Abstract
The purpose of this study was to investigate and compare the feasibility of rat sodium iodide symporter (rNIS) and human sodium iodide symporter (hNIS) as reporter genes for noninvasive monitoring of rat bone marrow mesenchymal stem cells (rBMSCs) transplanted into infarcted rat myocardium. rBMSCs were isolated from rat bone marrow. Adenovirus (Ad) was reconstructed to contain rNIS-enhanced green fluorescent protein (eGFP) or hNIS-eGFP. The transfection efficiency of Ad/eGFP/rNIS and Ad/eGFP/hNIS to rBMSCs was measured by real-time polymerase chain reaction, flow cytometry, Western blot, and immunofluorescence staining. The transfected rBMSCs were transplanted into infarcted rat myocardium followed by a single-photon emission computed tomography (SPECT) study with 99mTc-pertechnetate as the radiotracer and by autoradiography. The isolated rBMSCs were CD29, CD44, and CD90 positive and CD34, CD45, and CD11b negative. The expression of rNIS and hNIS in the transfected rBMSCs at both gene and protein levels was obviously higher than that without transfection. The myocardium of rats transplanted with transfected rBMSCs could be visualized by SPECT owing to the accumulation of 99mTc-pertechnetate in rBMSCs mediated by exogenous NIS genes. The accumulation of 99mTc-pertechnetate in myocardium mediated by rNIS was higher than that by hNIS, which was also confirmed by autoradiography. Both rNIS and hNIS are useful reporter genes to monitor BMSCs transplanted into infarcted myocardium in vivo with rNIS being superior to hNIS as the reporter gene.
WITH INCREASED STANDARD OF LIVING, myocardium infarction becomes one of the diseases with the highest attack rates and mortality. Stem cell transplantation is an effective means of treating infarcted myocardium. Bone mesenchymal stem cells (BMSCs) have been found to stimulate myocardial repair or actual regeneration because of the ability to expand many log-fold in vitro and unique immune characteristics allowing their use as an allogeneic graft.1–3 To promote the effect of repairing infarcted myocardium, BMSCs have been modified in many different ways. For example, the survival number of BMSCs was elevated with Bcl-24 or plasminogen activator inhibitor l 5 ; BMSCs were modified with angiopoietin-1 to improve remodeling after acute myocardial infarction onset 6 ; and BMSCs adhered more strongly to ischemic myocardium with integrin-linked kinase. 7 The exact mechanism of how BMSCs heal infarcted myocardium remains to be elucidated. More recently, it was believed that BMSCs may induce angiogenesis and improve left ventricular function by a paracrine mechanism.8–11
In the process of curing myocardial infarction, it is very important to find a noninvasive method to monitor the transplanted BMSCs at real time in vivo. At present, there are three primary methods to track transplanted BMSCs in vivo, namely, magnetic resonance imaging (MRI),12–14 optical imaging,15–18 and radionuclide imaging.19–21 Radionuclide imaging with high sensitivity, relatively good resolution, and strong penetration power is well suited for imaging rodents, large animals, and humans. The herpes simplex virus type 1 thymidine kinase (HSV1-tk) is a commonly used radionuclide reporter gene using 124/131I-2′-fluoro-2′-deoxy-5-iodo-1-beta-D-arabinofuranosyluracil (FIAU), 18F-9-(4-fluoro-3-hydroxy-methyl-butyl) guanine (FHBG), or 18F-9-[(3-fluoro-1-hydroxy-2-propoxy) methyl] guanine (FHPG) as the reporter probe. 22 Roelants and colleagues monitored BMSCs with HSV1 mutant tk (HSV1-sr39tk) as a positron emission tomography (PET) reporter gene and suggested that transfected BMSCs could be noninvasively visualized by small-animal PET using 18F-FHBG as the tracer. 23 Sodium iodide symporter (NIS) is another popular reporter gene that has unique characters and readily available probes. In addition to iodide, NIS can also deliver other anions, such as 99mTc-pertechnetate, which can be inhibited by sodium perchlorate. Miyagawa and colleagues performed gamma-camera imaging with 123I− and 99mTcO4− as tracers after direct injection of adenoviral particles carrying NIS reporter gene into the myocardium of rats and found that both tracers were suitable for imaging the NIS reporter gene in vivo. 24 NIS reporter gene imaging has also been reported to monitor rat cardiac-derived stem cells transplanted into rat myocardium by single-photon emission computed tomography (SPECT) with 99mTcO4− and PET with 124I−. 25
In this study, we investigated the feasibility of rat sodium iodide symporter (rNIS) and human sodium iodide symporter (hNIS) as reporter genes to monitor the fate of rat bone mesenchymal stem cells (rBMSCs) transplanted into infarcted rat myocardium by using SPECT and 99mTcO4−.
