.7 MBq (CT26) of [18F]FLT. After tracer injection, the catheter was flushed with 0.05 mL of saline. [18F]FLT was synthesized as described elsewhere. 20
In Vivo PET Imaging and Data Analysis
PET images were acquired on a microPET scanner (Inveon, Siemens Medical Solutions, Knoxville, TN) with a spatial resolution of 1.4 mm in the reconstructed PET images. 21 We used IAW, versions 1.0.4.0, 1.2.2.2, and 1.4.3.6 (Siemens Healthcare, Knoxville, TN). Acquired list-mode data were reconstructed by using the iterative two-dimensional ordered subset expectation maximization (OSEM2D) algorithm with 4 iterations and 16 subsets. The calibration factor is included in the normalization file and is hence applied during reconstruction of the emission data. Images were normalized to each other to the injected dose. No attenuation correction was applied.
Residual activity in the tail and the whole-body dose were not excluded from our data analysis because the regions of interest (ROI: arthritic ankles, CT26 carcinomas) were placed in the center of the field of view; therefore, whole-body imaging was not performed.
In the arthritis model, the data were analyzed by drawing defined ROI of 3.5 mm in diameter on three adjacent image slices of the normalized PET images. ROI covered the area of maximum activity in the inflamed and healthy ankles (Figure S1A, online version only).
In the CT26 model, the ROI were matched to the tumor size on all slices of the normalized PET images. As a control region, three ROI were placed over three slices in the shoulder muscle tissue on the opposite side of the tumor (Figure S1B). To quantify the PET results, the mean of the measured counts in the ROI was calculated and is reported as a percentage of the injected dose per cubic centimeter (%ID/cm3). Image analysis was performed with the microPET ASIPro VM software package (Siemens Preclinical Solutions, Knoxville, TN).
Histologic Analyses
Mice were sacrificed and ankles from GPI-arthritic mice and CT26 tumors were fixed in 4% buffered formalin and embedded in paraffin. Tissues were cut into 3 to 5 μm slices and stained with hematoxylin and eosin (H&E) by standard procedures. 19 We performed immunohistochemistry using an automated immunostainer (Ventana Medical Systems, Inc., Tucson, AZ) according to the manufacturer's protocols for open procedures, with slight modifications. All of the slides were stained with a Ki-67-specific antibody (SP6, Thermo Fisher Scientific, MI), and to confirm the adequacy of the staining, appropriate positive controls were used. A grid ocular objective was used to count 300 cells over three high-power fields (x40), and the percentage of positive cells was reported from 0 to 100%.
Statistical Data Analysis
Differences in the [18F]FLT uptake values (%ID/cm3) in CT26 colon carcinomas or arthritic ankles from mice that underwent different anesthesia protocols were compared using the Dunnett test. In the GPI-arthritis model, only swollen ankles were analyzed. For mice with two swollen ankles, we calculated the mean of the [18F]FLT uptake values, and for mice with only one swollen ankle, we used the [18F]FLT uptake value of that arthritic ankle for statistical analysis. The two-tailed Student t-test was used to analyze the differences in [18F]FLT uptake (%ID/cm3) in muscle tissue between the CT26 colon carcinoma–bearing mice that were anesthetized or conscious during tracer uptake. Tumor- to muscle ratios were compared using the Tukey-Kramer test, and p values < .05 were considered to be statistically significant. All of the data represent the mean ± 1 SD.
Results
Type of Anesthesia Influences [18F]FLT Pharmacokinetics
We first evaluated the kinetics of [18F]FLT uptake in CT26 colon carcinomas and arthritic and healthy ankles of ketamine/xylazine-, medetomidine/midazolam-, or isoflurane-anesthetized mice. Animals were investigated 10 to 14 days after the subcutaneous injection of 5 × 105 CT26 colon carcinoma cells, when the average tumor volume was 117 ± 67 mm3, or 5 to 7 days after control or GPI serum injection, a point in time with maximum ankle swelling 14 (Figure S2, online version only). For dynamic scans, room air/medical air–breathing mice were anesthetized and injected intravenously at the PET bed with [18F]FLT and continuously investigated by PET for 120 minutes (carcinomas) or 90 minutes (arthritis) under temperature-controlled conditions (see Figure 1A). Static PET scans were performed in spontaneously room air/medical air–breathing mice for 10 minutes after anesthetized or conscious [18F]FLT uptake over 30 (arthritis) or 115 (CT26 colon carcinoma) minutes (see Figure 1, B and C).
