Abstract
Nano-sized particles are widely regarded as a tool to study biologic events at the cellular and molecular levels. However, only some imaging modalities can visualize interaction between nanoparticles and living cells. We present a new technique, pulsed magnetomotive ultrasound imaging, which is capable of in vivo imaging of magnetic nanoparticles in real time and at sufficient depth. In pulsed magneto-motive ultrasound imaging, an external high-strength pulsed magnetic field is applied to induce the motion within the magnetically labeled tissue and ultrasound is used to detect the induced internal tissue motion. Our experiments demonstrated a sufficient contrast between normal and iron-laden cells labeled with ultrasmall magnetic nanoparticles. Therefore, pulsed magnetomotive ultrasound imaging could become an imaging tool capable of detecting magnetic nanoparticles and characterizing the cellular and molecular composition of deep-lying structures.
UNRAVELING OF THE molecular pathways of diseases and thus detecting pathology at early stages of development are possible through monitoring the cellular and molecular mechanisms of diseases. Thus, significant efforts have been directed toward the design and development of new imaging techniques capable of visualizing biologic events at the molecular and cellular levels. Molecular imaging, in contrast to classic diagnostic imaging, aims to probe specific molecules or cells that are the basis of disease at the early stages of development. 1 Molecular imaging methods are based on detecting a contrast between background tissue and specifically targeted cells or molecules. Therefore, using contrast agents that can bind to a specific biologic entity is an essential requirement in any molecular imaging technique. These contrast agents should be biocompatible, be able to overcome biologic delivery barriers (vascular, interstitial, cell membrane), bind to a specific cell or molecule, be retained long enough to be detected, and provide a clear contrast between normal cells and cells with desired targeted molecules. 2
Nanoparticles are attracting considerable interest as contrast agents in many molecular imaging modalities. The small size of nanoparticles makes them proper for binding to a biologic entity of interest. In addition, the development of biocompatible nanoparticles that have the desired molecular targeting moiety provides a controllable “tagging” mechanism. Metal-based nanoparticles are being extensively used as molecular-specific contrast agents for various imaging modalities, such as optical imaging and magnetic resonance imaging (MRI). 3
Among various types of particles, magnetic nanoparticles are already used in a wide range of biomedical applications. Their controllable small size and the magnetic response to an external magnetic field gradient made them a suitable tool in several applications of biomedicine, such as molecular MRI, cell labeling and magnetic separation, drug delivery, and hyperthermia. 4 During the past decade, several different types of magnetic nanoparticles have been introduced as MRI contrast agents. Because of their small size, magnetic nanoparticles can cross the leaky tumor microvasculature, accumulate in tumors, and bind to a specific cell. 5 Moreover, these nanoparticles remain in the blood circulation system without causing blood vessel embolism. The magnetic properties of these nanoparticles above a certain temperature (blocking temperature) are identical to those of superparamagnetic materials with a large magnetic moment and thus large susceptibility. These superparamagnetic nanoparticles do not magnetically interact with each other in the absence of an external magnetic field because their net magnetic moments are randomized and vanish macroscopically. Therefore, magnetic nanoparticles are very stable under physiologic conditions and thus are very attractive for biomedical applications. 6
Currently, radionuclide imaging, MRI, and optical imaging are the major molecular imaging modalities. Positron emission tomography (PET) and single-photon emission computed tomography (SPECT) use radionuclide probes to provide quantitative information on the cellular and molecular content of tissues. These techniques can visualize very low concentrations of radionuclide probes. However, the spatial and temporal resolutions of radionuclide imaging are limited. Moreover, in radionuclide imaging, biologically important chemicals are labeled with radioactive materials; therefore, there are concerns about the biologic safety of tissues exposed to radioactive materials. MRI molecular imaging uses superparamagnetic agents to label the molecules and cells and detect them through the change in the magnetic properties (ie, relaxation time) of tissue. Although molecular MRI has high spatial resolution, it is relatively expensive and not a real-time imaging modality. Optical molecular imaging systems use optical detectors to detect the light emitted by fluorescent and bioluminescent probes in visible or near-infrared wavelengths. Unfortunately, the light can travel only a few hundreds of micrometers in tissue before it scatters; therefore, the optical imaging is depth limited. Consequently, there is a need for a robust functional and structural molecular imaging technique that can overcome the limitations of current molecular imaging modalities.7–9
Ultrasound imaging, a nonionizing, real-time, cost-effective imaging modality, can be a suitable candidate for molecular imaging. However, ultrasound imaging is not capable of visualizing events at the molecular and cellular levels because of limited spatial resolution and contrast. Ultrasound contrast agents such as microbubbles, perfluorocarbon droplets, and liposomes have been introduced to enhance and improve the quality of ultrasound images, but these agents are not ideal for cellular and molecular ultrasound imaging. Microbubbles are too large to cross the microvasculature and bind to specific cells. They also have short life spans in the circulation system and may collapse when exposed to ultrasound pressure waves. Perfluorocarbon droplets and liposomal contrast agents do not produce strong ultrasound reflections and thus cannot provide significant contrast in ultrasound images.2,10 Finally, metal nanoparticles cannot be directly used as molecular probes in ultrasound imaging because of their small size and weak ultrasound reflectivity.
