Abstract
To visualize the norepinephrine transporters (NETs) in various brain diseases, we developed radioiodinated (2S,αS)-2-(α-(2-iodophenoxy)benzyl)morpholine ((S,S)-IPBM). This radioligand achieved the basic requirements for NET imaging. In this study, we assessed the potential of radioiodinated (S,S)-IPBM as an imaging biomarker of NET to obtain diagnostic information about depression in relation to NET expression in the brain using a rat depression model. The ex vivo autoradiographic experiments using the (S,S)-[125I]IPBM showed significantly lower accumulation of radioactivity in the locus coeruleus (LC) and the anteroventricular thalamic nucleus (AVTN) of the depression group than in those of the control group. Consequently, in vitro autoradiographic experiments showed that NET maximum binding (Bmax) values in the LC and AVTN, known as NET-rich regions, were significantly decreased in the rat model of depression when compared to those of the control rats. In addition, there was an extremely good correlation between NET Bmax and (S,S)-IPBM accumulation (r = .98), an indication of radioiodinated IPBM as a quantitative NET imaging biomarker. The reduction in(S,S)-[125I]IPBM accumulation in the rat model of depression correlated with that of NET density. These results suggest that (S,S)-[123I]IPBM has potential as an imaging biomarker of NET to obtain diagnostic information about major depression.
A RADIOIODINATED REBOXETINE ANALOGUE, (2S,αS)-2-(α-(2-iodophenoxy)benzyl)morpholine ((S,S)-IPBM), has been developed as an imaging agent of cerebral norepinephrine transporter (NET) expression. 1 (S,S)-IPBM shows high affinity and selectivity to NET in vitro and in vivo, and the regional accumulation of radioiodinated (S,S)-IPBM in the brain correlates well with NET expression in the brain.
NET, located on presynaptic terminals of the noradrenergic nervous system, terminates the action of norepinephrine via reuptake into the presynaptic neurons. NET not only regulates neurotransmission in the noradrenergic nervous system but also plays a very important role in presynaptic and postsynaptic homeostasis.2–4 NET has been implicated in the pathophysiology of numerous neuropsychiatric and mood disorders, including major depression, 5 arousal, anxiety, and attention-deficit/hyperactivity disorders.6–8 About 16% of the population is estimated to be affected by major depression one or more times during their lifetime. 9 Therefore, the association between major depression and NET has been under intense investigation from the point of view of elucidating the etiologic mechanism of major depression and the prediction of therapeutic effects of antidepressants. However, the relationship between major depression and NET remains unclear. For example, NET gene variation has been investigated in major depression, but with contradictory results.10–12 With regard to protein expression of NET in depression patients, only Klimek and colleagues showed reduced expression of NET in the locus coeruleus (LC) compared to age-matched normal control subjects using brain tissue collected postmortem from patients diagnosed with major depression. 5 Therefore, it is important to image the function of NET in depression patients to understand the etiology of depression and the mechanisms of antidepressants.
In this study, we investigated the potential of radioiodinated (S,S)-IPBM as an imaging agent of NET for the diagnosis of depression. As a depression model, a reserpine-treatmented rat model 13 was selected, and an ex vivo autoradiographic study using radioiodinated (S,S)-IPBM was performed for comparison of regional NET expression.
Materials and Methods
General
[3H]Nisoxetine (3.16 TBq/mmol) was purchased from PerkinElmer Life Sciences, Inc. (Boston, MA). (S,S)-[125I]IPBM was prepared according to the previously reported method. 1 All chemicals used in this study were of reagent grade.
Animals
Animal studies were conducted in accordance with our institutional guidelines, and the experimental procedures were approved by the Kyoto University Animal Care Committee. Male rats of the Wistar strain weighing 200 to 220 g were used. On arrival at the facility, the rats were housed four to a cage at a constant temperature of 25°C under a 12-hour light/dark cycle, with food and water available ad libitum for at least 1 week prior to the experiment.