ROI were drawn to calculate the [18F]FLT time-activity curves (TACs; 7,200 seconds) of CT26 colon carcinomas (see Figure 2 and Figure S1B), muscle tissue, and arthritic and healthy ankles (see Figure 3 and Figure S1A). The TACs of CT26 colon carcinomas of mice that underwent isoflurane anesthesia (n = 3) (see Figure 2B) yielded slow but continuous [18F]FLT accumulation until the end of the measurements, in contrast to the TACs from CT26 colon carcinomas of ketamine/xylazine-anesthetized mice (see Figure 2A), which reached a plateau until the end of the dynamic PET scans. Medetomidine/midazolam-anesthetized mice showed a rapid uptake of [18F]FLT and a tendency for a slight but not statistically significant washout after a 1-hour scan time (see Figure 2C). The TACs of muscle tissue showed an initial fast uptake over the first 1,000 seconds followed by a slight decrease independent of the type of anesthesia. The muscle tissue of ketamine/xylazine-anesthetized CT26 colon carcinoma–bearing mice displayed a higher uptake over the entire scan time than the muscle tissue of isoflurane- or medetomidine/midazolam-anesthetized mice. Continuous [18F]FLT tracer uptake in CT26 colon carcinomas demonstrated the high proliferative state within the tumor, proven by the establishment of a plateau.

Time-activity curves of dynamic (7,200 seconds) [18F]FLT-PET measurements of CT26 colon carcinomas and muscle tissue of (A) ketamine/xylazine- (n = 3), (B) isoflurane- (n = 3), or (C) medetomidine/midazolam-anesthetized air-breathing mice (n = 3).

Time-activity curves of dynamic (5,400 seconds) [18F]FLT-PET measurements of arthritic ankles and healthy control ankles of (A) ketamine/xylazine- (glucose-6-phosphate isomerase [GPI] arthritis: n = 4; control: n = 1), (B) isoflurane- (GPI arthritis: n = 8; control: n = 3), or (C) medetomidine/midazolam-anesthetized air-breathing mice (GPI arthritis: n = 3; control: n = 1).
The [18F]FLT PET TACs of arthritic ankles (5,400 seconds) indicated differences due to the type of anesthesia (see Figure 3). After an initial peak, [18F]FLT uptake declined slightly but significantly regardless of the type of anesthesia used (see Figure 3). The decline was significantly less pronounced in the arthritic ankles of [18F]FLT-injected mice that underwent ketamine/xylazine (n = 4), medetomidine/midazolam (n = 3), or isoflurane (n = 8) anesthesia (see Figure 3). The [18F]FLT TACs of healthy ankles from control serum–injected mice (ketamine/xylazine: n = 1, isoflurane: n = 3, medetomidine/midazolam: n = 1) showed no perfusion peak and reached a plateau shortly after intravenous [18F]FLT tracer injection. Differences in the [18F]FLT TACs between CT26 colon carcinomas and arthritic ankles were predominantly identified in the pharmacokinetics, in which the CT26 TACs showed slower uptake. In contrast, the TACs of the arthritic mice were characterized by an [18F]FLT uptake peak within the first 500 seconds, followed by a steady decrease. The initial [18F]FLT uptake peak in arthritic mice reflects the increased perfusion characteristic of inflammation.
