Abstract
Different from regular small molecule contrast agents, nanoparticle-based contrast agents have a longer circulation time and can be modified with ligands to confer tissue-specific contrasting properties. We evaluated the tissue distribution of polymeric nanoparticles (NPs) prepared from human serum albumin (HSA), loaded with gadolinium–diethylenetriaminepentaacetic acid (Gd-DTPA) (Gd-HSA-NP), and coated with folic acid (FA) (Gd-HSA-NP-FA) in mice by magnetic resonance imaging (MRI). FA increases the affinity of the Gd-HSA-NP to FA receptor–expressing cells. Clinical 3 T MRI was used to evaluate the signal intensities in the different organs of mice injected with Gd-DTPA, Gd-HSA-NP, or Gd-HSA-NP-FA. Signal intensities were measured and standardized by calculating the signal to noise ratios. In general, the NP-based contrast agents provided stronger contrasting than Gd-DTPA. Gd-HSA-NP-FA provided a significant contrast enhancement (CE) in the brain (p = .0032), whereas Gd-DTPA or Gd-HSA-NP did not. All studied MRI contrast agents showed significant CE in the blood, kidney, and liver (p < .05). Gd-HSA-NP-FA elicited significantly higher CE in the blood than Gd-HSA-NP (p = .0069); Gd-HSA-NP and Gd-HSA-NP-FA did not show CE in skeletal muscle and gallbladder; Gd-HSA-NP, but not Gd-HSA-NP-FA, showed CE in the cardiac muscle. Gd-HSA-NP-FA has potential as an MRI contrast agent in the brain.
Contrast media are critical to improve powerful imaging methods such as magnetic resonance imaging (MRI). Conventional small molecule contrast media mainly enhance contrast of the intravascular compartment. Contrasting of pathologies in the tissue is frequently limited by biologic barriers, mainly tight endothelia, and by rapid renal clearance of conventional contrast agents from the circulation, thus confining diagnosis to late stages of the diseases. For instance, conventional MRI contrast agents do not cross the blood-brain barrier (BBB).1,2 Contrast agents that can cross the BBB might be highly useful to improve imaging of pathologic alterations of the brain such as Alzheimer disease, early-stage tumors, multiple sclerosis, and inflammation.3–5
There is large interest in developing tissue- or tumor-specific contrast agents to improve radiologic diagnosis. Owing to their unique size and accessibility to surface functionalization to confer the desired characteristics, nanoparticles (NPs) are particularly suitable to overcome biologic barriers. 6 NPs have a prolonged circulation half-life and can be conjugated with tissue-specific ligands or with reporter molecules that increase the signal in medical imaging for actively targeting contrast agents. 7 The same NPs can be loaded with drugs for targeted therapeutic drug delivery in combination with simultaneous monitoring of their biodistribution (theragnostics). 8
A promising ligand conferring tissue-specific targeting is folic acid (FA). The expression of folate receptors (FRs) is confined to certain normal tissues and pathologic tissues (eg, some cancer entities and activated macrophages). 9 Recently, FA-conjugated human serum albumin (HSA)-based NPs have been shown to cross the BBB. 10 These NPs are nontoxic and biodegradable 10 and might be suitable for clinical application. Thus, FA-conjugated HSA-NP might be suitable to cross biologic barriers and may thus enable tissue-specific imaging or imaging of pathologies. A considerable number of studies investigated the use of folate and FR for designing FR-targeted contrast agents to improve tumor detection and characterization and for tumor therapy.11–14
In the present study, we investigated the biodistribution of FA-conjugated gadolinium–diethylenetriaminepentaacetic acid (Gd-DTPA)-loaded HSA-NP (Gd-HSA-NP-FA) and unconjugated Gd-DTPA-HSA-NP as novel potential MRI contrast agent in mice. This pilot study indicates that Gd-HSA-NP-FA can cross the intact BBB and thus has potential as an MRI contrast agent in the brain.