Reserpine was injected daily for 14 days at a dose of 0.1 mg/kg subcutaneously. 13 To minimize the influence of circadian rhythms, the experiments were performed between 13:00 and 15:00 hours. 14
Ex Vivo Autoradiographic Experiments
Sixty minutes after the last injection of reserpine, the rats were intravenously injected with 2.22 MBq of (S,S)-[125I]IPBM (control group: n = 4; reserpine-treated group: n = 4). At 180 minutes after the injection, the rats were decapitated under light ether anesthesia, and their brains were rapidly removed, frozen, and cut into sections 20 μm thick with a cryostat microtome. The sections were thaw-mounted on precleaned silane-coated slides. The slides were placed on a Fuji imaging plate (BAS-SR, Fuji Photo Film, Tokyo, Japan) for 24 hours along with calibrated [125I]labeled external standards ([125I]Microscales, Amersham, UK) to obtain (S,S)-[125I]IPBM autoradiograms. Densitometric analyses of the autoradiograms were performed using a bioimaging analyzer and image gauge. Regional tissue uptake was quantified in terms of %ID/g based on [125I]labeled external standards. Regions of interest were manually placed over the LC, hippocampus, thalamus, hypothalamus, striatum, frontal cortex, and anteroventricular thalamic nucleus (AVTN).
In Vitro Autoradiographic Experiments
An in vitro autoradiographic experiment was performed according to the method reported previously. 15 The frozen brains were serially cut into 20 μm thick coronal sections with a cryostat; the sections were mounted on 0.2% gelatin/0.03% chromium potassium sulfate–coated slides and kept at −80°C until use (control group: n = 5; reserpine-treated group: n = 5). The sections (two slides per animal, that is, one for total binding and one for nonspecific binding) were preincubated for 15 minutes at 25°C in 50 mM Tris-HCl buffer at pH 7.4 containing 120 mM NaCl and 5 mM KCl. The incubations were carried out with 3.0 nM [3H]nisoxetine at 4°C for 4 hours, and nonspecific binding was determined with 100 μM (final concentration) nisoxetine. The slides were then washed in ice-cold (0–4°C) buffer (3 × 5 minutes for [3H]nisoxetine), rinsed in cold deionized distilled water and dried under a stream of cold air. The sections and [3H]Microscales standards were placed on tritium-sensitive imaging plates (BAS-TR, Fuji Photo Film) for 24 hours. Densitometric analyses of the autoradiograms were performed using a bioimaging analyzer (BAS3000, Fuji Photo Film) and image gauge (Fuji Photo Film). Regions of interest were manually placed over the LC, hippocampus, thalamus, hypothalamus, striatum, frontal cortex, and AVTN.
Optical density values were converted to fmol/mg of protein using a computer-generated regression analysis that compared imaging plate densities produced by tissue sections and radioactive standards. The concentration values were then multiplied by (KD + c)/c to obtain maximum binding (Bmax) 16 (KD = equilibrium dissociation constant of [3H]nisoxetine reported previously [1.0 nM] 17 ; c = incubation concentration of [3H]nisoxetine).
Statistical Analysis
Results are expressed as mean ± SD. For statistical data analysis, one-way analysis of variance with a post hoc Tukey test was applied for multiple comparisons. The unpaired alternate Welch t-test was applied for two group comparisons. A probability value of less than .05 was considered to indicate a significant difference.
Results
Reserpine Treatment
The changes in body characteristics on day 14 induced by chronic treatment with reserpine are shown in Table 1. There were significant differences in rectal temperature and body weight between the rats treated with reserpine and the control rats. This is consistent with previous reports that the rectal temperature was decreased in reserpine-treated rats. 18
Reserpine-Induced Changes in Body Characteristics of Rats on Day 14

Each value is the mean ± SD.
p < .001 versus control rats.
Ex Vivo Autoradiographic Experiments
Ex vivo autoradiographic experiments of (S,S)-[125I] IPBM were performed in the control and reserpine-treated rats. Figure 1 shows the representative autoradiograms in both groups. The uptake of (S,S)-[125I]IPBM in the reserpine-treated rats was significantly lower in the LC and AVTN than that in the control rats (LC, control: 1.40 ± 0.09, reserpine: 0.74 ± 0.15%D/g, p < .001; AVTN, control: 0.93 ± 0.04, reserpine: 0.70 ± 0.04%D/g, p < .01; Figure 2).

Representative ex vivo autoradiograms of rat coronal brain sections in control (A) and depression model (B) rats. Rats were intravenously injected with 2.22 MBq of (S,S)-[125I]IPBM. At 180 minutes after the injection, the rats were decapitated under light ether anesthesia, and their brains were rapidly removed, frozen, and cut into sections 20 μm thick. AVTN = anteroventricular thalamic nucleus; Ctx = frontal cortex; Hip = hippocampus; Hyp = hypothalamus; LC = locus coeruleus; Str = striatum; Tha = thalamus.
