Abstract
Nuclear medicine techniques provide potential non-invasive tools for imaging infections and inflammations in the body in a precise way. These techniques are further exploited by the use of radiopharmaceuticals in conjunction with imaging tests such as scintigraphy and positron emission tomography. Improved agents for targeting infection exploit the specific accumulation of radiolabeled compounds to understand the pathophysiologic changes involved in the inflammatory process and correlate them with other chronic illnesses. In the recent past, a wide variety of radiopharmaceuticals have been developed, broadly classified as specific radiopharmaceuticals and nonspecific radiopharmaceuticals. New developments in positron emission (leveraging 18F and 18fluorodeoxyglucose) and heterocyclic/peptide chemistry and radiochemistry are resulting in unique agents with high specific activity. Various approaches to visualizing infection and inflammation are presented in this review, in an integral manner, that give a clear view of the existing radiopharmaceuticals in clinical practice and those under development.
EVER SINCE THEIR INCEPTION, nuclear medicine techniques have played a pivotal role in the visualization, detection, and localization of inflammation and infection. 1 They point to parts in the body where a particular metabolic process is ongoing, leading to elevated uptake of a radiopharmaceutical. In conjunction, other techniques, such as puncture, biopsy and culture, help to come to a definitive diagnosis. At present, several functional (planar scintigraphy, single-photon emission computed tomography [SPECT], and positron emission tomography [PET]) and morphologic (computed tomography [CT] and magnetic resonance imaging [MRI]) imaging modalities are being used to identify infectious and inflammatory foci of soft tissue and/or bone. All of these techniques have specific advantages and disadvantages and are listed in Table 1.
Imaging Techniques for Detection of Infection

For many inflammatory diseases, no unequivocal guidelines for the use of imaging procedures exist, but in some cases, there is sufficient evidence to advise on imaging procedures for optimal diagnosis or follow-up. Moreover, the diagnosis of a specific inflammatory disease depends on the type of suspected disease and the clinical presentation. Also, with the aging population and increasing use of implants and transplants, the number of immunocompromised patients is growing, mainly because of the frequent use of chemotherapeutic agents leading to neutropenia. Furthermore, the increased use of antibiotics leads to insensitivity or resistance. Persistent infection and inflammation can be the root cause of several dreaded diseases as well. It is thus imperative that the pathophysiologic changes involved in the inflammatory process and the coexistence of other diseases, such as cancer, to select the most efficacious procedure are well understood. Therefore, imaging of infection and inflammation is becoming increasingly important.
The design and development of new radiopharmaceuticals for infection imaging are based on the definition and description of the ideal properties of a radiopharmaceutical for infection imaging, such as the following:
High sensitivity for inflammation or infection so as to have efficient accumulation and retention in inflammatory foci
Ability to differentiate between infection and nonmicrobial inflammation
Ability to differentiate between acute and chronic infection
Early diagnostic imaging
No toxicity and/or immunogenic response
High specificity and no accumulation in nontarget organs
Rapid clearance from circulation and thus from the background
No uptake in the gastrointestinal tract
Easy low hazard preparation and low radiation burden
Low cost and wide availability
Radiolabeled antiinfective agents can fulfill the majority of these requirements. The radionuclides currently in use for these techniques are shown in Table 2. With the current resurgence of interest in molecular imaging strategies, the design of target-specific molecular imaging probes based on better understanding of the biochemistry of the inflammatory response may ultimately lead to the development of an ideal agent for infection imaging.
Isotopes Used in Nuclear Medicine Techniques

Pathophysiology of Inflammation and Infection
We can define inflammation as a complex tissue reaction to any kind of injury that may be caused by physical, chemical, or immunologic agents or even by radiation. If the injury is caused by or involves living microbes, it leads to infection, which simply means “contamination with microorganisms.” 2 There can be infection without inflammation, as in the case of a severely immunocompromised patient. Especially in such cases, it is clear that for the purpose of imaging, one would need an agent that directly interacts with the microorganisms. Conversely, there can be inflammation without infection when tissue injury is not due to invasion by microorganisms but rather is caused by “stimuli” such as trauma, ischemia, neoplasm, or foreign particles (eg, asbestos). Whatever the cause may be, inflammation is a nonspecific response to an injury and is a protective response to the cause and the consequences of such an injury. In general, the inflammatory response is characterized by local hyperemia (rubor, calor), edema or swelling (tumor), and, in some locations, pain (dolor).
Inflammation may be classified broadly as acute or chronic depending on the duration of the inflammatory reaction (the interval since the initial injury) and on other pathologic and clinical features. 3 Acute inflammation is the early or an immediate response to injury that lasts for a short duration (8–10 days). It is mediated through the activation of the immune system and is usually beneficial for the host. The condition characterized as chronic inflammation is of longer duration, lasting for several weeks to even years. The systemic changes associated with inflammation are leukocytosis (caused by stimulation by complement component C3a and colony-stimulating factors), fever, and an increase in plasma antiinflammatory proteins such as C-reactive protein, fibrinogen, and haptoglobin. 4 Abscess formation is an occasional consequence, resulting in isolation of the inflammatory focus, which may result in hindering radiotracer access and hence reducing detection.
Acute Inflammation
Acute inflammation is characterized by regional and systemic changes, such as vasodilation of local arterioles and capillaries, increased vascular permeability, and exudate formation. Increased vascular permeability results because of (1) widening of intercellular gaps, (2) leukocyte-mediated endothelial injury, (3) endothelial necrosis and detachment, and (4) formation of new capillaries (angiogenesis) that are leaky. Formation of exudate, an inflammatory extravascular fluid with a high protein (globulins and fibrin) content and cell debris, results as a consequence of vasodilation and increased capillary permeability. These events are followed by local cellular events, such as chemotaxis, leukocyte margination, and emigration (diapedesis), most of which are regulated by chemical mediators produced endogenously by cells at the infectious foci or present in circulation.4,5
Chemotaxis is the process of leukocyte migration to the site of injury. Leukocytes accumulate at the site of infection as a result of leukocyte margination and emigration. First, they adhere to the vascular endothelium owing to locally enhanced expression of adhesion molecules (rolling, arrest, and adhesion). Subsequently, they pass through the endothelium and the basal membrane (diapedesis) and migrate into the inflammatory focus (chemotaxis). Several inflammatory cells, particularly polymorphonuclear leukocytes (PMNs), accumulate and aggregate at the site of inflammation/infection. As part of the response to infectious material, PMNs and macrophages ingest foreign particles and bacterial and tissue debris in a process known as phagocytosis. In the PMN, this is accompanied by the release of peroxides (the superoxide burst) that destroy bacteria.
Chronic Inflammation
After acute inflammation, healing of the injured tissue may lead to resolution (restoration of normal structure and function) or repair (formation of a scar consisting of collagen). Chronic inflammation is a progressive stage of acute inflammation that lasts for a longer period of time. It is characterized by a reduction in the number of PMNs and an increased infiltration of macrophages, lymphocytes, plasma cells, and fibroblasts. Chronic inflammation may predispose the host to various chronic illnesses, including cancer.