Materials and Methods
Preparation of the Contrast Agents
HSA-NP were prepared using a previously described desolvation procedure and stabilized by chemical crosslinking with glutaraldehyde.10,15 Briefly, 200 mg HSA in 2 mL of 10 mM NaCl solution, adjusted to pH 8.4, was formed into NPs by continuous (1 mL/min) addition of 8 mL desolvating agent ethanol under constant stirring at room temperature. After protein desolvation, 235 μL of an 8% aqueous glutaraldehyde solution was added to achieve particle crosslinking. The resulting NPs were purified by three cycles of differential centrifugation (16,100g, 10 minutes) and redispersion of the pellet to the original volume in water. The NP content of the suspension was determined by gravimetry. Unmodified NPs were spherical and had diameters of 156 ± 25 nm.
FA was covalently coupled to amino groups on the surface of the activated HSA-NP by the use of N-(3-dimethylaminopropyl)-N-ethylcarbodiimide (EDC). Subsequently, Gd-DTPA was also attached using EDC. 10 The Gd-HSA-NP were washed to remove free Gd-DTPA, and loading was determined by MRI (box analysis). The particles remained stable at 4°C for several days.
Transmission Electron Microscopy
Transmission electron microscopy (TEM) samples were prepared by air-drying NPs of formvar/carbon-coated copper grids and observed using a Libra 120 transmission electron microscope (Carl Zeiss, Göttingen, Germany) operating at 80 kV.
In Vitro MRI Analysis
In vitro MRI contrasting properties of the suspended nanoparticles were determined by scanning of the NP suspension in a clinical 3.0 T scanner (Siemens Magnetom Trio, Siemens Medical Solutions, Erlangen, Germany). The parameters used were identical to those used for in vivo MRI.
In Vivo MRI
The animal experiments described in this study were approved by the governmental committee and our institutional animal research review board (permit number F34/07). Male mice weighing 34 to 36 g were anesthetized by intraperitoneal injection using ketamine (70 mg/kg) and xylazine (12 mg/kg). Contrast agents were administered via retrobulbar intravenous injection of Gd-HSA-NP or Gd-HSA-NP-FA (8 μmmol Gd-DTPA/kg body weight in 200 μL) or of 200 μL of Gd-DTPA (0.057 mmol/kg). The injection flow was 0.25 mL/s. Twenty minutes after the injection of the contrast agent, MRI was performed. The mice were scanned with or without contrast agent. While the experiments were performed, no side effects of the used contrast agents were observed.
Imaging scans were performed using a 3.0 T scanner (Siemens Magnetom Trio). The mice were positioned inside a multifunctional coil pair with a flexible holder (NORAS 8 Ch Multifunctional Coil, NORAS MRI Products GmbH, Hoechberg, Germany) in a supine position. Each coil is configured as a four-channel array and was centered on the liver of each mouse.
The animals were imaged in the coronal plane using a T1-weighted, three-dimensional FLASH sequence with the following parameters: repetition time (TR) 8.67 ms; echo time (TE) 3.31 ms; flip angle 16°; in-plane matrix 640 × 480; field of view 100 × 78 mm; fat suppression, fat saturated. The slice thickness was 0.3 mm, and the bandwidth was 180 Hz/Px.
Image Analysis
For the image analysis, a commercially available workstation and the program Centricity RIS 4.1i Plus, version 4.1 (General Electric), were used.
Quantitative Image Analysis
The quantitative MRI analysis of this study is based on the measurement of signal intensities (SIs). To determine the SI values of the organs, circular areas called operator-defined regions of interest (ROI) were drawn into the MRIs. The SI values of the ROI were displayed as average values with a standard deviation. Five ROI were put in: the brain at the level of the ventricles, the musculature in the area of the shoulder, the cardiac muscle of the right ventricle, the vascular lumen of vena cava, the cortex and medulla renalis, the lumen of the gallbladder, and the liver in the areas with regular branching of blood vessels.
To standardize the measurement, all ROI were as large as possible, of the same size and shape, and placed in one image slice as possible.
As SIs were measured in the present study to investigate the contrast enhancement in a cross-sectional scanning technique, it was relevant to estimate the image noise. For the purpose of this study, the common method of measuring the SI and its standard deviation (SD) of the background noise in the air outside the body of the mice was used.
