Abstract
Owing to increased needs for positron emission tomography (PET), high demands for a wide variety of radiolabeled compounds will have to be met by exploiting novel radiochemistry and engineering technologies to improve the production and development of PET probes. The application of microfluidic reactors to perform radiosyntheses is currently attracting a great deal of interest because of their potential to deliver many advantages over conventional labeling systems. Microfluidics-based radiochemistry can lead to the use of smaller quantities of precursors, accelerated reaction rates, and easier purification processes with greater yield and higher specific activity of desired probes. Several proof-of-principle examples along with the basics of device architecture and operation and the potential limitations of each design are discussed. Along with the concept of radioisotope distribution from centralized cyclotron facilities to individual imaging centers and laboratories (“decentralized model”), an easy-to-use, stand-alone, flexible, fully automated, radiochemical microfluidic platform can provide simpler and more cost-effective procedures for molecular imaging using PET.
Molecular Imaging with Positron Emission Tomography
POSITRON EMISSION TOMOGRAPHY (PET) is an exquisitely sensitive, noninvasive imaging technique for visualizing molecular processes in humans and animals.1,2 It measures the spatial and temporal distribution of positron emitters in the human body by coincidence detection of annihilation photons on positron decay. Based on data collected from those events, PET systems generate three-dimensional pictures from tracer locations in the tissue or organs of a patient. In addition, PET allows real-time studies of pharmacokinetics and pharmacodynamics of tracers or radiolabeled drugs inside the human body. Clinically, PET is heavily used in oncology for tumor imaging and detection of metastases. In neurology, PET has found application for diagnosis of certain diffuse brain abnormalities and neurodegenerative diseases. Equivalently, PET has become an important research tool to map human brain and heart function and visualize specific biologic processes in normal or diseased animal models to facilitate drug discovery and development. Over the past four decades, advances in instrumentation, basic biochemical research, and clinical investigations, as well as radiochemistry for probe production, have merged to make PET a truly powerful scientific and clinical tool to directly evaluate many critical in vivo biologic functions.
To obtain PET images, a radiolabeled probe targeted toward a specific biologic entity or incorporated into certain metabolic pathways is administered to the subject under study. So specific PET probes are developed to meet the criteria required for imaging selected biochemical and physiologic processes in vivo. Several short-lived positron emitters, such as 15 O, 13 N, and 11 C (half-lives [T1/2] = 2, 10, and 20.4 minutes, respectively), are isotopes of key elements of living organisms that extend the possibility of synthesizing radiolabeled endogenous tracers for imaging studies of specific biochemical processes. Alternatively, radiohalogens, such as 18 F, 76 Br, and 124I, with longer half-lives (110 minutes, 16 hours, and 4.3 days, respectively), allow different synthetic strategies and imaging protocols. Positron-emitting metals, such as 64 Cu (T1/2 = 12.8 hours), produced by charged-particle nuclear reactions, and 68 Ga (T1/2 = 68 minutes), obtained from a generator, are also gaining interest and use.3,4 Despite not being ideal for all applications, 18 F is easily produced in biomedical cyclotrons and its half-life is sufficient to permit complex or multistep radiochemical syntheses, delivery to off-site imaging centers, and extended in vivo PET. Therefore, currently, 18 F is widely used clinically because its low positron energy (0.64 MeV) limits the patient's absorbed dose and offers high-quality PET images.
2-[
18
F]Fluoro-2-deoxy-
Current Technologies for PET Probe Production and Their Limitations
Owing to the realization of PET as a powerful molecular imaging tool for clinical studies in disease diagnosis and receptor mapping, automated syntheses of radiopharmaceuticals labeled with positron emitters are becoming increasingly important. Driven by the necessity to carry out repetitive synthetic steps and cycles, many different types of automated radiochemistry synthesis modules and platforms have been designed and implemented to produce PET probes in an efficient, reproducible, and safe manner. Generally, because of the short half-lives of these PET probes, their syntheses must be accomplished within two half-lives after the isotope is produced. Besides achieving the high specific activity required for certain probes, large amounts of radioactivity are often used to compensate for radioactive decay and, sometimes, low radiochemical yields of the final products. Careful consideration of shielding, remote operations, and automation are all necessary and integrated into the experimental planning and production protocols. Therefore, in spite of the advantages PET offers, reliable and routine production of PET probes is indeed a major challenge to radiochemists and engineers.
Presently, common PET radiopharmaceuticals are mainly produced by specialized automated synthesis modules. In fact, the use of those modules has enabled large-scale [ 18 F]FDG production and made a substantial contribution to the widespread use of [ 18 F]FDG-PET in cancer diagnostics. For example, typical large-scale [ 18 F]FDG production can provide enough doses for 50 to 100 patients that can supply several PET centers. After quality control and assurance (QC/QA) tests, [ 18 F]FDG is distributed for clinical uses from the production site to imaging facilities that are within a distance of 10 to 100 miles (ie, 0.5–2 hours' transportation time).
These automated synthesis modules are designed to (1) reduce personnel exposure to harmful levels of radiation; (2) increase user efficiency and minimize operator error by reducing manual operation; (3) increase batch-to-batch reproducibility; and (4) facilitate compliance with good laboratory practices (GLPs) and current good manufacturing practices (cGMPs) for PET drugs. However, there are several drawbacks to current practices. First, radiolabeling reactions are inherently carried out on a small scale owing to the minute amounts of radioactive material (nano- to microgram masses) produced from a biomedical cyclotron. The reactor volume of these modules is typically 0.3 to 3.0 mL, and the entire system is designed to handle macroscale volume. The reaction rate is significantly reduced by the dilution of radioisotopes. To compensate for short half-lives, a large (10 2 - to 10 6 -fold) excess of the probe precursor is applied to promote rapid and efficient incorporation of the radioisotope into the target radiotracer. These conditions present a challenge for rapid purification or separation of an extra-small quantity of radioactive product from a mixture composed of a large excess of unreacted or decomposed precursor or by-products (mg scale). Second, automated radiochemistry modules are generally expensive and must be hosted in a lead-shielded hot cell or mini-cell for operation by highly skilled personnel. Third, expanding the capability of PET imaging requires the development of new PET probes for imaging novel biologic targets or unique biochemical processes relevant to specific diseases.
