Abstract
As leukotriene D4 receptor CysLT1R upregulation is an early event in inflammatory processes, specific detection of CysLT1R via molecular imaging might be a promising diagnostic tool for inflammatory diseases. We coupled a specific anti-CysLT1R IgG antibody to near-infrared (NIR) hemicyanine fluorophore DY-734. The fluorophore was also coupled to unspecific rabbit-IgG antibody or corresponding Fab fragments. Expression of CysLT1R in HL-60 human promyelocytic leukemia cells in vitro could be proven by reverse transcriptase—polymerase chain reaction (PCR), real-time PCR, and flow cytometry. Detection of the probes by flow cytometry showed that CysLT1R*DY-734 probe binds distinctly stronger to HL-60 cells than IgG*DY-734. Induction of ear edema in mice was conducted to test signaling of the synthesized probes in vivo. A markedly higher fluorescence intensity was observed in the edematous region than in the healthy region by a whole-body imaging system. Semiquantitative analysis showed that CysLT1R*DY-734 and Fab-CysLT1R*DY-734 probes bind 1.9- and 1.2-fold stronger, respectively, than the unspecific probes. Biodistribution studies revealed an enrichment of full-length IgG probes in liver and spleen, whereas Fab-containing probes are mostly found in liver and kidneys. Taken together, we present an approach that might improve early diagnosis of inflammatory diseases in the long term.
DIAGNOSIS OF chronic inflammatory diseases such as rheumatoid arthritis, arteriosclerosis, or edema is currently achieved with methods such as radiography, ultrasonography, computed tomography, or magnetic resonance imaging. However, these methods can only detect morphologic alterations that do not occur until advanced stages of the disease. To identify the localization of the infection in earlier stages and thus improve treatment of these diseases, it is important to demonstrate changes on a molecular level in vivo. The design of contrast agents consisting of a target-binding module, for example, a specific antibody against a cellular structure involved in inflammatory processes on the one hand and an imaging module on the other hand, is an interesting approach for the imaging of inflammatory diseases on a molecular level. 1 As imaging modules, hemicyanine or cyanine fluorophores showing near-infrared fluorescence (NIRF) are appropriate candidates as near-infrared (NIR) light allows relatively deep penetration into tissue. 2 Such NIR fluorophores have been successfully applied for detection of tumors in vitro.3,4 In the field of NIRF imaging of inflammation, we could already show that NIR fluorophore Cy5.5 is a suitable tool for detection of antigen-induced arthritis. 5 By labeling F4/80 monoclonal antibodies with Cy5.5, we designed a probe that allowed targeted detection of macrophages displaying F4/80 antigen on their surface. 6 Here we used hemicyanine NIR fluorescent dye DY-734 (Dyomics GmbH, Jena, Germany), which is eligible for the intended application owing to its suitable fluorescence properties (λexc. /λem. = 736/759 nm).
A decisive step in inflammatory processes comprises the recruitment of leukocytes.7,8 This event is regulated and mediated by many different cytokines, growth factors, complement factors, peptides, and eicosanoids. A highly active group of mediators encompasses the class of leukotrienes, which, belonging to the eicosanoids, are synthesized by the arachidonic acid pathway. 9 Leukotrienes display their functions by binding to specific receptors. For example, leukotriene D4 (LTD4) shows high affinity to the G protein-coupled 10 cysteinyl leukotriene receptor 1 (CysLT1R).11,12 As it has already been shown that targeting CysLT1R synthesis leads to remarkable therapeutic success,13,14 labeling of this receptor might also be an appropriate method to track early signs of inflammatory diseases. In this study, we developed and assessed specific CysLT1R-targeting NIRF contrast agents as a new diagnostic tool for molecular imaging of inflammatory diseases at early stages. Therefore, we coupled NIR fluorophore DY-734 to a polyclonal rabbit IgG anti-CysLT1R antibody (CysLT1R*DY-734) as well as to Fab fragments gained from the same antibody (Fab-CysLT1R*DY-734). As control for probe specificity, a full-length unspecific polyclonal rabbit-IgG antibody (IgG*DY-734) and its corresponding Fab fragments (Fab-IgG*DY-734) were coupled to DY-734. Binding properties of the probes were shown in vitro in HL-60 human promyelocytic leukemia cells, which were proven to express CysLT1R. The synthesized contrast agents were also tested in vivo in mice with zymosan A-induced ear edemas using a whole-body small-animal NIR imaging system.