Abscess Formation
Both acute and chronic inflammation can be accompanied by abscess formation, which is generally caused by bacterial infection and is defined as a collection of pus in tissues, organs, or confined spaces. During this period of development, inflammation is characterized by hyperemia, leukocytosis (increased production of leukocytes), and edema, with or without cellular necrosis (also known as phlegmon). During the period of abscess formation, there is a vigorous PMN response, but, after stabilization of the abscess, depending on the degree of isolation of the abscess contents, there is a diminution in the pathophysiologic response; hence, functional imaging procedures may be less sensitive.
Inflammation and Cancer
Inflammation is regarded as a “secret killer” for diseases such as cancer. It has been linked to cancer by epidemiologic and experimental data and confirmed by antiinflammatory therapies that show efficacy in cancer prevention and treatment. Tumors may arise at the sites of chronic inflammation, and there has been evidence of the presence of inflammatory cells within them. Oxidative stress has been shown to be a causative phenomenon leading to chronic inflammation, which in turn could mediate chronic diseases such as cancer. 6 Oxidative stress can activate inflammatory pathways by activating a variety of transcription factors, including nuclear factor kB, Activator Protein-1 (AP-1), p53, hypoxia-inducible factor 1a, peroxisome proliferator–activated receptor γ, β-catenin/Wnt, and NF-E2 related factor-2 (Nrf2).7,8 Activation of these transcription factors can lead to the expression of over 500 different genes, including those for antiinflammatory molecules. Thus, oxidative stress can lead to transformation of a normal cell to a tumor cell, tumor cell survival, proliferation, chemoresistance, radioresistance, invasion, and angiogenesis. 9
Types of Inflammation
Although the basic pathophysiology of infection/inflammation has a characteristic pattern, there are subtle differences in the microenvironment at different sites of soft tissue and bone infections.
Abdominal Infection/Inflammation
Abdominal inflammation may involve an organ diffusely, as in hepatitis, colitis, or peritonitis, or focally, as in intraperitoneal abscess, retroperitoneal abscess, or visceral abscess (hepatic, pancreatic, and splenic). Several factors, such as hyperemia and fibrin formation, may predispose infection/inflammation in the abdominal cavity to abscess formation. 4 The pathophysiology may include inflammation of the mucosal lining of the intestinal tract, causing ulceration, edema, bleeding, and fluid and electrolyte loss.
Pulmonary Infection/Inflammation
The common chest inflammatory conditions include bacterial and viral pneumonia, fungal infections, infection involving organisms that are usually nonpathogenic, such as Pneumocystis carinii pneumonia, granulomatous disease (either infectious, such as tuberculosis, histoplasmosis, or coccidiomycoses, or presumably noninfectious, such as sarcoidosis), and diffuse interstitial inflammation. Bacterial and even viral pneumonias are associated with an acute inflammatory process and a vigorous PMN response. The injured tissue becomes vulnerable to secondary infection involving other organisms, thus complicating the specific cellular content of the inflammatory response. In this setting, labeled leukocytes are quite sensitive and provide images with the greatest contrast between infected/inflamed tissue and background. Pulmonary sarcoidosis starts as a diffuse interstitial alveolitis, followed by characteristic granulomas, which are present in the alveolar septa and in the walls of bronchi and pulmonary arteries and veins. During periods of active inflammation, the response is characterized by macrophages and other elements typical of chronic inflammation.
Renal Inflammation/Infection
Two kinds of acute renal infection mainly found are pyelitis (confined to the renal pelvis) and pyelonephritis (involving both the pelvis and parenchyma). Acute pyelonephritis is a bacterial infection and most often occurs because of ascending urinary tract infection. The kidney is usually enlarged and edematous, with small foci of abscess formation in the cortex. The interstitial tissue is predominantly infiltrated by lymphocytes and plasma cells.
Skeletal Inflammation
Osteomyelitis is an infection involving the cortical bone and the myeloid (bone marrow). Skeletal infection may be limited to the periosteum (periosteitis) without involvement of cortex and marrow, but when the cortex is involved, it is called osteitis and osteomyelitis. Osteomyelitis may be classified based on several factors, such as the route of infection hematogenous or non-hematogenous), underlying etiology (diabetic foot), and age at onset (infantile). 4 These classifications are relevant as they also represent differences in the pathophysiologic responses involved and hence influence the choice of radiopharmaceutical selected to identify the inflammatory process. One of the consequences of osteomyelitis is reactive new bone formation resulting in increased blood flow. Chronic osteomyelitis is characterized by less marked infiltration of inflammatory cells than seen in the acute state and may exhibit variable amount of necrotic tissue. Osteomyelitis in the diabetic foot is a unique clinical and pathologic problem. It is a common complication of diabetes and generally occurs as a result of the spread of infection from adjacent foot ulcers. 4
Neuroinflammation
Neuroinflammation is a process in which the brain responds to infections, diseases, and injuries. This kind of inflammation involves these immune cells: lymphocytes, monocytes, and macrophages of the hematopoietic system and microglial cells of the central nervous system (CNS). Neuroinflammation disrupts the blood-brain barrier (BBB), allowing cells from the hematopoietic system to leave the bloodstream and come in contact with the site of injury. The immune cells respond to injuries by eliminating debris and synthesizing and releasing a host of powerful regulatory substances, such as the complements, cytokines, chemokines, glutamate, interleukins, nitric oxide, reactive oxygen species, and transforming growth factors. The substances have both beneficial and harmful effects on the cellular environment, creating further damage. Mature astrocytes are also activated following injury to the CNS. Astrocytic activation is believed to be necessary for containing the immune response, repairing the BBB, and attenuating further neuronal death.
Specific and Nonspecific Tracers for Imaging
During recent decades, many radiopharmaceuticals have been developed for the diagnosis of infection, each with its own advantages and disadvantages. However, all radiopharmaceuticals may not meet the prescribed criteria in a perfect way. In clinical practice, the choice of the imaging agent is based on careful examination of each individual case. Early diagnostic imaging is preferable but not always necessary, as in the case of osteomyelitis or in infected bone/joint prostheses. Imaging with high radiation doses or imaging that entails the occurrence of minor transient side effects after the administration of the radiopharmaceutical is in general unfavorable and should be considered only when significant clinical benefit is to be expected. Several radionuclides are used to prepare radiopharmaceuticals, such as 18F, 111In, 123I, 67Ga, and 99mTc. However, technetium 99m (99mTc)-labeled antibiotics have come up in a big way with their potential to differentiate sterile inflammation from infection. 99mTc-labeled antiinfectives are ideal as infection-seeking agents because of their direct and fast accumulation. Clinical studies already undertaken and further evaluation with different pathogen types, such as viruses, fungi, parasites, and intracellular pathogens, in humans will improve the potential of these compounds. Depending on their mechanism of action, radiopharmaceuticals may be classified as specific and nonspecific. Table 3 elaborates this classification with examples.
