Abstract
Matrix metalloproteinases (MMPs) are extracellular proteolytic enzymes involved in acute lung inflammation in response to cigarette smoke exposure (CSE). We present the in vivo detection of MMP activity using a specific MMP-activatable, near-infrared, polymer-based proteolytic probe in strains of mice with different susceptibility to developing smoking-induced emphysema (susceptible mice, C57BL/6j, and resistant mice, 129S2/SvHsd) to characterize the distinctive profile of CSE-induced acute inflammation. In vivo imaging of pulmonary inflammation expressing MMPs revealed a significantly different median ratio twofold higher in smoker than in nonsmoker susceptible mice (C57BL/6j) and no significant differences between the smoker and the nonsmoker group in resistant mice (129S2/SvHsd). Ex vivo imaging of the lungs of each group of mice confirmed the same in vivo experiment results obtained for both strains of mice. In the biochemical study of lung tissue, the proteolytic signal colocalized with the endogenously expressed MMP protein levels, with MMP-9 levels that are 2.2 times higher than in the nonsmoke-exposed group in C57BL/6j mice and no significant differences in the 129S2/SvHsd mice. The MMP-activatable probe provides a useful reagent for the in vivo and ex vivo detection of MMP-selective proteolytic activity. We are able to distinguish between susceptible and resistant strains of mice in terms of the profile of MMP activity in the early stages of pulmonary disease.
CHRONIC OBSTRUCTIVE PULMONARY DISEASE (COPD) is a major cause of illness and death throughout the world. It affects about 10% of the general population, 1 but its prevalence among heavy smokers can reach 50%. 2 Etiologically, COPD is largely associated with tobacco smoking, and epidemiologic studies show an increased risk of respiratory disease even in young, asymptomatic smokers. Although the main risk factor for developing COPD is tobacco, only 15–20% of smokers have been reported to develop the disease. 3 The reason for this is unknown, but individual factors, such as different sensitivity to oxidants and differences in the immune or regenerative capacity of lung cells, are most likely to predispose individuals to smoke-induced lung damage.4–6
Alveolar macrophages degrade and remodel tissue extracellular matrix through production of proteolytic enzymes, among which there are matrix metalloproteinases (MMPs), such as MMP-2 −3, −9, −12, and −13, that initiate and propagate inflammatory responses.7,8 Previous studies have suggested that macrophage-derived MMPs are the critical mediators of cigarette smoke–induced emphysema9–11 and have demonstrated that MMP-2 and MMP-9 and their active forms are present in increased amounts in COPD sputum. 12
Mice represent an important model for studying the factors influencing susceptibility to lung disease. 13 Animal models also afford strong arguments for the significant role of proteinases, such as MMPs, in emphysema. Recent studies detail knowledge of the involvement of MMP-2 and MMP-9 in acute lung injury and acute respiratory distress syndrome.14,15 In animal models of lung injury, authors have previously demonstrated that gene-targeted mice deficient in macrophage elastase (MMP-12) are entirely protected from cigarette smoke-induced emphysema 16 and acute cigarette smoke exposure (CSE)-induced pulmonary inflammation, with major participation of MMP-12 by alveolar macrophages. 17 Previous studies have revealed that there are differences in the responses to CSE among different strains of mice. Mice of the strain C57BL/6j responded to chronic CSE with an acceleration in the development of spontaneous emphysema, assessed with morphometric and biochemical methods18–21; responded to subchronic CSE modulating several clusters of genes 22 and appeared to undergo necrosis of bronchoalveolar lavage fluid cells 23 ; and responded to acute CSE, augmenting the pattern of inflammatory and oxidant responses compared to resistant mouse strains 24 and increasing neutrophils and keratinocyte chemoattractant levels and MMP-9 gene expression in the airways. 25 Mice of the strain 129S2/SvHsd, which produce low levels of tumor necrosis factor a, were resistant to lung inflammatory and oxidant responses to acute inflammation,24,25 showing no inflammatory response to smoke at 24 hours.26,27 Thus, an important point in the mice models would be the identification of predictive biomarkers in emphysema-susceptible and -resistant mice strains. We intended to identify common and distinct features of the tobacco response in two strains of mice with different sensitivity to cigarette smoke by imaging the profile of CSE-induced acute inflammation.