To accelerate the discovery process, a highly flexible synthesis platform is required to perform various radiochemistry processes to make a reasonable number of probe candidates that can be applied for both in vitro and in vivo screening and characterization. Currently, most commercial radiochemical synthesizers are probe specific and often have very limited flexibility for adopting other synthetic protocols for known or new PET probes. Although some custom-built robotic platforms have a certain degree of flexibility, they generally require a long reconfiguration interval for a new synthetic process. 26 The lack of highly versatile radiochemical systems, which can handle a variety of precursor in minute quantity and small volumes to facilitate diverse radiochemical operations, poses a major bottleneck in the discovery and development of new PET probes.
Microfluidics-Based Radiochemical Synthesis Technologies
Microfluidic reactors are generally defined as devices consisting of a network of micron-sized channels (typically 10–500 μm) embedded in a solid substrate such as glass, metal, or plastic. They are capable of controlling and transferring small amounts of liquids that allow chemical processes and biochemical assays to be integrated and carried out on a microscale. Microfluidic platforms have been widely used in the fields of biology and chemistry. For example, in biology, applications using microfluidic devices include immunoassays, polymerase chain reaction amplification, DNA/RNA analyses, chemical gradient formation, cell manipulation, separation, and patterning.27–29 Many applications are aimed at improving aspects of critical basic biologic analyses and clinical diagnosis. On the chemistry side, many examples have demonstrated that microfluidic reactors are extremely useful in performing a wide variety of chemical reactions with purer products, higher yields, greater selectivities, and shorter reaction times than those obtained in batch-scale reactions. Furthermore, as one of the major attributes of the microfluidic technology, more benign and milder reaction conditions can be found for certain reactions within microfluidic devices. Manufacturing processes to fabricate microfluidic devices are relatively inexpensive and easily amenable to mass production, even with highly elaborate, complicated device designs. Analogous to microelectronics, microfluidic technologies enable the implementation and production of highly integrated yet relatively inexpensive and disposable devices for performing several different functions on the same chip. Biology and chemistry carried out in integrated microfluidic chips have great promise as a foundation for new chemical technology and processes.
A typical microfluidic platform 30 has a “lab-on-chip” configuration that can manipulate small volumes (from femtoliter to microliter) of fluids constrained within microfluidic channels. In general, platforms have a series of generic components for (1) introducing and removing multiple reagents and samples within microfluidic channels; (2) efficient mixing of separate reagents; (3) performing reactions with precise temperature control; and (4) carrying out other functionalities, such as purification for chemical syntheses or detection for product characterization. At the microscale, heat and mass transfer are very rapid owing to high surface area to volume ratios and a large heat exchange coefficient, so reaction conditions within such an environment can be precisely controlled. As a consequence, the yield and selectivity of many biologic reactions and chemical syntheses carried out in microfluidic channels can be dramatically improved, whereas the consumption of reagents and reaction or process time is significantly reduced.31,32 In addition, unlike devices used in macroscale chemical and biologic reactions, microfluidic platforms exhibit a high degree of scalability, reproducibility, modularity, and automaticity.
Miniaturization of PET radiosynthesis devices is an emerging technology that has the potential to address many of the current issues associated with PET probe production.31–35 For example, it allows the use of smaller quantities of expensive precursors than currently required in conventional radiosyntheses, thus reducing production cost while simplifying purification processes. With improved yield and specific activity, much less starting radioactivity is required, lowering the shielding cost while enhancing safety. In addition, the ability to manipulate reagent concentrations, reaction interfaces, and rapid mixing in both space and time within the microchannel network of a microfluidic device provides the fine level of control that is highly desired in PET probe production processes. As a result, the microfluidic platform is very practical for synthesizing PET probes 33 on demand because the quantity of radioisotope required for single-patient injection is extremely minute. In general, microfluidic radiochemistry platforms can offer several advantages: (1) reliable production of PET probes with high radiochemical purity (RCP) and radiochemical yield (RCY); (2) stand-alone and self-shielded devices without full-sized hot cells; (3) production of various probes using custom-designed microfluidic chips; (4) use of lower starting activity and less probe precursor; and (5) a significant reduction in separation challenge during purifications. Most important, this approach provides a suitable direction for the development of cost-effective, interchangeable, disposable, and quality-assured microfluidic chip–based radiochemistry processes.
There are two major types of microfluidic platforms used for radiochemical preparation of PET probes: continuous-flow systems and batch-based reactor systems.
Continuous-Flow Microfluidics
A continuous-flow microfluidic platform36–39 is based on internally connected microfluidic channel networks and is perhaps the simplest microfluidic configuration. Given that no valve or other isolation mechanism is involved, it is easily constructed by mechanical assembly of capillary tubes or an “on-chip” microfluidic network fabricated using common lithography techniques. Generally, the individual microfluidic channel is made of glass, metal, silicon, or plastics that are resistant to organic solvents. In a typical operation, reagents and solvents are introduced into a continuous-flow microfluidic reactor by syringe or peristaltic pumps or by simply applying back-pressure through tube connections. After reagents are mixed, reactions can occur by flowing a reaction mixture through the heating or cooling areas with well-controlled temperature and residence time. Finally, the products are collected on the other end of the channels. Continuous-flow microreactors are particularly well suited to conducting multiphase gas–liquid or gas–liquid–solid reactions because of their capability to sustain high pressure. The pressurization allows reactions to be increased in rate at an elevated temperature beyond the solvent boiling point and facilitates the dissolution of reactant gas into the flow stream. Therefore, the characteristics of a continuous-flow microfluidic system confer great efficiency and reproducibility for various chemical and radiochemical reactions. However, without valve control, it is rather difficult to isolate processes from one another to avoid cross-contamination. Even though the application of external valves can provide a certain degree of controllability, serious detrimental effects, such as possible contamination, transfer delays, or material loss in dead volumes, can jeopardize the entire operation. 34
Batch-Based Microfluidic Reactors
In contrast to a continuous-flow system, a batch-based microfluidic reactor platform40–44 represents a scalable integration of microfluidic channel networks with functional microfluidic modules, enabling the execution and automation of complicated chemical reactions and biologic operations within a single device. Among the current designs, pioneered by Quake and colleagues, the poly (dimethylsiloxane)- (PDMS, a silicone polymer) based45–48 microvalve-controlled integrated system 49 is one of the most representative examples. Such microvalve-based chips offer several advantages over continuous-flow microfluidics: (1) precise reagent loading and delivery; (2) facilitated mixing with on-chip peristaltic pumps 50 ; (3) isolation of distinct regions in a microreactor to avoid cross-contamination; and (4) automated programmable control for sequential synthesis. Several multistep radiochemical reactions have been successfully conducted using batch-based microfluidic reactors. Despite those proof-of-concept demonstrations, some issues still remain. Currently, batch-based microfluidic reactors use PDMS as the chip-making material because it is inexpensive and easy to fabricate and the microvalve systems require a flexible membrane. However, the current chip material and architecture lack high-pressure capability and cannot be used in radiochemical reactions under high pressure. In addition, PDMS is hydrophobic and swells in many organic solvents, restricting its use to reactions that do not involve organic solvents or reagents that cause swelling or damage.51,52 Another major concern, especially after heating in the presence of organic solvents, is that radiolabeled probes, unreacted 18 F complex/salt, and solvent can be extracted from solution and absorbed into PDMS crevices. These detrimental effects can potentially cause dose loss and reduce reaction yield.