Radiopharmaceuticals Used for Imaging Inflammatory Processes

Specific Radiopharmaceuticals
Specific processes of accumulation comprise a number of possible interactions between the radiopharmaceutical and the target, for example, receptor binding and antibody-antigen binding. Several pathways can be distinguished in infection imaging based on specific processes of accumulation of the radiopharmaceutical, as shown in Table 4. Leukocytes preferentially target infection by chemotaxis and can therefore be used to transport radionuclides to the infected area (pathway A). With respect to pathways B and C, vasoactive or chemotactic mediators of the inflammatory process either can be targeted in vivo by radiopharmaceuticals or can be radiolabeled in vitro and injected. So a particular component that is already present in or goes to the inflamed area is either labeled in vivo or is injected in a radiolabeled form in the mediator itself. Pathway D can be said to be truly infection imaging because it uses radiopharmaceuticals that specifically target microorganisms. A completely different mechanism of accumulation at the inflammatory site underlies PET scans using fluorodeoxyglucose (FDG), namely enhanced uptake of radiolabeled glucose by infiltrated granulocytes and tissue macrophages with increased metabolic requirements (pathway E). The accumulation of FDG in cells with increased glucose metabolism is specific. However, FDG is also taken up by, for example, tumor cells.
Strategies in Infection Imaging with Specific Tracers

FDG-PET = fluorodeoxyglucose–positron emission tomography.
Leukocyte Labeling
It has been common practice in nuclear medicine to use radiolabeled leukocytes for imaging infectious/inflammatory lesions. 10 The principal mechanism of uptake of radiolabeled leukocytes at the sites of infection is by cellular migration and target-specific localization. Leukocytes move largely to the site of infection and localize there in great numbers. Detection of infection can be accomplished by direct labeling of leukocytes (ex vivo labeling) or by labeling leukocytes indirectly (in vivo labeling).
Ex vivo labeling requires withdrawal of blood from the patient containing a mixed leukocyte population, purification of leukocytes, and labeling and reinjection of the radiolabeled cells with appropriate radionuclides such as 111In or 99mTc. 111In- or 99mTc-labeled leukocytes have been useful in scintigraphic detection of infected or inflammatory lesions in a number of clinical pathologies, such as fever of unknown origin, inflammatory bowel disease, pulmonary and abdominal infections, osteomyelitis, infected artificial joints, infected vascular grafts, bacterial endocarditis, and inflammatory joint disorders such as rheumatoid arthritis. Leukocyte imaging provides up to 90% sensitivity for both acute and chronic infection. 11 Oxine or tropolone complexes of 111In are used in labeling leukocytes with 111In. Both of the agents are lipid soluble and allow for passive diffusion through the cell membrane after forming 3:1 complexes with indium. 111In-oxine shows higher labeling efficiency in saline than in plasma, whereas tropolone has the advantage of labeling 111In in the presence of plasma. Leukocytes labeled in plasma were shown to accumulate in abscesses to a greater extent compared to cells labeled in the absence of plasma. Of the 99mTc-labeled complexes, such as 99mTc–gentisic acid, 99mTc–hexamethylpropyleneamine oxime (HMPAO), and 99mTc–poly-D-lysine modified by N-acetyl homocysteine, 99mTc-HMPAO is the most widely used agent in routine application. It labels granulocytes predominantly; thus, complicated cell separation techniques used for 111In-oxine and 111In-tropolone are not necessary. 12 Use of autologous leukocytes labeled with 111In or 99mTc can lead to positive imaging because leukocytes, even after ex vivo labeling, have the capacity to migrate to the inflamed area. 111In leukocytes have been found to be more stable in vivo and better for infection imaging than 99mTc leukocytes owing to their more optimal radiation and biodistribution characteristics and are preferred for evaluation of the kidneys, bladder, and gallbladder and also in the case of chronic infection. The latter may provide early diagnosis (2–4 hours), but physiologic 99mTc activity in the abdominal area may be seen, resulting in false-positive images. 12 One important advantage of this method of labeling is that the nontarget localization is considerably reduced because the radio-contaminants are removed before reinjection, but this is accompanied by the time-consuming and laborious preparation of the radiopharmaceutical.
In vivo labeling of leukocytes can be based on antibody– antigen interactions (eg, radiolabeled antigranulocyte monoclonal antibodies) or on leukocyte receptor binding (eg, radiolabeled chemotactic peptides and cytokines). Given that radiolabeled leukocytes are specific to detect infection and inflammation, 99mTc-labeled antigranulocyte antibodies have been developed to label leukocytes in vivo.13,14 The murine anti-NCA-95 IgG1 antibody (BW 250/183) recognizes the nonspecific cross-reacting antigen 95 (NCA-95) expressed on human granulocytes, myelocytes, and promyeloctes (Kd: 2 × 109). It has been used successfully to image various infections and inflammatory processes, including subacute infectious endocarditis, lung abscesses, and septic loosening of hip and knee prostheses. The anti–stage-specific embryonic antigen 1 (anti-SSEA-1) IgM (Neutrospec) recognizes CD15 antigens on granulocytes with high affinity (Kd = 10−11 mol/L). The in vivo binding exceeds 50%, and the 99mTc-labeled anti-SSEA-1 IgM has been successfully used in patients with various inflammatory and infectious diseases, such as osteomyelitis, diabetic foot ulcers, and postsurgical infection with similar diagnostic accuracy when compared to radiolabeled leukocytes. 10 Imaging with 99mTc-anti-SSEA-1 IgM has also proved to be a highly sensitive test for the detection of appendicitis in equivocal cases. 15 However, the high molecular weight of monoclonal antibodies could prove to be disadvantageous, resulting in slow diffusion into sites of inflammation, a long plasma half-life, and uptake in the liver owing to clearance by the reticuloendothelial system. Because of this, a long interval is often required between administration of radiolabeled antibodies and acquisition of images to improve target to background ratios.