Effects of Anesthesia on [18F]FLT Uptake in Carcinomas
The [18F]FLT uptake in CT26 colon carcinomas of mice that were anesthetized during the entire tracer [18F]FLT uptake period was slightly higher than uptake in mice that were conscious during tracer uptake followed by a short 10-minute period of anesthesia during the static PET scan (ketamine/xylazine: anesthetized 4.3 ± 0.9 %ID/cm3 [n = 14] vs conscious 3.8 ± 1.6 %ID/cm3 [n = 17]; isoflurane: anesthetized 6.4 ± 1.5 %ID/cm3 [n = 7] vs conscious 5.9 ± 0.4 %ID/cm3 [n = 7]; medetomidine/midazolam: anesthetized 7.0 ± 1.5 %ID/cm3 [n = 7] vs conscious 6.8 ± 0.4 %ID/cm3 [n = 4]) (Figure 4). In ketamine/xylazine-anesthetized mice that were conscious during the [18F]FLT tracer uptake phase (see Figure 4A), we detected a significantly reduced absolute level of [18F]FLT in CT26 colon carcinomas compared to the uptake in isoflurane- (5.9 ± 0.4 %ID/cm3, n = 7, p = .002; see Figure 4B) and medetomidine/midazolam (6.8 ± 0.4 %ID/cm3, n = 4, p = 0.0002; see Figure 4C). In line with the [18F]FLT uptake data for CT26 colon carcinomas, the uptake in the muscle tissue of mice that were conscious during tracer uptake (1.8 ± 0.7 %ID/cm3) and anesthetized only for 10 minutes with ketamine/xylazine was significantly lower (p = .0002) than the uptake in mice that were anesthetized with ketamine/xylazine (2.7 ± 0.4%ID/cm3) during the entire tracer uptake time (see Figure 4A). A similar but not statistically significant tendency was observed in isoflurane- and medetomidine/midazolam-anesthetized mice (see Figure 4, B and C). Figure S2A shows that the differences in [18F]FLT uptake were not related to differences in the volumes of the CT26 colon carcinomas of the experimental groups. 14
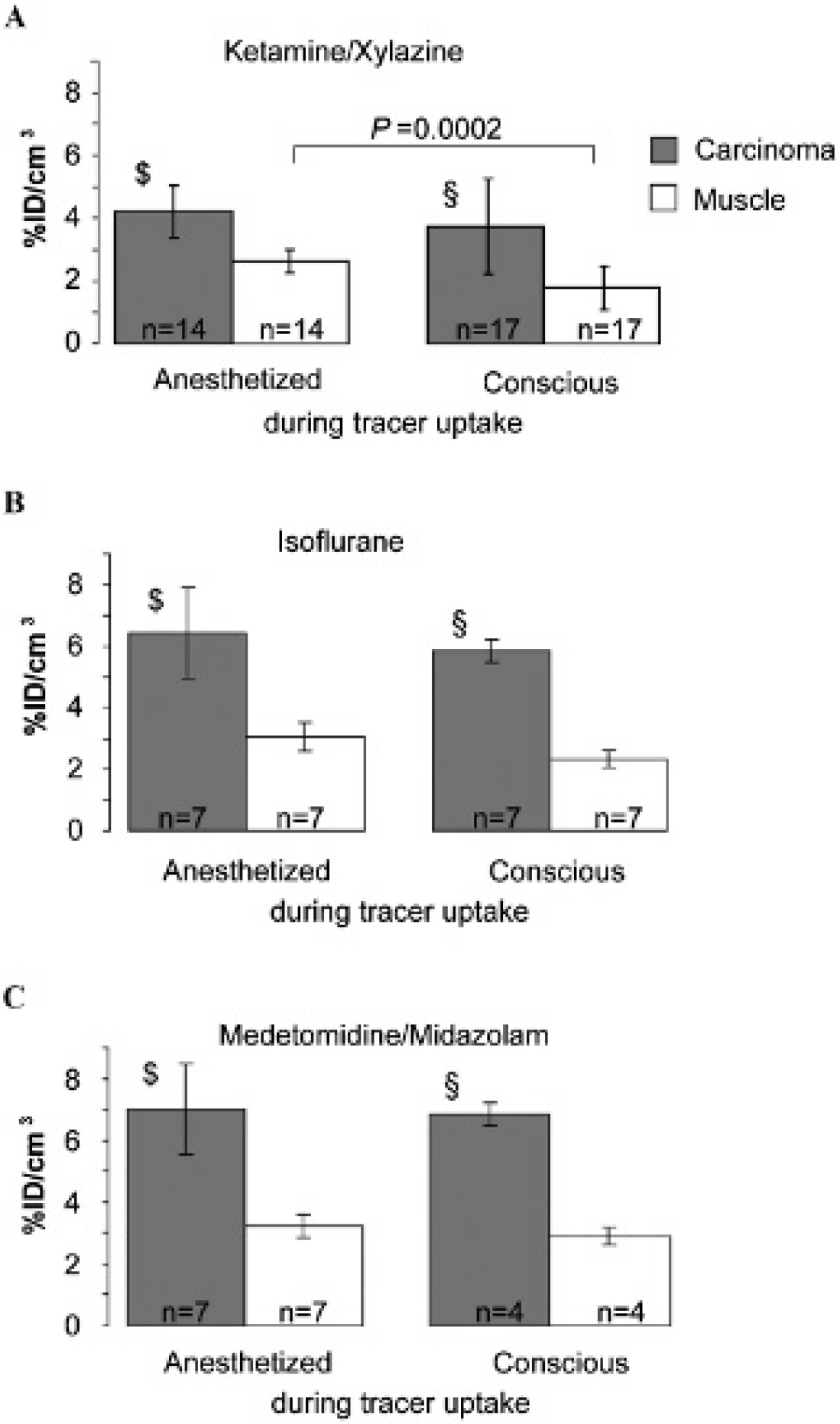
Quantitative analysis of pooled static [18F]FLT-PET scans of CT26 carcinomas and muscle tissue of mice that were conscious or awake during [18F]FLT uptake. [18F]FLT uptake of CT26 carcinomas and muscle tissue in (A) ketamine/xylazine-, (B) isoflurane-, or (C) medetomidine/midazolam-anesthetized mice. The [18F]FLT uptake values (%ID/cm3) for CT26 colon carcinomas of ketamine/xylazine-anesthetized mice were compared to the [18F]FLT uptake values for isoflurane- and medetomidine/midazolam-anesthetized mice using the Dunnett test. Mice that were anesthetized ($) or conscious (§) during tracer uptake showed significant differences between ketamine/xylazine versus isoflurane and medetomidine/midazolam: p < .004.