Single Patient Dose on Demand
PET assessment provides the modern physician with unprecedented opportunities for disease diagnosis and therapeutic monitoring at the molecular level. 1 As personalized medicine continues to evolve into more specific, tailored treatment regimens to target particular molecular characteristics of a patient's disease, specific PET probes to improve diagnosis and guide treatment selection are crucial and will become indispensable. However, this goal cannot be achieved without easy and reliable access to different PET probes specific to particular diseased states. At present, the PET probe production based on a cyclotron-centered distribution model (ie, “centralized model”) allows [ 18 F]FDG and several other common probes to be synthesized in large quantity within radiopharmacies equipped with biomedical cyclotrons and automated synthesis modules and then transported to imaging centers where PET scans are performed (Figure 1). Given that each scan is performed on one patient at a time, at approximately 0.5- to 1-hour intervals, significant radioactive decay can occur between patient administration from nominal storage and transport from the radiopharmacy. At the same time, the RCP of PET probes might decrease continuously. For example, it is well known that [ 18 F]FDG can undergo radiolysis on storage, and this might confound imaging results and application potential. 53 Therefore, as the demand for a diverse array of PET probes specific to certain biologic processes continuously increases, it may become impractical to produce many different PET probes in high activity within centralized facilities and dispense single to a few patient doses for individual PET imaging without increasing the burdens of isotope production and risk of radiolysis.
Single dose on demand has emerged as a concept to overcome these limitations, whereby probe synthesis is conducted for each patient (or study) just prior to imaging. In this way, patients can be scheduled according to imaging need rather than dose availability. This also reduces the amount of decayed products and probe radiolysis.53,54 One idea to fulfill this concept of single dose on demand is to employ a “decentralized” model whereby probe production is transitioned to individual PET imaging centers or preclinical research facilities with the supply of radioisotopes, such as 18 F, from off-site cyclotrons (see Figure 1). This approach can be realized through the use of novel on-site microfluidic radiochemistry platforms capable of performing different radiochemical syntheses to yield desired probes in a microscale environment. In addition, the microfluidic on-site PET probe production platform can also enable biologists and clinicians in the basic research community along with research and development scientists in the pharmaceutical and biotechnology sectors to synthesize small molecule–based tracers or prosthetic groups to label biologic molecules or drugs of interest by technicians.

Schematic illustration of two PET probe distribution models: (1) the centralized model for delivery of PET probes (left) and (2) the decentralized model for delivery of radioisotopes (right).
Ideal PET Probe Production Platform
So what are the requirements for a versatile, microscale, on-site PET probe production platform? It should fulfill the following specifications: (1) ability to handle liquids in small volumes to produce single patient dose on demand; (2) kit-based disposable reactors, fluid tubing, and reagent cartridges to avoid contaminations; (3) universal base platform for different probes to reduce instrument cost and simplify operation; (4) easy to use, low maintenance, and reliable mechanical performance; (5) minimum and inexpensive shielding; and (6) possible automated purification, formulation, and final QC/QA assessment.
Microfluidics allows decreased reaction volumes, avoids reagent waste, reduces costs, increases reaction speed, and enables new probe distribution models that are not possible at the macroscopic scale. Given that many of the milestones in delineating biochemical processes and mapping receptor density in the human body and brain have involved 11 C- and 18 F-labeled probes, we focus on discussing microfluidic radiochemistry using these two isotopes.
Microfluidics in 11 C Radiochemistry
The progress of making PET a powerful imaging technique in nuclear medicine and drug research and development is driven by an increasing demand for novel imaging probes, especially for those labeled with 11 C probes.55,56 The unique feature of 11 C in PET studies comes from its short half-life of 20.4 minutes, a time interval that permits repeated PET studies and, to some extent, simple radiochemical transformations. Additionally, unlike 18 F-labeled probes, which often required functional group replacement during the labeling process (eg, 18 F to a hydroxyl group) and can change the target binding affinity or other pharmacologic properties, 11 C-labeled probes can be very similar, if not identical, to the original compound to be labeled. Therefore, radiolabeling through substitution of a carbon atom with 11 C makes the corresponding 11 C-labeled tracers indistinguishable from their stable counterparts within the biologic system.
On the other hand, the short half-life of 11 C constrains the synthetic strategy of 11 C-labeled compounds. For example, it is preferred to introduce the 11 C radioisotope at the latest stage of syntheses, if possible. In addition, the final 11 C-labeled probes have to be purified and pass quality control rapidly before being used in vivo. Although a wide variety of 11 C-labeled PET probes exist (eg, [ 11 C]methionine,57,58 [ 11 C]choline,59,60 [ 11 C]acetate,61,62 [ 11 C]raclopride,63–65 [ 11 C]-2β-carbomethoxy-3β-(4-fluorophenyl)tropane [β-CFT, WIN 35,428],66–68), they can be routinely available only for those privileged PET imaging centers equipped with on-site cyclotron and radiochemistry facilities.