The chemotactic peptide formyl-methionyl-leucyl-phenylalanine (fMLP) is one of the earliest peptides studied in inflammation imaging. This peptide binds to the specific receptors expressed on the surface membrane of granulocytes and monocytes with high affinity. Diagnostic applications have shown 99mTc-fMLP to be superior to 111In leukocytes in rabbits. 16
Interleukin-8 (IL-8) is a small protein (8.5 kDa) belonging to the CXC subfamily of chemokines or chemotactic cytokines in which the first two cysteines are separated by one amino acid. IL-8 binds with high affinity (0.3−4 × 10−9 mol/L) to two different receptors (CXCR1 and CXCR2) expressed on granulocytes and promotes chemotaxis of these cells. 123I- and 99mTc-labeled IL-8 were developed as target-specific molecular probes to image infection. 17 Recent clinical studies in patients with suspected infection demonstrated the diagnostic potential of 99mTc-IL-8 with no side effects. 18
99mTc-MDP
Bone scintigraphy using 99mTc-radiolabeled diphosphonates such as 99mTc-methylene diphosphonate (99mTc-MDP) has historically shown good success in detecting bone infections such as osteomyelitis. 99mTc-MDP binds to the hydroxyapatite bone crystal and therefore gives signal depending on both blood flow and rates of bone turnover. 19 Bone scintigraphy has a high sensitivity (> 80%) but a limited specificity (up to 50%). 20 99mTc-MDP imaging only denotes regions of active bone turnover and therefore cannot discriminate osteomyelitis from other causes of increased local bone remodeling. 21 Therefore, the specificity of this technique is less in patients with underlying skeleton conditions due to uptake of the radiopharmaceutical at all sites of increased bone metabolism irrespective of the underlying cause, which may otherwise induce a positive finding on the bone scan. However, specificity can be increased by using this technique in combination with 67Ga imaging or SPECT/CT. 22
Ciprofloxacin (Infecton)
Ciprofloxacin, a fluoroquinolone antimicrobial agent, binds to the deoxyribonucleic acid (DNA) gyrase enzyme present in all dividing bacteria, even to those resistant to ciprofloxacin. Fluoroquinolones are thought neither to bind to dead bacteria nor to accumulate in nonmicrobial inflammatory processes such as Crohn disease. Given that ciprofloxacin binds only to living bacteria, 99mTc-labeled ciprofloxacin theoretically allows discrimination between infection and sterile inflammation. 23 Localization, however, appears to be based primarily on extravasation and stasis at the sites of increased vascular permeability. It is rapidly cleared from circulation by the kidneys and shows no uptake in bone marrow and minimal localization in the liver and abdominal area. In clinical studies, ciprofloxacin has shown acceptable sensitivity and specificity in identifying a wide variety of infectious foci, such as bone and joint infections, infections of hip or knee prostheses, and osteoarticular infections24–28 but does not appear to distinguish between infection and sterile inflammation. 29
Nonspecific Radiopharmaceuticals
Increased blood supply, increased vascular permeability, and enhanced transudation are processes that are used during nonspecific accumulation of tracers. It must be noted that all radiopharmaceuticals accumulate to some extent in this nonspecific way at the site of infection. Examples of nonspecific tracers for the detection of infection based on increased vascular permeability include the following:
Radiolabeled Nanocolloids and Liposomes
Inflammatory lesions can be visualized with nanometer-sized particles that have prolonged blood clearance. Owing to their small size, nanoparticles remain in circulation and make contact with inflammatory/infected cells for relatively longer periods of time. Typical nanoparticles include 99mTc-albumin nanocolloids30–33 and liposomes.34,35
Liposomes, lipid vesicles that are formed as a result of thermodynamic arrangements where an aqueous core is surrounded by two phospholipid layers, have been investigated for infection imaging. 36 Their size varies between 0.1 and 0.5 mm. The exact mechanism for accumulation is speculated to be extravasation at the inflammatory sites as a result of enhanced capillary permeability; however, the role of bacteria and leukocytes is not yet clear. Liposomes are removed from the circulation by the mononuclear phagocytic system (MPS) by being recognized as foreign particles. The short circulation time resulting from their rapid uptake by MPS has been an obstacle in their clinical applications. If the surface of the liposomes is coated with a hydrophilic polymer such as polyethylene glycol (PEG), they circumvent recognition by the MPS, leading to a prolonged residence time in the circulation and enhanced uptake at pathologic sites by extravasation owing to locally enhanced vascular permeability. 37 Such stabilized PEG liposomes can be labeled with 111In-oxinate and 99mTc either using HMPAO as an internal label or via hydrazinonicotinamide (HYNIC) as an external chelator.38,39 99mTc-PEG liposome scintigraphy has shown high sensitivity (94%) and specificity (89%), and visualization of musculoskeletal and abdominal pathology has been shown to be better than with 111In-IgG.
Radiolabeled Macromolecules
Radiolabeled macromolecules such as proteins (albumin, immunogammaglobulin, monoclonal antibodies, and avidin or streptavidin),40–46 polysaccharides (dextran), and derivatized polyamines (methoxy poly(ethylene glycol)-poly-L-lysine–diethylenetriaminepentaacetic acid [DTPA]) 47 have been proposed for imaging inflammatory and infectious lesions.
Some agents that have small molecular weight, such as 67Ga-citrate, 99mTc-porphyrin, 48 and 99mTc-glucose phosphate, combine with plasma proteins after intravenous injection and thus become macromolecules. Radiolabeled macromolecules have prolonged blood residence. Macromolecules leak out of the injured blood vessels and get entrapped at the interstitial space owing to their large size. They might bind to extracellular proteins at the site of inflammation. The most investigated among these are human immunoglobulin (HIG) and streptavidin/avidin–111In-biotin complex. Recently, there has been considerable interest in 99mTc-dextran in this respect owing to the low cost and easy labeling procedure.
111 In and 99m Tc-Labeled Nonspecific Human IgG. According to earlier studies, it was proposed that HIG labeled with 111In or 99mTc is retained at sites of infection and inflammation because of a specific interaction (binding) of HIG with Fc-γ receptors expressed on leukocytes. 49 Subsequently, it was shown that radiolabeled HIG localization at the infectious foci is mainly attributable to nonspecific extravasation or leakage (similar to 67Ga-transferrin complex) of the labeled protein owing to increased vascular permeability. For clinical use, HIG has been labeled with 111In as well as 99mTc. HIG was initially labeled with 111In with the use of DTPA-conjugated HIG (111In-HIG). Subsequently, HIG labeled with 99mTc (99mTc-HIG) with hydrazinonicotinamide as the chelator to complex the metal became available. Both agents have slow blood clearance and physiologic uptake in the liver, spleen, and kidneys. The 99mTc-labeled preparation has the known ideal radiation characteristics, whereas the 111In-labeled preparation allows imaging at time points beyond 24 hours postinjection. In various subacute infections, 111In-HIG scintigraphy showed a slightly but significantly better overall accuracy compared to 111In–white blood cell imaging. 111In- or 99mTc-labeled HIG has been extensively tested in a large number of clinical studies. It has shown excellent performance in the localization of musculoskeletal infection and inflammation. 50 Radiolabeled HIG appeared to be clinically useful for infection imaging studies in patients with musculoskeletal infections and inflammation 51 or rheumatoid arthritis and in pulmonary infection, particularly in immunocompromised patients1,49,51,52 and abdominal inflammation. 53
Avidin-Biotin System. Scintigraphic detection of infectious/inflammatory lesions can also be carried out by a two-step approach in which a nonradioactive compound is pre-targeted followed by a radiolabeled high-affinity partner. Biotin is a compound of low molecular weight that can be easily radiolabeled. Avidin and streptavidin (a family of proteins present in the eggs of amphibians) (molecular weight 66,000 and 60,000 kDa, respectively) bind to biotin with extremely high affinity (Kd = 10−15 M). The avidin-biotin approach is based on the fact that avidin (or streptavidin) will nonspecifically localize at sites of infection by transcapillary leakage and entrapment in the interstitial edema while clearing from blood and nontarget tissues. Avidin (or streptavidin) is injected as a pretargeting agent, followed hours later by a second injection with radiolabeled biotin. Good diagnostic accuracy has been demonstrated in studies of vascular infection 54 and chronic osteomyelitis. 55
99m Tc-Dextran. 99mTc-dextran is a potential radiopharmaceutical for infection/inflammation imaging owing to its easy labeling procedure and economical pricing. Dextran (average molecular weight 60,000–90,000 kDa) can be labeled almost quantitatively with 99mTc by Sn+2 reduction according to a method modified 56 from Henze and colleagues. 57 99mTc-dextran has been evaluated in both experimental abscesses in mice 58 and arthritis in rabbits 59 and in patients with inflammatory lesions, including ulcerative colitis.60–62 The blood clearance of 99mTc-dextran in normal rabbits has been shown to be faster in comparison with other macromolecules, such as HIG and 67Ga-citrate. Compared to HIG, it had the advantages of early detection of the lesions (1 hour vs 6 hours), faster blood clearance (T1/2 = 70 minutes vs 300 minutes), consequently resulting in lower background, and lower cost. 62 The main mechanism of its accumulation in inflammatory lesions is increased capillary permeability. Its superiority over other agents was attributed to its smaller size and neutral charge, facilitating higher outflow to the lesion from the vascular pool compared to negatively charged proteins such as Human Serum Albumin (HSA) and HIG.56,62
Radiolabeled Small Molecules and Ions
The accumulation of pertechnetate ion in inflamed synovium was demonstrated almost four decades ago. Since then, localization of other small molecules and ions in various inflammatory and infectious lesions has been reported. These include ionic and neutral complexes of 99mTc with ligands such as DTPA, citrate, gluconate, glucoheptonate, and pentavalent dimercaptosuccinic acid (DMSA).58,63 They are small water-soluble complexes with low plasma protein binding used as renal agents in routine practice. Owing to their small molecular size, they can penetrate the injured capillaries more easily than macromolecules.
