Abstract
Epidermal growth factor receptor (EGFR) signaling inhibition represents a highly promising arena for the application of molecularly targeted cancer therapies. EGFR conjugated metal chelates have been proposed as potential imaging agents for cancers that overexpress EGFR receptors. Through improved understanding of EGFR biology in human cancers, there is anticipation that more tumor-selective therapy approaches with diminished collateral normal tissue toxicity can be advanced. We report here on the results with a thermodynamically stable chelate, 1,4,7-tris(carboxymethyl)-10-(2-aminoethyl)-1,4,7,10-tetraazacyclododecane (DO3A-EA) and anti-EGFr (ior egf/r3) conjugate to develop immunospecifc imaging agent. Conjugation and labelling with anti-EGFr was performed using standard procedure and subjected to purification on size exclusion chromatography. The conjugated antibodies were labeled with a specific activity 20-30 mCi/mg of protein. Labeling efficiencies were measured by ascending paper chromatography on ITLC-SG strips. Radiolabeling of the immunoconjugate was found to be 98.5 ± 0.30%. 99mTc-DO3A-EA-EGFr conjugate was studied in athymic mice bearing U-87MG, MDA-MB-468 tumors following intravenous injection. Pharmacokinetic and biodistribution studies confirmed long circulation times (t1/2(fast)= 45 min and t1/2(slow)=4 hours 40 min) and efficient accumulation in tumors. Biodistribution studies in athymic mice grafted with U-87MG human glioblastoma multiforme and Hela human cervical carcinoma tumors revealed significant localization of 99mTc-labeled antibodies conjugate in tumors and reduced accumulation in normal organs. This new chelating agent is promising for immunoscintigraphy since good tumour-to-normal organ contrast could be demonstrated. These properties can be exploited for immunospecifc contrast agents in nuclear medicine and SPECT imaging.
CONJUGATED ANTIBODY METHODS provide the most direct and comprehensive route to the identification of antibody-targetable differences between tumor cells and nontumor cells. Conjugated monoclonal antibodies (mAbs) have additional mechanisms of action related to the conjugate. 1 Immunoconjugates to radioisotopes were extensively investigated initially as tumor detection 2 and imaging agents 3 and later as agents for delivering cytotoxic radiation to tumors. 4 A variety of conjugates are being developed to allow mAbs to serve as vehicles for tumor targeting of radioisotopes and chemotherapeutic drugs.5–8
Tumor targeting with radioisotope-labeled antibodies is being used for cancer radioimmunotherapy (with 90Y3+ as a β-emitter), 9 cancer diagnosis (with mIn3+ and 67Ga3+ as a γ-emitter),10–12 and in vivo magnetic resonance imaging (MRI) (with Gd3+ as a paramagnetic contrast agent). 12 Central to the success of such approaches is the development of bifunctional chelating agents containing both a reactive functionality for covalent attachment to the antibody and a strong binding group capable of forming rapidly and selectively an inert complex with the radionuclide.9,13–20
Radiolabeled antibodies can be detected easily by external scintigraphy; thus, it is noteworthy to mention the additional application of radiolabeled antibodies for imaging. 21 The demonstration of cancer targeting with a radiolabeled antibody fragment to carcinoembryonic antigen resulted in the development of radiolabeled antibodies for cancer imaging. 22 Antibody-based vascular targeting compounds, especially antibody-cytokine fusions and radiolabeled antibodies, have been proven to be highly efficacious in various preclinical models of cancer.23,24 99mTc and 111In radioconjugates have been commonly used for this application, but with the advent of positron emission tomography (PET), investigators are now beginning to take advantage of this technologically superior imaging system by radiolabeling tumor-associated antibodies with positron emitters.25,26
Antibodies naturally have long serum half-lives. For immunotherapy, this property is helpful because the antibody is maintained in the body fluids, where it can continually interact with its target.27,28 For other targeting strategies, most notably with radioconjugates, to reduce the radiation damage to normal organs and tissues, radioimmunotherapy under physiologic conditions requires formation of a stable and inert complex of the chelate-antibody conjugate. The bifunctional chelating approach was used to radiolabel monoclonal antibody. For an effectual monoclonal antibody–chelate complex, the binding affinity should not be compromised with unaltered distribution and catabolism of the monoclonal antibody, and the bifunctional chelating agent should have a high stability constant with the radiometal's ion to avoid unacceptable toxicity and reduced efficacy.29,30 To evade demetalation of radionuclides with appropriate radiophysical properties, DO3A-EA (1,4,7-tris(carboxymethyl)-10-(2-aminoethyl)-1,4,7,10-tetraazacyclododecane), a bifunctional chelate, was used to increase the stability of the immunoconjugate. The targeted pathway for tumor detection is the presence of selective growth factor receptors (eg, HER2, epidermal growth factor receptor [EGFR], cyclooxygenase), folate receptors, and an amino acid transporter system, among many others.