Currently, the most dominant source of 11 C generated in biomedical cyclotrons is [ 11 C]CO2. Several reactions, such as [ 11 C]methylation via [ 11 C]CH3I and [ 11 C]carbonylation via [ 11 C]CO, have been carried out using microfluidic reactors because [ 11 C]CH3I and [ 11 C]CO are the most popular secondary precursors derived from [ 11 C]CO2. It matches well with the requirements and characteristics of 11 C radiochemistry. The promising initial results reported by Lu and colleagues 69 and Brady and colleagues 70 make the microfluidics-based 11 C-labeling reaction a very attractive approach for producing 11 C-labeled PET probes. Once widely used, microfluidics-based 11 C radiochemistry will certainly have significant impacts on radiopharmaceutical chemistry and drug discovery in the future. We have summarized important examples of 11 C-labeled compounds generated by microfluidic reactors in Table 1.
[ 11 C]Methylation in Microfluidic Reactors
In 2004, Lu and colleagues reported the synthesis of 11 C-labeled carboxylic ester via [ 11 C]methylation using a simple hydrodynamically driven microreactor. 69 The design of this T-shaped glass chip is illustrated in Figure 2. It was composed of two inlet ports for reagent delivery: a microfluidic channel for mixing and radiolabeling (with dimensions 220 μm [width] × 60 μm [height] × 14 mm [length] and total volume of approximately 0.2 μL) and one outlet port for product collection. Precise control of flow rate was attained by syringe pumps that delivered [ 11 C]CH3I and 3-(3-pyridinyl)propionic acid accurately into the microchannel. The best RCY (the RCY mentioned here is “decay-corrected”) of the corresponding [ 11 C]methyl ester 4 was 88% when the infusion rate of reagents was set at 1 μL/min at room temperature. This microreactor was further applied to prepare compound 5, a brain peripheral benzodiazepine receptor ligand via 11 C-methylation. The highest RCY was 65% when the infusion rate was also at 1 μL/min. The same device can be used for the synthesis of 18 F-labeled esters as well (described below). This simple device demonstrated the unique advantages and promises of using a microfluidic device for PET imaging probe synthesis.
Summary of 11 C-Labeled Compounds Generated within Microfluidic Reactors

DMF = N,N-dimethylformamide; Pd = palladium; RT = room temperature.
Infusion rate = 10 mL /min.
Infusion rate = 1 mL/min.
Brady and colleagues also described a similar glass microreactor for 11 C-labeling of various substrates containing −NH, −OH, and −SH functional groups in their patent, which also described the direct application of [ 11 C]CH3I for 11 C-methylation. 70 For example, the reported RCYs of 1 to 3 were 5 to 19% in the case of substrates containing secondary amines with the infusion rate from 10 to 100 μL/min. It should be noted that the radioactive reaction mixtures obtained from the microreactor were easily and rapidly separable on an analytical high-performance liquid chromatography (HPLC) column because only low amounts of material are present in a low volume. The radiotracer may be obtained from the HPLC in a smaller volume, which, in turn, facilitates easier formulation for safe intravenous administration. These results exemplify some of the potential advantages of this methodology for radiotracer synthesis, which, in successive generations, should be amenable to greater sophistication to encompass entire radiosyntheses in a versatile high-throughput manner.

11 C-Carbonylation in Microfluidic Reactors
For PET chemistry, 11 C-carbonylation reaction using [ 11 C]CO is a very effective means to introduce a 11 C-labeled carbonyl group into a probe because many biologically active substances often contain carbonyl groups or functionalities that can be derived from a carbonyl group, such as amides, esters, lactams, and lactones. 71 However, direct introduction of gaseous [ 11 C]CO into a microfluidic reactor is challenging. For example, the low [ 11 C]CO trapping efficiency and poor reagent or substance contact in reaction mixture often results in a low RCY. In addition, the harsh reaction conditions, such as the elevated temperature, high pressure, and use of organic solvents, make the current plastic-based (eg, PDMS) microfluidic reactors suboptimal. Therefore, until now, only the microfluidic reactors based on a microcapillary format have shown promise for [ 11 C]CO-based radiochemistry.
Recently, Miller and colleagues described a simple, low-cost, and effective microreactor employing a reusable silica-supported palladium catalyst packed into a standard Teflon tube 72 for the rapid multiphase 11 C-carbonylation reactions (Figure 3A). 73 It combined three phases: gaseous [ 11 C]CO, substrates in solution, and solid-phase catalyst in the 11 C-labeling process. The main component of the microreactor is the catalyst-immobilized Teflon microtube (1 mm [diameter] × 45 cm [length]) connected with precision syringe pumps or injectors and a mass flow controller to meter the flow of [ 11 C]CO through a mixing T-shaped connector (see Figure 3A). The active catalyst within the microtube is a palladium-phosphine complex attached to the silica support material, where the surface area to volume is drastically increased. It is estimated to be about 20,000 times larger than that by coating only the catalyst inside the surface of the microchannel. A feature such as this has dual properties in contributing to the improved yields of 11 C-labeled amides. It provides a large active surface area (coated with palladium catalyst) for the catalytic reaction to occur. As well, it increases the turbulence of both [ 11 C]CO and substrate solution, enhancing the mixing efficiency. This platform is, indeed, a very elegant and highly efficient design.

In a typical experiment, the substrate solution (aryl halide and benzylamine in tetrahydrofuran) was infused into the microtube reactor with a steady stream of [
11
C]CO gas controlled by a mass-flow controller through the T-shaped piece. Temperature in the microtube was maintained at 80°C for 6 minutes to generate the target
11
C-labeled compounds. Several aryl halides were investigated to test the applicability of this microtube reactor to [
11
C]CO labeling for amide formation (Figure 3B). The
11
C-labeled amides
In conclusion, a microtube reactor packed with palladium-supported catalyst (which proved to be reusable) is an effective method for continuous-flow carbonylation reactions. This methodology has been successfully applied toward radiolabeling by [ 11 C]CO carbonylative cross-coupling reactions and obtained modest to good RCYs and RCPs of labeled amides. RCYs can be improved by increasing the residence time of the substrates within the microtube reactor to give better synthetic and radiochemical output. The microtubule reactor has great potential to be a very useful methodology for discovering a diverse array of 11 C-labeled PET probes from [ 11 C]CO radiolabeling.