Materials and Methods
Adenoviral Vector Construction
The rNIS plasmid was a gift from Prof. Carrasco. The hNIS plasmid was provided by Prof. Fang and Prof. Tan. The plasmid was excised by EcoRI/HindIII and connected with pShuttle-eGFP-CMV(-)TEMP, and then transferred to a pAdxsi vector. Both adenovirus (Ad)/enhanced green fluorescent protein (eGFP)/rNIS and Ad/eGFP/hNIS plasmids were transferred into HEK293 cells for amplification. The viral particles were collected and the titer was determined following the manufacturer's operating instructions (Sinogene, Beijing, China) after depuration.
BMSC Culture and Characterization
Male Sprague-Dawley rats were obtained from the central animal facility of Tongji Medical College of Huazhong University of Science and Technology. All animal experiments were approved by the Institutional Animal Care and Use Committee. The rats were about 8 weeks old and specific pathogen free (SPF) and weighed 200 ± 15 g. They were sacrificed by cervical dislocation and soaked into 75% alcohol for 10 minutes. The femur, tibia, and humerus were harvested with sterile operation. The bone marrow was collected from the medullary cavity and isolated by density gradient centrifugation using Ficoll separation solution (Biochrom AG) and cultured in Dulbecco's Modified Eagle's Medium (DMEM) including 10% fetal bovine serum (FBS), 100 U/mL penicillin, and 100 mg/mL streptomycin. Cells were cultured at 37°C in an incubator with humidified atmosphere containing 95% air and 5% CO2.
Flow Cytometry Analysis
The rBMSCs were collected when they proliferated to reach about 80% confluence and incubated at room temperature for 30 minutes with 5 µUL monoclonal antibody solution of CD34 (Santa Cruz Biotechnology); CD44 (Serotec); and CD11b, CD29, CD45, and CD90 (Biolegend), respectively. Cells incubated without the antibodies served as control for analysis by flow cytometry (FACSCalibur, BD Biosciences, Becton and Dickson Company, San Jose, CA).
Differentiation of rBMSCs
To induce osteogenic differentiation of rBMSCs, the common DMEM was replaced by osteogenesis culture medium containing DMEM, 10% FBS, glutamic acid, 100 U/mL penicillin, 100 mg/mL streptomycin, 20 mM ascorbic acid, 1 M β-glycerophosphate, and 1 mM dexamethasone for 20 days. Then the cells were stained by alizarin red and observed under a microscope. To investigate the potential of BMSCs to differentiate into adipose, the common DMEM was changed with adipogenic culture medium containing 5 mg/mL insulin, 250 mM 3-isobutyl-l-methylxanthine, 100 mM indomethacin, and 200 µUL 1 mM dexamethasone for 16 days. Then the cells were stained by oil red O and observed under a microscope.
Toxicity of Ad/eGFP/rNIS and Ad/eGFP/hNIS to BMSCs
To study the toxicity of Ad/eGFP/rNIS and Ad/eGFP/hNIS to rBMSCs, the cells were seeded in a 96-well plate for 24 hours, and then the Ad/eGFP/rNIS and the Ad/eGFP/hNIS solutions with different multiplicities of infection (MOI) (0, 100, 200, 400, 800, 1,000, 1,600, 2,000, 3,200, and 4,000) were added and incubated for 4 hours and then replaced with the common DMEM. Forty-eight hours later, methylthiazol tetrazolium 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) (20 µUL/well, 10 mg/mL) was added to the cells. The supernatant was removed and changed with dimethyl sulfoxide (150 µUL/well), and the cells were further incubated for 4 hours. The values of photoabsorption (A) were measured by spectrophotometer at 490 nm wavelength.
Transfection Efficiency of Ad/eGFP/rNIS and Ad/eGFP/hNIS to rBMSCs
To determine the optimal MOI for transfection, cells were seeded in six-well plates for 24 hours before adding the Ad/eGFP/rNIS solution with different MOI (0, 100, 200, 400, 800, 1,000, 1,600, and 2,000) to detect the transfection efficiency of adenovirus to rBMSCs. After 4 hours of incubation, the medium was changed with the common DMEM. After 24 hours, the cells were observed under a fluorescent microscope. The cells were then digested and the transfection efficiency was detected by flow cytometry. The cell-transfected efficiency was measured by flow cytometry after incubation with Ad/eGFP/rNIS at optimal MOI for different times (1, 2, 3, 5, 7, and 9 days). The transfection efficiency of Ad/eGFP/rNIS and Ad/eGFP/hNIS at the optimal MOI for 1 and 2 days was further studied by visualizing eGFP fluorescence and flow cytometry quantification.
NIS Gene and Protein Expression in rBMSCs
The gene and protein expression of rNIS and hNIS in rBMSCs was detected with real-time polymerase chain reaction (PCR) and Western blot, respectively, after Ad/eGFP/rNIS and Ad/eGFP/hNIS were transfected into rBMSCs with an MOI of 1,600 for 48 hours. The transfected rBMSCs were incubated with the appropriate primary antibodies (Santa Cruz Biotechnology) for 24 hours at 4°C, and after adding Cy3-labeled secondary antibody (Sigma-Aldrich), the cells were incubated in the dark for 1 hour. Hoechst 33258 was then added to stain the cellular nuclei for 8 minutes. Finally, the cells were observed under a laser confocal microscope to localize the two NIS antibodies.