16
The SD was divided through 0.66
Each signal detected from the scanned animals in the MRI is interfered with by an image noise signal. To describe the quality of the signal and to standardize the SI measurements by taking the image noise into account, the signal to noise ratio (SNR) was used. Furthermore, the employment of SNR data allows a better comparison of the results among different mice. The SNR was determined using the following formula: SNR = SI(of an organ) /image noise. 17 SNRs of noncontrast mice and mice with contrast agents were determined to evaluate the contrast enhancement of the different contrast agents.
Statistical Analysis
The present study was designed as a proof of principle. For this reason, each experimental group contained two mice.
Data are presented as means ± SD. The acquired data were compared for significant differences between the groups using analysis of variance (ANOVA) with an α level (error of the first kind) of .05 per comparison. The ANOVA assumptions were ensured using the Kolmogorov-Smirnov test, and, where necessary, multiple comparisons by Scheffé were performed. All statistical computations were performed using the software BiAS for Microsoft Windows, version 9.05 (Epsilon Publishing, Hochheim-Darmstadt, Germany).
Results
Preparation and Characterization of the NPs
HSA-NP were conjugated with FA and loaded with Gd-DTPA. The particle diameter as determined by TEM was between 100 and 200 nm (Figure 1). Loading of the washed HSA-NP with Gd-DTPA was verified by in vitro MRI. Gd-HSA-NP and Gd-HSA-NP-FA showed signal enhancement, whereas NPs that had not been loaded with gadolinium provided no contrast enhancement (Figure 2).

Transmission electron microscope image of Gd-HSA-NP showing their size and morphology. The arrows point to individual HSA nanoparticles.
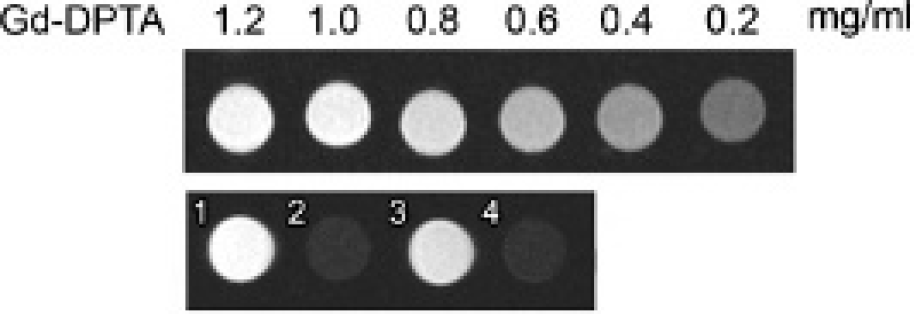
Magnetic resonance image of the box of the studied contrast media. The solutions were separately filled in Eppendorf cups and scanned in the magnetic resonance tomograph. All contrast agent solutions with incorporated gadolinium showed a contrast enhancement (1 = Gd-HSA-NP-FA; 2 = HSA-NP-FA [without Gd]; 3 = Gd-HSA-NP; 4 = HSA-NP [without Gd]). Upper line = calibrating array with different concentrations of Gd-DTPA.
In Vivo Biodistribution of Contrast Agents in Mice
Gd-HSA-NP (n = 2), Gd-HSA-NP-FA (n = 2), or Gd-DTPA was administered intravenously with NPs corresponding to 8 μmol Gd-DTPA/kg body weight or 0.057 mmol Gd-DTPA/kg of Gd-DTPA. Each mouse was scanned with or without contrast agent. Imaging scans were performed using a clinical 3.0 T scanner. The mean SNR values of the tissues of noncontrast mice and of those with administered contrast agents are displayed in Table 1. The p values resulting from the statistical analysis and the comparison of the experimental groups are shown in Table 1 and Table 2.
SNR Mean Values (± SD) of the Tissues of Noncontrast Mice and Mice with Different Injected Contrast Agents (Gd-DTPA, Gd-HSA-NP, or Gd-HSA-NP-FA)

Gd-DTPA = gadolinium–diethylenetriaminepentaacetic acid; Gd-HSA-NP = nanoparticles prepared from human serum albumin and loaded with Gd-DTPA; Gd-HSA-NP-FA = nanoparticles prepared from human serum albumin and loaded with Gd-DTPA; SNR = signal to noise ratio.
SNR data were compared between the different experimental groups for significant differences using analysis of variance. At p < .05, differences were considered to be significant. The p values shown represent the differences between mice with contrast agent and noncontrast mice.