EGFR is a transmembrane with an intracellular tyrosine kinase domain, which is overexpressed in the cells of a wide range of solid tumors.31,32 The EGFR is the prototypical member of the class I (ErbB, HER) family of receptor tyrosine kinases, which includes EGFR, HER2 (ErbB2), HER3 (ErbB3), and HER4 (ErbB4). As a target antigen, EGFR is a readily accessible cell surface receptor 33 and, when overexpressed, provides a basis for selective antibody-based targeting of tumor cells. 34 Modified antibody specifically binds to the external domain of the receptor with an affinity comparable to the natural ligand and inhibits epidermal growth factor–induced activation of receptor protein kinase and cell proliferation.35,36
We report here on the results with a bifunctional chelate, DO3A-EA conjugate, with anti-EGFR in the quest to develop an immunospecific imaging agent.
Materials and Methods
Chemicals
Acetonitrile, methanol (high-performance liquid chromatography [HPLC] grade) was obtained from E. Merck Germany Ltd., L-phenylalanine, stannous chloride dihydrated (SnCl2.2H2O), trifluoroacetic acid were purchased from Sigma-Aldrich Co (Germany). N-Hydroxysulfosuccinimide (sulfo-NHS) and 1-ethyl-3-[3-dimethylaminopropyl] carbodiimide hydrochloride (EDC) were obtained from Fluka (Germany). DO3A-EA was synthesized as reported previously. 6 99mTc was procured from the Regional Center for Radiopharmaceuticals (Northern Region), Board of Radiation and Isotope Technology, Department of Atomic Energy, India.
Cell Culture
Monolayer cultures of human malignant glioma cells, U-87MG (obtained from National Institute of Mental Health and Neuro Sciences, and the human cervical cell line, HeLa, were maintained at 37°C in a humidified CO2 incubator (5% CO2, 95% air) in Dulbecco's Modified Eagle's Medium (Sigma-Aldrich) supplemented with 10% fetal calf serum (Biological Industries, Israel), 50 U/mL penicillin, 50 μg/mL streptomycin sulfate, and 2 μg/mL nystatin. Cells were routinely subcultured twice a week using 0.05% trypsin (Sigma) in 0.02% ethylenediaminetetraacetic acid (EDTA).
Monoclonal Antibody
Anti-EGFR antibody (ior egf/r3, a gift from the Center of Molecular Immunology) and antirabbit IgG–fluorescein isothiocyanate (FITC) were procured from Invitrogen.
Instrumentation
The 1H nuclear magnetic resonance spectra were determined using a nuclear magnetic resonance spectrometer (Bruker Spectro spin DPS 400 MHz). Mass spectrum was recorded on an Agilent 1100 mass spectrometer using m-nitrobenzyl alcohol as the matrix in electrospray ionization-positive mode. HPLC analyses were performed on a Waters Chromatograph efficient with 600 coupled to a Waters 2487 photodiode array ultraviolet (UV) detector. The C-18 RP Beckman column (5 μ, 1 mm × 1.25 cm), Shimadzu ODS-AQ (5 μ, 4.6 mm × 250 mm) column, and Sepharose SW 3000 column were used, applying the elution system described herein. The receptor binding data were analyzed using the iterative curve-fitting program EQUILIBRATE software from GraphPad (La Jolla, CA). Radioimaging and biodistribution studies were done using a planner gamma camera hawk eye and single-well-type γ-scintillation counter from Capintec.