Over the last century, research on the early detection of COPD has moved from invasive (postmortem) histologic studies to noninvasive anatomic techniques, such as spirometry, radiography, computed tomography, magnetic resonance imaging, and positron emission tomography, which are also used to measure ventilation, perfusion, and pulmonary function parameter. However, there is a need for more efforts to take advantage of imaging methods and to develop and validate lung disease–specific biomarkers as valuable adjuncts to diagnosis and assessment of treatment response. 28 Optical imaging is the method of choice for functional, real-time, in vivo monitoring because it provides an “easy” approach for measuring enzymatic activities, and fluorescence molecular imaging is a technique developed to visualize deeper into the living animal and is herein considered a noninvasive molecular lung imaging tool for small animals.29–32 Several proteases are overexpressed or activated during lung inflammatory response progression and are thus attractive targets for the so-called “activatable” probes. Activatable means that a change in signal intensity or wavelength can be detected in response to the interaction of the probe with its target.33,34
Here we describe the use of an injectable pan-MMP-targeted optical sensor that specifically and semiquantitatively resolves MMP-2, −3, −9, and −13 activity in the lungs of mice with experimental acute CSE-induced airway inflammation. The purpose of this study was to compare, via fluorescence molecular imaging, the effects of CSE on pan-MMP-mediated responses in susceptible and resistant mice strains. We hypothesized that noninvasive molecular imaging of MMP activity might provide information on inflammatory disease severity. The combination of sensitive enzyme-targeted sensors in different mice strains with noninvasive molecular imaging approaches makes it possible to evaluate the severity of airway inflammation. It could be used as a model to rapidly screen for new drug effects 35 and has the potential to be applied in the clinic.
Material and Methods
Animals
Adult (22–25 g body weight) inbred male mice susceptible to smoking-induced emphysema (C57BL/6j, Charles River Laboratories, France) and resistant to smoking-induced emphysema (129S2/SvHsd, Harlan Laboratories, UK) were housed in the Inhalation Core Facility at the Fundación Jiménez Díaz for 1 week of acclimatization before CSE. After CSE, mice were returned to their cages, where a diet of alfalfa-free rodent food (Harlan Teklad) and water was provided ad libitum. Studies described herein were performed under a project license issued by the Ministry of Innovation and Science of Spain, and protocols were approved by the Local Ethical Animal Research Committee at IIS-Fundación Jiménez Díaz, Madrid.
Cigarette Smoke Exposure
Twelve- to 16-week-old mice (six to eight mice per group) were divided into a sham-exposed group (nonsmokers) and a cigarette smoke–exposed group (smokers). CSE was performed in a single exposition using smoke from three consecutive (with 5-minute breaks between them) standard research nonfiltered cigarettes (2R1, University of Kentucky, Lexington, KY; 11.7 mg tobacco particulate matter, 9.7 mg tar, and 0.85 mg nicotine per cigarette). Cigarette smoke was generated by an exposure system and was drawn into the chambers via a peristaltic pump. 36 CSE was delivered in 1 puff/min of 2 seconds' duration and 35 mL volume with fresh air being pumped in for the remaining time. Nonsmoking mice were exposed to filtered air in an identical chamber according to the sample protocol described for CSE. Thirty minutes after the last exposure, blood samples were collected and analyzed for carboxyhemoglobin levels to confirm exposure. No significant change in body weight between the strains of mice was observed in response to exposure to three consecutive cigarettes (data not shown). Animals were sacrificed 24 hours after the third cigarette with an overdose of terminal anesthetic (sodium pentobarbital, 100 mg/kg body weight, intraperitoneally, Abbott Laboratories, Abbott Park, IL) followed by lung extraction in block.
Carboxyhemoglobin Level Measurement
Immediately after removal of the animals from the smoke chamber, blood samples (≈50 μL) were collected in a heparinized tube via puncture of the retro-orbital sinus of each animal. Carboxyhemoglobin levels were measured spectrophotometrically with an IL 682 CO-Oximeter (Instrumentation Laboratory, Lexington, MA).
In Vivo Imaging of Acute Pulmonary Inflammation Model
After 24 hours of tobacco exposition, mice were anesthetized during image acquisition with a mixture of 2% isoflurane and 2 L/min oxygen using an inhalation anesthesia system (Harvard Apparatus, Holliston, MA), and hair from the thorax was removed by shaving and chemical depilation. Approximately 24 hours prior to imaging, mice were injected via the tail vein with MMPSense 680 (2 nmol/150 μL phosphate-buffered saline per mouse; VisenMedical, Woburn, MA). MMPSense 680 is a protease activatable fluorescence in vivo imaging agent that fluorochromes emit in the near-infrared (excitation 680 nm; emission 700 nm), a spectral window, whereas hemoglobin and water absorb minimally so as to allow photons to penetrate for several centimeters in tissue. MMPSense 680 is activated by key MMPs, including MMP-2, −3, −9, and −13; is optically silent in its unactivated state; and becomes highly fluorescent following the protease-mediated activation.