Material and Methods
Coupling of NIR Fluorescent Probes and Preparation of Fab Fragments
For preparation of fluorescent probes, polyclonal rabbit anti-CysLT1R (Cayman Chemicals, Ann Arbor, MI; distributed by Biozol, Munich, Germany) or IgG from rabbit serum (Sigma-Aldrich GmbH, Steinheim, Germany) was used. Fab fragments of both antibodies were gained by cleavage with immobilized papain (Pierce Chemical Company, Rockford, IL) according to a protocol adapted from the manufacturer's instructions. Accordingly, papain mixture was equilibrated with 20 mM NaH2PO4*H2O, 10 mM ethylenediaminetetraacetic acid (EDTA), 20 mM Cystein*HCl, pH 7.0. IgG (2 μg/μL) and 20 mM NaH2PO4*H2O, 10 mM EDTA, pH 7.0, were added and volume was reduced by centrifugation in Microcon Centrifugal Filter Devices (Millipore Corporation, Billerica, MA). IgG solution was added to papain and incubated for 20 hours at 37°C on a shaker. Reaction was stopped by addition of 10 mM Tris*HCl, pH 7.5. IgG fragments were isolated from papain gel, and Fab fragments were purified by protein A chromatography. Protein A was soaked in buffer A (20 mM NaH2PO4, 150 mM NaCl, pH 8.0). A 1:2 solution of protein A and buffer A was prepared and then transferred to a spin column previously washed with buffer A. The probe was applied to the column, and the eluate was collected and reapplied five times. Finally, Fab fragments were eluted in 10 fractions with buffer A. N-Hydroxysuccinimidyl (NHS) ester derivative of fluorescence dye DY-734 (Figure 1; λexc./λem. = 736/759 nm; molar absorbance 240,000 M−1cm−1; molecular weight 1,092.14 g*mol−1) was provided by Dyomics GmbH (Jena, Germany) and coupled to the antibodies and their corresponding Fab fragments according to the manufacturer's instructions. Briefly, IgG antibodies were dissolved in 100 mM NaHCO3, 500 mM NaCl, pH 8.0. DY-734 dissolved in dimethyl formamide (5 μg/μL) was added in a molecular weight ratio of 12:1 (dye:IgG), and the mix was incubated for 2 hours at room temperature on a shaker. The fluorescent probes were purified on a Sephadex G-25 column (Amersham Biosciences AB, Uppsala, Sweden). In this context, Sephadex G-25 was soaked with phosphate-buffered saline (PBS) (3 mM KCl, 137 mM NaCl, 1.5 mM KH2PO4, 8 mM Na2HPO4 [water free]; pH 7.4) for 2 hours and packed on a 2 mL column. The mixture was applied, and the eluate containing the purified probe was collected in a 1.5 mL tube. Labeling efficiency (dye to protein ratio) was determined by recording the absorption spectrum of the coupled probes in an Ultrospec 3300 pro spectrophotometer (Amersham Biosciences AB). The following ratios (number of bound DY-734 molecules per IgG molecule) were measured for the synthesized probes applied in this study: 0.51 (CysLT1R*DY-734), 1.06 (IgG*DY-734), 0.31 (Fab-CysLT1R*DY-734), and 1.15 (Fab-IgG*DY-734).

Chemical structure of DY-734.
Cell Culture
To assess if the designed probes bind to the target in cell systems in vitro, HL-60 human promyelocytic leukemia cells (ECACC, Sigma-Aldrich GmbH) were employed. CysLT1R-negative BJ human foreskin fibroblasts (ATCC-Nr. 2522, LGC Standards GmbH, Wesel, Germany) served as a negative control for quantitative real-time polymerase chain reaction (PCR). One to 5 × 106 HL-60 cells were cultivated in RPMI 1640 culture medium (Gibco BRL, Paisley, Scotland) containing fetal calf serum (FCS) (Gibco BRL; 10% [v/v]), 1% (w/v) 100 mM Na-pyruvate, and 0.1 mM Minimum Essential Medium (MEM) nonessential amino acids (Gibco BRL) at 37°C in a 5% CO2 atmosphere; 1 × 106 BJ cells were grown in MEM containing 10% FCS. Determination of cell number was carried out with a CASY TT cell counter (Innovatis AG, Reutlingen, Germany).