Microfluidics in 18 F Radiochemistry
Significant research efforts have been directed to developing microfluidic reactors capable of producing PET probes, especially for [ 18 F]FDG. As with 11 C-radiolabeling, microfluidic devices provide an excellent match for using minute amounts of precursors to yield 18 F-labeled PET probes. In the following section, we highlight some of the most notable examples of 18 F-radiofluorination reactions performed in microfluidic devices.
[18F]FDG Synthesis Using Microfluidic Devices
[
18
F]FDG, an
18
F-labeled glucose analogue, is the most widely available PET probe for imaging normal and elevated glucose use states of disease processes in brain, heart, and cancer.
74
Its radiosynthesis is typically composed of three sequential steps75,76 (Figure 4): (1) concentration and drying of the [
18
F]fluoride from [
18
O]H2O solution (1–10 ppb), which is produced in a biomedical cyclotron using proton bombardment; (2) radiofluorination of the precursor (
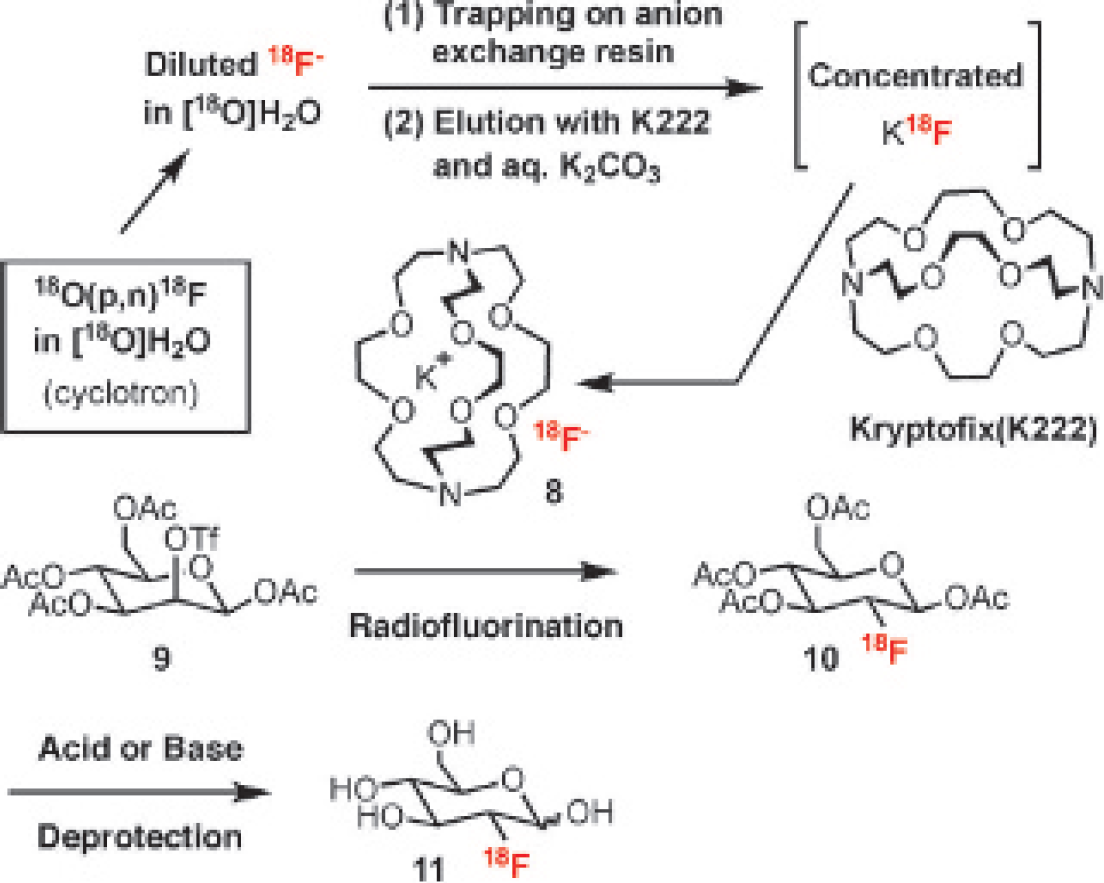
Continuous Flow–Based Microfluidics for [18F]FDG Synthesis
Gillies and colleagues reported using two identical disk-shaped polycarbonate microreactors for [
18
F]FDG synthesis (Figure 5A).78,79 This device was constructed by three layers of polycarbonate bonded together using SU-8, an ultraviolet-sensitive polymer, as an adhesive to form microfluidic channels and a reaction chamber. The top layer was used for reagent delivery. The middle and bottom layers were etched by hydrofluoric acid to form disks 100 μm in diameter and 1 mm in depth with a total internal volume of approximately 16 μL in each microreactor. These three layers were thermally bonded into a single microfluidic device and connected in sequence by fused silica capillary to allow multistep reactions. Reagents
Steel and colleagues reported the automated synthesis of [ 18 F]FDG using a two-stage, serpentine-channeled microfluidic device (Figure 5B). 80 With a flow rate of 250 μL/min, 40% RCY of [ 18 F]FDG is obtained. The on-chip reaction time includes 2 minutes for 18 F-fluorination at 70°C and 2 minutes for deprotection at 20°C. Using a wide range of starting activities, the authors claimed successful radiosyntheses of [ 18 F]FDG without any observed radiolysis. This system demonstrated the feasibility of using a microfluidic design for routine [ 18 F]FDG production and resulting RCY comparable to commercially available [ 18 F]FDG synthesizers.

Schematic illustration of two continuous-flow microfluidic (A) polycarbonate-based and (B) glass-based microreactors for [ 18 F]FDG synthesis. RT = room temperature.
Wester and colleagues recently reported a simple and low-cost capillary tube–based microfluidic device that can be integrated into a fully automated module for fast batch production of [
18
F]FDG.
81
The synthesis module includes several features: (1) a simple T-shaped mixer to mix reagents for radiofluorination and deprotection reaction; (2) a microtube-based reactor for in-capillary radiofluorination; and (3) on-column basic deprotection via an ion-exchange cartridge. The diameter of the capillary tube is approximately 300 μm, its length is 0.7 m, and the internal volume is approximately 50 μL. The capillaries can be made of chemoresistive plastics, such as polytetrafluoroethylene (PTFE), fluoroethylenepropylene (FEP), or polyetheretherketone (PEEK), without affecting the radiofluorination. It is reported that the optimized RCY of
Batch-Based Integrated Microfluidic Device for [18F]FDG Synthesis
In contrast to the continuous-flow microfluidic platform, Lee and colleagues reported a batch reactor–based microfluidic platform using PDMS chips (Figure 6A).