67 Ga-Citrate. 67Ga-citrate is used in clinical practice in several pathologic conditions, including infection and many skeletal disorders, 64 owing to its accumulation in soft tissue tumors and uptake in inflammatory lesions. Because 67Ga is a cyclotron-produced radionuclide, it is essentially carrier free and is available in very high specific activity (10–16 Ci/μmol). As a result, the routine clinical dose of 67Ga-citrate (370 MBq) represents < 50 ng. Once injected in the circulation, 67Ga citrate binds to transferrin and is distributed generally within the body. This complex extravasates at the site of infection owing to the locally enhanced vascular permeability and is partly bound there to lactoferrin excreted by leukocytes or to siderophores produced by microorganisms. The agent is excreted partly via the kidneys (especially during the first 24 hours after injection) and via the gastrointestinal tract. Physiologic uptake of the radiolabel occurs in liver, bone, bone marrow, and bowel. Although 67Ga-citrate scintigraphy has high sensitivity for both acute and chronic infection and noninfectious inflammation, 65 the specificity of the technique is low owing to physiologic bowel excretion and accumulation in malignant tissues and areas of bone modeling.66,67 In addition, its unfavorable imaging characteristics (long physical half-life and high-energy gamma radiation) in combination with the development of newer radiopharmaceuticals have narrowed the clinical applications for gallium scintigraphy.
Pentavalent DMSA. 99mTc (V) DMSA was initially proposed for tumor imaging and has been investigated for imaging inflammatory lesions and various malignancies. 68 It can be instantly prepared from ready kits at basic pH. Its chemical and biologic properties are different from the renal radiopharmaceutical 99mTc (III) DMSA, which localizes predominantly in the renal cortex. Pentavalent DMSA exhibited less plasma protein binding; in mice, greater bone and muscle uptake and lower kidney and liver uptake were observed with respect to trivalent DMSA. 69 99mTc (V) DMSA has been evaluated for imaging inflammatory lesions in experimental animals in comparison with 99mTc(III) DMSA and 99mTc-HIG. 70 Early results for 99mTc-DMSA scintigraphy in neonates with symptomatic urinary tract infections were helpful in ruling out later development of permanent renal damage but were not predictive of the absence of dilating vesicoureteral reflux. 71
PET Radiopharmaceuticals. Infection imaging with FDG-PET is based on the fact that all activated leukocytes (granulocytes, macrophages, and lymphocytes) use glucose as an energy source after activation during the metabolic burst. When activated through infection, metabolism and thus FDG uptake increase because of increased cellular expression of glucose transporters.72–74 18F-FDG-PET has demonstrated significant diagnostic potential and advantage compared to standard nuclear medicine tracers (67Ga-citrate and radiolabeled leukocytes) in patients with soft tissue and bone infections.1,75 The usefulness of FDG-PET for the imaging of infections has been demonstrated in several patient studies.76–78 It can be used for a wide variety of infections, including lesions of bacterial, tuberculous, or fungal origin; soft tissue infections; bone infections; and chronic inflammatory diseases, such as cancer. Sensitivity and specificity using 18F-FDG have generally exceeded 90%. 18F-FDG has been especially successful in cases of osteomyelitis.79,80 The high spatial resolution allows differentiation between osteomyelitis or inflammatory spondylitis and infection of the soft tissue surrounding the bone, 81 and the absence of artifacts from metallic implants allows the imaging of patients with suspected infection in hip and knee prostheses. The accuracy of FDG-PET in evaluating prosthetic joint infections, however, is higher in hip prostheses than in knee prostheses, perhaps because attenuation correction in imaging knee prostheses can introduce artifacts that lead to false-positive results. A negative result of PET study essentially rules out osteomyelitis because of its high sensitivity in detecting infection; however, this may lead to increased false-positive results. A drawback of FDG-PET is the lack of anatomic landmarks, which may make it difficult to assign lesions to a particular structure. High spatial resolution and rapid accumulation into infectious foci are significant advantages over conventional imaging techniques such as the use of labeled leukocytes. In addition, because of the superior signal provided by 18F and the utility in both acute and chronic inflammation, 18F-FDG may become the agent of choice for the initial screening of patients suspected of having occult inflammation.
Limitations in the Clinical Use of Nonspecific Radiopharmaceuticals
Infectious foci can be visualized using radiopharmaceuticals without their specific interaction with a tissue component in the infectious focus by means of a nonspecific process of localization owing to the locally enhanced vascular permeability. Prolonged high blood levels are needed to allow sufficient diffusion into the target tissue ([macro]molecules diffuse into blood vessels by a relatively slow process). However, high blood levels entail relatively high background levels, especially in well-perfused tissues. Moreover, in chronic inflammation, the vascular permeability tends to normalize. Given that nonspecific agents accumulate owing to a common feature of infection and inflammation, these agents cannot distinguish between infection and inflammation. Thus, nonspecific tracers are in principle limited in their ability to detect (and discriminate between) infections and inflammations.