Animal Models
Animal protocols were approved by the institutional animal ethics committee. New Zealand rabbits, 2 to 3 kg, and BALB/c mice, 25 to 28 g, were used for blood clearance, imaging, and biodistribution. Mice and rabbits were housed under conditions of controlled temperature of 22 ± 2°C and fed a normal diet. BALB/c mice were inoculated subcutaneously with 0.1 mL of cell suspension (1.5 × 107 Ehrlich ascites tumor [EAT]/U-87MG cells) into the right thigh under sterile conditions.
Immunoconjugate
For coupling, 25 μL of 20 mM of chelate solution was added to 300 μL of a solution containing 1 mg of antibodies in 0.1 M sodium phosphate buffer, pH 7.4. Saturated trisodium phosphate solution (40 μL) was added to bring the pH to approximately 8.5. The reaction mixture was incubated at 37°C for 14 hours and then subjected to centrifuged column gel chromatography, which removed the unreacted chelate and changed the buffer to 0.1 M sodium acetate, pH 5.5. UV absorbance at 280 nm for the centrifuged column effluent was used to determine the concentration of protein. 37 Immunoconjugate formation was assessed by size exclusion HPLC (Sepharose SW 3000 column).
Radiochemical Synthesis of DO3A-EA-ior egf/r3
The immunoconjugates of anti-EGFR were labeled with 99mTc using stannous chloride (30 μL; 1 mg dissolved in N2 purged in 1 mL 10% acetic acid) followed by the addition of freshly eluted (< 1 hour old) 99mtechnetium pertechnetate (5 mCi). The final pH was adjusted to 7 to 8. The labeling solution was incubated at room temperature (22°C) for 20 minutes. After incubation radioimmuno-conjugate was eluted on an exclusion-diffusion column, purification was performed using G-50 Sephadex (in 0.1 M NaOAc, pH 7.0, buffer). Quality control was done at 30, 60, and 120 minutes. The radiochemical purity of radio-immunoconjugates was performed using instant thin-layer chromatography-silica gel (ITLC-SG; Gelman Sciences, Ann Arbor, MI), and purified antibodies were analyzed on HPLC using the SW 3000 size exclusion column.
DTPA Challenge
Radiolabeled modified mAbs and native mAb were incubated at different concentrations in 10 mM diethylene-triaminepentaacetic acid (DTPA) and maintained at 37°C up to 24 hours. Periodically, samples were removed and spotted on a 10 cm ITLC-SG strip and developed in 0.9% NaCl. Once the solvent front had reached the end of the strip, it was removed from the solvent and cut at a retention factor (Rf) of 0.5. The two portions were assayed for radioactivity, and the amount of intact chelate was determined.
Cobalt Binding Assay
A 57Co assay was used to obtain the bound chelate concentration; 1 nmol of conjugated antibody, 2 nmol of 59Co, and a tracer of 57Co were mixed and incubated at pH 5.5 at room temperature for 30 minutes. An aliquot was applied to thin-layer chromatography with ITLC-SG (Gelman Sciences) with ammonium acetate/CH3OH (1:1 v/v) solution as an eluant. Free cobalt migrated to Rf 5 1.0, whereas labeled cobalt with immunoconjugate remained at Rf = 0.17–19
Determination of Average Number of Chelate Conjugated to an Antibody Molecule
The average number of chelate to antibody ratio was quantitated by liquid scintillation counting of the chelate, and measurement of protein concentration was determined using the bicinchoninic acid (BCA) protein assay. A measured volume was counted, and the radioactivity associated with the protein was corrected for specific activity of the chelate and volume, thus yielding the amount of chelate present. The DOTA/Ab ratio in prepared conjugates was calculated as the ratio of DOTA concentration (μM) to IgG concentration (μM).