Reverse Transcriptase and Real-Time PCR
Isolation of messenger ribonucleic acid (mRNA) from HL-60 and BJ cells was carried out by the guanidine isothiocyanate-phenole-chloroform method using Trizol reagent (Invitrogen GmbH, Karlsruhe, Germany). The concentration and purity of ribonucleic acid (RNA) were determined with a NanoDrop spectrometer (NanoDrop Technologies, Wilmington, NC). Preparation of complementary DNA (cDNA) was performed from 1 μg RNA using a cDNA Synthesis Kit (Bioline GmbH, Luckenwalde, Germany) according to the manufacturer's instructions. All primers were purchased from Jena Bioscience (Jena, Germany). CysLT1R cDNA was amplified with two primer pairs encoding for a 545 bp product (sense: 5′-GTG CCG CCT CAG CAC CTA TGC-3′; antisense: 5′-CGG ACT TCT GCA TTC TAA GGA CAG-3′) and a 225 bp product (sense: 5′-CTG CCA CAT GCC ATG ACA CTA-3′; antisense: 5′-ATA GAC CAC ACG GAG AGG CAG T-3′). Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) primer sequences were 5′-TCG GAG TCA ACG GAT TTG GTC GTA-3′ (sense) and 5′-ATG GAC TGT GGT CAT GAG TCC TTC-3′ (antisense), leading to a 520 bp product. PCR products were separated and visualized on a 1% (w/v) agarose gel. For real-time PCR, a QuantiFast SYBR Green PCR Kit (Qiagen, Hilden, Germany) was performed using a Roche Light Cycler (Roche, Mannheim, Germany). Values for CysLT1R were normalized on GAPDH values.
Flow Cytometry Analysis
Two × 105 HL-60 cells were collected in FACS buffer (PBS with 2% [v/v] BSA and 5 mM EDTA) and stained with polyclonal rabbit anti-CysLT1R and fluorescein isothiocyanate (FITC)-conjugated goat anti-rabbit IgG (secondary antibody; Chemicon International Inc., Temecula, CA). Cells were washed and analyzed in a FACSCalibur Flow Cytometer, and the results were evaluated with Cell/Quest Pro software (both Becton Dickinson, Heidelberg, Germany). Probe binding was determined accordingly by replacing 1 μg primary antibody with 2 μg DY-734-coupled IgG or Fab-IgG or 4 μg DY-734-coupled CysLT1R antibody or Fab-CysLT1R antibody, respectively.
Animal Experiments
All procedures were approved by the regional animal committee and were in accordance with international guidelines on the ethical use of animals. Twenty-one male NMRI mice (Harlan Winkelmann GmbH, Borchen, Germany) divided into seven groups consisting of three animals each were housed under standard conditions with food and water ad libitum. The different groups received the following treatments: untreated (group 1), zymosan A injected (group 2), zymosan A injected and treated with pure DY-734 (group 3), CysLT1R*DY-734 (group 4), IgG*DY-734 (group 5), Fab-CysLT1R*DY-734 (group 6), and Fab-IgG*DY-734 (group 7). Seven to 14 days prior to the start of experiments, all mice received a low-pheophorbide diet (Altromin, Lage, Germany) to reduce autofluorescence of abdominal organs. At the beginning of the experiments, all animals were shaved in the head area.
In Vivo NIRF Imaging of Ear Edema in a Mouse Model
Edemas were induced in all mice except the native control group 1 by injection of 20 μL of a 1% (w/v) suspension of zymosan A (Sigma-Aldrich GmbH) into the root of the left ear according to the protocol of Kurnatowska and Pawlikowski. 15 Shortly afterward, equimolar amounts of probes or pure dye were injected intravenously into the tail veins of corresponding mice (amount corresponding to 1 pmol DY-734 per gram of body weight). Fluorescence intensity was measured before and after zymosan A treatment and at 0, 2, 4, and 6 hours after probe injection using the Maestro™ small-animal whole-body imaging system (CRi, Woburn, MA). Data were evaluated with the corresponding software. With this software, we were able to subtract autofluorescence and to conduct a semiquantitative analysis by manually selecting regions of interest. These regions were chosen to cover the complete inflamed regions and to measure the same region in each animal. This procedure allows a comparison between different regions of a mouse, for example, edematous and healthy ears. Therefore, we subtracted average signal/exposure time in the healthy region from the one in the edematous region of each mouse at each time point. The obtained values were used for comparison of different probes.
Ex Vivo NIRF Imaging of Organs
After measurement of in vivo fluorescence distribution 6 hours after probe injection, mice were sacrificed, several organs were isolated, and fluorescence of whole organs was assessed as described for in vivo experiments.