Chemical Leads for Antimicrobial Agents
Pyrimidines
Pyrimidines as Antibacterial Agents
The chemistry of pyrimidines and its derivatives is of great significance in medicine owing to their diverse pharmacologic properties. Pyrimidines are considered to be important not only because they form an integral part of the genetic material,. DNA and ribonucleic acid (RNA), as nucleotides and nucleosides but also because they impart numerous biologic properties. Pyrimidines are an important class of chemicals used as antibacterial agents; the simplest antibiotic based on pyrimidines is bacimethrin, 5-hydroxymethyl-2-methoxypyrimidine-4-amine (58), which was isolated from Bacillus megaterium. It has a broad spectrum of antibacterial activities against various yeasts, bacteria, and staphylococcal infections in vivo. Hitchings and Elion developed trimethoprim (TMP) (59) as a single agent for the oral treatment of uncomplicated urinary tract infections caused by susceptible bacteria. 82 The active component of this antibacterial agent is its structural similarity to the pteridine ring of dihydrofolic acid, which allows it to act as a competitive inhibitor of the bacterial enzyme dihydrofolate reductase.
Sulfonamides prove to be the drugs of choice in well-defined areas such as acute urinary tract infections or cerebrospinal meningitis or in the numerous patients sensitive to penicillins; in addition, most sulfonamides have excellent therapeutic indices, and their side effects, if any, disappear quickly on discontinuance of the drug. 83 Three sulfa pyrimidines, namely sulfadiazine, sulfamerazine, and sulfamethazine, are homologues in which the N′- substituent is 2-pyrimidinyl in sulfadiazine (60), 4-methyl-2-pyrimidinyl in sulfamerazine, and 4,6-dimethyl-2-pyrimidinyl in sulfamethazine. Several other metal analogues and new derivatives are being added to the list and are summarized in Table 5.
Pyrimidines as Antibacterial Agents

ACP = acyl carrier protein; DNA = deoxyribonucleic acid.
Numbers in parentheses refer to the structures given in Figure 1.

Structures of antibacterial pyrimidines 1–27.
Pyrimidines as Antifungal Agents
Pyrimidines act as novel lead molecules for designing antifungal agents. A series of pyrimidines that are being used against fungal cells are listed in Table 6. 5-Fluorocytosine (28), a pyrimidine antimetabolite, is widely used in systemic antifungal chemotherapy. Other pyrimidines known for their antifungal activity include natural products such as nikkomycins (29), 84 polyoxins (30), and their synthetic analogues. A pyrimidinyl ketone oxime ether derivative at 200 ppm completely controlled Cercospora beticola for beet seedlings. 2-(Pyrimidinyloxyphenyl)methacrylates gave complete control of seven weeds, for example, Pyricularia oryzae, at 100 ppm combined foliar spray and root drench. 4-(2-Pyrimidinyloxy and 2-pyrimidinylthio)quinoline N-oxide derivatives have been used as agrochemical fungicides. 2-Acylvinyl sulfide pyrimidine derivative at 200 ppm controlled > 95% P. oryzae on rice seedlings. Tetrahydropyrimidine derivatives gave > 80% control, for exanple, at 600 ppm, therapeutically against Erysiphe graminis on barley seedlings. In tests against Alternaria solani, Phytophthora infestans, Botrytis cinerea, and other phytopathogens under a variety of conditions, various salts have typically shown > 80% disease control.
Pyrimidines as Antifungal Agents

DNA = deoxyribonucleic acid; RNA = ribonucleic acid.
Numbers in parentheses refer to the structures given in Figure 2.
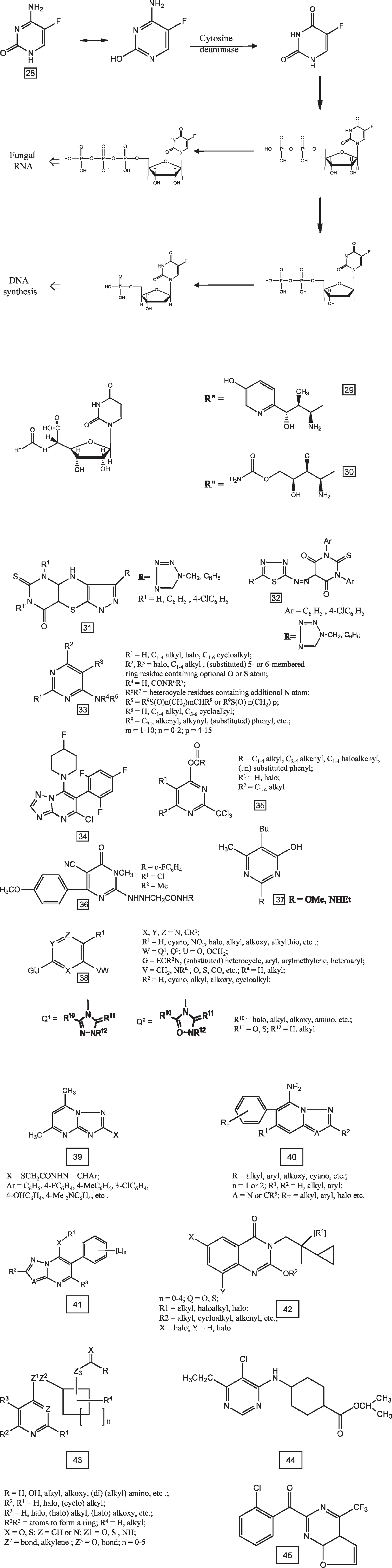
Structures of antifungal pyrimidines 28–45.
Imidazoles and Triazoles as Antifungal Agents
The azoles are synthetic compounds containing one or more azole rings with a complex side chain attached to one of the nitrogen atoms. Azole-based antifungal agents in clinical use contain either two or three nitrogens in the azole ring and are thereby classified as imidazoles (eg, ketoconazole and miconazole, clotrimazole) or triazoles (eg, itraconazole and fluconazole), respectively. 85 With the exception of ketoconazole, use of the imidazoles is limited to the treatment of superficial mycoses, whereas the triazoles have a broad range of applications in the treatment of both superficial and systemic fungal infections. Another advantage of the triazoles is their greater affinity for fungal rather than mammalian cytochrome P-450 enzymes, which contributes to an improved safety profile.
The imidazoles can be used to treat invasive diseases but are mostly effective as topical therapies against cutaneous and mucocutaneous infections. 86 The imidazole and triazole groups of antifungal drugs inhibit the enzyme cytochrome P-450 14α-demethylase (also referred to as 14α-sterol demethylase or P-450DM).87–90 This enzyme converts lanosterol to ergosterol and is required in fungal cell membrane synthesis. This enzyme also plays an important role in cholesterol synthesis in mammals. 89 Examples are drugs such as miconazole, bifonazole, ketoconazole, butoconazole, clotrimazole, and econazole, which also block steroid synthesis in humans.