Immunoreactivity
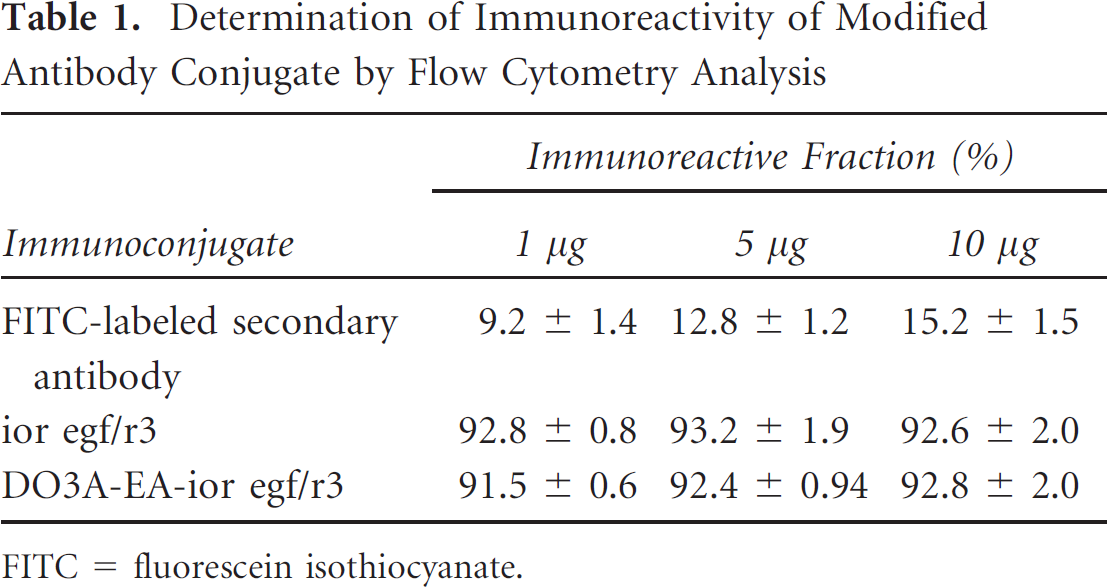
The reactivity of the immunoconjugate to the U-87MG cell line was determined by flow cytometry analysis. Twenty-four hours before analysis, 0.1 × 106 cells/petri dish (U-87MG) were plated. Cells were washed twice with phosphate-buffered saline containing 0.05% Tween and 1% bovine serum albumin (BSA). Monolayer cultures of the U-87MG cell line were incubated with 1, 5, and 10 mg of unmodified antibody, modified DO3E-EA-ior egf/R3. Unmodified mAb ior egf/r3 was included as a positive control, and FITC-labeled secondary antibody was used as a negative control (Table 1).
Determination of Immunoreactivity of Modified Antibody Conjugate by Flow Cytometry Analysis
FITC = fluorescein isothiocyanate.
Human Serum Stability Evaluation
Human serum was prepared by allowing blood collected from healthy volunteers to clot for 1 hour at 37°C in a humidified incubator maintained at 5% carbon dioxide, 95% air. Then the samples were centrifuged at 400g and the serum was filtered through a 0.22-micron syringe filter into sterile plastic culture tubes. The radiolabeled antibody conjugate and antibody serum samples were immediately placed in a CO2 chamber incubated at 37°C and then analyzed to check for any dissociation of the complex. The percentage of free pertechnetate at a particular time point that was estimated using acetone and pyridine, acetic acid, and water (3:5:1.5) as a mobile phase represented the percent dissociation of the complex at that particular time point in serum.
Blood Kinetics
A normal rabbit, weighing about 2 to 2.5 kg, was injected with 10 MBq of 99mTc-DO3A-EA-ior egf/r3 intravenously through the dorsal ear vein. Blood was withdrawn from the other ear vein at different time intervals, from 4 minutes to 24 hours. The persistence of activity in the circulation was calculated, assuming total blood volume as 7% of the body weight.
Receptor Binding Studies
Exponentially growing cells, 0.1 × 106 cells/petri dish (U-87MG and HeLa), were plated at a uniform cell density and incubated overnight. Monolayer cultures (U-87MG and HeLa) of the cell lines were washed twice for 2 minutes with ice-cold binding buffer (25 mM HEPES, 10 mM MgCl2, and 1% BSA). The cell line cultures were then incubated for 40 minutes with labeled antibody (0.01 nM to 1 μM) in the absence and presence of the 100-fold excess unlabeled antibody for estimation of total and nonspecific binding, respectively. Specific binding was obtained by subtracting nonspecific binding from total binding. At the end of each experiment, the cells were washed with ice-cold binding buffer three times for 3 minutes. The cells were lysed with 200 μL of lysis buffer. The cell-associated radioactivity was determined by γ-scintillation counting. Scatchard plot analysis was done using Equilibrate software.