Results of the Statistical Analysis

Gd-DTPA = gadolinium–diethylenetriaminepentaacetic acid; Gd-HSA-NP = nanoparticles prepared from human serum albumin and loaded with Gd-DTPA; Gd-HSA-NP-FA = nanoparticles prepared from human serum albumin and loaded with Gd-DTPA.
Signal to noise ratio data of the different experimental groups compared to the analysis of variance. At p ≤ .05, differences were considered to be significant.
Brain
Gd-HSA-NP-FA provided a statistically significant contrast enhancement in the brain (p = .0032) (Figure 3), whereas Gd-HSA-NP and Gd-DTPA did not. For MRIs and graphic representation of the distribution of the SNR values in the brain, see Figure 3 and Figure 4. Statistical comparisons of SNRs in this organ showed significant differences in SNR values between Gd-HSA-NP-FA and Gd-HSA-NP or Gd-DTPA (see Table 2).
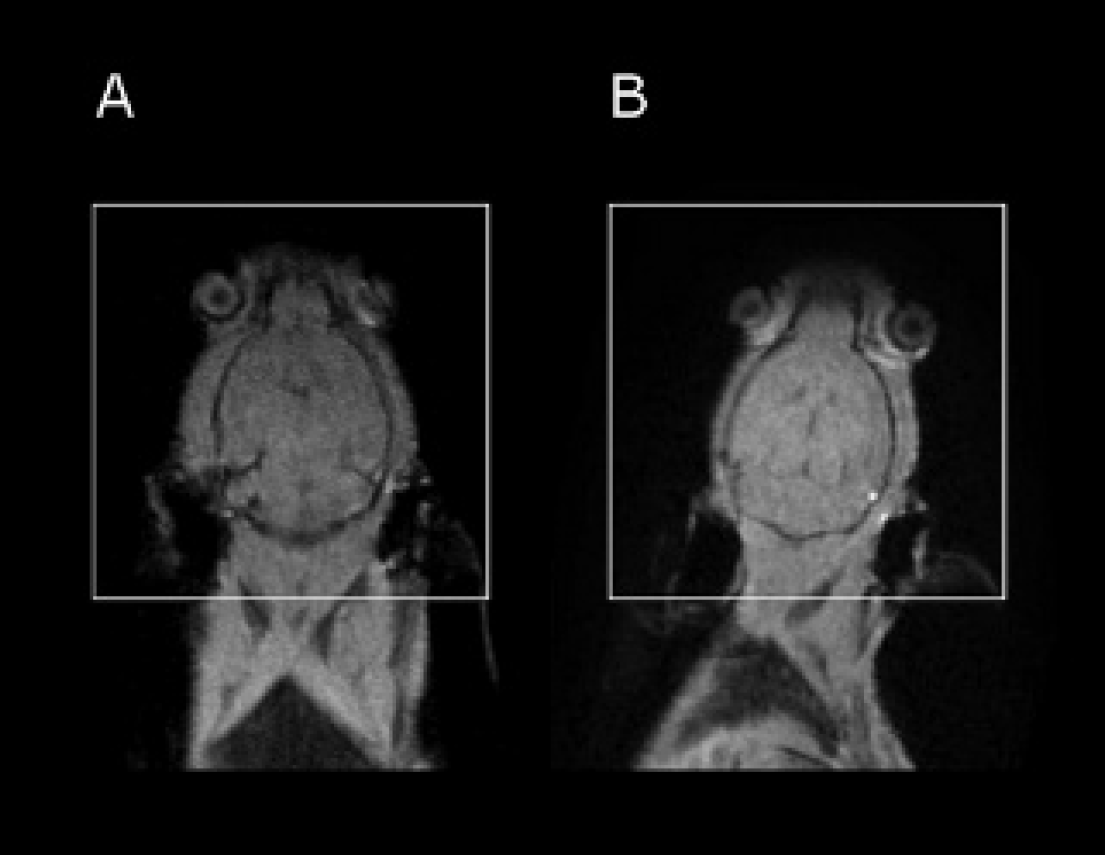
T1-weighted magnetic resonance images (TR = 8.67 ms; flip angle = 16°) in the coronal plane view of a mouse in the supine position before (A) and after (B) injection of Gd-HSA-NP-FA. Marked are the brains (white rectangles) for representation of the clearly visible contrast enhancement provided by the studied contrast agent Gd-HSA-NP-FA.