However, ultrasound-based imaging techniques can be used to image molecularly targeted nanoparticles. Magneto-motive ultrasound (MMUS) imaging,11,12 which is capable of visualizing magnetic nanoprobes through their dynamic response to an externally applied magnetic field, is an example of such an imaging technique. In MMUS imaging, a high-strength continuous-time harmonic magnetic field is used to induce motion within the magnetically labeled tissue and ultrasound imaging is used to detect the internal tissue displacement. However, imaging of deep-lying tissue structures with higher sensitivity requires stronger magnetic excitation because the induced tissue motion is proportional to the magneto-motive force, which, in turn, is proportional to the magnetic flux density squared. 13 Unfortunately, a harmonic magnetic field of finite amplitude can generate heat in magnetically labeled cells and tissue; indeed, thermal damage is one of the possible concerns associated with the safety of harmonic MMUS imaging. Consequently, using MMUS to image deeper tissue structures is limited by thermal constraints. 14 Furthermore, several hardware limitations are associated with the relatively long operation time in harmonic MMUS that generates an enormous amount of heat within both the current amplifier and the magnetic coil. Moreover, the strong harmonic magnetic field requires a high current to drive the coil, which is not feasible at higher frequencies owing to coil inductance. To overcome these limitations, we developed a pulsed magneto-motive ultrasound (pMMUS) imaging approach capable of high-sensitivity imaging of magnetic nanoparticles at significant depth. In pMMUS imaging, a pulsed magnetic excitation is used instead of a continuous-time field, whereas the contrast mechanism remains the same as in MMUS imaging.
When a magnetically labeled tissue is exposed to an external time-varying magnetic field, the magnetic moments of nanoparticles are aligned with the magnetic field. Therefore, the magnetic nanoparticles and thus the associated tissue tend to move toward lower magnetic potential. The motion of a magnetic particle depends on the spatiotemporal characteristics of the magneto-motive force and on the viscoelastic properties of the surrounding tissue. The magneto-motive force (Fm) acting on a magnetic nanoparticle and causing displacement of tissue can be expressed as
