Abstract
Superparamagnetic iron oxide particles are used as potent contrast agents in magnetic resonance imaging. In histology, these particles are frequently visualized by Prussian blue iron staining of aldehyde-fixed, paraffin-embedded tissues. Recently, zinc salt-based fixative was shown to preserve enzyme activity in paraffin-embedded tissues. In this study, we demonstrate that zinc fixation allows combining in situ zymography with fluorescence immunohistochemistry (IHC) and iron staining for advanced biologic investigation of iron oxide particle accumulation. Very small iron oxide particles, developed for magnetic resonance angiography, were applied intravenously to BALB/c nude mice. After 3 hours, spleens were explanted and subjected to zinc fixation and paraffin embedding. Cut tissue sections were further processed to in situ zymography, IHC, and Prussian blue staining procedures. The combination of in situ zymography as well as IHC with subsequent Prussian blue iron staining on zinc-fixed paraffin-embedded tissues resulted in excellent histologic images of enzyme activity, protease distribution, and iron oxide particle accumulation. The combination of all three stains on a single section allowed direct comparison with only moderate degradation of fluorescein isothiocyanate–labeled substrate. This protocol is useful for investigating the biologic environment of accumulating iron oxide particles, with excellent preservation of morphology.
SUPERPARAMAGNETIC IRON OXIDE NANOPARTICLES were developed as sensitive probes for magnetic resonance imaging (MRI). The majority of them are based on steric stabilization with polymer coats such as dextran and polyethylene glycol. In contrast, very small iron oxide particles (VSOPs) are stabilized electrostatically by an anionic citrate acid shell and were first introduced for magnetic resonance angiography.1,2 Recently, a variety of other MRI probes with specific molecular targeting capabilities were designed on the basis of VSOPs. 3
Prussian blue iron staining is the most widely used method for evaluating iron oxide particle accumulation in histologic sections. For studies of the mechanisms of particle accumulation in target tissues, it is highly desirable to have compatible histologic methods that allow sophisticated biologic analysis.
In the present study, we chose to supplement particle staining with the evaluation of matrix metalloproteinases (MMPs) by immunohistochemistry (IHC) and in situ zymography. MMPs play a crucial role in normal physiology by digesting extracellular matrix proteins, for example, in tissue development and homeostasis, as well as in many pathologic conditions, for example, tissue repair, cancer, and chronic inflammatory vascular and joint diseases.4–7 Most MMPs are secreted as latent proenzymes and become activated on enzymatic cleavage. Two prominent members of this family, the gelatinases MMP-2 and MMP-9, are highly expressed in several types of human cancers. They are also produced in relatively large amounts by tissue macrophages, controlling disruption and removal and remodeling of connective tissues, and granulocytes. Smaller amounts are released by stimulated T and B cells. 8
In situ zymography is a method for assessing enzymatic activity and is usually done in fresh frozen tissues.9,10 Gelatin in situ zymography localizes MMP-2, MMP-9, and other potential gelatin-degrading enzymes. Recently, this method has been demonstrated to be feasible in zinc-fixed, paraffin-embedded tissues. 11 Compared to fresh frozen sections, this fixation method, unlike the widely used aldehydes, not only offers superior tissue morphology but also preserves the antigens for immunohistologic staining without cross-linking of reactive functional groups in proteins. How this zinc salt–based fixative functions is still unclear, but it appears to ensure good preservation of tissue morphology and immunoreactivity of processing-sensitive cell markers.11–13
In the present report, we show that zinc fixation of paraffin-embedded splenic tissue allows sensitive in situ zymography and IHC analysis with subsequent iron staining. This method may be useful for thoroughly characterizing other target tissues, such as atherosclerotic plaques, liver, or tumor. For precise signal colocalization, it is even possible to do triple staining on a single slice.
Materials and Methods
VSOP Administration
BALB/c athymic nude mice (Charles River, Wilmington, MA) received a single injection of VSOPs14,15 (30 mmol/kg Fe, hydrodynamic size 10 nm, T1 relaxivity 24 mM−1s−1,T2 relaxivity 64 mM−1s−1) under inhalation anesthesia (isoflurane 1–2%). Three hours after administration, animals were sacrificed, and the spleens were processed to fixation.