Shown are the results of the descriptive statistics that were performed using signal to noise ratio (SNR) data of the brain calculated using signal intensities (SIs). SI values of the brain were measured in the area of the ventricles of mice before and after administration of contrast agent (Gd-DTPA, Gd-HSA-NP, or Gd-HSA-NP-FA). *Significantly different SNR data (p ≤ .05).
Musculature
In the musculature, the NP-based contrast agents provided no significant contrast enhancement compared to the noncontrast mice (Gd-HSA-NP: p = .4208; Gd-HSA-NP-FA: p = .9975). In contrast, Gd-DTPA showed significant contrast enhancement in this tissue (p = .0025) (see Table 1 and Table 2).
Vena Cava
In the blood, all studied agents showed contrast enhancement (p < .05) with significant differences between the single contrast agents. Gd-HSA-NP-FA yielded a higher contrast enhancement than Gd-HSA-NP (p = .0069), and Gd-HSA-NP had a higher contrast enhancement than Gd-DTPA (p = .0407) (see Table 1 and Table 2). For the graphic representation of the distribution of the SNR values, see Figure 5.

Biodistribution of Gd-DTPA, Gd-HSA-NP, and Gd-HSA-NP-FA in the vena cava. Shown are the results of the descriptive statistics that were performed using signal to noise ratio (SNR) data calculated using signal intensities measured in the lumen of the vena cava before and after contrast agent administration. *Significantly different SNR data (p < .05).
Cardiac Muscle
Measurements of the SIs and the calculated SNRs showed significant increased values for Gd-DTPA and Gd-HSA-NP in this tissue (see Table 1 and Table 2). In contrast, Gd-HSA-NP-FA did not show contrast enhancement in the cardiac muscle (p = .0926); that is, FA-modification reduced contrast enhancement of Gd-DTPA-HSA-NP in the cardiac muscle (see Table 2).
Kidney
All injected imaging agents provided a statistically significant contrast enhancement in the cortex and medulla renalis (see Table 1 and Table 2). In both tissues, the SNR values of Gd-HSA-NP-FA did not differ from that of Gd-HSA-NP (see Table 2). Gd-DTPA showed a higher contrast enhancement in the medulla renalis than Gd-HSA-NP-FA (p = .003) (see Table 2).
Gallbladder
No studied contrast agent showed a significant contrast enhancement in the gallbladder. Gd-DTPA showed a trend to a contrast enhancement in this organ (p = .0519) (see Table 1).
Liver
All studied contrast agents yielded a contrast enhancement in this organ (see Table 1). There was no significant difference between Gd-HSA-NP and Gd-HSA-NP-FA (p = .4218) and between Gd-DTPA and the NPs (see Table 2).
Discussion
NP-based MRI contrast agents offer a number of advantages in comparison with conventional small molecule contrast agents. Owing to the high molecular weight, Gd-DTPA-NP circulate much longer in the blood than Gd-DTPA. Moreover, the modification of the Gd-DTPA-NP with targeting ligands offers the possibility of tissue-specific targeting. Here we investigated the biodistribution of FA-conjugated Gd-DTPA-loaded HSA-NP and conjugated Gd-DTPA-HSA-NP as novel potential MRI contrast agents in mice. The data show that Gd-HSA-NP-FA can cross the intact BBB and thus has potential as a brain-specific contrast agent.