Biodistribution Studies in Mice
Normal BALB/c mice were inoculated with EAT, and athymic mice were xenografted with U-87MG human glioblastoma multiforme in the right flank. Animal handling and experimentation were carried out according to the guidelines of the institutional animal ethics committee. For biodistribution studies, BALB/c mice, bearing tumors of the EAT cell line and complex of the 99mTc-labeled antibody conjugate, were used as the animal model. An equal dose of 0.4 MBq of 99mTc-DO3A-EA-ior egf/r3 in a volume of 100 μL was injected through the tail vein of each mouse. Mice were dissected at 1, 4, and 24 hours postinjection; different tissues were taken out, weighed, and counted in a μ-counter calibrated for 99mTc energy. The actual amount of radioactivity administered to each animal was calculated by subtracting the activity left in the tail from the activity injected. Radioactivity accumulated in each organ was expressed as the percent administered dose per gram (%ID/g) of tissue. Total volume of the blood was calculated as 7% of the body weight.
Scintigraphy in Tumor-Bearing Nude Mice
Tumor imaging was performed in tumor-bearing nude mice implanted with the U-87MG cell line, injected with 20 mCi of the 99mTc-labeled conjugate. Images were obtained at different time intervals, from 15 minutes to 24 hours postinjection of the 99mTc-labeled conjugate.
Results
Antibody Conjugation
There are several approaches to conjugate DO3A-EA with mAb. By using EDC and sulfo-NHS for activating the carboxylic group of antibody, we conjugated DO3A-EA with free NH2 at pH 9. 19 An average of 4.02 ± 1.04 DO3A-EA molecules could be randomly conjugated to ior egf/r3, as estimated by cobalt binding assay, 19 with negligible loss of immunoreactivity. Similar ratios were obtained (5 ± 0.20) when calculated as the ratio of DOTA concentration (μM) to IgG concentration (μM) through protein estimation.
Immunoreactivity
The immunoreactivity of the mAb conjugate showed high and specific binding ability to target cells, the U-87MG human tumor cell line at 5 mg of the antibody conjugate shown in Table 1, which was revealed by flow cytometry on the U-87MG cell line. Biologic activity of DO3A-EA-ior egf/r3 immunoconjugate on the U-87MG cancer cell line was determined by indirect immunofluorescence analysis. The biologic activity and immunoreactivity remained unaltered after modification. The percentage of positive cells at the concentrations of 1.0, 5.0, and 10.0 μg/mL was higher than 90% for the immunoconjugate. The EGFR overexpressed on U-87MG human cancer cells was recognized by the modified antibodies, and the immunoconjugate after modification kept the biologic activity constant.
Quality Control
ITLC of 99mTc-radiolabeled antibody before purification by a Sephadex column was 92.4%, and bound activity in purified immunoconjugate was 99.8%. The 99mTc loading of the mAb conjugate was achieved rapidly (< 1 hour) under mild conditions (pH 7 at 25°C), thereby avoiding degradation of the protein. HPLC of the 99mTc-labeled conjugate showed a single peak at 25 minutes. Radio-labeled immunoconjugate was challenged with 10 mM DTPA to test the stability of the radiolabeled antibodies. Approximately more than 97% of the radioactivity remained associated with the antibody after a 2-hour challenge at 37°C with DTPA. Challenge studies demonstrated 3% of transcomplexation of 99mTc to 10 mM DTPA after 2 hours of incubation at 37°C for modified antibody (Figure 1).

In vitro stability study of 99mTc-labeled anti-EGFR antibody ior egf/r3 () and DO3A-EA-ior egf/r3 conjugate (◆) challenged with DTPA.
Radiolabeled Immunoconjugate Stability Evaluation
In vitro stability as assessed by size exclusion HPLC (Sepharose SW 3000 column) showed that the metal ion was intact under physiologic conditions. Unbound 99mTc was less than 2.3% in 6 hours. Labeling efficiency was measured by ascending paper chromatography on ITLC-SG strips. Preliminary complexation with 99mTc was found to be sufficiently stable under physiologic conditions (Figure 2). The DO3A-EA-ior egf/r3–modified antibodies were labeled with a specific activity of 20 to 30 mCi/mg of protein. Radiolabeling in 0.9% saline was found to be 98.6% (± 0.02). ITLC-SG results in acetone showed that 2 to 3% or less free pertechnetate ran at the solvent front (Rf = 0.8–1.0), indicating that pertechnetate was reduced almost quantitatively. Radiocolloids were less than 2.0% in all cases. Furthermore, the radiolabeled immunoconjugate was analyzed on an Omniscan EZ-TLC scanner, which showed peak at Rf = 8.7 (3%) for free technetium and Rf = 4.6 (97%), confirming radioactivity associated with the immunoconjugate.