Effects of Anesthesia on [18F]FLT Uptake in Inflammation
In contrast to the significantly reduced [18F]FLT uptake values in CT26 colon carcinomas of ketamine/xylazine-anesthetized mice, we could not detect significant differences in [18F]FLT uptake in the arthritic ankles of isoflurane-, ketamine/xylazine-, or medetomidine/midazolam-anesthetized mice, regardless of whether the mice were conscious or anesthetized during the tracer uptake phase (Figure 5A: ketamine/xylazine: anesthetized 3.5 ± 0.8 %ID/cm3 [n = 4] vs conscious 3.7 ± 0.8 %ID/cm3 [n = 4]; Figure 5B: isoflurane: anesthetized 2.8 ± 0.4 %ID/cm3 [n = 8] vs conscious 2.9 ± 0.6 %ID/cm3 [n = 7]; Figure 5C: medetomidine/midazolam: anesthetized 3.7 ± 0.7 %ID/cm3 (n = 4) vs conscious 3.4 ± 0.6 %ID/cm3 [n = 4]). The lowest [18F]FLT uptake values in arthritic ankles were observed in isoflurane-anesthetized mice. Moreover, we did not observe any significant alterations in [18F]FLT uptake in the healthy ankles of control serum–injected mice. The [18F]FLT uptake values of healthy ankles were similar in all of the experimental groups breathing room air/medical air (1.1 ± 0.3 %ID/cm3, n = 14), regardless of whether the mice were conscious or anesthetized during the [18F]FLT tracer uptake period. Figure S2B shows that the differences in [18F]FLT uptake were not related to the differences in ankle swelling. 14

Quantitative analysis of pooled static [18F]FLT-PET scans of arthritic and healthy ankles of mice that were conscious or awake during [18F]FLT uptake. [18F]FLT uptake in arthritic and healthy ankles of (A) ketamine/xylazine-, (B) isoflurane-, or (C) medetomidine/midazolam-anesthetized mice. The [18F]FLT uptake values (%ID/cm3) for arthritic ankles of ketamine/xylazine-anesthetized mice were compared to the [18F]FLT uptake values for isoflurane- and medetomidine/midazolam-anesthetized mice using the Dunnett test; the differences were not significant. GPI = glucose-6-phosphate isomerase.