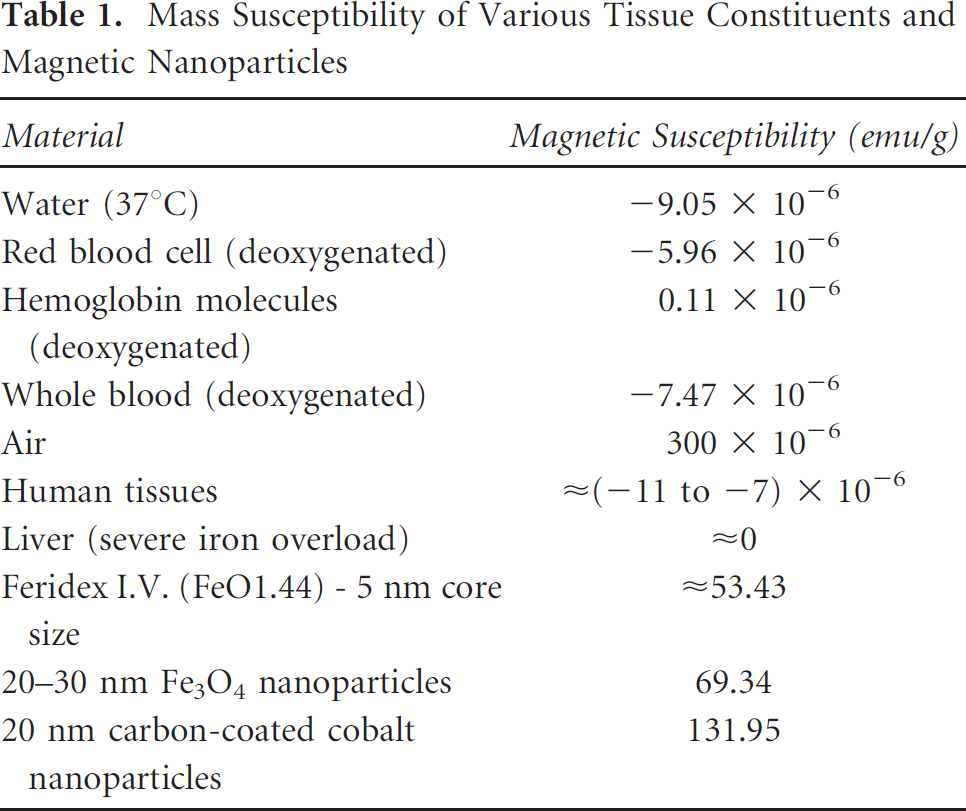
The larger magneto-motive force can be obtained by using materials that exhibit larger magnetic susceptibility. Table 1 compares the magnetic properties of several types of tissues or cells with magnetic materials that are commonly used in the synthesis of magnetic nanoparticles. Negative values for mass magnetic susceptibility indicate the material with diamagnetic properties. The mass magnetic susceptibility for magnetic nanoparticles was measured using a superconducting quantum interference device (SQUID) susceptometer. To compare the magnetic properties of soft tissue and the magnetic nanoparticles quantitatively, the volume magnetic susceptibilities of various human soft tissue were converted to mass magnetic susceptibilities, assuming that the densities of red blood cells (deoxygenated), hemoglobin molecules (deoxygenated), whole blood (deoxygenated), and human tissue (including liver) are 1,093 kg/m3, 1,335 kg/m3, 1,057 kg/m3, and 1,050 kg/m3, respectively. 15 The significant difference between the magnetic susceptibility of normal tissue and the magnetic nanoparticles is the basis of the contrast mechanism in pMMUS imaging.
Mass Susceptibility of Various Tissue Constituents and Magnetic Nanoparticles
Materials and Methods
In our studies, superparamagnetic iron oxide (SPIO) nanoparticles (Feridex I.V., Bayer Healthcare, Inc., Pittsburgh, PA) were used as magnetic nanoprobes. The size and morphology of the magnetic nanoprobes were characterized by transmission electron microscopy (TEM) and dynamic light scattering (DLS). Whereas the TEM-measured iron oxide core size of particles was about 5 nm (Figure 1A), the DLS-measured volume-averaged hydrodynamic diameter of the nanoprobes, including the dextran coating, was about 87 nm (Figure 1B). The induced magnetization of the nanoprobes was measured at 300 K using a SQUID. The sharp increase in magnetization (from 0 to 80% saturation) at a relatively low (0–1 T) field strength along with the saturation value of 53.43 emu/g of iron oxide is typical for this type of small nanoprobe (Figure 1C).

A, High-resolution TEM image of the nanoprobes. B, DLS measurement of the nanoprobes. C, SQUID measurements of magnetization of the nanoprobes at a different applied field.