Statistical Analysis
Values are expressed as mean ± standard error of fluorescence intensities obtained from three animals. The in vivo results of mice treated with specific and unspecific probes were tested for statistical significance by Student t-test with p < .05 accepted as the level of significance.
Results
For synthesis of CysLT1R-directed contrast agents applicable in molecular imaging, NHS ester of DY-734 (see Figure 1) was used as the imaging module. This hemicyanine fluorophore was selected because of its fluorescence properties, which make it detectable with NIRF.
To test the in vitro binding properties of the probes, human promyelocytic leukemia cells HL-60 were used. Expression of CysLT1R on mRNA level was tested by reverse transcription (RT) and real-time PCR using two different CysLT1R-specific primer pairs amplifying a 225 and a 545 bp transcript, respectively (Figure 2). A 520 bp GAPDH product was amplified as control. Both CysLT1R-specific primer pairs delivered products of the expected size, which was shown by agarose gel electrophoresis (see Figure 2A). Accordingly, quantification of CysLT1R mRNA expression by real-time PCR delivered a 25-fold higher amount of mRNA in HL-60 than in CysLT1R-nonexpressing BJ fibroblasts, which were used as a reference (see Figure 2B). These results were confirmed by investigation of CysLT1R protein expression (Figure 3). Flow cytometry analysis of HL-60 cells incubated with anti-CysLT1R antibody revealed that fluorescence was markedly increased compared to cells incubated with polyclonal rabbit-IgG antibody.
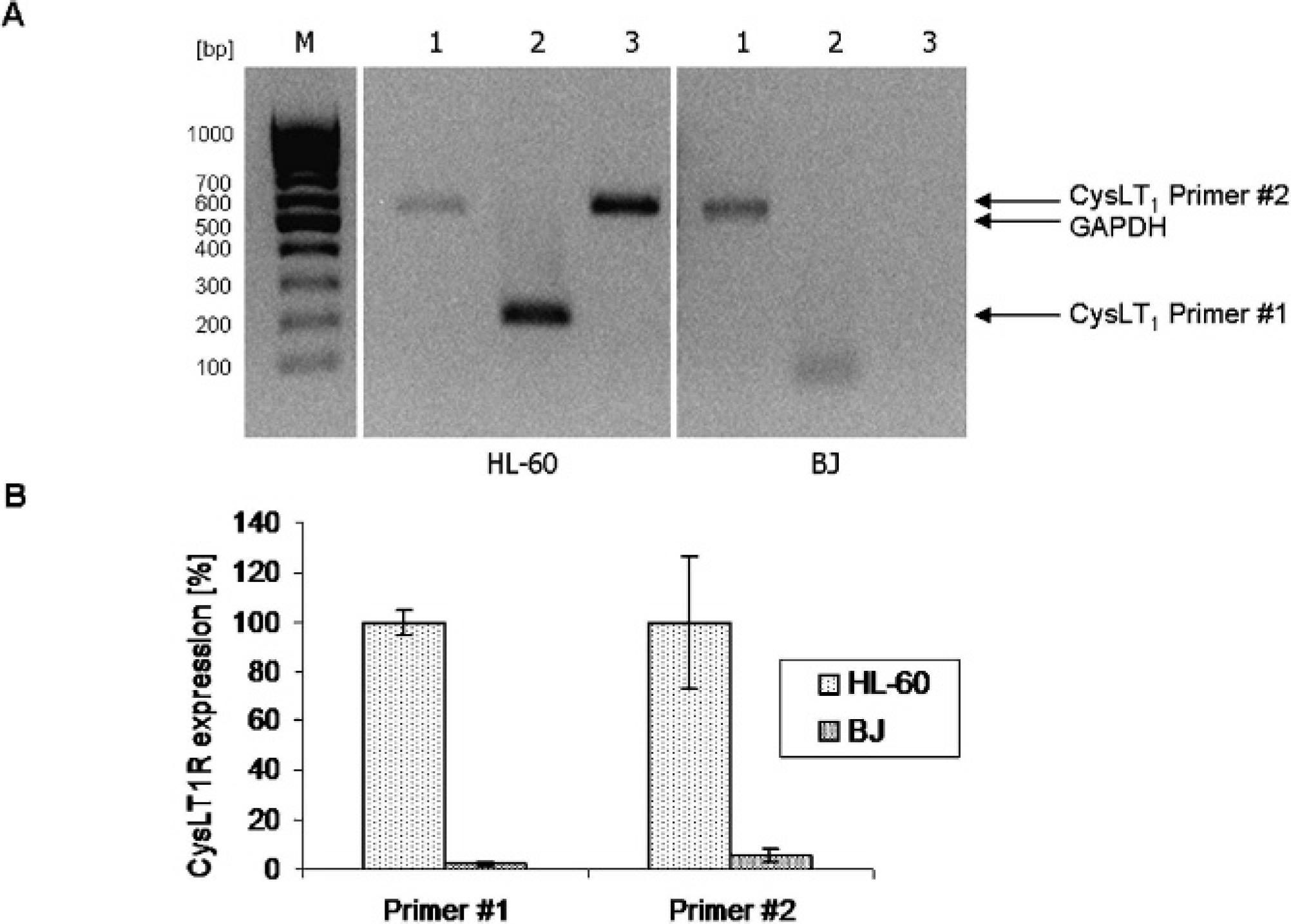
LTD4 receptor CysLT1R is expressed in HL-60 promyelocytic leukemia cells. A, CysLT1R mRNA was translated to cDNA by RT-PCR. cDNA was amplified by specific primer pairs for a 520 bp GAPDH transcript as loading control (1), a 225 bp (2), and a 545 bp (3) CysLT1R transcript; M: DNA ladder 50 to 500 bp (Jena Bioscience, Jena, Germany). B, Relative CysLT1R mRNA expression in HL-60 cells and CysLT1R-negative BJ fibroblasts as a reference was measured by real-time PCR with the primer pairs 1 (225 bp transcript) and 2 (545 bp transcript). Values for CysLT1R were normalized on GAPDH values.

CysLT1R protein is expressed in HL-60 cells. For flow cytometry analysis, cells were incubated with an unspecific polyclonal rabbit-IgG antibody (green) or a polyclonal rabbit-IgG anti-CysLT1R antibody (red; Cayman Chemicals) and detected by an FITC-coupled secondary goat-anti-rabbit-IgG antibody.