Zinc Fixation and Paraffin Embedding
Up to 3 mm thick spleen tissue pieces of nude mice were fixed in zinc fixative (BD Pharmingen, Heidelberg, Germany) for 24 hours at room temperature, washed in distilled water for 1 hour, and then dehydrated and embedded in paraffin using the following standard procedure: 1 hour 70% ethanol; 1 hour 80% ethanol; 2 × 1 hour 96% ethanol; 3 × 1 hour 100% ethanol; 1 hour 90% xylene + 10% ethanol; 3 × 1 hour 100% xylene; 2 × 1 hour melted paraffin at 60°C; and embedding into histology cassette at 60°C. Cooled embedded tissues were cut into 6 μm sections and placed on Superfrost Plus slides (Thermo Fisher Scientific, Schwerte, Germany). Sections were dried overnight at 37°C and further used for IHC and in situ zymography experiments.
Immunohistochemistry
Primary polyclonal rabbit antibodies against human and mouse MMP-2 (Abcam, Cambridge, UK) and MMP-9 (Abcam) at 1:500 dilution were used in one mix. For double staining of macrophages and gelatinases, this mix was supplemented with monoclonal rat antibodies against mouse peritoneal macrophages (Abcam, RM0029-11H3). Secondary Cy5-antirabbit antibody (Abcam) and fluorescein isothiocyanate (FITC)-antirat antibody (Abcam) at 1:300 dilution were used for the labeling of primary targets. Paraffin-embedded slices were heated for 1 hour at 58°C, deparaffinized in xylene (2 × 5 minutes), rehydrated in four graded alcohol baths (100%, 96%, 80%, and 70% ethanol, each 5 minutes), and then brought to distilled water. Next, the sections were washed for 5 minutes in phosphate-buffered saline (PBS) and incubated with blocking solution (10% fetal calf serum, 0.5% bovine serum albumin, 0.025% Triton in PBS, pH = 7.4) for 45 minutes to reduce unspecific staining. After that, mixed primary antibodies, diluted in blocking solution, were applied for 1 hour at room temperature (for staining of one section) or for 24 hours at 4°C (for staining of parallel sections). Following this step, slices were washed in PBS three times and incubated with secondary antibody for 1 hour at room temperature. After rinsing two times in PBS, slices were incubated with Hoechst solution (Sigma, Seelze, Germany) (1 μg/mL in PBS) for 45 minutes for nuclear labeling, rinsed in water, and mounted with Fluoromount (Sigma, Seelze, Germany). For negative control, PBS replaced the primary antibodies.
In Situ Zymography
The gelatinolytic activity of MMP-2 and MMP-9 in tissues was examined using in situ zymography, as recently described. 11 Briefly, tissue sections were prepared as described, heated at 58°C for 1 hour before the procedure, deparaffinized in xylene, and rehydrated in four graded alcohol baths (as described for IHC). One milligram DQ gelatin (Invitrogen, Karlsruhe, Germany) was dissolved in 1 mL Milli-Q water and further diluted 1:50 in a reaction buffer containing 50 mM Tris-HCl, 150 mM NaCl, and 5 mM CaCl2 (pH 7.6). This substrate for gelatinases was dropped at tissue sections, covered with glass slides, and incubated in a dark humidity chamber at 37°C for 2 hours. For negative control, the sections were incubated at −20°C. Then cover glasses were gently removed by immersing sections in Milli-Q water; the sections were washed again with Milli-Q water and fixed in 4% buffered Paraformaldehyde (PFA) solution for 10 minutes in the dark. After that, for staining on parallel sections, slices were washed two times in PBS, counterstained with Hoechst solution (1 μg/mL in PBS) for 45 minutes, and mounted with Fluoromount. For staining on one section, slices were processed directly after fixation and washing to the IHC procedure.
Prussian Blue Iron Staining
Iron deposits were identified using Prussian blue (Perl) staining. Tissue sections were first processed to IHC staining, in situ zymography, or both of these methods. After documentation by fluorescence microscopy, the cover glass and water-soluble mounting medium were removed by keeping slices in water for 72 hours. Following that, tissue sections were immersed in 2% potassium ferricyanide and 2% HCl solution for 25 minutes at room temperature, washed twice in distilled water, counterstained with 0.1% nuclear fast red solution (Merck, Darmstadt, Germany) in 5% aluminum sulfate (Sigma) for 20 seconds, dehydrated in four graded alcohol baths (70%, 80%, 96%, and 100% ethanol, each 2 minutes), cleared in xylene, and mounted with Entellan (Merck).