Histology and Ki-67 Immunohistochemistry
In the CT26 colon carcinoma model, approximately 40% of carcinoma cells stained positive for the immunohistochemical proliferation marker Ki-67 without an indication of infiltrating proliferating leukocytes in the H&E stains. In contrast, in arthritic ankles, approximately 30% of resident cells (eg, synovial fibroblasts, dendritic cells) and infiltrating cells (eg, lymphocytes, macrophages) stained positive for Ki-67 (Figure 6). The number of positive cells was resported in %. Thus, in CT26 colon carcinomas, we expect [18F]FLT uptake predominantly in resident nonmigrating proliferating carcinoma cells, and in arthritic ankles, we expect [18F]FLT uptake in proliferating resident synovial fibroblasts and macrophages and infiltrating leukocytes. H&E staining of arthritic ankles showed massive inflammation, invasion of leukocytes, and pannus formation compared to healthy controls (see Figure 6).

Hematoxylin-eosin (H&E) histology and Ki-67 immunohistochemistry of CT26 colon carcinomas (n = 3) on day 13 after tumor inoculation and of arthritic and healthy ankles (n = 3) on day 8 after glucose-6-phosphate isomerase (GPI) serum or control serum transfer (original magnifications: x50, x100, and x400); H&E stains of CT26 colon carcinomas indicated no infiltrates of leukocytes. Approximately 40% of carcinoma cells stained positive for Ki-67. In contrast, H&E stains of arthritic ankles showed a dense leukocyte infiltration in the synovium and the subsynovial pericapsular tissue, a strong increase in the fibrous connective tissue, and pannus formation, whereas healthy ankles displayed normal joint structures. In arthritic ankles, approximately 30% of resident cells and infiltrating leukocytes stained positive for Ki-67. C = cartilage; JS = joint space; S = synovium.
Thus, the [18F]FLT uptake in CT26 colon carcinomas (3.8–7.0 %ID/cm3: range of all anesthesia protocols) and the [18F]FLT uptake in arthritic ankles (2.8–3.7 %ID/cm3: range of all anesthesia protocols) correspond quite well with the percentage of Ki-67-positive cells (Ki −67 index: CT26, approximately 40%; GPI-arthritis, approximately 30%).
Discussion
The type and duration of anesthesia and the choice of breathing gas can impact the reproducibility and reliability of PET imaging studies and thus have to be properly selected prior to the start of new research projects. Therefore, knowledge of the impact of anesthetics, the anesthesia duration, and breathing on the physiologic parameters of laboratory animals is crucial to make imaging studies from different laboratories comparable and to avoid unnecessary animal experiments.
In our experiments, we identified differences in [18F]FLT TACs and [18F]FLT uptake values in CT26 colon carcinomas and arthritic ankles when using different anesthesia protocols. The variations were more severe in the CT26 tumor model than in the rheumatoid arthritis model. To further elucidate the anesthesia-induced differences in [18F]FLT uptake in CT26 carcinomas and arthritic ankles, we focused on the mode of action and on the specific side effects of isoflurane, ketamine/xylazine, and medetomidine/midazolam anesthesia (see Table S1). Ketamine is described as a unique drug because of its hypnotic, analgesic, amnesic, and stimulatory cardiovascular effects. Ketamine binds to α2B receptors of smooth muscle cells of the vasculature and induces vasoconstriction and, consequently, an initial increase in venous and arterial blood pressure and a decrease in the heart rate.22,23 Xylazine, an α2-adrenoreceptor agonist, exhibits skeletal muscle relaxation and hypnotic and analgesic effects. During long-term anesthesia, xylazine causes a decrease in the mean arterial pressure and cardiac output 24 and increased breathing depression caused by other anesthetics. However, combined ketamine/xylazine anesthesia causes increased blood pressure 25 and a decreased respiration rate. The α2-adrenoreceptor agonist medetomidine is similar to xylazine and also exhibits skeletal muscle relaxation and hypnotic and analgesic effects. Medetomidine decreases the heart rate, provokes arrhythmia, and causes breathing depression. 12 The benzodiazepine midazolam has potent anxiolytic, amnesic, hypnotic, anticonvulsant, skeletal muscle relaxant, and sedative properties. Isoflurane is a commonly used inhalation anesthesia for small-animal imaging and has hypnotic and skeletal muscle relaxant effects but no analgesic effects. Isoflurane anesthesia induces a dose-dependent depression of breathing, 26 shows no alterations in tissue perfusion or oxygenation, 23 and triggers antiinflammatory effects. In addition, it decreases cardiac output and increases the heart rate. Isoflurane, ketamine, and midazolam are also known to have antiinflammatory effects,27–30 which might reduce [18F]FLT uptake in different experimental models (see Table S1).
