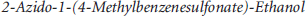Abstract
The (R)- and (S)-enantiomers of 2-amino-3-[1-(2-[18F]fluoroethyl)-1H-[1,2,3]triazol-4-yl]propanoic acid (4) were synthesized and evaluated in the rat 9L gliosarcoma brain tumor model using cell uptake assays, biodistribution studies, and micro-positron emission tomography (microPET). The (R)- and (S)-enantiomers of [18F]4 were radiolabeled separately using the click reaction in 57% and 51% decay-corrected yields, respectively. (S)-[18F]4 was a substrate for cationic amino acid transport and, to a lesser extent, system L transport in vitro. In vivo biodistribution studies demonstrated that (S)-[18F]4 provided higher tumor uptake and higher tumor to brain ratios (15:1 at the 30- and 60-minute time points) compared to the (R)-enantiomer (7:1 at the 30- and 60-minute time points). MicroPET studies with (S)-[18F]4 confirmed that this tracer provides good target to background ratios for both subcutaneous and intracranial 9L gliosarcoma tumors. Based on these results, the 1H-[1,2,3]triazole-substituted amino acid (S)-[18F]4 has promising PET properties for brain tumors and represents a novel class of radiolabeled amino acids for tumor imaging.
Amino acids enter cells via membrane-associated carrier transport proteins. Over 20 mammalian amino acid transport systems have been described with varying substrate specificities, pH dependence, sodium dependence, and regulatory mechanisms. A number of amino acid transport subtypes, including the L-type amino acid transporter 1 (LAT1) and system ASC amino acid transporter 2 (ASCT2), have been shown to be upregulated in various human tumors, including breast cancers and gliomas, and their presence may have prognostic significance.11–14 System L and, to a lesser extent, system A amino acid transport substrates have been the major focus of radiolabeled amino acid development for positron emission tomography (PET) and single-photon computed tomography (SPECT). Few radiolabeled amino acids are suitable for PET and SPECT that target amino acid transport systems other than system L and system A, and the utility of radiolabeled amino acids that target other transport systems for tumor imaging remains largely unexplored.
The synthesis and biologic evaluation of (R)- and (S)-2-amino-3-[1-(2-[18F]fluoroethyl)-1H-[1,2,3]triazol-4-yl]propanoic acid (4) was motivated by both radiosynthetic and biologic considerations. From a synthetic perspective, fluorine 18–labeled 1H-[1,2,3]triazole derivatives can be prepared through the Huisgen 1,3-dipolar cycloaddition reaction, also known as the “click” reaction.15–17 This reaction occurs between terminal alkynes and primary azides in the presence of a copper (I) catalyst under mild reaction conditions to provide 1H-[1,2,3]triazole substituents, typically in high radiochemical yield. In terms of biologic activity, data are very limited on the biologic transport of amino acids containing this group and its derivatives. The aromatic 1H-[1,2,3]triazole five-membered ring could act as a phenyl analogue, and if so, the resulting amino acids would be expected to be substrates for system L like other aromatic amino acids, such as
To determine the potential of 1H-[1,2,3]triazole-substituted amino acids for tumor imaging, (R)- and (S)-[18F]4 were prepared and evaluated in cell uptake assays, biodistribution studies, and microPET studies using the rat 9L gliosarcoma tumor model. A primary brain tumor model was selected because of the utility of other radiolabeled amino acids in human patients with brain gliomas. The 9L gliosarcoma tumor is an established model for human glioblastoma, the highest grade of gliomas, and has been used in the preclinical evaluation of other fluorine 18–labeled amino acids.18–22 Both enantiomers of [18F]4 were evaluated because stereochemistry is known to affect the transport of many radiolabeled amino acids.18,22–24
Experimental Methods
All reagents, reactions, and materials were purchased from commercially available sources. Chemicals were purchased from Aldrich Chemicals Co. (Milwaukee, WI) and Sigma Chemical Co. (St. Louis, MO) unless otherwise specified, and solvents were purchased from Aldrich Chemicals and Fisher Scientific Products (Pittsburgh, PA). Flash chromatography was carried out using Merck Kieselgel silica gel 60 (Sigma-Aldrich, St. Louis, MO) (230–400 mesh). Thin-layer chromatography (TLC) analyses were performed with 250 μm UV254 silica gel backing on glass plates (Aldrich Chemicals Co.). The TLC plates were developed with ninhydrin and/or phosphomolybdic acid stains. Alumina and C-18 SepPak cartridges were purchased from Waters, Inc. (Milford, MA). Ion-retardation resin (AG 11A8 50–100 mesh) was purchased from BioRad (Hercules, CA).