Flow cytometry analysis was conducted to investigate the probes' binding properties in vitro. Because DY-734 emits NIR light (λem. = 759 nm), it cannot be detected directly with a flow cytometer for technical reasons. Therefore, a FITC-coupled secondary goat-anti-rabbit-IgG antibody was applied for indirect identification of probe binding in HL-60 cells. A CysLT1R-specific (CysLT1R*DY-734) probe showed increased fluorescence intensity in HL-60 cells as well as IgG*DY-734, leading to the conclusion that full-length IgG probes bind in vitro to HL-60 cells (Figure 4). For the corresponding Fab fragment probes, no binding could be detected (data not shown).

Full-length IgG-containing probes bind in HL-60 cells, whereas Fab-containing probes do not. Binding was examined indirectly by flow cytometry. As a negative control, unspecific polyclonal rabbit-IgG antibody (blue) was detected by a FITC-coupled secondary goat-anti-rabbit antibody. A, DY-734-coupled probes with full-length IgGs (CysLT1R*DY-734 [red], IgG*DY-734 [green]) were used instead of pure primary antibodies to prove their binding ability in a coupled state. B, Positive control with primary polyclonal rabbit CysLT1R antibody (green).
Subcutaneous injection of zymosan A in the lobe of the left ear led to the development of ear edema in mice. Altogether, seven groups with three animals each were investigated. One group was left untreated, whereas the others were injected with zymosan A. Immediately after ear edema induction, mice were either left without further treatment or intravenously injected with pure DY-734, CysLT1R*DY-734, IgG*DY-734, Fab-CysLT1R*DY-734, or Fab-IgG*DY-734, respectively, and in vivo imaging was carried out using the Maestro whole-body small-animal NIRF imaging system. Representative images 6 hours postinjection (Figure 5) show, as expected, a distinct fluorescence spot at the edematous area of zymosan A injection. Semiquantitative analysis of relative fluorescence intensities (average signal divided by exposure time) in the edematous (infected) region and the contralateral (healthy) region before zymosan A injection, after zymosan A injection, and 0, 2, 4, and 6 hours after probe application was conducted. Values at the healthy region were subtracted from the ones measured at the infected region for each mouse at each time point. This procedure revealed that a specific increase in fluorescence intensity was significantly higher in CysLT1R*DY-734-treated than in IgG*DY-734-treated mice (1.9 fold, p = .034). Fab-CysLT1R*DY-734 accumulated 1.2-fold stronger in the infected region than Fab-IgG*DY-734 (Figure 6). This relative increase in fluorescence intensity was detectable shortly after probe application and maintained for at least 6 hours, whereas enrichment of pure DY-734 was much lower and started to decrease after 4 hours.