The antiinflammatory effects of isoflurane and ketamine have been demonstrated in various models. Molecular mechanisms involve activation of adenosine and β-adrenergic receptors. 31 In addition, inhibition of relevant proinflammatory cytokines such as tumor necrosis factor α and interleukin-1 contributes to the antiinflammatory effects of volatile anesthetics.32,33 Further antiinflammatory effects of isoflurane include blocking the oxidative burst of neutrophils and phagocytosis of monocytes/macrophages, both critical mechanisms of the innate immune system. 34 Importantly, antiinflammatory properties of volatile anesthetics are critically time dependent and occur in an early (4–6 hours) and a late (12–24 hours) time window, both of which include distinct molecular mechanisms.35–37 We measured an enhanced [18F]FLT uptake in CT26 colon carcinoma–bearing mice that were anesthetized during the tracer uptake phase compared to mice with a conscious tracer uptake. All measurements were performed before the defined time frames of the antiinflammatory effects were evident; therefore, we are convinced that the antiinflammatory effects of the anesthetics did not crucially impair [18F]FLT uptake in tumor and arthritic ankles. Consequently, the antiinflammatory effects of the anesthetics should not reduce leukocyte populations within 1 hour. Moreover, we expected no significant impairment of our longitudinal [18F]FLT PET studies because the mice were investigated in 24-hour time intervals, exactly at the time when every antiinflammatory effect should have disappeared. Finally, the antiproliferative effects of isoflurane, ketamine, xylazine, medetomidine, and midazolam have not been demonstrated yet.
In addition, it is important to mention that some of the CT26 colon carcinoma–bearing mice were investigated on 4 consecutive days and some of the arthritic mice on 3 consecutive days (see Table S2 and Table S3). Analysis of confounding effects on [18F]FLT uptake as a consequence of repetitive anesthesia—once daily—yielded an enhanced [18F]FLT uptake in CT26 colon carcinomas and a reduced [18F]FLT uptake in arthritic ankles. Importantly, this phenomenon did not impair the general conclusion of our investigations.
In addition to studies on [18F]FLT, various experiments have been conducted to evaluate the impact of different anesthetics on [18F]FDG uptake in mice and rats.6,38–40 For [18F]FDG PET imaging, the type and strength of anesthetic should be carefully selected based on the suitability for the visualization of the target tissue of an experimental model because different anesthetics, the nutritional state (fasted/not fasted), and the maintenance of the body temperature (warmed/not warmed) are factors that can influence [18F]FDG uptake in carcinomas or organs of interest in small-animal models. Consequently, these factors may also have an effect on the uptake of other tracers, such as [18F]FLT.
In our PET studies, the [18F]FLT TACs of CT26 colon carcinomas of isoflurane-anesthetized mice showed slow but continuous [18F]FLT accumulation until the end of the measurements, in contrast to the TACs of ketamine/xylazine-anesthetized mice, which displayed a plateau until the end of the dynamic PET scans (see Figure 2).