Melting points were measured with a MelTemp 3.0 apparatus (Barnstead International, Dubuque, IA) in capillary tubes and are uncorrected. Hydrogen nuclear magnetic resonance (1H NMR) spectra were recorded on a Varian 300 MHz spectrometer, and chemical shifts (δ values) are reported as parts per million (ppm) downfield from tetramethylsilane and coupling values in hertz. Elemental analyses were performed by Atlantic Microlabs, Inc. (Norcross, GA) and were within ± 0.4% of the theoretical values. The phrase “usual workup” refers to removal of residual water with anhydrous magnesium sulfate followed by rotary evaporation.
Chemistry
For both the (R)- and the (S)-enantiomers, the same reaction conditions were employed separately for the preparations of each enantiomer as depicted in Figure 1.

Synthesis of labeling precursors (R)- and (S)-
(R)-Propargyl glycine (500 mg, 4.42 mmol) was suspended in a solution of methanol (18 mL) and triethylamine (1.8 mL), and water (1.8 mL) was added to provide a homogeneous solution. Subsequently, di-tert-butyl dicarbonate (1.89 g, 8.66 mmol, 2 Eq) was added, and the reaction mixture was stirred at room temperature overnight. The reaction mixture was concentrated under reduced pressure, and the residue was partitioned between ethyl acetate (40 mL) and 0.2 M aqueous hydrochloric acid (30 mL). The organic phase was retained, and the aqueous phase was extracted with ethyl acetate (2 X 20 mL). The combined organic extracts were washed with water (2 X 30 mL) followed by the usual workup to provide a crude, colorless, thick oil (1.06 g) suitable for use in the next step without further purification. 1H NMR (CDCl3) δ 1.47 (s, 9H), 2.09 (t, J = 2.9 Hz, 1H), 2.75 to 2.86 (br m, 2H), 4.49 to 4.57 (m, 1H), and 5.34 (d, J = 8.1 Hz, 1H).
For the (S)-enantiomer, a 200 mg portion of (S)-propargyl glycine (200 mg, 1.77 mmol) provided crude (S)-
The crude N-Boc acid (R)-
For the (S)-enantiomer, the same reaction performed with (S)-
This reaction was performed based on conditions reported by Glaser and Arstad. 17 A portion of 1-(4-methylbenzenesulfonate)-2-fluoroethanol (200 mg, 0.917 mmol, 1.07 Eq) in N,N-dimethylformamide (4 mL) was stirred with a suspension of sodium azide (179 mg, 2.75 mmol, 3.2 eq.) at room temperature. After 48 hours, the solution was filtered, and the crude 1-azido-2-fluoroethane was used immediately in the next step without further purification.
In a separate flask, copper (I) iodide (873 mg, 4.58 mmol, 5.4 Eq) was suspended in methanol (2 mL) under an argon atmosphere with vigorous stirring. In rapid succession, (R)-
For the (S)-enantiomer, the reaction was performed as for the (R)-enantiomer with slightly higher excesses of reagents (1.5 Eq 1-(4-methylbenzenesulfonate)-2-fluoroethanol, 4.5 Eq of sodium azide, 6.4 Eq of copper (I) iodide, 6.4 Eq of triethylamine). Using this procedure, (S)-
A portion of (R)-3 (45 mg, 0.13 mmol) was dissolved in a mixture of methanol (2 mL) and 1 M aqueous hydrochloric acid (2 mL) and then sealed in a screw-top vial. The reaction mixture was heated at 60°C for 3 hours and then concentrated under reduced pressure. The residue was washed with diethyl ether (3 X 3 mL). The resulting oil was dissolved in a small amount of ethanol (≈ 0.5 mL), and the solid product was precipitated by adding diethyl ether (10 mL). The supernatant was removed to provide (R)-
For the (S)-enantiomer, (S)-
The enantiomeric purity of (R)- and (S)-
This reaction was performed based on the procedure of Demko and Sharpless with minor modifications.