Radionuclide Uptake of BMSCs In Vitro
The rBMSCs were seeded in 24-well plates (1 × 105 cells/well) and transfected by the two Ad/eGFP/NIS solutions with different MOI (0, 100, 200, 400, 800, 1,000, 1,600, 2,000) for 48 hours. 99mTc-pertechnetate solution (Atom Gaoke, Beijing, China) (740 MBq/mL, 10 µUL/well) was then added and the cells were incubated for 1 hour at 37°C. The cells were washed twice with ice-cold phosphate-buffered saline (PBS) and dissolved in 0.1% NaOH. The cell lysates were collected and the radioactivity accumulated in the cells was measured by a gamma counter (Perkin-Elmer, Santa Clara, CA). For the blocking experiment, adenovirus-transfected rBMSCs were incubated with 10 mM sodium perchlorate, a potent NIS inhibitor, and 99mTc-pertechnetate. After the workup as described above, cell uptake of 99mTc was measured by a gamma counter.
Myocardial Infarction Model
Twenty-five male Sprague-Dawley rats with 250 ± 25 g body weight were randomly divided into five groups (n = 5/group). After anesthesia with 3% napental, the rats were performed tracheal intubation and provided oxygen by small animal respirator. At the same time skin of limbs and chest of the rats were connected with the electrode of electrocardiogram (ECG). Then the thoracic cavity was opened and the heart was exposed. The left anterior descending coronary artery situated in an interventricular groove near the left coronary vein was ligated. A few drops of 1% lidocaine (0.5 mL) could be dropped on the surface of the heart if ventricular fibrillation appeared to recover sinus rhythm. When the pathologic Q wave was shown on the ECG after 1 hour of ligation, the rats in the control group were each injected with 1 × 106 cells without transfection into the edge of the infarcted myocardium marked with ligation using a nonwounding pin; the rats in the other groups were injected with the same number of cells transfected by Ad/eGFP/rNIS or Ad/eGFP/hNIS for 24 hours into the corresponding myocardium. The thoracic cavity of rats was then closed, and the respirator was removed. Rats were administrated intramuscularly with 200,000 U benzylpenicillin potassium for 3 days to prevent infection after the operation.
SPECT Studies In Vivo
SPECT was performed on all rats 24 hours after the operation was completed. The 10-minute static SPECT images were acquired at the 30-minute, 1-hour, 1.5-hour, and 2-hour time points after intravenous injection of Na99mTcO4 (≈222 MBq/rat). During the scan, the rats were placed in a supine position on a GE Millennium MPR SPECT system equipped with a low-energy, high-resolution collimator (256 × 256 matrices, zoom 1.6). In each image, 128 pixel-sized regions of interest were drawn around the heart and the right upper limb. Mean counts were obtained, and the values of Rheart/Rlimb (denoted as relative uptake ratio [RUR]) were calculated as RURmean. In the sodium perchlorate group, the rats were orally administered sodium perchlorate solution 24 hours before SPECT was performed.
Autoradiography
The rats were sacrificed and the hearts were harvested, rapidly washed with ice-cold PBS, and frozen on dry ice. Then 20 urn slices of the marked myocardium were cut by a cryomicrotome (LEICA - CM1900, Nussloch, Germany), exposed on a phosphorus plate, and then developed in a Cyclone Imager (Perkin Elmer). The images were quantified by the ImageTool IT310 (Perkin Elmer). The ratio of radioactivity in the anterolateral and contralateral walls was obtained.
Biodistribution
The rats were sacrificed at the end of SPECT scans (2-hour time point). The heart, liver, spleen, lung, kidneys, thyroid, stomach, intestine, muscle, bone, and blood were harvested and wet weighed, and radioactivity was measured by a gamma counter. The counts were converted to a percentage of radioactivity per gram of tissue in the total injected dose (%ID/g).
NIS Genes and Proteins Expression in Infarcted Rat Myocardium
The hearts of all rats were removed after SPECT was performed. The marked myocardiums were separated. Real-time PCR and Western blot were performed to test the expression of the rNIS gene and protein in the myocardiums, respectively.
Hematoxylin-Eosin Staining
The marked myocardium tissues were imbedded into the paraffin and then cut into slices with 5 μm thickness, deparaffinaged, hydrated, stained with hematoxylin for 5 minutes followed by staining with eosin for 2 minutes, and then observed under an optical microscope.
Location of Transfected BMSCs in Infarcted Myocardium
The marked myocardial tissue was cut by a cryomicrotome (LEICA - CM 1900) with 15 urn slices after the rats were sacrificed and the hearts were harvested, rapidly washed with ice-cold PBS, and frozen on dry ice. Then the myocardial tissue slides were observed under a fluorescent microscope.