In vitro human serum stability study of 99mTc-DO3A-EA-ior egf/r3 (▴) and 99mTc-ior egf/r3 (◆) under physiologic conditions.
Blood Kinetics
In vivo clearance in rabbits revealed a rapid washout of the conjugate from the circulation as only 10.8% of injected activity remained in the circulation at 30 minutes. The clearance followed a slow pattern, and at 24 hours, approximately 1.12% activity persisted in the blood (Figure 3). The biologic half-life was found to be T½ Fast = 45 minutes; T½ Slow = 4 hours 40 minutes.

Pharmacokinetic blood clearance of 99mTc-DO3A-EA-ior egf/r3.
Receptor Binding Studies
Ligands of high-affinity EGFR, including conjugates of radiopharmaceuticals and proteins, all display dose-dependent cell association. As shown in Figure 4, the cell uptake of 99mTc-DO3A-EA-ior egf/r3 was also found to be dependent on the extracellular concentration. Scatchard plot analysis revealed affinity of the DO3A-EA-ior egf/r3 on tumor cell lines. Kd was found to be 2.48 and 5.2 nM in U-87MG and HeLa cell lines, respectively (Figure 5).
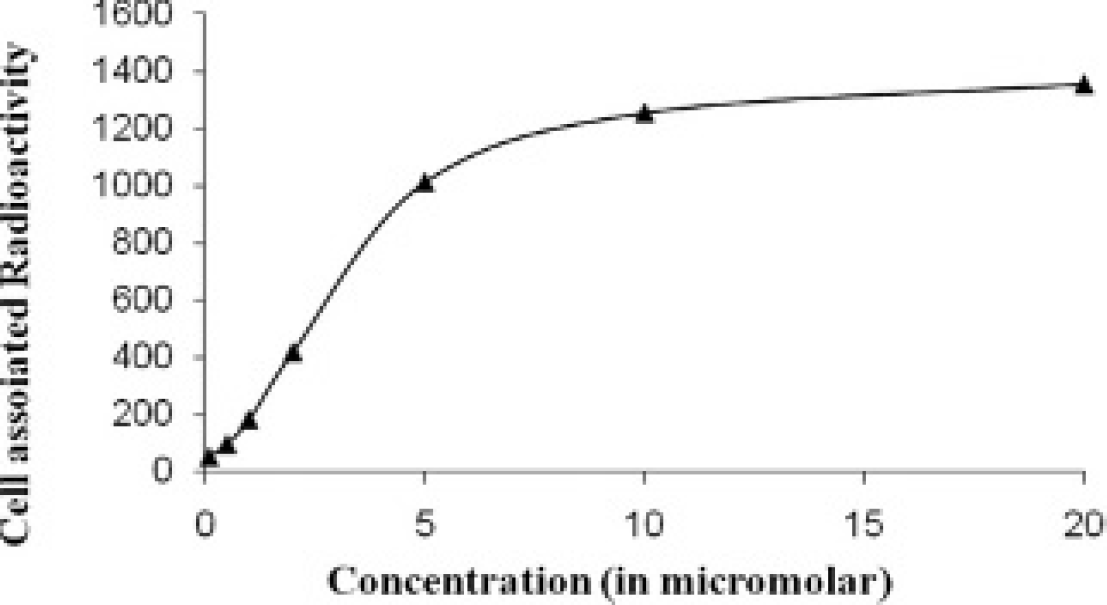
Concentration-dependent association of 99mTc-DO3A-EA-ior egf/r3 to U-87 human glioma cell line with increasing concentrations (0.01 nM to 10 μM) for 2 hours at 37°C. Nonspecific binding (blank) was determined in the presence of a 100-fold excess of unlabeled antibody. Each point represents the mean of triplicate determinations after subtracting the blank value.