Microscopy
Pictures were obtained using an Axio Observer Z1 microscope (Carl Zeiss, Jena, Germany) equipped with an ApoTome imaging device. Sections were analyzed after in situ zymography and IHC procedures, performed individually or combined. After that, sections were immersed in distilled water for 72 hours to remove cover glasses and mounting medium; then staining was performed, and the sections were analyzed again.
Results
First, we analyzed iron distribution in mouse spleen on two parallel sections. One section was subjected to IHC staining and the second section to in situ zymography (Figure 1). The red pulp of the spleen in particular is rich in MMP-secreting macrophages, which are also the primary target for VSOP uptake in this organ.
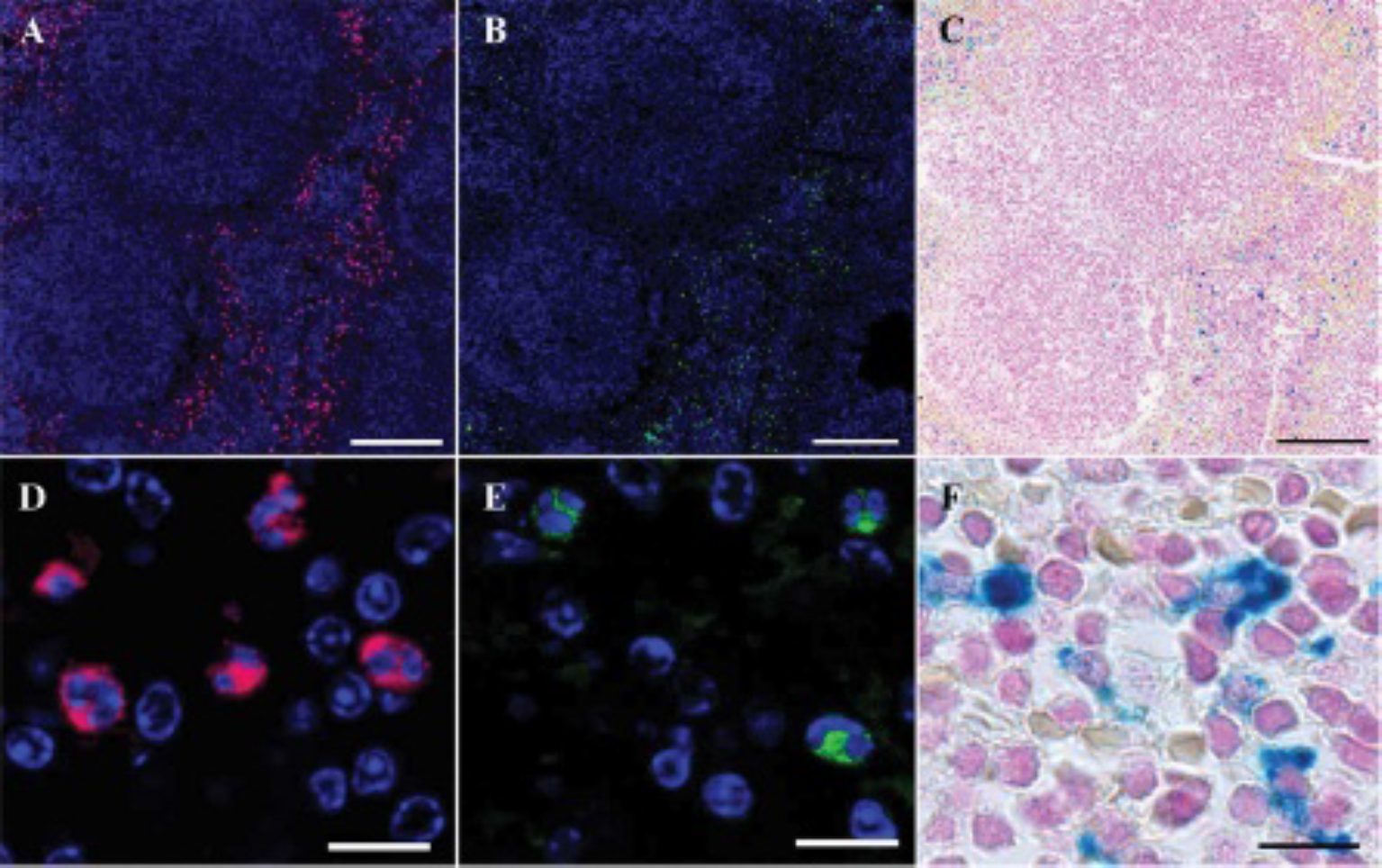
Immunohistologic MMP-2/MMP-9 staining, in situ zymography, and Prussian blue iron staining on zinc-fixed parallel sections of spleen after very small iron oxide particle administration. A and D, Combined MMP-2/MMP-9 antibody staining shows gelatinase-positive cells (secondary Cy5-fluorescence signal) in the red pulp and marginal zone. Note the kidney-shaped form of the nuclei (Hoechst nuclear staining), which is typical for macrophages. B and E, In situ zymography (DQ-gelatin-FITC) of a parallel slice demonstrating sites of gelatinase activity (MMP-2/MMP-9). C and F, Prussian blue iron staining of the same slice as for in situ zymography (B, E). Iron staining was done after microscopy of zymography results. Size bars: A–C, 200 μm; D–F, 10 μm.