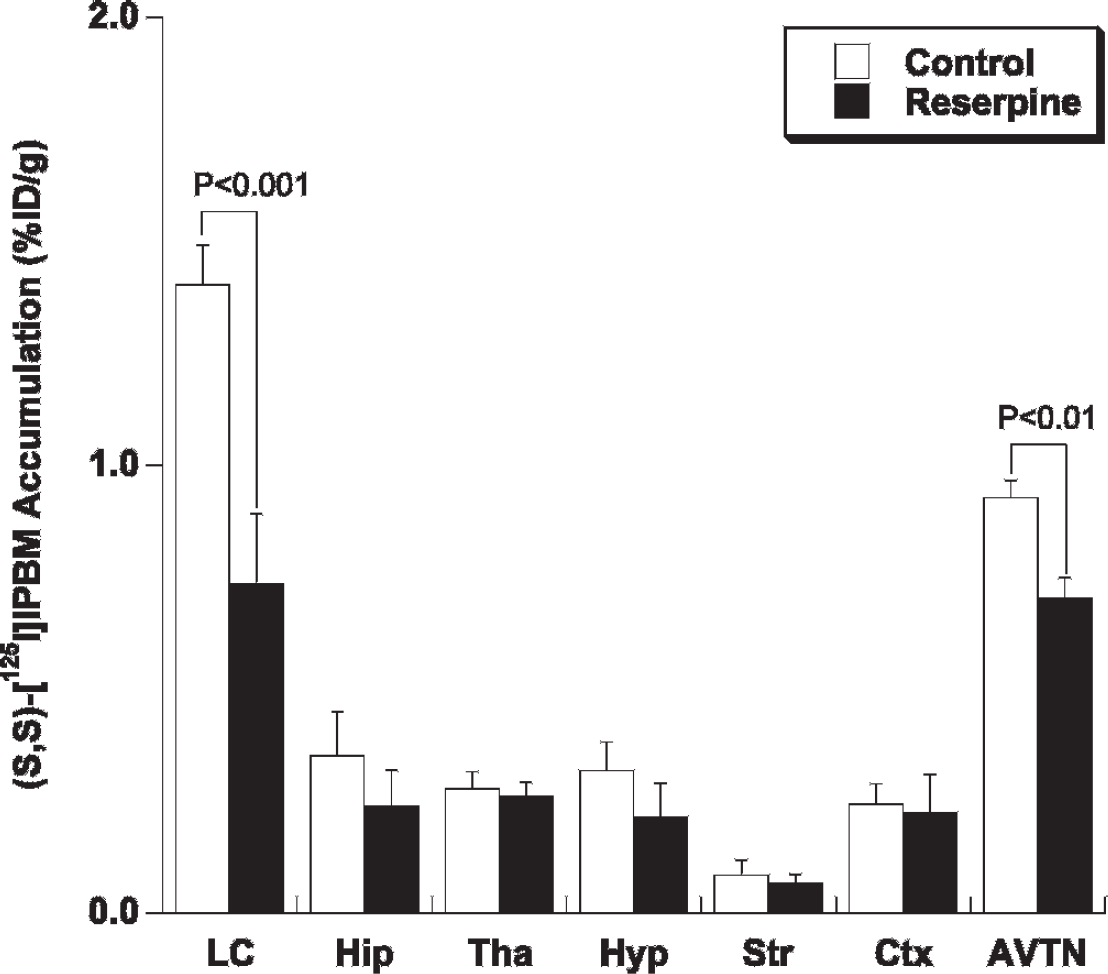
Regional uptake of (S,S)-[125I]IPBM in control (n = 4) and reserpine-treated rats (n = 4). The uptake was measured densitometrically by quantitative ex vivo autoradiography and image analysis. AVTN = anteroventricular thalamic nucleus; Ctx = frontal cortex; Hip = hippocampus; Hyp = hypothalamus; LC = locus coeruleus; Str = striatum; Tha = thalamus.
In Vitro Autoradiographic Experiments
To investigate regional NET expression, we performed in vitro autoradiographic experiments using [3H]nisoxetine. As shown in Figure 3, in the control group, the binding of [3H]nisoxetine was highest in the LC followed by the AVTN, known as NET-rich regions.19,20 There was a good correlation between the regional uptake of (S,S)-[125I]IPBM and the regional NET expression (r = .98) (Figure 4). Compared to the control rats, the nisoxetine binding in the reserpine-treated rats was significantly lower in the LC (control: 336 ± 36, reserpine: 164 ± 54 fmol/mg protein, p < .001) and AVTN (control: 171 ± 20, reserpine: 102 ± 59 fmol/mg protein, p < .05), and the binding of [3H]nisoxetine in other regions tended to decrease in the reserpine-treated rats.