Twenty-four hours after tobacco exposition and MMPSense 680 injection, mice were imaged using a cryogenically cooled charge-coupled device (CCD) camera, the IVIS-Lumina Imaging System (Caliper Life Sciences Inc., Hopkinton, MA), consisting of a 150-watt quartz tungsten halogen lamp with a dichroic reflector, which provides light for fluorescence excitation, and a supercooled CCD camera (at −90°C), with a field of view set at 7.5 cm above the sample shelf, mounted on a light-tight specimen chamber (dark box) and a Windows-based computer system. Grayscale-reflected images and fluorescence-colorized images were superimposed and analyzed using Living Image 3.1 software (Xenogen Corporation, Alameda, CA). Living Image software uses a spectral unmixing technique for reducing the effects of native tissue autofluorescence and calculates the respective contribution of each on each pixel in the image. 37 Regions of interest were created to measure the average radiance (photons/s/cm2/steradian). Signal distribution maps help separate autofluorescence from other probes in the raw images and hence provide better localization for each probe. The fluorescent signal detected from an animal depends on the amount of fluorophore present in the sample and the intensity of the incident excitation light. When we analyzed fluorescent images, the units were the ratio between emission light and excitation light. They were referred to as efficiency. For graphing, values were shown as efficiency from respective nonsmokers together with the standard error of the mean.
An additional image set was recorded every 6 hours for up to 24 hours postinjection of MMPSense 680 (data not shown). In vivo results were from six animals, each bearing control and MMP-expressing pulmonary inflammation, with ex vivo imaging from lungs removed from four of the same animals.
X-Ray Imaging of the Animals
Mice were anesthetized, and after fluorescence imaging, acquisition x-ray images were acquired using the ultrafast Lunar PIXImus Densitometer (GE Medical Systems, Salt Lake City, UT) to in vivo colocalize lung tissue with lung fluorescence signal.
Ex Vivo Imaging of Lung Tissue
Mice were anesthetized, and after fluorescence imaging and x-ray acquisition, animals were sacrificed with an overdose of terminal anesthetic, at which point their perfused lung blocks were removed and rinsed with ice-cold 1× phosphate-buffered saline (50 mL). Images were taken using a cryogenically cooled CCD camera (IVIS Lumina Imaging System, Caliper Life Sciences Inc.) to examine fluorescence signal from MMP cleavage. The acute inflammation and noninfiammation image data sets were shown.
Western Blotting Analysis of Lung Tissue
After protein extraction from lung tissue, samples (20 μg total proteins) were electrophoresed through a 10% sodium dodecyl sulfate–polyacrylamide gel and transferred to a nitrocellulose membrane. After blocking with 5% nonfat powdered milk in Tris-buffered saline, the blot was incubated with rabbit polyclonal anti-MMP-2 antibody, mouse monoclonal anti-MMP-13 antibody (1:1,000 for MMP-2 and 1:400 for MMP-13; Chemicon International, Inc., Temecula, CA), mouse monoclonal anti-MMP-3 antibody, goat polyclonal anti-MMP-12 antibody (1:250 for MMP-3 and 1:500 for MMP-12; R&D Systems, Minneapolis, MN), and goat polyclonal anti-MMP-9 antibody (1:500; Sigma-Aldrich Química SA, Madrid, Spain). After washing with Tris-buffered saline/polysorbate 20, they were incubated with each secondary antibody conjugated to horseradish peroxidase. The blot was developed using the enhanced chemiluminescence method (Amersham Biosciences, GE Medical Systems) according to the manufacturer's instructions. The membranes were reprobed with mouse monoclonal anti-α-tubulin antibody (Sigma-Aldrich Química) to normalize for protein loading. In general, MMPs are secreted as inactive proenzymes (latent form) and need activation by several mechanisms, including enzymatic cleavage, to be fully functional. 38 Active protein fragment of each anti-MMP antibody (65 kDa for MMP-2, 45 kDa for MMP-3, 92 kDa for MMP-9, 48 kDa for MMP-13, and 45 kDa for MMP-12) was scanned by densitometric analysis and normalized to α-tubulin. For graphing, values were shown as arbitrary units from nonsmoker group together with the range.