Statistical Analysis
The data were described as mean ± standard deviation. The Student t-test was applied to compare the statistical differences among groups. A p value < .05 was considered statistically significant.
Results
Characterization of rBMSCs
To characterize BMSCs isolated from Sprague-Dawley rats, we analyzed their cell surface markers. Based on the average of three fluorescence-activated cell sorter (FACS) analyses, rBMSCs were uniformly positive for extracellular matrix protein CD90 (98.07 ± 5.62%), β1 integrin CD29 (95.96 ± 2.80%), and hyaluronate receptor CD44 (96.15 ± 3.74%); a low to negligible level of hematopoietic progenitor cell marker CD34 (0.09 ± 0.02%); integrin αM chain GD11b (0.21 ± 0.05%); and leukocyte common antigen CD45 (0.49 ± 0.15%). Overall, the surface marker expression pattern of rBMSCs was consistent with those reported in the literature. 16
To examine the osteoblastogenic differentiation ability of rBMSCs, cells were incubated in osteoblastogenic medium for 7 days. Single cells with osteoblastic morphology were detected among the fibroblast-like cells, including polygon- and cube-shaped cellular elements with a major volume of cytoplasm and nucleus; their quantity increased throughout the observation period. Calcium content could be observed among cells after BMSCs were incubated for 20 days by alizarin red staining (Figure 1A).
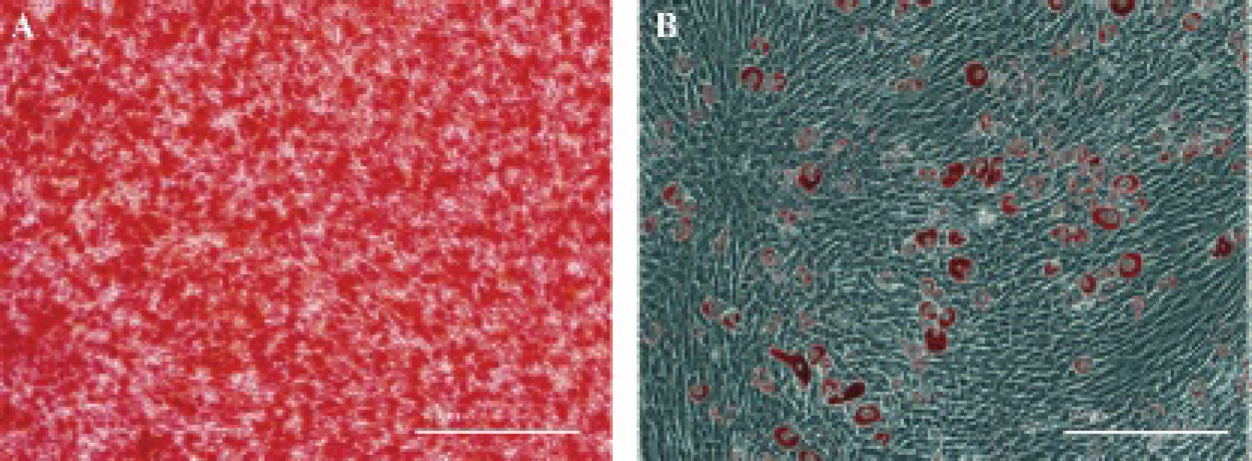
Osteogenic and adipogenic differentiation potential of bone marrow mesenchymal stem cells as assessed by alizarin S (A) and oil red O (B) staining (original magnification X 200).
To examine the adipogenic differentiation potential of rBMSCs, cells were induced to form adipocytes and were assessed by checking for the presence of lipid-rich vacuoles. As determined with this method, approximately 50% of rBMSCs contained granular and drop lipid endocellular actuations positively stained with oil red O after 16 days of adipogenic induction (Figure 1B).
Toxicity of Ad/eGFP/rNIS and Ad/eGFP/hNIS to rBMSCs
The toxic effect of Ad/eGFP/rNIS and Ad/eGFP/hNIS to rBMSCs with different titers is shown in Figure 2A. The cell viability was virtually unaffected when the MOI was less than 2,000 as determined by MTT colorimetric assay. A further increase in MOI induced cytotoxicity to rBMSCs.

The toxicity of Ad/eGFP/rNIS and Ad/eGFP/hNIS to rat bone mesenchymal stem cells (rBMSCs) at different multiplicities of infection (MOI) measured by MTT assay. Cell viability was unaffected until the MOI was more than 2,000. A, The transfection efficiency of different adenoviral vectors to rBMSCs. B, The cells were incubated with Ad/eGFP/rNIS for 1 day (a) and 2 days (b) and incubated with Ad/eGFP/hNIS for 1 day (c) and 2 days (d), respectively, at a MOI of 1,600 and visualized using a fluorescent microscope (X200).