Injection of the fluorescent CysLT1R probes in mice with zymosan A-induced ear edema led to increased fluorescence intensity in vivo, particularly in the edematous region. NIRF intensity was measured using the Maestro™ whole-body imaging system (NIR 710–760 nm Maestro excitation filter and NIR 800 nm longpass Maestro emission filter). Representative white light images of native mice (A-D) and NIRF images 6 hours after injection of zymosan A and contrast agents (E-H) are shown. Fluorescence is depicted in false colors as pictured by the “Treshold Mode” of Maestro software, with blue representing weakest and red representing strongest intensity. Injected probes: CysLT1R*DY-734 (A, E), IgG*DY-734 (B, F), Fab-CysLT1R*DY-734 (C, G), and Fab-IgG*DY-734 (D, H).
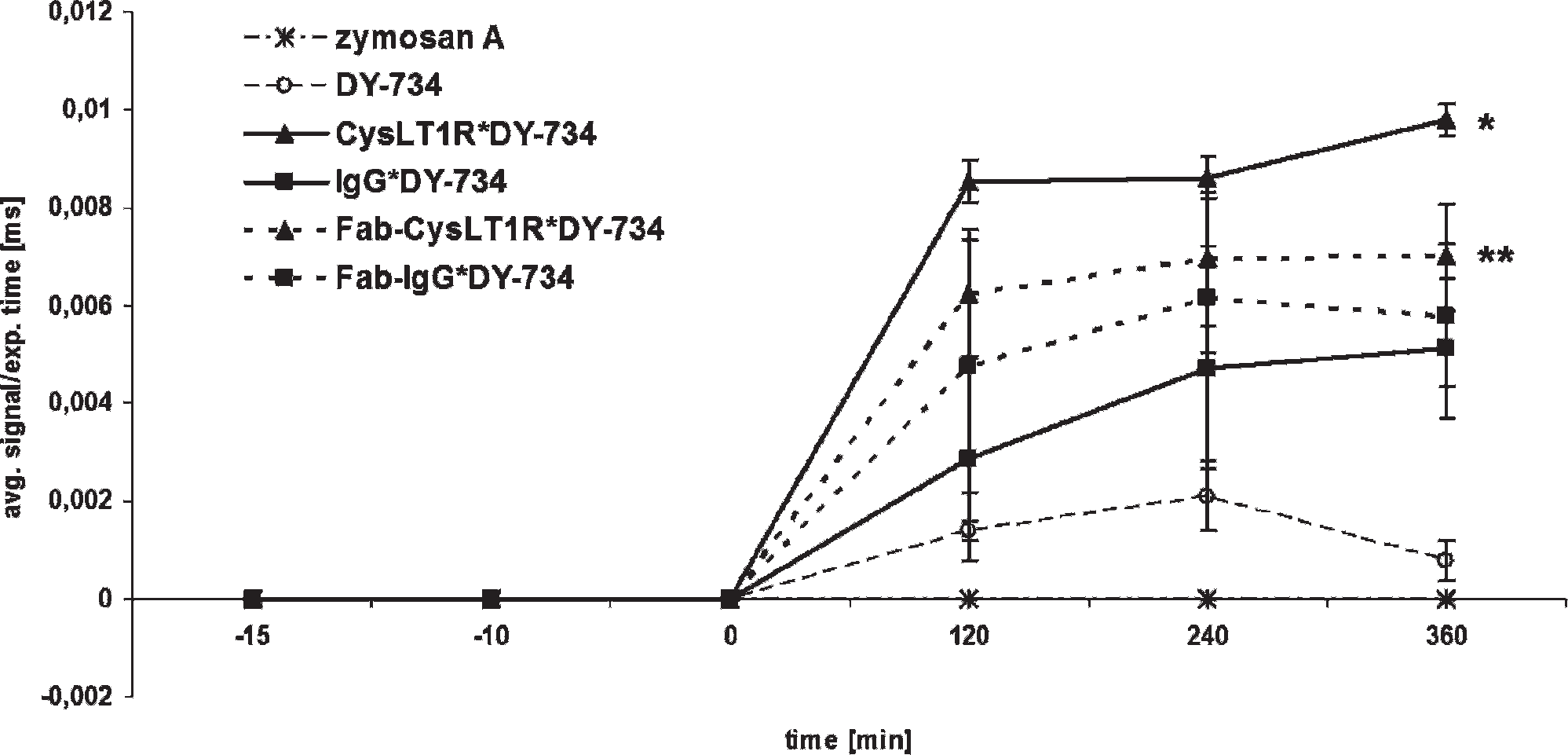
The increase in fluorescence intensity in the edematous to contralateral region is significantly stronger after injection with specific CysLT1R probes than with unspecific IgG probes. The time course of difference in fluorescence intensities (average signal divided by exposure time) between the edematous and contralateral regions from zymosan A induction up to 6 hours (time course depicted in minutes) after probe application in native (measured 15 minutes prior to probe injection), untreated (only zymosan A injected, measured 10 minutes prior to probe injection), and pure DY-734-, CysLT1R*DY-734-, IgG*DY-734-, Fab-CysLT1R*DY-734-, and Fab-IgG*DY-734-treated mice is shown. At the 6-hour time point, fluorescence intensity in the edematous compared to the healthy region is significantly higher after treatment with CysLT1R*DY-734 (*) than with IgG*DY-734 (p = .034). For Fab-containing probes, no significance could be detected (**p = .545). Fluorescence was measured with the Maestro™ whole-body imaging system; average values of three mice each with standard error are presented (semiquantitative analysis).