Scatchard plot of the specific binding data to the ratio of bound to free with increasing concentrations (0.01 nM to 1 μM) of U-87MG (◆) and HeLa (▴) cell lines.
Scintigraphy
Localization of 99mTc-labeled immunoconjugate in a nude mice implanted with a U-87MG cell line tumor over time, as determined by gamma camera imaging, is presented in Figure 6 at different time intervals: 15 minutes, 30 minutes, 1 hour, and 2 hours. The mice are depicted at the beginning of accumulation of activity in the tumor at 30 minutes, which reached a maximum at 2 hours (see Figure 6).

Whole-body scintigraphic image of radiolabeled antibody-chelate conjugate in nude mice grafted with a U-87MG cell tumor in the right thigh.
Biodistribution
Biodistribution studies in BALB/c mice implanted with EAT cell line tumors revealed that the major portion of the complex is excreted by renal routes because at 1 hour, accumulation in the kidneys was 14.0%/g. The 99mTc-labeled biodistribution study results are presented in Table 2. The tumor accumulation of labeled antibody conjugate was 3.2 ± 0.2%ID/g at 1 hour and 5.4 ± 1.02%ID/g at 4 hours postinjection. As the background activity gradually cleared from the body, there was an increase in target to nontarget ratios. Biodistribution studies in athymic mice conducted to quantify immunolocalization of the immunoconjugate of DO3A-EA-ior egf/r3 on U-87MG tumors showed significant accumulation of the radiolabeled immunoconjugate in the tumors 24 hours after injection (Table 2). For 99mTc DO3A-EA-ior egf/r3, tumor uptake was found to be 6.68 ± 0.26% and 9.84 ± 1.1% ID/g of tissue at 4 and 24 hours after administration of the radiopharmaceutica, respectively.
Biodistribution of 99mTc-DO3A-EA-ior egf/r3 in Mice with an EAT Cell Tumor Implant following Intravenous Injection

EAT = Ehrlich ascites tumor.
The elimination of radioactivity from plasma after administration of 99mTc-labeled mAb conjugates occurs primarily by metabolism of the immune conjugate and elimination of the radiolabeled mAb by renal and hepatobiliary excretion. Insignificant accumulation of technetium activity in the small intestine up to 24 hours postinjection confirmed in vivo stability. Animal studies have shown that DO3A-EA-ior egf/r3-labeled antibodies can be used for tumor imaging, with nonspecific liver retention being greatly reduced.
Conclusion
The use of nuclear medicine, in both diagnostics and therapeutics, relies implicitly on accurate delivery to the disease location. Within this field, the application of antibodies as delivery agents for the radioisotopes holds much promise. Radioisotope can be stably conjugated to an antibody without adversely affecting its biologic activity, which has led to the widespread evaluation of radioisotopes in both imaging (immunoscintigraphy) and radioimmunotherapy. Radiolabeled mAbs have been developed as alternative immunoconjugates for the delivery of radionuclide to target cells for the diagnosis and therapy of cancer.3,12,18,20–26,37
The mAbs against abundantly expressed antigens have proved to be effective tracer molecules for radioimmuno-diagnosis. The mAb ior egf/r3 recognizes EGFR and inhibits the binding to its ligand. This antigen has been used successfully as a target for imaging and therapy because of its overexpression in tumors of epithelial origin.38–41 Although normal cells also express EGFR, the elevated number of receptors on tumor cells confers a degree of targeting specificity. Therefore, the tumor cells can proportionately bind more amounts of radiolabeled mAbs than can the normal cells. In the present study, immunoconjugates of the anti-EGFR antibodies have been generated using DO3A-EA as a multifunctional chelate. The use of this chelating agent to prepare radioimmuno-conjugate made rapid clearance from blood circulation and normal organs compared to directly radiolabeled antibodies, as previously described. 42 DO3A-EA conjugation to the antibody ior egf/r3 does not interfere with the antibody's immunoreactivity and has a very high metal stability constant with many radionuclides of interest. With extended research, the in vitro and in vivo results suggest that DO3A-EA-antibody conjugate may be clinically useful as a noninvasive radiodiagnostic imaging agent for the detection of EGFR-positive human cancers.
Footnotes
Acknowledgment
Financial disclosure of authors and reviewers: None reported.