Transfection Efficiency of Ad/eGFP/rNIS and Ad/eGFP/hNIS to rBMSCs
In the preliminary experiment, the transfection efficiency of Ad/eGFP/rNIS and Ad/eGFP/hNIS to rBMSCs was gradually enhanced with the progressive increase in the titer. The transfection efficiency of Ad/eGFP/rNIS to rBMSCs at a MOI of 1,600 was 81.6% and at a MOI of 2,000 was 82.5% after cells were transfected for 24 hours, which was 92.5%, 93.0% 63.6%, 49%, and 13.2%, respectively, after being transfected with a MOI of 1,600 for 2, 3, 5, 7, and 9 days as determined by flow cytometry and confirmed by visualizing eGFP-positive cells under the microscope (data not shown). Given that a MOI of more than 2,000 induced toxicity to rBMSCs, as shown in Figure 2A, there was no significant difference in the transfection efficiency of the adenovirus to rBMSCs between a MOI of 1,600 and a MOI of 2,000 after being transfected for 24 hours, and the highest transfected efficiency appeared when cells were transfected for 48 hours, the optimal transfection condition was thus defined as a MOI of 1,600 and 2 days of transfection. The transfection efficiency of Ad/eGFP/rNIS and Ad/eGFP/hNIS solution to rBMSCs was 81.6%, 92.5%; 80.5%, and 90.7%, respectively, after being transfected with a MOI of 1,600 for 1 and 2 days, and the eGFP-positive cells could be visualized under fluorescent microscopy (Figure 2B).
Expression of NIS Genes and Proteins in Transfected rBMSCs
Real-time PCR showed that the levels of rNIS gene and hNIS gene expression in rBMSCs with a MOI of 1,600 were much higher than those with a MOI of 0 (Figure 3A). Western blot also indicated that the concentrations of the two NIS proteins with a MOI of 1,600 were higher than those with a MOI of 0 (Figure 3B). Under the laser confocal microscope, strong staining of NIS antibody (red) was seen in the endochylmal and cell membrane of transfected rBMSCs (Figure 3D). No positive staining was observed in BMSCs without transfection.

The expression of two NIS genes (A) and NIS proteins (B) in rat bone mesenchymal stem cells (rBMSCs) transfected with Ad/eGFP/rNIS and Ad/eGFP/hNIS at a multiplicity of infection (MOI) of 1,600 measured by real-time polymerase chain reaction and Western blot, respectively. C, The uptake of 99mTc-pertechnetate in rBMSCs transfected with Ad/eGFP/rNIS (•) and Ad/eGFP/hNIS (•) at different MOI. D, Laser confocal microscope visualization of rBMSCs transfected with Ad/eGFP/rNIS (left) and Ad/eGFP/hNIS (middle) at a MOI of 1,600. Strong staining of NIS antibodies (red) was seen in the endochylmal and cell membrane of transfected but not untransfected cells (right).
Radionuclide Uptake of BMSCs In Vitro
To measure the cell uptake of 99mTc-pertechnetate, rBMSCs with or without Ad/eGFP/rNIS or Ad/eGFP/hNIS transfection were incubated with 99mTc-pertechnetate for 1 hour and the accumulated radioactivity in the cells was measured by a gamma counter. Under the reaction condition as described in Materials and Methods, the transfected cells with Ad/eGFP/rNIS at a MOI of 1,600 had more than 90 times higher uptake of 99mTc-pertechnetate and those with Ad/eGFP/hNIS transfection had more than 32 times higher uptake of 99mTc-pertechnetate than the nontransfected wild-type cells (Figure 3C). There was also a significant difference in the uptake between cells transfected by Ad/eGFP/rNIS and by Ad/eGFP/hNIS (p < .001). The uptake can be completely blocked by adding NaClO4 (data not shown).
In Vivo Imaging
The accumulation of rBMSCs in the infarcted rat myocardium could be visualized at different time points after intravenous injection of 99mTc-pertechnetate in the two NIS groups where the infarcted rat myocardiums had intracardiac injection of rBMSCs transfected by Ad/eGFP/rNIS or Ad/eGFP/hNIS 48 hours before the tracer injection (Figure 4A). The RURmean for the rNIS group at 1.5 hours after tracer injection was 10.9 ± 1.1 and that for the hNIS group was 8.0 ± 1.2 (n = 5/group). There was also tracer accumulation in the thyroid and stomach as both organs express endogenous rNIS. 26 The radioactivity in the myocardium was higher than that in the thyroid but lower than that in the stomach. In the control group where the rBMSCs were not transfected with reporter gene, 99mTc was mainly found in the thyroid and stomach; the myocardium RURmean was 3.0 ± 0.2 under the same condition described above, which was significantly lower than that in the rNIS group (p < .001) and the hNIS group (p < .002). The myocardium RURmean in the rNIS group was significantly higher than that in the hNIS group (p = .016). In both the rNIS and hNIS groups, the uptakes in the thyroid, stomach, and myocardium were all effectively blocked by sodium perchlorate, with a myocardium RURmean of 3.2 ± 0.3 (n = 10), which was similar to that in the control group (Figure 4B).