15
2-Bromo-1-ethanol (4.0 g, 32.0 mmol) was added to water (10 mL) followed by sodium azide (2.5 g, 38.5 mmol), and the reaction mixture was heated at reflux overnight. After cooling, the reaction mixture was extracted with dichlormethane (3 X 10 mL), and the combined organic phases were dried over anhydrous magnesium sulfate and filtered. A 1.5 Eq portion of p-toluenesulfonyl chloride (9.15 g, 48.0 mmol) and 2.0 Eq portion of triethylamine (8.9 mL, 64 mmol) were added to the filtrate, and the reaction mixture was stirred at room temperature overnight. The reaction mixture was then washed with water (2 X 20 mL) followed by the usual workup. Purification by silica gel flash chromatography (2:8 ethyl acetate to hexane) provided
Radiosynthesis
The identical procedure was used for the radiosynthesis of (R)- and (S)-[18F]

Radiolabeling of (R)- and (S)-[18F]
The 2-[18F]fluoroethyl azide
The cycloaddition reaction was performed by adding a mixture of 1.5 M aqueous sodium
Purification of the intermediate [18F]
Two different methods were used for formulating the final product [18F]
The radiochemical purity, enantiomeric purity, and specific activity of the final product [18F]
Cell Uptake Assays
9L gliosarcoma cells were cultured in Earle's Minimal Essential Medium (MEM) with Earle's basic salt solution and 2 mM
Cell uptake assays were performed using the cluster tray method reported in the literature.
25
Two buffers were used for the assays: a phosphate-buffered saline solution and a sodium-free phosphate-buffered choline chloride solution. The sodium buffer consisted of 105 mM sodium chloride, 3.8 mM potassium chloride, 1.2 mM potassium bicarbonate, 25 mM sodium phosphate dibasic, 0.5 mM calcium chloride dihydrate, 1.2 mM magnesium sulfate, and 5.6 mM
The following inhibitors were added to the appropriate sodium or choline buffer: N-methyl-α-aminoisobutyric acid (MeAIB, 10 mM), a mixture of
Each 24-well plate was used for six assay conditions with each condition performed in quadruplicate. The cell uptake assays were initiated by rinsing the cells with 2 X 2 mL of the appropriate sodium or choline buffer without inhibitor at 37°C and then adding 0.4 mL of the appropriate assay buffer at 37°C with inhibitor and (R)- or (S)-[18F]
The amounts of radioactivity in each sample from each well and the standard counts for each condition were measured as counts per minute (cpm) using a gamma counter and decay corrected for elapsed time. The cpm values of each well were normalized to the amount of radioactivity added to each well and the protein concentration in the well and expressed as percent uptake relative to the sodium control condition. The data from each plate were analyzed with a one-way analysis of variance (ANOVA) with Tukey posttests using GraphPad Prism software (GraphPad Software, La Jolla, CA) with p values ≤ .05 considered statistically significant.
Biodistribution Studies with (R)- and (S)-[18F]4 in Rats with Subcutaneous 9L Tumors
All animal experiments were conducted under Institutional Animal Care and Use Committee-approved protocols in compliance with the National Institutes of Health (NIH) guidelines for the care and use of research animals established by the Washington University Medical School Animal Studies Committee. All animals were housed in accordance with the NIH and Association for Assessment and Accreditation of Laboratory Animals Care guidelines in facilities maintained by the Division of Comparative Medicine.