The block diagram of the custom-built pMMUS imaging system is shown in Figure 2. A focused high-frequency single-element ultrasound transducer and an ultrasound pulser/receiver were used to capture an ultrasound radiofrequency (RF) signal. The magnetic field generator was based on a commercially available magnetic pulser (MPG5, SOTA, Inc., Penticton, BC). To meet the imaging system requirements, the pulser was modified to output a trigger signal prior to the magnetic pulse. The pulser was connected to a coil consisting of 270 turns with the inductance of 2.5 mH and the DC resistance of 0.56 Ω at room temperature. The coil measured 60 mm in outer diameter, 20 mm in inner diameter, and 20 mm in height. To increase the magnetic flux density and to localize the magnetic field in the desired imaging region, a custom-built cone-shaped iron core, made of ferritic stainless steel, was embedded into the center of the coil. Ferritic stainless steels have soft magnetic properties such as high relative magnetic permeability, a low coercive force, and low residual induction. The core was cylindrically shaped with a 5 mm diameter and a 21 mm height. To better focus the field into a smaller region of interest, a symmetric conic frustum was cut at 56° on the top side. The conical region measures 3 mm tall, and the cone tapers up from the 5 mm cylindrical diameter to a diameter of 1 mm. The magnetic pulse strength, measured 5 mm above the iron-core tip using a digital gaussmeter (DSP 475, Lakeshore Inc., Westerville, OH), was 0.6 T. Prior to the experiment, the focal point of the ultrasound transducer and the iron-core tip were aligned. To obtain grayscale and magneto-motive ultrasound images, the samples were placed in a water cuvette and mechanically scanned over the region of interest using a motion axes system (Zaber Technologies Inc., Vancouver, BC). A microprocessor-based unit was used to control all of the timing and positioning signals. 16 Specifically, the trigger pulse, issued several milliseconds ahead of the magnetic pulse, was used to initiate the ultrasound pulser/receiver as well as the data acquisition unit (12-bit A/D converter operating at a 200 MS/s sampling rate). The same unit controlled the duration of the captured ultrasound pulse-echo RF signal. Furthermore, the unit was designed to send the signal to motion axis after the required data were captured at a certain position.

Diagram of a custom-built pulsed magneto-motive ultrasound (pMMUS) imaging system.
To measure the magnetically induced internal motion within the samples, pulse-echo ultrasound signals before, during, and after the application of the magnetic pulse were captured. The relatively short (6–10 ms) magnetic pulses caused the iron-laden tissue to move, reaching the maximum displacement within a few milliseconds after the magnetic pulse. Therefore, ultrasound pulse-echo RF signals were collected for 50 ms after application of the magnetic pulse. To ensure reliable measurement of the tissue displacement, the high (5 kHz) pulse repetition frequency was used to generate pulse-echo ultrasound signals every 200 μs. The tissue motion was detected using a block-matching, motion-tracking algorithm where cross-correlation between a reference RF signal captured before the magnetic pulse and the consecutive RF signals was used to find the tissue displacement. 17 In this correlation-based speckle-tracking method, the displacement is estimated by finding the time delay between the reference signal and the captured signal at different time points through a maximum likelihood estimator. 18 To eliminate the bulk motion of the sample, the bottom of the water cuvette was used as a stationary reference; the detected motion of the boundary was subtracted from the sample motion. The maximum displacement at each position within the imaging plane was then used to form the magneto-motive ultrasound image.
To test the sensitivity of the custom-built pMMUS system, experiments were performed using 2 mm diameter cylindrical tissue-mimicking samples made out of 8% polyvinyl alcohol (PVA) mixed with different concentrations of magnetic nanoprobes (3.5, 8.5, 17, and 34 μmol Fe per cubic centimeter of mixture). The control PVA sample had no magnetic nanoprobe. In addition, 0.5% of 15 μm silica particles were added to the samples to provide ultrasound contrast. The prepared solutions of PVA and additives underwent four 12-hour freeze/thaw cycles to produce viscoelastic samples closely resembling the acoustic and mechanical properties of tissue. 19 Tissue-mimicking samples were mechanically scanned using a single-element focused ultrasound transducer (48 MHz center frequency, 5.5 mm focal depth, f-number of 1.4). The size of the lateral steps was selected to be less than half of the ultrasound beam width (25 μm) to satisfy the sampling requirement.