Statistical Analysis
Results were expressed as mean ± standard error of the mean or mean with the range. Statistical significance was taken as a p value of less than .05 and denoted with an asterisk. Given that pairwise comparisons were of primary interest, a nonparametric (Mann-Whitney) method was performed comparing smoke-exposed animals (smokers) to corresponding time-matched, sham-exposed (nonsmokers) animals, followed by Monte Carlos exact methods within each set of comparisons, using SPSS software (SPSS Inc, Chicago, IL).
Results
Carboxyhemoglobin Levels in Blood of Acute Cigarette Smoke–Exposed Mice
Mice were exposed to the smoke from three cigarettes in a single day. Blood samples in each strain of mice were collected 30 minutes after the last exposure and analyzed for carboxyhemoglobin levels. Importantly, based on carboxyhemoglobin blood measures, levels of smoke exposure were similar to those reported clinically. 39 The carboxyhemoglobin levels for the smoke-exposed group were ≈12.6% ± 2.47% in C57BL/6j and ≈14.8% ± 1.23% in 129S2/SvHsd mice, confirming the correct exposure to tobacco smoke (n = 4).
In Vivo Detection of Cigarette Smoke–Mediated MMP Activity
In vivo detection fluorescence molecular imaging analysis of MMP activity (efficiency) in the lung parenchyma of different strains of mice revealed increased CSE-associated MMP activity in each animal with acute inflammation in comparison with the nonsmoker group (Figure 1A). This increment was significantly higher in the smoker group of C57BL/6j mice (1.93 ± 0.33 times the non-smoker group, n = 4, p = .007) than in the nonsmoker group. However, in the 129S2/SvHsd strain of mice, there were no significant differences between the smoker group and the nonsmoker group (Figure 1B).
X-Ray and Ex Vivo Imaging of MMP Activity in the Lungs of Mice
To colocalize lung position with lung fluorescence signal, x-ray imaging was acquired in a representative live animal after fluorescence imaging analysis (Figure 2A). To localize MMP activity in the pulmonary inflammation at the tissue level, the lung blocks were removed from the live animals 24 hours postinjection of the MMP-activatable fluorescence probe, and the images were acquired by fluorescence molecular imaging, which clearly showed that the fluorescence signal comes from the lung and not from the heart or other organs, such as the liver (data not shown) (Figure 2B).
Western Blotting Analysis for MMP Protein Levels in Lung Tissue
Acute lung inflammation expresses the protein level for several MMP family members, most notably MMP-2, −3, −9, −12, and −13, the expressions of which from lung homogenates were examined 24 hours after CSE (Figure 3). Densitometric analysis showed that MMP-2 active protein fragment levels were significantly increased in the smoker group compared with the nonsmoker group in each strain of mice: C57BL/6j (1.82 ± 0.21 times the nonsmoker group, n = 5, p = .036) and 129S2/SvHsd mice (1.49 ± 0.05 times the nonsmoker group, n = 4, p = .028), although these levels were more markedly increased in susceptible mice than in resistant mice. With respect to the active protein fragment levels of MMP-3, MMP-9, and MMP-13, they were significantly increased in the smoker group compared to the nonsmoker group only in susceptible mice (C57BL/6j): MMP-3, 1.84 ± 0.32 times the non-smoker group, n = 5, p = .009; MMP-9, 2.14 ± 0.28 times the nonsmoker group, n = 6, p = .027; and MMP-13, 1.31 ± 0.04 times the nonsmoker group, n = 5, p = .031. Regarding the MMP-12 active protein fragment levels, they were significantly increased in C57BL/6j mice (1.16 ± 0.02 times the nonsmoker group, n = 4, p = .005) and significantly decreased in 129S2/SvHsd mice (0.71 ± 0.02 times the nonsmoker group, n = 5, p = .021).