41
It exemplified a scalable integration of a simple microchannel network with functional microfluidic modules. This setup enables the execution and automation of complicated chemical reactions and biologic operations in a single device. The integrated microfluidic chips possess several critical features for conducting sequential chemical processes. First, microvalves provide precise control of liquid movement.
82
Second, reagent mixing is accomplished by a peristaltic pump improving the trapping efficiency and concentrating
18
F by nearly three orders of magnitude compared to the original solution. Third, the PDMS matrix is gas permeable, allowing solvent exchange directly within the microfluidic channel on heating. During the [
18
F]FDG synthesis, five processes occur in sequence (Figure 6B): (1)
18
F is concentrated via a miniaturized anion exchange column located in a square-shaped
18
F concentration loop: the diluted [
18
F]fluoride from a cyclotron is trapped by anion exchange beads, followed by releasing via aqueous potassium carbonate solution into a ring-shaped reaction loop; (2)
18
F drying and solvent exchange is accomplished by removing water via evaporation and solvent exchange into anhydrous CH3CN; (3) radiofluorination of the precursor
It is worthwhile to note that platform operation is highly computer automated, further improving and accelerating chemical synthesis with high RCY and RCP. The entire process takes about 14 minutes from loading the diluted [ 18 F]fluoride solution and produces 272 μCi [ 18 F]FDG with an RCY of 38% and an RCP of 97.6%. A simplified and enlarged microfluidic device was designed and fabricated for scaled-up production. 83 The device has a coin-shaped reactor with an internal volume of 5 μL. It has been reported that it can handle up to 100 mCi of [ 18 F]fluoride and produce up to 3 mCi of [ 18 F]FDG, an amount sufficient for several microPET studies with high RCP.
Comparison between Different Microfluidics Platforms in [18F]FDG Syntheses
Table 2 summarizes the main points of comparison between continuous flow–based microreactors and batch reactor–based integrated microfluidic devices for [
18
F]FDG synthesis. Continuous-flow microfluidic platforms can easily be constructed and have parallel array configurations for additional function. They can also be integrated with purification modules (solid-phase extraction or HPLC) for streamlining the entire PET probe production process. In most continuous-flow microfluidic chips, depending on the starting activity and reagent volume, the overall [
18
F]FDG synthesis time is generally less than 15 minutes, starting with radiofluorination reagent preparation [K222][
18
F]F
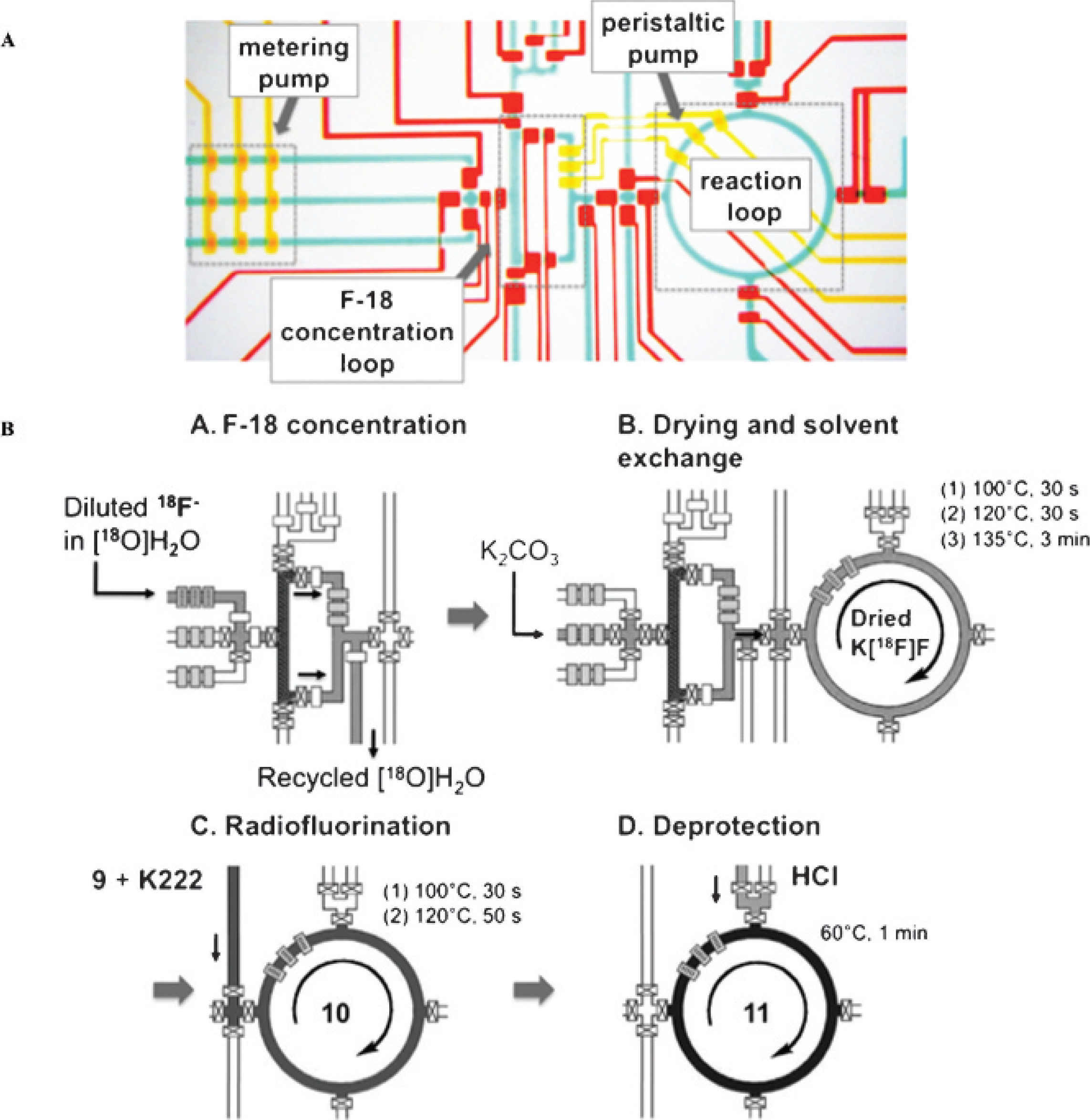
Batch reactor–based integrated microfluidic reactor for [ 18 F]FDG synthesis. A, Schematic representation of a PDMS-based microfluidic reactor used in the production of [ 18 F]FDG. B, Four sequential steps of the [ 18 F]FDG production performed in this device: (A) 18 F concentration; (B) drying/water evaporation; (C) [ 18 F]fluorination; (D) deprotection.