The different FR isoforms have a high affinity for FA and different affinities for the derivates of vitamin B9.9,11 These functional differences offer an additional selectivity for FR-targeted imaging agent transport and for drug delivery. 11 The FR isoform α (FR-α) is the most widely expressed isoform in normal tissues and has a high affinity for FA, which was used in our study for the synthesis of the FR-targeted contrast agent. 18 FR-α is expressed only on certain healthy tissues, including the proximal tubules of the kidney, intestinal epithelial cells, lung (type I and II pneumocytes), choroid plexus, submandibular salivary, bronchial glands, fallopian tube, uterus, ovary, epididymis, and trophoblasts in the placenta.9,18 The FRs expressed on these tissues are inaccessible to intravenously injected FR-targeted agents except the FRs in the kidney, which filter the FR-targeted agents from the bloodstream. 18 Consequently, in the present study, Gd-HSA-NP-FA provided contrast enhancement in the cortex and medulla of the kidney. This is in agreement with findings with another contrast agent. 9
Owing to their prolonged circulation time, nontargeted Gd-HSA-NP also showed significant contrast enhancement in the blood. Moreover, whereas conventional contrast agents diffuse across vascular endothelium outside the BBB, nontargeted NPs remain relatively strict in the vasculature. Similar to the present study, a recent study investigating the biodistribution of folate-coated, gadolinium-loaded, lipid-based NPs in tumor-bearing mice showed prolonged retention of NPs in the blood in vivo. 12 As the contrast enhancement provided by Gd-HSA-NP-FA was significantly higher than the contrast enhancement provided by Gd-HSA-NP, a nonspecific contrast enhancement of Gd-HSA-NP-FA is unlikely. Thus, this additional contrast enhancement of the FR-targeted NPs in comparison with the nontargeted NPs might be related to the FR in the blood macrophages. 11 These data suggest that Gd-HSA-NP-FA may have potential for blood-pool magnetic resonance angiography. Of course, to clearly evaluate the diagnostic potential of the NP formulations, further investigations providing information on the pharmacokinetics of the NP formulations in disease models, for example, are necessary.
The transport of most drugs and imaging agents over BBB into the brain is restricted by the tight junctions between endothelial cells of the blood vessels in the central nervous system. 19 The BBB-targeted chemical modification of drugs and imaging agents can enable delivery from the bloodstream into the brain. 20 Magnevist does not cross the intact BBB. 2 This was confirmed by no contrast enhancement of the brain after Gd-DTPA injection. The same result was observed after administration of uncoated Gd-HSA-NP. In contrast, Gd-HSA-NP-FA caused a statistically significant contrast enhancement, indicating that the FA modification of the HSA-NP enables penetration of the NPs across the BBB. Thus, the present study shows for the first time that FA-conjugated NPs can be used as a contrast agent in the brain in the absence of disturbance of the BBB. As brain-specific contrast agents are lacking, FA-conjugated, Gd-DTPA-loaded NPs may meet the need to detect brain tumor at early stages, when the BBB is intact. Gd-HSA-FA may detect brain tumors before induction of cerebral edema and its clinical symptoms. In support of our data, a previous study described a certain capacity of human and rat BBB endothelial cells for uptake of FA. 21
As Gd-HSA-NP-FA apparently crosses the BBB, it can potentially be used for contrasting in brain MRI and might be suitable for the detection of pathologically altered brain tissue, most notably cancer or sites of infection and inflammation (note the FR expression on brain tumors and activated macrophages). 9 These particles also may be used for drug transport into the brain.
FA-coupled NPs also may be relevant for the detection and treatment of several tumor types. The “angiogenic switch” and proliferation of the blood vessels are a well-known mechanism of cancerogenesis.22,23 Gd-HSA-NP-FA achieve high contrast enhancement of the blood and have the potential to detect tumor lesions using hypervascularization of the tumor. The enhanced tumor targeting effect of the folate-modified NPs is further augmented by the overexpression of FR on several human cancer types, such as malignant tumors of the brain and cancer of myeloid cells of hematopoietic origin, lungs, breast, kidneys, ovaries, endometrium, and colon. Imaging agents can be transported to these tumors without uptake by healthy tissues, which have only a limited expression of FR; thus, a high grade of tumor specificity can be achieved. 9
Successful administration and the biodistribution of Gd-DTPA and, for the first time, Gd-HSA-NP and Gd-HSA-NP-FA were demonstrated by the use of MRI and calculation of the SNR. Gd-HSA-NP-FA may potentially be used as a contrast agent in the brain.
Footnotes
Acknowledgment
Financial disclosure of authors: This work was supported by grants from the Wilhelm Sander Stiftung (2003.119.2), the Else Kröner-Fresenius Stiftung, and the Deutsche Forschungsgemeinschaft (GRK 1172).
Financial disclosure of reviewers: None reported.