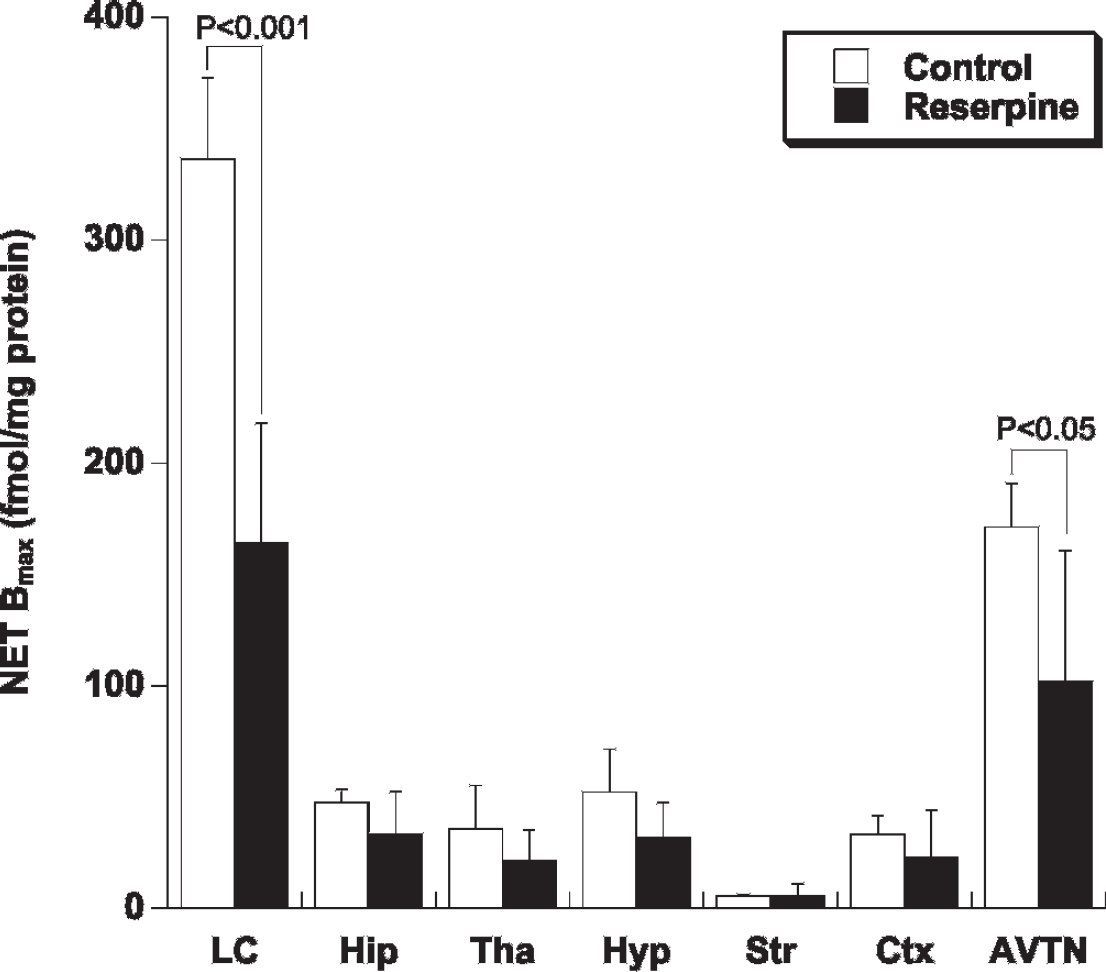
Regional norepinephrine transporter (NET) expression determined by in vitro [3H]nisoxetine autoradiography in the rat brain sections (control group: n = 5, reserpine-treated group: n = 5). The expression of NET was measured densitometrically by quantitative in vitro autoradiography and image analysis. AVTN = anteroventricular thalamic nucleus; Ctx = frontal cortex; Hip = hippocampus; Hyp = hypothalamus; LC = locus coeruleus; Str = striatum; Tha = thalamus.