Summary of [ 18 F]FDG Radiosyntheses in Microfluidics

DMF = N,N-dimethylformamide; NR = not reported; PC = polycarbonate; PDMS = poly(dimethylsiloxane); RCP/RCY = radiochemical purity/yield; RT = room temperature.
Starting activity measured from fluorination reagent [K222][ 18 F]F.
Starting activity measured from the 18 F solution.
Other 18 F-Labeled PET Probes Prepared by Microfluidics
Lu and colleagues also applied the same T-junction type microfluidic chip illustrated in Figure 2 to perform
18
F-fluoroethylation using 2-[
18
F]fluoroethyl tosylate.
69
3-(3-Pyridinyl)propionic acid and 2-[
18
F]fluoroethyl tosylate were passed by syringe pumps into the microfluidic channels via ports A and B, respectively. Initially, the radiofluoroethylation did not proceed at ambient temperature. Then the device was heated to 80°C and product
Recently, the same group reported an efficient one-step fluorination approach to prepare N-[
18
F]fluoroacetyl-N-(2,5-dimethoxybenzyl)-2-phenoxyaniline
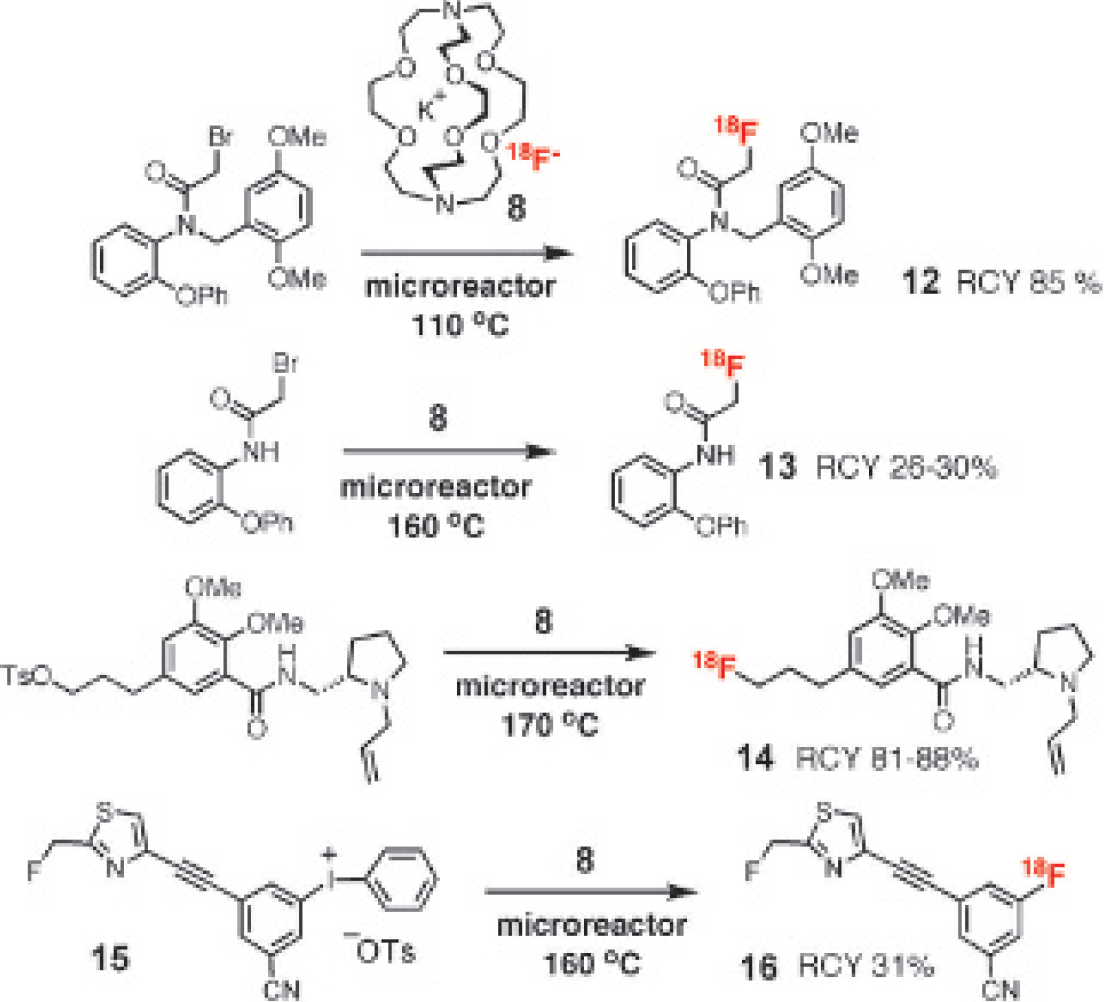
Furthermore, 3-[
18
F]fluoro-5-[(2-(fluoromethyl)thiazol-4-yl]ethynyl]benzonitrile ([
18
F]SP203B,
Other Positron Emitter–Labeled PET Probes Prepared by Microfluidics
With the dramatic expansion of PET applications in clinical diagnosis and preclinical research and the increasing demand of different types of PET probes over recent years, several other positron-emitting radionuclides have become available in high yields in small biomedical cyclotrons, such as nonmetallic 76 Br and 124I and metallic 68 Ga, 94mTc (T1/2 = 4.9 hours), 64 Cu, and 86 Y (T1/2 = 14.7 hours), that hold different half-life and chemical features. They are drawing great attention because either their half-lives or their production sources make their delivery significantly easier than 18 F-labeled ones.3,4 Their labeling conditions, especially for the metallic positron–emitting radionuclides, are often mild, which is very suitable for using PDMS-based microfluidic reactors.