To demonstrate the capability of pMMUS as a cellular/molecular imaging technique, a set of experiments was performed using mouse kidney tissue injected with magnetically labeled macrophages suspended within 10% gelatin. The murine macrophages (J774A.1 cell line) with a high rate of nonspecific uptake were cultured in Dulbecco's Modified Eagle's Medium supplemented with 5% fetal bovine serum at 37†C in a 5% CO2 environment. To magnetically label cells, macrophages were incubated in a suspension of Feridex I.V. solution diluted to 0.2 mg/mL Fe. After 24 hours' incubation, cells were harvested and mixed with 10% gelatin and 0.5% of silica particles. The mixture, kept at 35†C temperature, was injected into a parenchyma of a fresh mouse kidney, and the sample was then cooled down to allow the gelatin implant with cells to harden. Prior to the imaging experiment, the kidney was positioned and restrained in a water cuvette. Ex vivo experiments were performed using a single-element ultrasound transducer (7.5 MHz center frequency, 50.8 mm focal depth, 12.7 mm diameter aperture, f-number of 4) scanned laterally with 200 μm steps. Tissue displacements less than a certain threshold (15 μm) were considered background noise and were eliminated from the magnetomotive ultrasound image.
Results and Discussion
The grayscale ultrasound and magneto-motive ultrasound image of tissue-mimicking samples are presented in Figure 3, A and B, respectively. Ultrasound attenuation is increasing at higher frequencies and is significant at 48 MHz. 20 The ultrasound signals received from the lower portion of the inclusions are more attenuated compared to signals from the upper part of the inclusion. In addition, the nonuniformity of the ultrasound backscattered signal could be attributed to nonuniform distribution of silica particles within the inclusion. As evident from images in Figure 3A, ultrasound imaging cannot differentiate between inclusions containing different concentrations of magnetic nanoprobes. However, the magneto-motive ultrasound imaging (Figure 3B) not only identifies the iron-laden inclusions but also demonstrates the dependence of pMMUS signal on the concentration of magnetic nanoprobes. Figure 3C represents the average displacement within each inclusion containing different concentrations of nanoprobes. There was a small (< 25 μm) displacement measured within the control (no nanoprobes) sample; this is due to the mechanical vibration transmitted to the sample via the water cuvette. The variation in measured displacements is attributed to several factors, including the nonuniform distribution of magnetic agents within the mixture, pulse-to-pulse variations of magnetic field, and small error in correlation-based motion estimator. Nevertheless, the sensitivity of the pMMUS method is high and can be further improved by increasing the strength of the applied magnetic field and by using magnetic nanoprobes with higher magnetic susceptibility.

(A) Grayscale ultrasound image and (B) pulsed magnetomotive ultrasound image of tissue-mimicking samples with different concentrations of SPIO (left to right: 34, 0.0, 8.5, 3.5, and 17 μmol Fe/mL mixture). Motion within the samples was induced by a magnetic field measuring 6,000 gauss near the inclusions. The field of view in both images is 20 mm by 7.78 mm. C, The mean and standard deviation of the displacement measured in samples with different concentrations of magnetic nanoprobes.
Figure 4 summarizes the results of the ex vivo pMMUS imaging of a tissue sample with magnetically labeled cells. The dark-field microscope images of control (see Figure 4A) and labeled (see Figure 4B) cells clearly demonstrate the uptake of iron oxide nanoprobes by cells. Although the ultrasound image (see Figure 4C) identifies the location of the gelatin inclusion within the background tissue, the contrast in this image is not related to the presence of the nanoprobes. Indeed, the size of nanoprobes is too small compared to ultrasound wavelength, and the low concentration of nanoprobes does not significantly affect the acoustic properties of the tissue (see, for example, Figure 3A). However, the magneto-motive ultrasound image (see Figure 4D) clearly detects the presence of magnetically labeled cells within tissue background.