Ergosterol also has a hormone-like (“sparking”) function in fungal cells, which stimulates growth and proliferation.87,90 This function may be disrupted when ergosterol depletion is virtually complete (0.99%). 87
The azoles act through an unhindered nitrogen, which binds to the iron atom of the heme, preventing the activation of oxygen that is necessary for the demethylation of lanosterol. In addition to the unhindered nitrogen, a second nitrogen in the azoles is thought to interact directly with the apoprotein of lanosterol demethylase. It is thought that the position of this second nitrogen in relation to the apoprotein may determine the specificity of different azole drugs for the enzyme. The resulting depletion of ergosterol alters the fluidity of the membrane, and this interferes with the action of membrane-associated enzymes. The overall effect is an inhibition of replication (ie, the azoles are fungistatic drugs). A further repercussion is the inhibition of transformation of candidal yeast cells into hyphae—the invasive and pathogenic form of the parasite.
A number of promising new azole and triazole derivatives are currently being developed by several pharmaceutical companies. Three of these agents—voriconazole, ER-30346, and D0870—are derivatives of fluconazole, and another agent, SCH 56592, is a hydroxylated analogue of itraconazole. All of these new agents are active following oral administration; voriconazole and T-8581 may also be administered intravenously, and SCH 56592 shows topical activity. With the exception of D0870, UR-9746, and UR-9751, which show little activity against Aspergillus spp., the new triazole derivatives possess potent, broad-spectrum, antifungal activity. These agents have promise for the treatment of a wide variety of superficial and systemic infections owing to both opportunistic and endemic fungal pathogens. As per earlier studies, only voriconazole was in phase III clinical trials; SCH 56592 was in phase I/II studies, the other new azoles were in the preliminary stages of development, and D0870 has been withdrawn from development. Thus, any proposed clinical uses are based for the most part on in vitro data and the results of in vivo studies in experimental animal infection models. The chemical structures, proposed routes of administration, and potential clinical uses of these compounds are listed in Table 7.
Important New Azole Antifungal Agents under Development

Peptides as Infection Imaging Agents
Antimicrobial peptides (molecular weight = 5–7 kDa) are produced by phagocytes, endothelial cells, and many other cell types and are an important component of the innate immune system. The basis of their antimicrobial activity is their direct interaction with the bacterial plasma membrane by electrostatic and hydrophobic interaction. Owing to this preferential binding, these peptides potentially can be used to discriminate bacterial infections from nonbacterial infections and sterile inflammations. One of the earliest peptides studied in inflammation imaging is the chemotactic peptide fMLP, 16 which binds to the specific receptors expressed on the surface membrane of granulocytes and monocytes with high affinity. Another molecule, 99mTc-UBI 29-41, a fragment of the antimicrobial peptide ubiquicidin (UBI 29-41) labeled with 99mTc, is now under evaluation in in vitro and preclinical and clinical settings. 91 It binds preferentially to bacteria in vitro and not to activated leukocytes. It binds to the negatively charged groups present on the bacterial cell envelope by electrostatic interaction, and a specific mechanism is responsible for intrabacterial 99mTc-UBI 29-41 accumulation. 92 This 99mTc-UBI 29-41 showed some specific uptake (1–2%ID) in the infected thigh muscle at 30 minutes postinjection, with coinjection of an excess of unlabeled UBI 29-41 peptide showing significantly lower uptake of 99mTc-UBI 29-41. In addition, uptake in muscle tissue with inflammation induced by inoculation of dead bacteria was also significantly lower. 93 UBI scintigraphy was shown to be 100% sensitive in acute postoperative prosthetic joint infection. 94 Similarly, human neutrophil peptide 1 (HNP-1) labeled with 99mTc was characterized in mice with inflamed thigh muscles. 95 The abscess to background ratios obtained with this tracer were low (3–5) and decreased with time. In the peritoneal cavity of the infected mice, 99mTc-HNP-1 bound preferentially to bacteria rather than to leukocytes. These peptides can be used to monitor the efficacy of antimicrobial therapy regimens and to distinguish between bacterial infection and sterile inflammation.
Many proteins and peptide drugs can now be produced relatively economically. A number of physiologic and biologic barriers are associated with these peptides that limit their use as therapeutic agents. However, most protein drugs have a short half-life in vivo, requiring multiple treatments to provide efficacy. They are also unstable in biologic fluids and not fully absorbed because of their high molecular weight. To overcome these disadvantages associated with protein drugs, microspheres are beneficial as long-term dosage forms.
Antibacterial peptides, which were originally described in insects (Hyalophora cecropia) and plants and subsequently identified in vertebrates, constitute a key component of the innate immune system. Most of the known antibacterial peptides (currently more than 850 characterized) are active against multiple microorganisms, such as viruses, bacteria, fungi, and protozoa, and can be ascribed to one of three main groups: (1) α-helical peptides without cysteine, (2) peptides with three disulfide bonds, or (3) peptides rich in proline or tryptophan 95 (Table 8). However, in most cases, peptide sequences are unique to each species and even to tissues within a species. Consequently, in nature tremendous sequence diversity exists among bacteria-killing peptides. Antibacterial peptides exert their activity by both intracellular and extracellular killing of microbes, attacking multiple molecular targets to cooperatively penetrate and disrupt microbial surfaces and membrane barriers, resulting in membrane depolarization, a loss of pH gradient, impaired osmotic regulation, and inhibition of respiration. 96 When bacterial cells are exposed to antibacterial peptides, within minutes more and more peptide molecules dock to the surface of the bacterial membrane. Charge-based interactions between negatively charged bacterial wall moieties such as lipopolysaccharide (LPS) and lipoteichoic acid (LTA) and positively charged sites on the peptides initiate adhesion of antibacterial peptides to bacterial surfaces, 97 and hydrophobic interaction may further enhance binding. Although electrostatics is assumed to be the critical force driving attachment of antibacterial peptides, some antibacterial peptides permeate both zwitterionic and negatively charged phospholipid vesicles. 98 The hydrophobicity of these molecules does not ensure their binding to phospholipids, 99 but it does favor their penetration into phospholipid monolayers if docking is enabled, thereby destabilizing membranes of both eukaryotic and prokaryotic cells. 100
Structure of Some Representative Antimicrobial Peptides

For amino acids, the one-letter code is used. Subscript numbers represent amino acids that are joined by disulfide bridges.
A new class of antibacterial agents is those that act through the inhibition of the metalloenzyme peptide deformylase (PDF).101,102 Nature itself has confirmed the importance of PDF as an antibacterial target with the discovery that the antimicrobial natural product actinonin is a potent PDF inhibitor.103,104 Some new inhibitors have been designed on the basis of substrate analogues that demonstrated a high affinity for the enzyme, which are summarized in Table 9.
Antibacterial Peptides as Potent PDF Inhibitors

PDF = peptide deformylase.

Structures of antibacterial peptides 46–61.