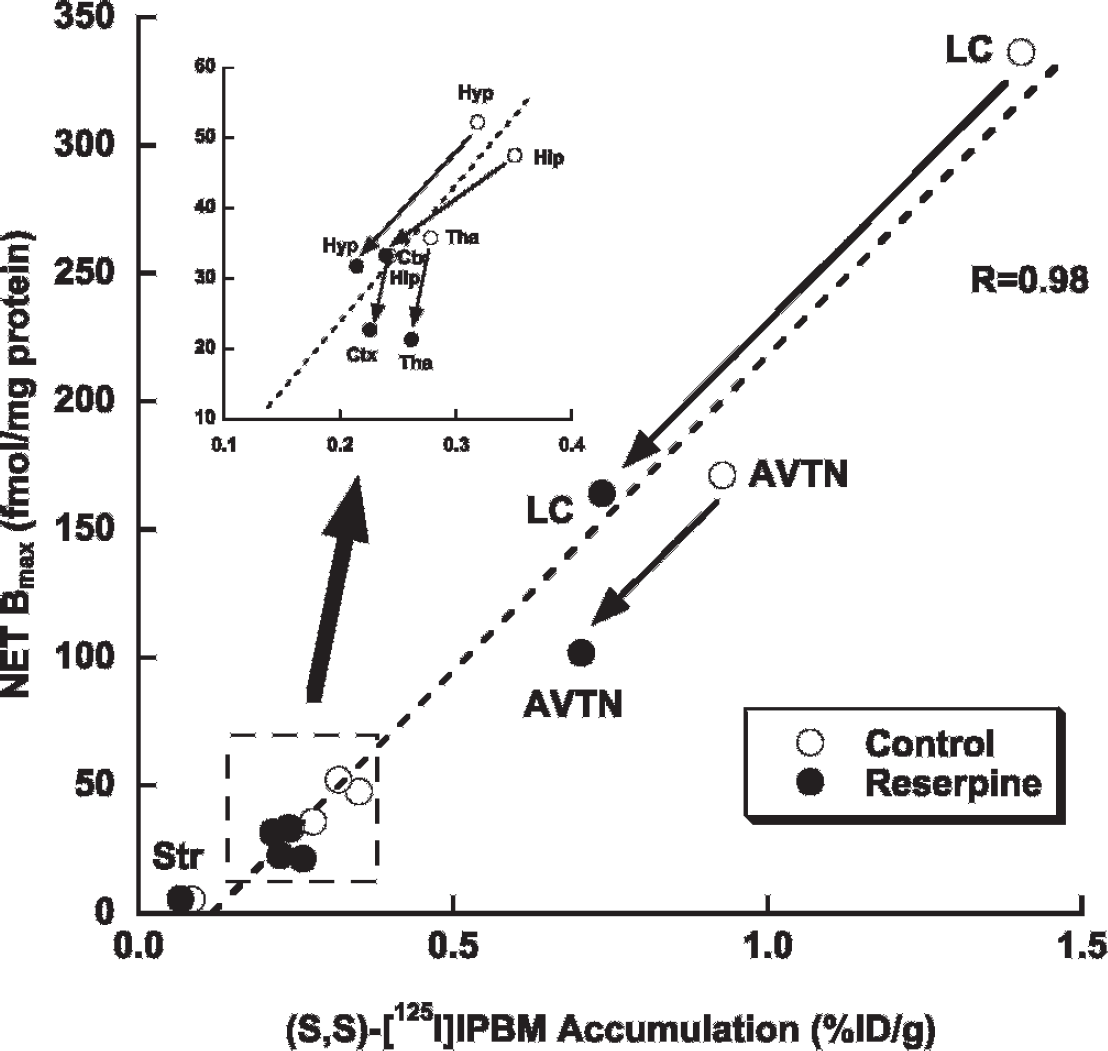
Correlation between the regional uptake of (S,S)-[125I]IPBM and the expression of norepinephrine transporters (NETs) in control and reserpine-treated rats. AVTN = anteroventricular thalamic nucleus; Ctx = frontal cortex; Hip = hippocampus; Hyp = hypothalamus; LC = locus coeruleus; Str = striatum; Tha = thalamus.
Discussion
In this study, we demonstrated that radioiodinated (S,S)-IPBM has good potential as a quantitative imaging biomarker of regional NET expression in normal as well as diseased brains. The regional uptake of (S,S)-[125I]IPBM in both normal and depression model rat brains correlated closely with the regional NET expression.