Six hours after contrast agent injection, mice were sacrificed and several organs, including lung, kidney, liver, spleen, and brain, were taken to analyze fluorescence intensities with the Maestro system ex vivo (Figure 7). As expected, a different biodistribution for both full-length antibody and Fab probes was observed as all probes accumulated in liver, but only Fab probes could also be detected in kidneys. Only weak fluorescence could be observed in brain and spleen. Nevertheless, full-length antibody probes showed a stronger accumulation in spleen than Fab-containing probes. Almost no fluorescence was seen in lung.

NIRF ex vivo images of mice with zymosan A-induced ear edema show different biodistribution for full-length IgGs and Fab fragment-containing probes. Six hours after injection of the probes, untreated (healthy), native (only zymosan A injected), and pure DY-734-, CysLT1R*DY-734-, IgG*DY-734-, Fab-CysLT1R*DY-734-, and Fab-IgG*DY-734-injected mice were sacrificed and organs were measured for fluorescence intensity with the Maestro™ whole-body imaging system. Fluorescence intensities (average signal/exposure time) in lung, kidney, liver, spleen, and brain are shown; average values of three mice each and standard error of the mean are depicted (semiquantitative analysis).
Discussion
Our study is the first to show that targeting LTD4 receptor CysLT1R with a specific IgG antibody coupled to NIR hemicyanine fluorophore DY-734 is an appropriate tool for detection of CysLT1R in vitro as well as in vivo using a mouse ear edema model. CysLT1R was particularly chosen as a target because LTD4, the strongest leukotriene representative, displays a high affinity to this receptor, 16 thus emphasizing the relevance of CysLT1R in inflammatory processes. Interference with CysLT1R synthesis has already been proven to be a successful therapeutic tool.13,14 Therefore, targeting of this receptor with a fluorescent contrast agent might be a helpful instrument to allow an earlier detection of inflammatory processes, which leads to earlier and hence more effective therapy. For imaging of CysLT1R, NIRF is adequately located within the diagnostic window between 650 and 900 nm. In this range, hemoglobin and water show minimal absorbance 17 ; therefore, a relatively deep tissue penetration up to several centimeters is possible.2,18 NIR fluorophores are preferable on the widely used Cy5.5 as their absorbance and emission maxima are located better within this window. Currently, the only clinically approved NIR fluorophore is indocyanine green. Nevertheless, we chose hemicyanine dye DY-734 for the imaging module of our contrast agents as several asymmetric cyanine dyes of the DY series displayed a red shift of absorption and emission bands, higher fluorescence quantum yields, enhanced thermal stability, and reduced cytotoxicity in endothelial cells and macrophages in comparison with indocyanine green. 19
Probe specifity in vitro was shown on HL-60 promyelocytic leukemia cells. This cell system has already been proven to express the seven transmembrane domains containing CysLT1R receptor on their surface, particularly after stimulation with interleukin-5. 20 Nevertheless, we could show CysLT1R expression in HL-60 on the mRNA level by RT and real-time PCR with BJ human foreskin fibroblasts as a negative reference as well as on a protein level by flow cytometry without further stimulation.