The [18F]FLT TACs of arthritic ankles of isoflurane-anesthetized mice displayed a slight decrease after an initial injection peak over the entire time of the investigation, whereas the [18F]FLT TACs of medetomidine/midazolam- or ketamine/xylazine-anesthetized mice yielded an initial injection peak followed by a constant plateau until the end of the investigation (see Figure 3). In CT26 colon carcinomas without inflammatory cell infiltration, we mainly expect [18F]FLT uptake primarily by resident nonmigrating proliferating carcinoma cells. In contrast to CT26 colon carcinomas, in arthritic ankles, we expect [18F]FLT uptake primarily by proliferating resident synovial fibroblasts and infiltrating leukocytes (see Figure 6). Activated proliferating lymphocytes migrate into the inflamed ankles (endothelial rolling, firm adhesion, and transmigration in postcapillary venules) and reside or die there as a consequence of the oxidative burst caused by the neutrophils or migrate (eg, dendritic cells) into the regional lymph nodes. The influx of activated leukocytes could be impaired by an increased or reduced flow rate within the postcapillary venules and arterioles caused by the different anesthetics. This difference in flow rate might explain the differences in the [18F]FLT TAC between CT26 colon carcinomas and arthritic ankles (see Figure 2 and Figure 3). The enhanced heart rate in isoflurane-anesthetized mice27–30 and the ketamine anesthesia–induced vasoconstriction and increase in the venous and arterial blood pressure22,23 could explain some of the differences in the [18F]FLT TACs of CT26 colon carcinomas and arthritic ankles. These factors could also account for the reduced [18F]FLT uptake in CT26 colon carcinomas in ketamine/xylazine-anesthetized mice and the reduced uptake in arthritic ankles of isoflurane-anesthetized mice (see Figure 4A and Figure 5B). We hypothesize that the decreased [18F]FLT uptake in arthritic ankles of isoflurane-anesthetized mice could be the consequence of anesthesia-specific effects. Although our data do not allow final conclusions regarding the underlying mechanisms, it is tempting to speculate that the impaired influx of leukocytes might account for the reduced [18F]FLT uptake in arthritic ankles. Moreover, the increase in venous blood flow may cause enhanced [18F]FLT uptake in muscle tissue and reduced uptake in CT26 colon carcinomas of mice that were ketamine/xylazine anesthetized during tracer uptake. Interestingly, a tendency toward increased ratios between healthy and arthritic ankles and between CT26 colon carcinomas and muscle tissue was found in all of the experimental groups that were conscious during tracer uptake (Table S4, online version only). In line with our quantitative results (%ID/cm3) (see Figure 4), the tumor tomuscle ratios of ketamine/xylazine-anesthetized mice that were anesthetized during [18F]FLT uptake were significantly reduced compared to isoflurane- and medetomidine/midazolam-anesthetized mice (see Table S4).
Hence, for PET imaging studies, isoflurane anesthesia provided a stable [18F]FLT uptake under room air/medical air breathing conditions, regardless of whether the mice underwent a conscious or an anesthetized tracer uptake. Furthermore, isoflurane as an inhalation anesthesia provides an easy handling and a proper dose regulation of anesthesia, especially for long-term studies. Compared to ketamine/xylazine and medetomidine/midazolam injection anesthetics, redosing is not required.
Conclusions
Various anesthetics impact the kinetics and absolute [18F]FLT tracer uptake in CT26 colon carcinomas and arthritic ankles in different ways. To render studies comparable and to avoid variations in [18F]FLT uptake due to different imaging protocols and different anesthetics, we recommend the adoption of standardized anesthesia protocols. Thus, for preclinical in vivo [18F]FLT PET studies, we recommend isoflurane anesthesia and air breathing to ensure stable tracer uptake conditions.
Footnotes
Acknowledgments
We thank Maren Koenig, Mareike Lehnhoff, Daniel Bukala, Nadine Bauer, Ursula Kohlhofer, Dennis Thiele, and Stefan Wiehr for excellent technical and practical assistance.
Financial disclosure of authors: This work was supported by the Deutsche Forschungsgemeinschaft (SFB 773), the Bundesministerium für Bildung und Forschung (BMBF, FKZ 0314103), and the Werner Siemens-Foundation.
Financial disclosure of reviewers: None reported.