Biodistribution studies were performed with both enantiomers of [18F]
MicroPET with (S)-[18F]4 in Rats with Subcutaneous and Intracranial 9L Gliosarcoma Tumors
MicroPET studies were performed in two rats implanted with subcutaneous 9L tumors and one rat implanted with an intracranial 9L tumor. The subcutaneous implantations were performed as in the biodistribution studies except that the tumors were allowed to grow for 11 days prior to imaging. The intracranial tumor was implanted in the left midcerebrum using a template method as described previously
26
using a total of 5 X 104 cells suspended in 5 μL total volume. Anesthesia was achieved with a mixture of 1% isoflurane/oxygen during image acquisition. Computed tomographic (CT) images were acquired with a MicroCAT II System (ImTek Inc., Knoxville, TN), and dynamic PET data were acquired with MicroPET-FOCUS 120 and 220 scanners (Concorde MicroSystems, Knoxville, TN) for 120 minutes after the intravenous tail vein injection of 8.9 to 11.5 MBq of (S)-[18F]
Results and Discussion
Chemistry and Radiosynthesis of (R)- and (S)-[18F]4
The (R)- and (S)-enantiomers of nonradioactive
The initial step of the radiosynthesis of (R)- and (S)-[18F]
The crude intermediate (R)- or (S)-[18F]
The total synthesis time was approximately 2.5 hours with (R)- and (S)-[18F]
The estimated enantiomeric purity of both the (R)- and the (S)-[18F]
The decision to purify the intermediates (R)- and (S)-[18F]
Cell Uptake Assays with (R)- and (S)-[18F]4
Cell uptake assays were performed with 9L gliosarcoma cells using both enantiomers of [18F]
In Vitro Uptake of (R)- and (S)-[18F]4 by 9L Gliosarcoma Cells in the Presence and Absence of Amino Acid Transport Inhibitors

ASC =
The uptake data were normalized based on the amount of activity added to each well and to the amount of protein present in each well. The data are expressed as percent uptake relative to control conditions with standard deviation. See the Methods section for experimental details.
The Na sucrose and Cho sucrose conditions represent controls for the sodium-containing and sodium-free conditions, respectively.