Gillies and colleagues also expanded the applications of their three-layer polycarbonate microfluidic platform (see Figure 5A) using other isotopes, such as 124I.78,79 The device was used for the preparation of the apoptosis imaging probe 124I-labeled annexin-V. Three separate syringes were respectively loaded with the annexin-V in buffer solution, [124I]NaI in buffer solution, and Iodogen (an oxidizing agent) in acetonitrile which were pumped through the microfluidic reactor simultaneously. The labeling efficiency was about 40% after 2 minutes and comparable to conventional methods over the same time frame. The anticancer drug doxorubicin was labeled with 124I via the one-step radioiodination of the corresponding butyl-tin precursor in a similar way within the microfluidic chip. The tributyl stannous precursor [124I]NaI and oxidizing agent N-chlorosuccinamide were pumped through the chip, and the iodinated [124I]doxorubicin iodobenzoate (I-DOXIB) with approximately 80% labeling efficiency was obtained within 2 minutes.78,79
Although reports on the preparation of PET probes labeled with metallic positron–emitter radioisotopes are scant in the literature, we believe that the highly flexible batch-based integrated microfluidic reactors fabricated by PDMS will play an important role in this category because of their mild labeling conditions in aqueous solution.
Summary and Outlook
In the last few years, the use of microfluidic reactors in synthesizing PET probes has attracted great attention as a novel means of streamlining radiolabeling protocols. Various examples of microfluidics-based PET radiochemistry have focused on reactor design and proof-of-principle reactions, as in the reported syntheses of 11 C- and 18 F-labeled probes. Furthermore, the multistep radiosyntheses of [ 18 F]FDG have been accomplished by several groups using microfluidic reactors with different designs. Using microfluidic reactors offers many benefits, such as increased product yields and purities; accelerated reaction kinetics; superior reaction control; high production reliability; improved safety; more efficient use of hot cell space or less shielding material; use of less precursor for saving precious material and a reduced separation challenge and waste; low-cost, interchangeable, disposable, and quality-assured reagent cassettes and microreactors for radiochemical processes; facile automation and integration with downstream processing; and decreased use of reaction volumes to increase concentration of radioisotope that is difficult to achieve at macroscales. The combination of all of these features is critically important and essential for cGMP and future GMP for PET drug production of clinical applications. Additionally, an entirely new avenue for studying fundamental radiochemical reactions with similar stoichiometry of both radioisotope and precursor is opened.
The development of microfluidic reactors for performing continuous-flow organic reactions on a small scale has obvious potential in the area of radiolabeling for PET. Although some platforms are amenable to routine use, several performance issues should be rectified prior to commercial deployment. For example, an inherently low Reynolds number causes low reagent mixing efficiency, which requires additional mixing apparutus. 87 In addition, before establishing a desired continuous-flow “equilibrium state,” significant amounts of reagents can be wasted. Furthermore, the possibility of cross-contamination between different reactions prevents sequential syntheses of different high-purity radiopharmaceuticals.88,89 The development of batch-based microfluidic reactors can provide potential solutions to issues found in continuous-flow platforms. However, despite their advantages over the continuous-flow systems, batch-based reactors are usually made of plastic elastomeric materials, such as PDMS, which are not compatible with most organic solvents or reagents and thus cannot sustain harsh chemical reaction conditions. 51 Additionally, because of the serial nature of the system, the total amount of product generated is limited to one probe for a single-batch production. Nevertheless, once the material requirements can be met, the direct transition to new chemoresistive elastomers or plastics can be made. It is conceivable that a highly automated batch-based microfluidic platform is likely to be the most promising solution for “single patient dose on demand” for on-site syntheses and will truly enable the decentralized model to fulfill clinical, translational, and research PET needs.
An interesting idea to explore is to confer digital control over a continuous-flow system to generate sizable droplets whose volumes are similar to those of a batch microreactor. This can be accomplished by incorporating integrated microvalves into a microchannel network, thus enabling easy generation and manipulation of single droplets and optimizing reaction conditions within the microenvironment. 90 In a single-droplet mode, this platform would work the same as batch microreactors; in a multiple-droplet mode, it can assimilate continuous-flow reactions and can easily be scaled up to produce multiple single-patient doses. This hybrid design could be modular and suitable materials can be chosen for different areas of the platform to fulfill operational requirements. This design is possible to circumvent the need to satisfy a broad range of very stringent requirements within a single material.
The overarching purpose of microfluidic technologies for PET probe production is to create scalable, flexible, and programmable platforms capable of preparing various specific PET probes to target and understand human diseases. Although a number of issues remain, many aspects of microfluidics have great potential and are an ideal platform for performing the rapid productions of a wide spectrum of PET imaging probes. With continuous improvements in microfluidic technology, cost-effective, integrated, easy-to-use microfluidic radiochemistry platforms are beginning to enable a number of different medical and biologic fields formerly limited from PET use. Along with a new distribution model supported by existing radiopharmacy networks to deliver common radioisotopes, such as 18 F, the concept of “single dose on demand” can indeed be realized. Next-generation microfluidic platforms will have highly innovative designs and be composed of a number of advanced materials that satisfy diverse radiochemistry requirements for PET probe discovery. By enabling function of these technologies, a number of valuable existing and new probes will come to play an important role in advancing PET. This transition of current operational and logistical practices to accommodate decentralized PET probe production and distribution and the use of automated modular microfluidic platforms can potentially revolutionize the development and use of the next generation of PET probes. Further development of microfluidic technology could empower biologists and clinicians to routinely use radiolabeled probes in their research, which will be of tremendous benefit to fundamental science and medical research. This review is merely to herald a new era of microfluidics-based radiosyntheses. In summary, the numerous attributes of the microfluidics-based platforms suggest that microfluidics could become the core technology underlying the next generation of radiochemical synthesizers, making the outlook for microfluidics-based radiochemistry extremely promising.
Footnotes
Acknowledgments
Financial disclosure of authors: The authors are grateful for the financial support from the US Department of Energy (DE-PS02–09ER09–08 and DE-PS02–09ER09–18), a UC Discovery Grant (bio07–10665 and bio08–129095), and a UCLA Jonsson Comprehensive Cancer Center seed grant.
Financial disclosure of reviewers: None reported.