On the first section, the presence of gelatinases in tissue was shown by IHC staining using mixed primary antibodies (against MMP-2 and −9) and secondary Cy5-fluorescence antibodies. Many cells with strong MMP-2/MMP-9 signal were found in the red pulp and marginal zone of the white pulp (see Figure 1, A and D), typically rich in macrophages, which play a major role in immune response, producing large amounts of MMP-9 on activation. 16 There is evidence that MMP-9 can accumulate on the cell surface, so it possibly acts on membrane proteins. 17 It should be mentioned that nude mice are deficient in T lymphocytes, but B-cell follicles, marginal zone, and red pulp are relatively normal. 18
On the next parallel slide, enzymatic activity of both gelatinases was examined using in situ zymography. Highly defined FITC fluorescence patterns of digested DQ gelatin in spleen sections correspond to double immunostaining for MMP-2 and MMP-9 (see Figure 1, A and B). Some of the cells expressing MMP-2/-9 (eg, see the upper left corner of Figure 1A) demonstrate only low or no gelatinase activity (see Figure 1B). Single-cell analysis demonstrates detailed morphology and precise spatial localization (see Figure 1E) matching IHC staining typical of macrophages (see Figure 1D). After microscopic documentation, the sections were processed for iron staining (see Figure 1, C and F). Clearly visible iron deposits ensure the compatibility of the Prussian blue iron staining technique with zinc-fixed tissue, enabling detection of enzymatic activity and iron oxide particles on a single slice. Subsequent iron staining after IHC labeling yielded a similar result (not shown).
Figure 2 (A, D, and G) reveals that almost all cells producing gelatinases are positive for macrophage antibodies. Only a few macrophages do not express MMP-2/MMP-9 (Figure 2, B, E, and H). Some of the cells expressing gelatinases do not double-stain with the macrophage marker (Figure 2, C, F, and I). These cells are presumably neutrophilic granulocytes.

Immunohistologic double-staining of macrophages and MMP-2/MMP-9 on zinc-fixed spleen tissue. A–C, Merged image of spleen section, double-stained for macrophages (FITC, green; also G–I) and MMP-2/MMP-9 (Cy5, magenta, D–F). Almost all cells are positive for both signals (yellow), with only some macrophages not expressing gelatinases (B, E, H, arrow). Some other MMP-2/MMP-9-positive cells are not marked as macrophages (C, F, I, arrow), presumably neutrophilic granulocytes. Size bars: A, D, G, 50 μm; others, 5 μm.
Second, we performed all three staining techniques on a single tissue section. After in situ zymography, IHC staining was performed on the tissue section. Fluorescence analysis (Figure 3) shows a partially unspecific zymography signal when examined at low magnification (see Figure 3B) in comparison with the individual procedure (see Figure 1B). IHC MMP-2/MMP-9 localization seems not to be affected (see Figure 1A and Figure 3A). For single-cell investigation, at higher magnification, combining these two methods provided more defined results after adjusting for the relatively strong background. Figure 3 specifically demonstrates cells positive for antibody-labeled gelatinases (D and F), which are active, as confirmed by in situ zymography (E and F). Further iron staining of the same slice also yielded strong signals, confirming the feasibility of performing all three staining techniques under investigation on a single tissue section.