Although NET is generally considered to be an important target in studies of major depression, there have been a few positron emission tomographic (PET) tracers targeting NET.21–23 In clinical studies, (S,S)-[11C]MeNER is often used.24–26 However, Logan and colleagues reported that problems associated with (S,S)-[11C]MeNER included a low specific to nonspecific ratio and slow uptake into target regions. 24 Takano and colleagues reported that (S,S)-[18F]FMeNER-D2 was superior to (S,S)-[11C]MeNER27 but still had some limitations. The defluorination of (S,S)-[18F]FMeNER-D2 caused high accumulation of [18F]fluoride in the skull, which resulted in bad quantification in the cortical regions. 27 Besides the regions that highly express NET, the regions with medium expression of NET (eg, frontal cortex, hippocampus, thalamus, and hypothalamus) are considered to be important because the functions in these regions control the majority of human systems. Furthermore, it may be difficult to analyze regions with high expression of NET (eg, LC and AVTN) by PET and single-photon emission computed tomography (SPECT) in clinical studies owing to the small sizes of these regions. Therefore, quantification of NET in the cortical regions could be essential for studies of major depression, and radioiodinated (S,S)-IPBM has the possibility to quantify NET in the cortical regions with SPECT because radioiodinated (S,S)-IPBM has a high affinity for and selectivity to NET and metabolites of radioiodinated (S,S)-IPBM do not accumulate in the skull. 1 However, there is the limitation using the reserpine-treated model of concluding that radioiodinated (S,S)-IPBM is useful in the cortical regions because there were no significant differences in the uptake of radioiodinated (S,S)-IPBM in those regions between the control and reserpine-treated model rats. The expression of NET in the cortical regions in our mode was not decreased, but Lee and colleagues reported that the Bmax value of NET in the cerebral cortex was decreased in reserpine-treated rats, 28 and this contradiction with our data may be due to the dose (0.1 mg/kg in our study versus 0.5 mg/kg in Lee and colleagues study) and the duration of treatment (14 days in our study versus 18 days in Lee and colleagues study). Therefore, if their model had been selected, the uptake of radioiodinated (S,S)-IPBM would significantly decrease in the cortex. Further studies regarding the dose and duration of reserpine are needed. Furthermore, the sensitivity of radioiodinated (S,S)-IPBM is also important for detecting subtle changes in the expression of NET in cortical regions. The reported Kd values from in vitro saturation binding assay of [3H]nisoxetine (1 nM) 17 and (S,S)-[125I]IPBM (1.3 nM) 1 are similar. Tejani-Butt reported the region-specific binding of [3H]nisoxetine by in vitro autoradiographic experiments. 20 The minimum specific binding was observed in the caudate putamen and the maximum binding was observed in the LC. In this range, the regional uptake of (S,S)-[125I]IPBM was well correlated with the specific binding of [3H]nisoxetine. 1 Therefore, (S,S)-[125I]IPBM could detect subtle changes in the cortical regions.
Furthermore, this is believed to be the first report regarding regional changes in NET expression in the brain after reserpine treatment. The reserpine-treated depression model is one of the most frequently used models of depression.13,18,29,30 There have been some reports about the expression of NET in the brain of rats after reserpine treatment to date.28,31 However, these reports were forcused on a particular region of the brain. Cubells and colleagues reported that the messenger ribonucleic acid of NET was decreased in the LC region of rats treated with reserpine. 31 In addition, Lee and colleagues reported that the Bmax value of NET in the cerebral cortex was decreased in reserpine-treated rats. 28 Although the Bmax value of the cerebral cortex in the present study was not significantly decreased, our data on changes in NET expression were largely consistent with previous reports. The reduction in NET expression might be a compensatory mechanism for the depletion of norepinephrine caused by the treatment with reserpine; a similar phenomenon was observed in the brains of depression patients. 5 Therefore, it is important to detect the expression of NET in the brain for elucidating the role of NET in depression. Radioiodinated (S,S)-IPBM could be a useful tool for this purpose.
Conclusion
The reduction in the radioiodinated (S,S)-IPBM uptake in the rat model of depression correlated with that of the NET expression. (S,S)-[123I]IPBM would be useful for elucidating the etiologic mechanism of major depression and the prediction of therapeutic effect of antidepressants by SPECT, although further studies are needed.
Footnotes
Acknowledgment
Financial disclosure of authors: This study was supported by the Program for Promotion of Fundamental Studies in Health Sciences of the National Institute of Biomedical Innovation and a Grant-in-Aid for Scientific Research on Priority Areas (Research on Pathomechanisms of Brain Disorders) from the Ministry of Education, Culture, Sports, Science and Technology, Japan.
Financial disclosure of reviewers: None reported.