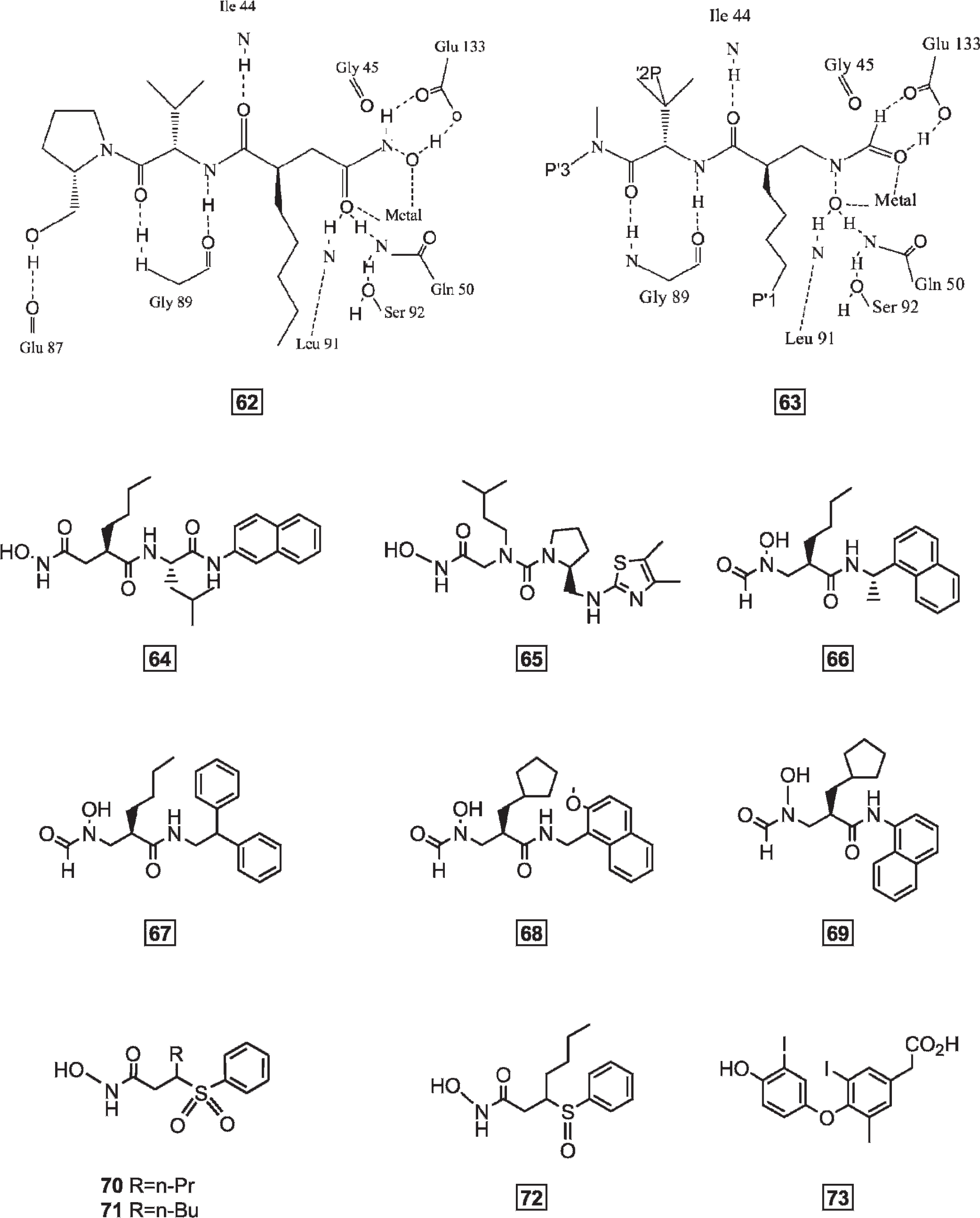
Structures of antibacterial peptides 62–73.
Limitations of Nuclear Medicine Imaging Techniques
Nuclear medicine procedures can be time consuming. It can take hours to days for the radiotracer to accumulate in the part of the body under study, and imaging may take up to several hours to perform, although in some cases, newer equipment is available that can substantially shorten the procedure time. But at the same time, nuclear medicine techniques have become of the utmost importance in evaluating patients with infection and inflammatory diseases. SPECT and PET are accurate methods for diagnosing infection and related abnormalities but often do not provide the anatomic landmarks needed to precisely localize lesions. MRI and CT, on the other hand, offer excellent anatomic detail but are less sensitive because they do not provide functional detail. Fusion imaging combines functional studies with morphologic ones, overcoming the drawbacks of these existing modalities. Software-based fusion of independently performed scintigraphic and radiologic images has proven time consuming and impractical for routine use. Recently, dual-modality integrated imaging systems (SPECT/CT and PET/CT) have been developed. Although these are in their nascent stages, they provide great hope in the field of nuclear medicine for imaging infection and inflammation.
Conclusions
During the last several decades, a gradual shift has taken place in the field of nuclear medicine from basic to advanced techniques (radiolabeled leukocytes) to more intelligent approaches based on small agents that bind to their targets with high affinity. Classes of agents can be designed to specifically distinguish between infection and noninfectious inflammation and between acute and chronic processes. Antifungal/antibacterial drug research has grown considerably over the past decade. The use of the currently available azoles in combination with other antifungal agents with different mechanisms of action is also likely to provide enhanced efficacy. 99mTc as a radionuclide offers several advantages, as already mentioned in this review; however, many more can still be explored. Labeled leukocytes provide the greatest specificity to identify infection but have the limitation that they are most sensitive to detect acute inflammation, that is, the inflammatory response dominated by polymorphonuclear leukocytes (granulocytes). Chronic and acute inflammation are nevertheless readily detectable with either 67Ga-citrate or 18F-FDG because the accumulation of both occurs with localized pH reduction. It is appropriate to briefly consider the choice or preference between these two tracers and the differences observed between 111In- and 99mTc-labeled leukocytes. A summarized list is given on the basis of their mechanism of action in Table 10. Although a shorter radionuclide half-life is favorable because it allows for the administration of a larger dose, labeling with high specific activity will reduce the doses used so that undesirable agonistic activities will become nonexistent. At present, however, although 18F-FDG is available as an imaging agent for neurologic, oncologic, and cardiac indications, it is not yet approved as an agent for the identification of inflammatory/infectious foci. Having said this, it should be mentioned that positron-emitting isotopes have enormous potential for inflammation imaging owing to their ability to be used with various tracers with different specificities and the long time spans necessary for imaging and diagnosis.
Known Radiopharmaceuticals for Inflammation/Infection Imaging

DNA = deoxyribonucleic acid; FDG = fluorodeoxyglucose; HMPAO = hexamethylpropyleneamine oxime; MDP = methylene diphosphonate; NCA = nonspecific cross-reacting antigen; SSEA = stage-specific embryonic antigen.
Footnotes
Acknowledgments
We would like to thank Dr. R.P. Tripathi, director, Institute of Nuclear Medicine and Allied Sciences, for providing all of the facilities as well as support during the writing of the manuscript. We also thank Dr. Anupama Datta for valuable discussions on the subject.
Financial disclosure of authors and reviewers: None reported.