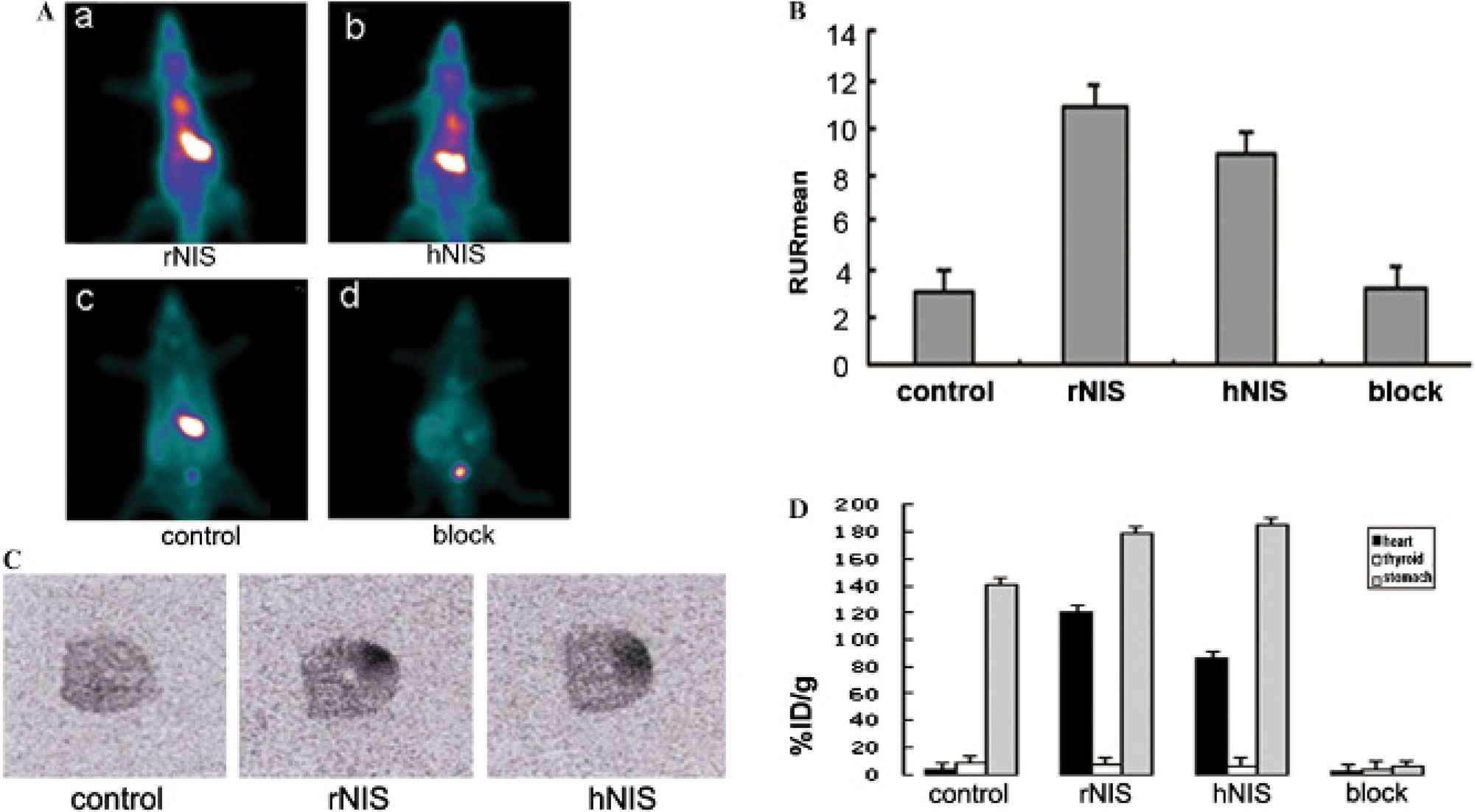
A, Representative single-photon emission computed tomographic plane images of Sprague-Dawley rats at 1.5 hours postinjection of 99mTc-pertechnetate and intracardiac injection of 1 × 106 rat bone mesenchymal stem cells (rBMSCs) 48 hours before tracer injection. (a) rNIS, rBMSCs transfected with Ad/eGFP/rNIS; (b) hNIS, rBMSCs transfected with Ad/eGFP/hNIS; (c) control, rBMSCs without transfection; (d) block, rBMSCs transfected with either Ad/eGFP/rNIS or Ad/eGFP/hNIS; rats were fed with NaClO4. B, The value of the myocardium RURmean in the control, rNIS, hNIS, and block groups. C, An autoradiography study showed increased activity in the anterolateral wall compared to that in the contralateral wall in the rNIS and hNIS groups, whereas no remarkably increased activity was seen in the control group. D, The heart, thyroid, and stomach uptakes of 99mTc expressed as %ID/g in different groups were calculated from direct tissue sampling and gamma-counting, respectively.