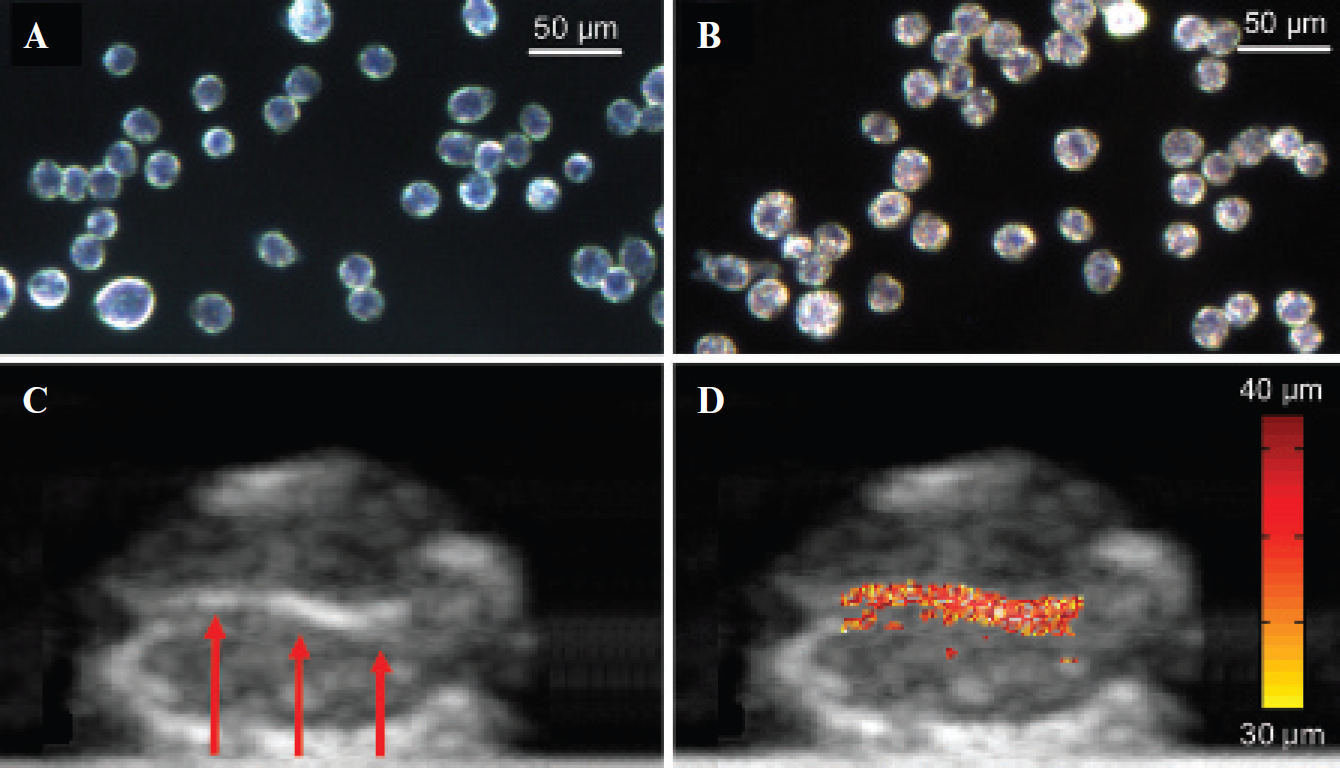
Optical dark-field reflectance images of (A) intact murine macrophages and (B) cells labeled with SPIO. (C) B-scan ultrasound image and (D) combined ultrasound and pulsed magneto-motive ultrasound image of a murine kidney with inclusion. The arrows in the ultrasound image indicate the location of magnetically labeled cells injected into the kidney.
In this experiment, the kidney tissue served as the control because of negligible magnetic susceptibility of kidney tissue compared to the magnetically labeled cells. The small difference between the kidney and injected cells in the B-scan ultrasound image is related to structural differences between the kidney tissue and cells within gelatin. Contrarily, the magneto-motive ultrasound image exhibits significant contrast between the cells and background tissue based on the presence of magnetic nanoparticles within the labeled cells. The tissue motion is primarily confined to the inclusion, although there is negligible motion near the inclusion owing to the mechanical coupling between the inclusion and background tissue. However, the background motion is rapidly reduced in regions that are farther away from the inclusion. The average displacement measured within the relatively stiff gelatin inclusion was tens of micrometers. A softer background is anticipated in in vivo experiments. Therefore, the results presented in Figure 4 suggest that pMMUS imaging has sufficient sensitivity to localize the magnetic nanoprobes in tissue.