(S)-[18F]
(R)-[18F]
(S)-[18F]
(R)-[18F]
A variety of inhibitor conditions were used to determine which amino acid transport system or systems were responsible for cellular uptake of (R)- and (S)-[18F]
Initial cell uptake assays were performed with MeAIB, BCH, and ASC inhibitor conditions primarily to evaluate the role of system A and system L transport in the uptake of both enantiomers of [18F]
The initial assays with (S)-[18F]
These conditions inhibited the uptake of (S)-[18F]
In contrast, the same uptake assays performed with (R)-[18F]
In summary, the in vitro uptake of (S)-[18F]
Biodistribution of (R)- and (S)-[18F]4 in Rats with Subcutaneous 9L Gliosarcoma Tumors
The biodistribution results obtained with (R)- and (S)-[18F]
Biodistribution of (S)-[18F]4 in Fischer Rats with Subcutaneous 9L Gliosarcoma Tumors
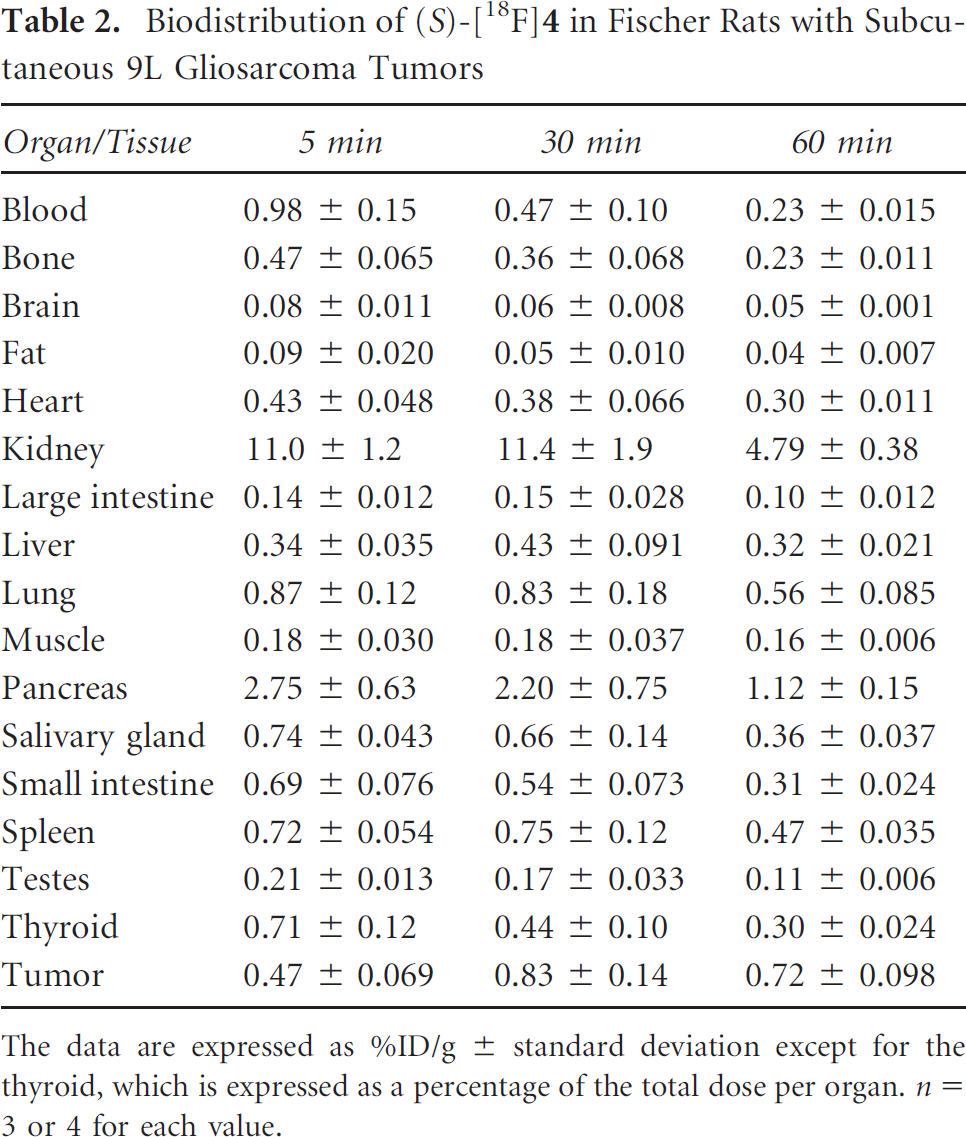
The data are expressed as %ID/g ± standard deviation except for the thyroid, which is expressed as a percentage of the total dose per organ. n = 3 or 4 for each value.
Biodistribution of (R)-[18F]4 in Fischer Rats with Subcutaneous 9L Gliosarcoma Tumors

The data are expressed as %ID/g ± standard deviation except for the thyroid, which is expressed as a percentage of the total dose per organ. n = 3 or 4 for each value.
The distribution of (R)- and (S)-[18F]
Tumor to Organ Ratios Observed with (R)-[18F]4 and (S)-[18F]4

The data are expressed as the average ratios of tumor uptake (%ID/g) divided by the corresponding normal organ/tissue uptake (%ID/g) for each animal at each time point ± standard deviation. n = 3 or 4 for each value.
Tissue to Blood Ratios Observed with (R)-[18F]4 and (S)-[18F]4
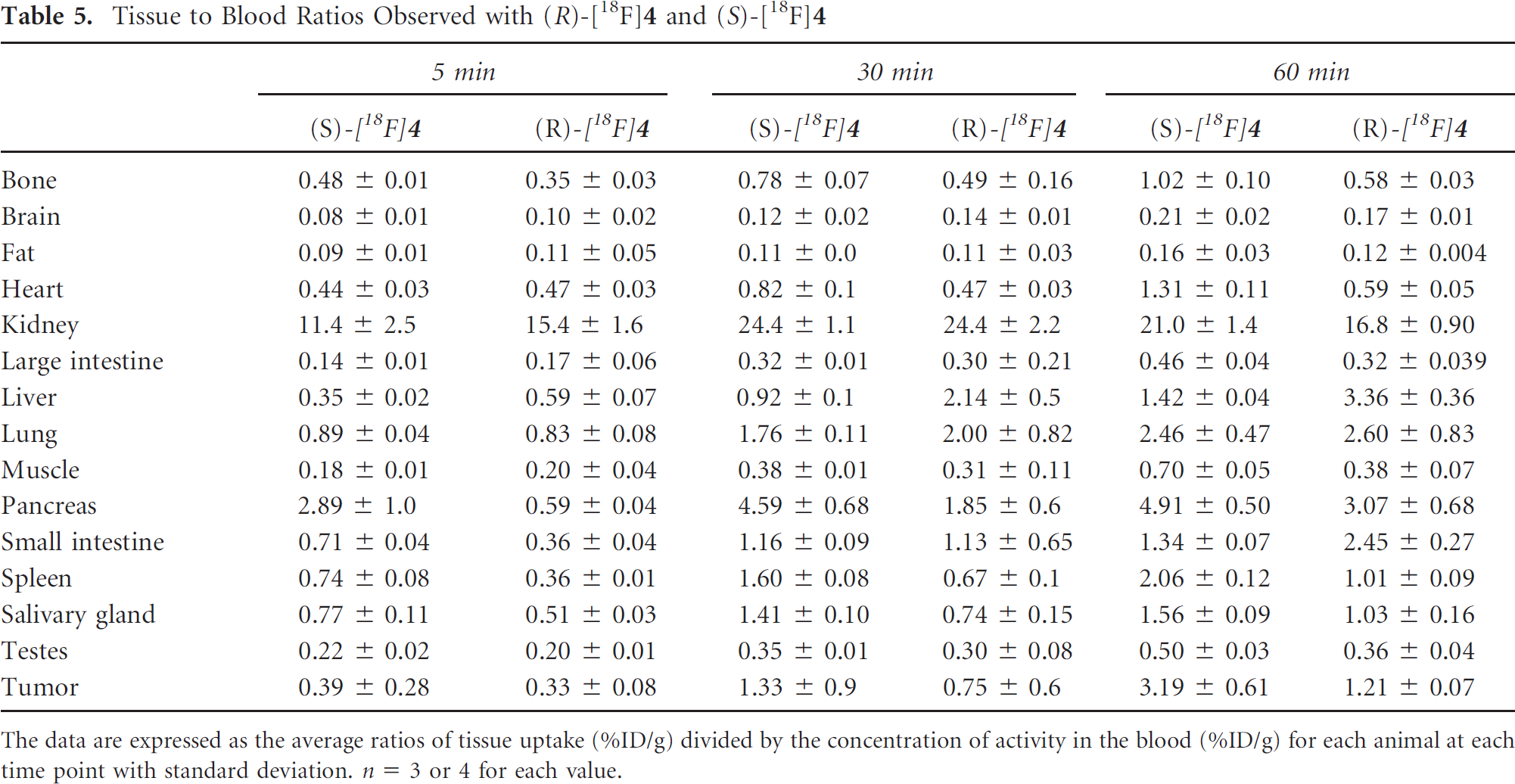
The data are expressed as the average ratios of tissue uptake (%ID/g) divided by the concentration of activity in the blood (%ID/g) for each animal at each time point with standard deviation. n = 3 or 4 for each value.