Immunohistologic MMP-2/MMP-9 staining, in situ zymography, and Prussian blue iron staining on a single zinc-fixed spleen section after very small iron oxide particle administration. A, D, and G, Combined MMP-2/MMP-9 antibody staining (Cy5 channel, magenta), performed after the in situ zymography procedure, demonstrates gelatinase-positive cells in the red pulp and marginal zone of the mouse spleen. B and E, In situ zymography (DQ-gelatin-FITC) of the same section demonstrating sites of active gelatinases (MMP-2/MMP-9). B has the same exposure parameters as Figure 1B; note the relatively strong unspecific background and low specific signal at this magnification compared to well-recognizable FITC labeling in high magnification on the single-cell level in E. F, Merged image of D and E. C and H, Prussian blue iron staining of the same slice. Iron staining was done after documentation by fluorescence microscopy of in situ zymography and IHC results. Note in A to C and G to H that MMP-2/MMP-9 signal overlaps mainly with iron staining. Arrows in G and H demonstrate three cell groups, expressing gelatinases and partially or full loaded with iron. Bars: A–C, 200 μm; D–H, 10 μm.
Discussion
When tissue is fixed in widely used neutral-buffered formalin, enzymes are irreversible inactivated, and globular proteins are cross-linked with methylene bridges, resulting in poor immunoreactivity. 19 Enzymatic or heat-induced antigen retrieval,20,21 mostly used to improve the accessibility of antigens in IHC protocols, can cause tissue damage, leading to increased background staining and detachment from the object slide, and is not helpful for fixation-sensitive cell surface markers. Gelatin in situ zymography is usually performed using fresh frozen tissue sections because formalin fixation blocks enzyme activity in tissue. 9 Since Beckstead published a new zinc-based fixative that retains cytohistologic tissue details comparable to formaldehyde and preserves immunoreactivity and genomic deoxyribonucleic acid (DNA), 12 an increasing number of investigators have confirmed these properties of the new fixative.11,13,19,22–25 This alternative to formalin-fixed paraffin tissue embedding and frozen sectioning is trying to gain acceptance in scientific research laboratories and pathology units in hospitals. The fixating mechanism of zinc salts at the molecular level is still under discussion. It has been suggested that the combined effects of the zinc and acetate ions create structural changes or cause mild denaturation of proteins in the specimen, bringing the cell membrane proteins into a more rigid layer. 19
In our study, we chose spleen tissue for microscopic analysis because it contains significant amounts of macrophages, which take up iron oxide particles and express gelatinases. Spleens were cut into thin 3 mm pieces to optimize penetration of fixative into the tissue, since zinc salt fixatives have poor penetration properties and can induce well-fixed edges but not equally fixed interiors. 24 However, there is evidence that this fixative allows normal IHC processing of samples.19,22 Our fluorescence IHC and in situ zymography results, along with Prussian blue iron staining, corroborate this conclusion. Zinc salt–based fixation yields clear, sharp, and well-detectable fluorescent signal using standard IHC procedures. In situ zymography also revealed a clearly defined distribution with very well-preserved morphology. Interestingly, tissue sections processed with both techniques were suitable for subsequent Prussian blue staining and yielded good results, allowing precise observation of the distribution of nanoparticles and the protein of interest, in our case, latent progelatinases and their active forms. Gelatinolytic activity in the spleen (see Figures 1 and 3), predominantly expressed by macrophages (see Figure 2), overlapped mainly with the IHC staining pattern of Zn2+-dependent MMP-2/MMP-9. Some cells positive for MMP-2/MMP-9-expression showed no or weak gelatinolytic activity, suggesting that these cells contain mainly nonactivated MMP proenzymes. These results demonstrate the specificity of the method despite higher Zn2+ concentrations of the zinc fixative.
On the single-cell level, even the combination of IHC, in situ zymography, and iron staining demonstrated well-evaluable signal quality, despite higher background and unspecific signal at lower magnification, probably caused by partially degraded DQ-gelatin after subsequent IHC staining. One crucial hindrance to the use of this fixative in routine hospital procedures is the incomplete inactivation of pathogens. 13
Conclusion
Zinc salt–based fixation provides an excellent method for combining fluorescence IHC, in situ zymography, and subsequent iron staining to obtain high-quality results for research on mechanisms of iron oxide particle accumulation in tissues of interest.
Footnotes
Acknowledgment
All animal experiments were performed in accordance with laboratory animal care guidelines of Charite University Medicine Berlin and the State Office of Health and Social Affairs Berlin (Registered project number G 0246/08). The work was supported by Deutsche Forschungsgemeinschaft (DFG SCH 1416/2-1). We thank Bettina Herwig for language editing.