The current imaging system was based on a mechanically scanned single-element ultrasound transducer and, therefore, was not capable of real-time imaging; the postprocessing algorithms were required to reconstruct both ultrasound and pMMUS images. Real-time magneto-motive ultrasound imaging is possible by using a high-frequency array-based ultrasound imaging system along with a modified magnetic field generator that provides a strong magneto-motive force in the imaging region. Furthermore, normalizing the tissue motion with respect to the magneto-motive force can provide more accurate information on the magnetic content of the tissue.
The sensitivity of the pMMUS system depends on several parameters, including the magnetic excitation field, magnetic properties of the nanoprobes, concentration of nanoprobes within the object, and viscoelastic properties of the surrounding medium. The strength of the magnetic field can be enhanced by using a high-power amplifier that can drive the high inductive coils with a large amount of current. Furthermore, the shape and duration of the magnetic pulse can also affect the sensitivity of pMMUS imaging. Using nanoprobes with higher magnetization, such as iron-cobalt nanoparticles, can lead to higher sensitivity owing to the larger magneto-motive force and thus larger tissue displacement.21–26 Increasing the concentration of magnetic nanoprobes in the desired region can also improve the signal to noise ratio in pMMUS images. The concentration of nanoparticles can be increased by using magnetic nanoprobes that have the desired targeting moiety.27–30 Furthermore, the magnetic properties of nanoprobes provide the opportunity to deliver more nanoprobes to a desired site by using an external static magnetic field.31–34 Consequently, pMMUS imaging can be suitable for in vivo applications where magnetic nanoprobes are targeted to specific biomarkers. For in vivo applications, one of the main challenges will be to separate the magnetically induced displacement from the tissue displacement induced by the cardiac and respiratory systems. Several studies have shown that “motion gating” algorithms can effectively minimize the effect of cardiac and respiratory motion during in vivo imaging.35–37 In pMMUS imaging, the magnetically induced motion within the tissue is time controlled through triggering of the magnetic excitation pulse and therefore can be similarly time-gated, thus helping to differentiate between natural and induced tissue motion. Moreover, the relatively rapid magnetically induced motion of the iron-laden tissue with well-known temporal behavior can be separated from slowly varying background tissue motion introduced by respiratory or cardiac activity. In clinical implementation of the pMMUS system, an ultrasound transducer array will allow electronic scanning of the ultrasound beam, eliminating the necessity of any mechanical movement. Furthermore, the magnetic field may be produced by an external coil or a coil incorporated within a handheld ultrasound probe.
Interestingly, given that the dynamic behavior of magnetic nanoprobes depends on the mechanical properties of the tissue, the measured motion can be used to assess the mechanical properties of the surrounding tissue. 38 Therefore, pMMUS can be developed into an imaging system capable of “remote palpation” of tissue, allowing in vivo assessment of mechanical properties of soft tissues.
In conclusion, pMMUS imaging, augmented by small, nontoxic magnetic nanoprobes, was introduced as a new imaging modality capable of visualizing events at the molecular and cellular levels. The pulsed nature of pMMUS imaging allowed a higher magnetic field to be applied to tissue, thus enabling the imaging of deeper tissue structures with sufficient sensitivity.
Footnotes
Acknowledgments
The authors are grateful to Mr. Russell Torlage (SOTA Inc.) for technical help with customizing the magnetic pulser and Dr. Keith P. Johnston, Dr. Konstantin Sokolov, Mr. Li L. Ma, and Ms. Kimberly Homan for the helpful discussions and characterization of magnetic nanoprobes.
Financial disclosure of authors: This work was partially supported by the National Institutes of Health under grant EB 008821.
Financial disclosure of reviewers: None reported.