Autoradiography
An autoradiography study showed increased activity in the anterolateral wall compared to the contralateral wall in both the rNIS and hNIS groups, whereas no remarkably increased activity in the control group was found (Figure 4C); these findings confirmed the accumulation of 99mTc-pertechnetate in infarcted myocardium transplanted by transfected rBMSCs. The ratio of the anterior wall to the contralateral wall was 21.5 ± 3.4, 15.4 ± 2.6, and 1.4 ± 0.6 in the rNIS group, the hNIS group, and the control group, respectively. The ratio of the anterolateral wall to the contralateral wall was significantly higher in the rNIS group than that in the hNIS group (p = .0009).
Biodistribution
The tissue and organ uptake of 99mTc expressed as %ID/g was calculated from direct tissue sampling and gamma-counting right after the SPECT scans were completed. Similar to what was found in the SPECT studies, the uptakes in the heart, thyroid, and stomach were 120.0 ± 21.0, 7.4 ± 2.1, and 177 ± 15.3 %ID/g for the rNIS group; 85 ± 25.4, 6.8 ± 2.0, and 184 ± 31.5 %ID/g for the hNIS group; 3.5 ± 1.0, 8.8 ± 1.5, and 141.2 ± 14.4 for the control group (n = 5/group); and 2.9 ± 0.5, 2.6 ± 0.5, and 5.6 ± 0.8 for the two NaClO4 blocking groups (n = 10) (Figure 4D). The myocardium uptake in the control group was significantly lower than that in both the rNIS and hNIS groups (p < .001). It was also of note that the myocardium uptake in the rNIS group was higher than that in the hNIS group (p < .001).
Expression of Two NIS Genes and NIS Proteins in the Infarcted Myocardium
The expression of rNIS and hNIS at the gene level was high, with real-time quantitation (RQ) values of 47.7 and 38.7, respectively, whereas the endogenous rNIS level in the control group was rather low (RQ = 5.3) (Figure 5A). The protein expression level in the two NIS groups was also much higher than that in the control group (Figure 5B).

A, The expression of the two NIS genes. The expression of the NIS gene in both the rNIS and hNIS groups was higher than that in the control group. B, The expression of the two NIS proteins in the infarcted rat myocardium transplanted with 1 × 106 rat bone mesenchymal stem cells (rBMSCs). (a) The expression of rNIS protein in the control and rNIS groups and that of hNIS protein in the hNIS group, respectively. (b) β-Actin. C, Hematoxylin-eosin staining of rat myocardium after rBMSC transplantation. (a) The normal cardiac muscle fibers arranged regularly, which presented fasciculated distribution. (b) The infarcted cardiac muscle fibers engorged and arranged disorderedly. They had many vacuoles. Some fibers disrupted, whereas some appeared to have ischemic necrosis. (c) The transverse striation of the fibers disappeared. The nucleus of the cardiocyte disappeared, and the sarcoplasm became an irregular coarse grain. D, The myocardial tissue transplanted with untransfected rBMSCs could not be seen by a fluorescent microscope in the control group (a). Those transplanted with transfected rBMSCs were visualized in the rNIS group (b) and the hNIS group (c) (original magnification X 200).
Hematoxylin-Eosin Staining
Hematoxylin-eosin staining confirmed the success of the myocardial model. Under the optical microscope, the normal cardiac muscle fibers aligned regularly, which presented fasciculated distribution. The infarcted cardiac muscle fibers were engorged and arranged disorderedly with many vacuoles and some disrupted fibers. Ischemic necrosis was also seen. The transverse striation and the nucleus of cardiocytes disappeared. The sarcoplasm became irregular coarse grain (Figure 5C).
Location of Transfected BMSCs in Infarcted Myocardium
Under the fluorescent microscope, the infarcted myocardial tissue transplanted with untransfected rBMSCs could not be seen in the control group. They were visualized in the rNIS group and hNIS group because the rBMSCs transfected by Ad/eGFP/rNIS or by Ad/eGFP/hNIS in the infarcted myocardium expressed eGFP (Figure 5D).
Discussion
Stem cell transplantation is an effective means of treating infarcted myocardium that has been used in the clinic. It is important to monitor the distribution and fate of the stem cells at real time in vivo with noninvasive technology for physicians. Reporter gene imaging with radionuclide as a tracer is suitable for monitoring the transplanted stem cells in vivo. 20 , 23 NIS was a perspective reporter gene with a distinctive character. It is known that not only iodide but also 99mTc-pertechnetate and other negative ions can be accumulated into cells mediated by the NIS gene. Lee and colleagues transfected NIS into ARO human thyroid cancer cells and compared the accumulation extent among 125I, 99mTc, and 188Re in the transfected cells. 27 They found that the cancer cell uptake of 125I in transfected cells was 109 times higher than in nontransfected cells and that of 99mTc-technetium was 21 times and that of 188Re was 47 times higher than nontransfected cells. Zuckier and colleagues further investigated the biodistribution and uptake of these three anions in mice and found a similar pattern. 28 In the current study, we also found high accumulation of 99mTc-pertechnetate in both rNIS- and hNIS-transfected rBMSCs. No active transport was demonstrated in the nontransfected BMSCs or after perchlorate challenge. The transiently transfected rBMSCs injected into infarcted myocardium can also be visualized by SPECT and 99mTc-pertechnetate after cell transplantation, demonstrating the success of our rNIS and hNIS reporter genes approach to tracking mesenchymal stem cell engraftment.