These biodistribution results in conjunction with the cell uptake assays indicate that 1H-[1,2,3]triazole-substituted amino acids are biologically active and represent a promising class of nonnatural amino acids for tumor imaging. To date, substrates for cationic amino acid transport systems have not been systematically evaluated as potential tumor imaging agents. Although (S)-[18F]
The component of system L transport observed with (S)-[18F]
These effects of the α-carbon stereochemistry on the mechanism of transport and biodistribution of (R)- and (S)-[18F]
MicroPET Studies with (S)-[18F]4 in Rats with Subcutaneous and Intracranial 9L Gliosarcoma Tumors
The (S)-enantiomer of [18F]

A, Time-activity curve from a microPET study performed with (S)-[18F]

A, PET (left) and fused PET-CT (right) images from a microPET study performed with (S)-[18F]
The tumor to normal tissue ratios measured in the microPET studies were in good agreement with those observed in the biodistribution studies. The subcutaneous tumor to paraspinal muscle microPET uptake ratio was approximately 5.0:1 at 30 minutes compared to the biodistribution tumor to muscle ratio of 4.6:1 at 30 minutes. Similarly, the tumor to bone (spinal column) ratio was 2.5:1 at 30 mintes in the microPET study compared to a tumor to bone (femur) ratio of 2.2:1 in the biodistribution study at the same time point. Finally, the maximal tumor to brain ratios in the microPET study were approximately 9:1 compared to 15:1 in the biodistribution study. This difference in tumor to brain ratios between the intracranial and subcutaneous tumors is due at least in part to partial volume effect in the microPET study. The relatively small size of the intracranial tumor and the low uptake in adjacent normal brain are expected to accentuate partial volume effects with the intracranial tumor.
The blood-brain barrier (BBB) may play an important role in the relatively high tumor to brain ratios observed with (S)-[18F]
Conclusions
The radiolabeled amino acid (S)-[18F]
Footnotes
Acknowledgment
Financial disclosure of authors and reviewers: The radiotracers reported in this article are subject to an option for licensing from Washington University in St Louis (J McConathy and RH Mach, inventors) by Isotrace Technologies, Inc. (St. Charles, MO).