Fluorescence molecular imaging of matrix metalloproteinase (MMP) activity in the lungs of living mice. A, Lung imaging in vivo of each group of animals. Male mice were anesthetized and imaged using the IVIS Imaging System 24 hours after exposure to the smoke of three cigarettes. The entire efficiency signal from the thorax was measured with Living Image software using a spectral unmixing technique. One representative smoker mouse and one representative nonsmoker mouse of each strain are shown. B, MMP activity (efficiency from the nonsmoker group) in the lungs from the smoker group (▪) in C57BL/6j susceptible mice and 129S2/SvHsd resistant mice compared to those in the nonsmoker group (□) and expressed as mean ± standard error of mean. *p < .05 or lower versus the respective nonsmoker group (n = 4/group).
Discussion
In this study, we investigated, by imaging techniques, the inflammatory response to acute CSE in terms of the production of MMPs by alveolar macrophages in two strains of mice. For this purpose, mice were exposed to mainstream cigarette smoke using a whole-body exposure system. Our data, obtained by fluorescence molecular imaging, revealed that the in vivo profile of MMP activation within the lung typifies the inflammatory response to tobacco in strains of mice with different susceptibility to developing emphysema, being able to distinguish in the early stages of pulmonary disease susceptible and resistant mice strains. To our knowledge, this is the first recorded study that compares the MMP inflammatory profile in strains of mice with different sensitivity to cigarette smoke by fluorescence molecular imaging. The data presented herein serve to further evince molecular imaging as a technology highly appropriate for research on lung inflammation.
Enzyme-based activatable probes for fluorescence-mediated imaging have proven valuable for the detection of tumor-associated lysosomal protease activity in an animal model30,40 but have never been used to detect MMP activity in lung tissue after acute CSE. We choose a pan-MMP sensor for imaging of enzymes because the lung disease mobilizes multiple MMPs. The presence of these MMPs activated in the airway during inflammation suggests the important role of MMPs in the acute inflammatory response and in the determination of early susceptibility to the development of cigarette smoke–induced emphysema. With the help of an activatable sensor, which reports activity, we have demonstrated that MMP activity, visualized by molecular imaging in the lungs of living mice, is significantly increased in mice strains susceptible to smoking-induced emphysema. This finding is in agreement with the previous data obtained from the analysis performed in an acute model showing early matrix destruction in the first hours after smoke exposure in C57BL/6j mice related to the presence of MMPs. 41 Interestingly, our data demonstrate that two strains of mice with different susceptibility to developing emphysema in chronic CSE have different and distinct inflammatory profiles in the early stages of lung inflammation by fluorescence molecular imaging. The activity of MMPs measured with imaging appears to be related to the amount of each active protein fragment of MMP-2, −3, −9, and −13 measured levels, included in the activatable fluorescence probe. Specifically, lung tissue levels of MMP-9 were more significantly increased in the airways of susceptible mice (C57BL/6j) after acute CSE than leftover MMPs studied according to the immunohistochemical studies that revealed that MMP-2 and MMP-9 expression were important in mediating lung injury. 42 In the case of the resistant mice (129S2/SvHsd), although MMP-9 active protein fragment levels were slightly lower (not significantly) than those in respective nonsmoker group, the overall balance sheet, along with the rest of the MMPs (−2, −3, and −13) detected by fluorescent probe, makes global activity of MMPs, measured by in vivo imaging techniques, greater than control (although not significantly). Imaging of MMP-associated airway inflammation entails several attractive possibilities for translational medicine.

X-ray and ex vivo lung fluorescence signal. A, To colocalize lung position with lung fluorescence signal, x-ray images were acquired in a representative live animal after fluorescence imaging analysis. B, To corroborate the lung fluorescence signal, whole cardiopulmonary block was extracted from the representative live animal and showed the signal of the lung and heart by ex vivo fluorescence imaging.
The results of our study of various strains of mice with different susceptibility to cigarette smoke–induced emphysema attest to the importance of noninvasive fluorescence molecular imaging in discriminating between different levels of inflammatory burden and may suggest a possible biomarker of early sensitivity to tobacco smoke in acute response. These results need to be interpreted with caution because the experiments performed here are short term and the exact correlations between acute connective tissue breakdown and long-term appearance of emphysema are not known. Future studies will be needed to determine whether this technique is also applicable to screening for protease activity in airway pathologies such as COPD.