There is a debate as to whether PET or SPECT is preferred for imaging the NIS reporter gene. In general, PET is considered to be more sensitive than SPECT. 29 However, several reports found that the resolution of microPET with 124I tracer was no better than that of SPECT with 99mTc-pertechnetate. 25 , 30 124I is a high-energy emitter with 25.6% positron efficiency and a relatively long positron range, leading to resolution degradation. 31 , 32 The 4.2-day half-life of 124I and the organification of iodine are other major concerns related to the unfavorable radiation dosimetry and its inability to perform longitudinal scans with multiple injections of the 124I tracer. 99mTc-pertechnetate, on the other hand, is a low-energy gamma-emitter with a relatively short half-life (6.01 hours), does not affect the function of thyroid, and appears to be highly suitable for NIS reporter gene imaging. Our in vivo SPECT results clearly demonstrated the ability of SPECT and 99mTcO4−to follow the fate of transplanted BMSCs through imaging of the two NIS reporter genes.
It is of note that we compared rNIS and hNIS in our study. In vitro uptake of 99mTc-pertechnetate in rNIS-transfected rBMSCs was 90 times higher than that in nontransfected cells, and the uptake was 32 times higher in hNIS-transfected cells than in nontransfected cells. Heltemes and colleagues previously compared the uptake of radioiodide in several cancer cell lines transfected by rNIS and hNIS and found higher uptake in rNIS-expressing cells than in hNIS-expressing cells. 33 The mechanism has not been clearly elucidated but is likely to be related to the transcriptional regulation of the NIS gene. The NIS upstream enhancer (NUE) presents the most important aspect in NIS gene regulation. There is over 94% sequence homology between rat NIS upstream enhancer (rNUE) and mouse NIS upstream enhancer (mNUE), whereas there is only 69% between rNUE and human NIS upstream enhancer (hNUE). 34 In our in vivo study, infarcted myocardium transplanted by rBMSCs expressing rNIS had significantly higher concentration of 99mTc-pertechnetate than those expressing hNIS by SPECT planar scanning, autoradiography, and biodistribution. The use of rNIS in the study of stem cell engraftment to the rat myocardium is thus more relevant than the use of hNIS.
Given that the adenovirus vector cannot be integrated into the deoxyribonucleic acid (DNA) of BMSCs and would be slowly cleared out following the metabolism of the cells, the reporter gene can stay in the cells for less than half a month. In our preliminary study after intracardiac injection of Ad/eGFP/rNIS-transfected rBMSCs, the heart could be clearly visualized 1 day after the cell transplantation with the best heart-to-background contrast on days 2 and 3, and the heart uptake of 99mTc was subsequently gradually decreased to the background level by day 12. No obvious leakage of transplanted cells was observed (data not shown). Despite the effectiveness of SPECT and 99mTc to track the disposition and the short-term fate of Ad/eGFP/rNIS-transfected BMSCs, the transient transfection with adenoviral vector causes some complications in interpreting the results. It is unclear whether the faded SPECT signal in vivo at late time points after stem cell transplantation is due to cell death, the clearance of transiently transfected adenoviral vectors, or both. Our future work would involve the transfection of BMSCs with a lentiviral vector, which could be integrated into the DNA of target cells, resulting in continued expression of reporter gene and/or therapy gene for up to several months, to monitor the therapeutic effect of cells/gene to infarcted myocardium. A combination of bioluminescence imaging 16 , 35 and rNIS gene imaging would be another option to better visualize the distribution, homing, and differentiation of injected BMSCs.
Conclusion
rBMSCs can be transfected with rNIS and hNIS via an adenoviral vector to accumulate pertechnetate. The disposition and fate of the cells transplanted to infarcted rat myocardium can be visualized by longitudinal SPECT in the presence of 99mTcO4−. The rNIS gene is more powerful than the hNIS gene in reporter gene imaging.
Footnotes
Acknowledgments
We thank Prof. Carrasco for providing the rNIS plasmid and Profs. Peihua Fang and Jian Tan for providing the hNIS plasmid and Drs. Weijing Xie and Wei Li for their excellent technical support.
Financial disclosure of authors: This study was supported, in part, by the China Postdoctoral Award (20090451053) and the Open Foundation of Hubei Province Key Laboratory of Molecular Imaging (2009-27).
Financial disclosure of reviewers: None reported.