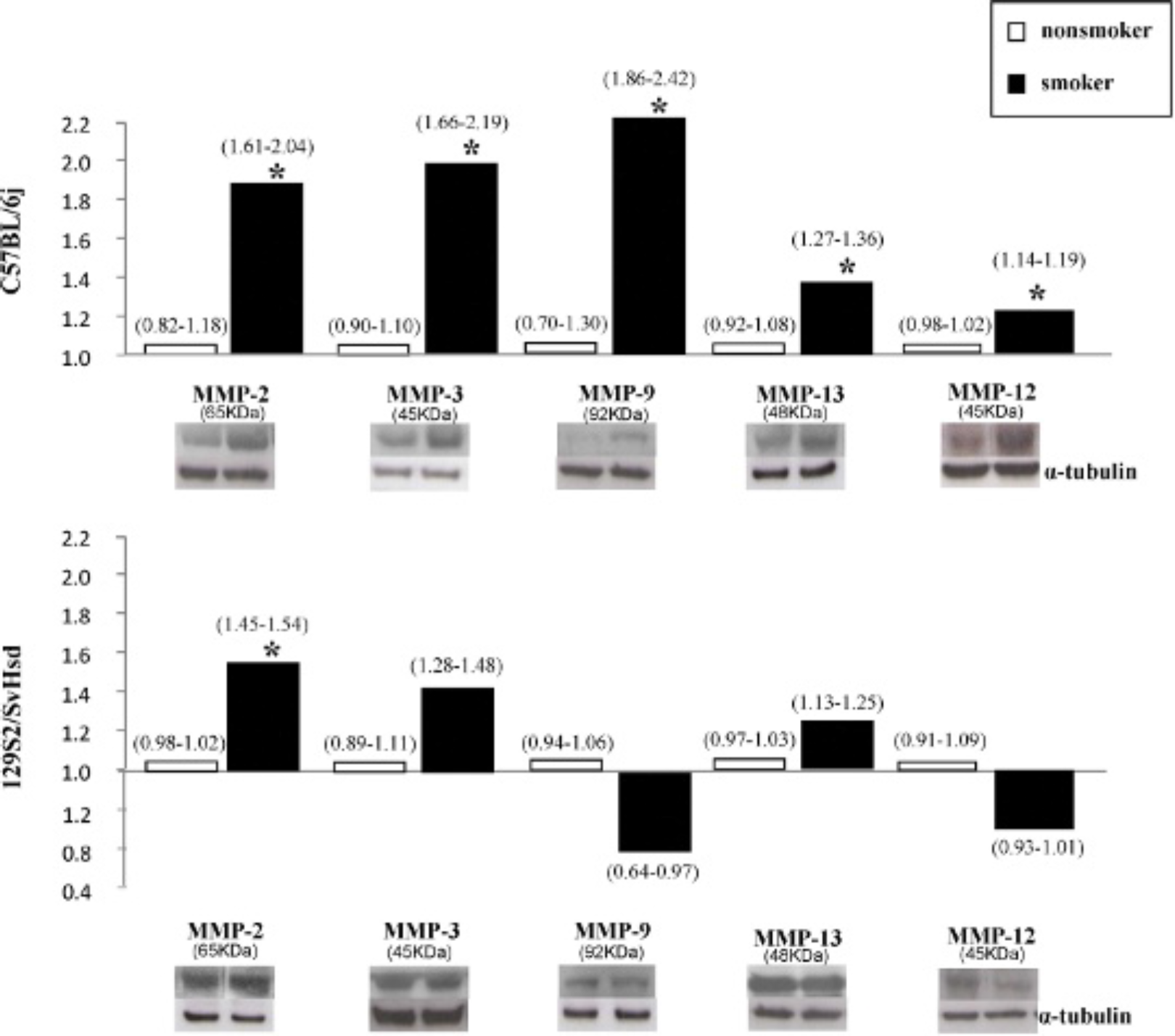
Matrix metalloproteinase (MMP) protein levels from the lung tissue of mice by Western blotting. Detection of MMP-2, −3, −9, −13, and −12 protein levels in lung tissue homogenates was performed. The active protein fragment of each MMP antibody was measured by densitometric analysis and normalized with α-tubulin. Results are expressed as mean (arbitrary units from the respective nonsmoker group) with the range. For each group, n = 4 to 6, *p < .05 or lower versus the respective nonsmoker group.
Footnotes
Acknowledgments
Financial disclosure of authors: This study was supported by the Spanish “Ministerio de Ciencia e Innovatión” (SAF2008-05412-C02- 02), “Sociedad Española de Neumología y Cirugía torácica” (SEPAR-768), and “Centro de Investigatión Biomédica en Red de Enfermedades Respiratorias” (CIBERES06/06/0009).
Financial disclosure of reviewers: None reported.