Flow cytometry revealed increased affinity of CysLT1R*DY-734 and IgG*DY-734 to HL-60 cells in comparison with unlabeled IgG. The fact that unspecific IgG*DY-734 shows an effect as well is probably mainly due to the phagocytic activity of HL-60 cells, which occurs under certain conditions.21,22 Another reason might be the fact that DY-734 seems to bind stronger to unspecific IgG than to CysLT1R-IgG, as the dye to protein ratios of the probes (0.51 for CysLT1R*DY-734 in contrast to 1.06 for IgG*DY-734) suggest. The lack of binding capacity observed for Fab fragments (data not shown) could be attributed to the fact that the corresponding epitopes were eliminated during papain cleavage.
However, our in vivo results reveal that Fab fragments indeed show an increased affinity to cells involved in the inflammatory process, in contrast to our in vitro results. All investigated probes accumulated in the edematous region at the left ear lobe of mice. Much less accumulation could be detected in the corresponding healthy region in the proximity of the right ear. Although there was visible signaling when using unspecific IgG probes, it is evident that CysLT1R*DY-734 and Fab-CysLT1R*DY-734 displayed larger time-dependent differences in fluorescence intensities between edematous and healthy regions. A targeted accumulation of the probes in the inflamed region obviously took place as pure DY-734 alone could not enrich in the edematous region. Further studies are needed to find a way to distinguish unspecific, phagocytosis-derived signals from specific CysLT1R binding. The observed accumulation of unspecific IgG and Fab-IgG probes could be attributed to the phagocytic activity of leukocytes, particularly of macrophages,7,8 at the edematous region. The recruitment of phagocytically active cells is known to be a fundamental event in every inflammation process. Interestingly, it has recently been discovered that macrophages are recruited from a splenic reservoir 23 ; thus, they might internalize the probes in the blood system on their way to the infected area. Consequently, this process could have influenced the amount of actually free probe molecules available in the blood pool for specific binding to CysLT1R. In general, this unspecific phagocytic activity of cells involved in inflammatory processes still remains a challenge for the design of probes directed at molecular targets, and the course and mechanisms of these processes need to be elucidated. Moreover, as expected, different pharmacokinetics for full-length IgG- and Fab fragment-containing probes in vivo were observed. With regard to the fluorescence intensities of Fab probes, there seems to be a tendency for highest levels at 4 to 6 hours after probe injection, whereas full-length IgGs still show an increase at 6 hours after probe injection. As binding of the probes at Fc receptors of macrophages is possible for full-length antibodies but not for Fab fragments, the prolongation of fluorescence detection in full-length antibodies might at least partly be due to the unspecific processes described above. This is likely as the zymosan A-induced edema model is characterized by a massive recruitment of eosinophils and macrophages, which peaks after 4.5 hours.15,24 Labeled macrophages with fluorescent liposomes applied in this model also displayed maximal fluorescence intensity in the edematous region 4 hours after zymosan A injection but after 6 hours still showed elevated intensity in comparison with the healthy region. 25 Thus, 6 hours after zymosan A injection, macrophages are definitely present at the site of inflammation, and internalization of fluorescent probe molecules by macrophages contributes to NIRF signals obtained at this time point.
Fab fragments were also used because they display improved pharmacokinetics, such as shorter intravascular half-time, 26 compared to full-length antibodies. By fluorescence measurement of isolated organs ex vivo, we could, as expected, observe a different biodistribution of full-length IgG- and Fab fragment-containing probes. Although all probes enriched in liver, suggesting a hepatic elimination pathway, Fab probes additionally accumulated in kidney. Here the accumulation of the full-length probes was comparatively lower. As Fab fragments display only about a third of the size of a full-length IgG antibody, they are likely eliminated in a renal pathway. In spleen, although enrichment is very low in general, full-length IgG probes accumulate distinctly stronger than Fab probes. Pharmacokinetics described for radiolabeled Fab fragments 27 might not be completely transferable to these probes as the presence of DY-734 in the Fab fragment leads to comparatively larger molecular sizes and expectedly different molecular conformations, which might change pharmacokinetics such as plasma protein binding and biodistribution. In all other organs, such as lung, brain, or heart, skeletal muscles, intestine, stomach, fat, and bone marrow (data not shown), little or no increased fluorescence could be observed. Native mice or zymosan A-injected mice with no further treatment or pure DY-734 treatment displayed no or only very basal fluorescence in the examinated organs. Nevertheless, this method cannot be applied to quantify inflammation in organs such as as liver or kidneys because of interfering metabolic effects caused by systemic probe application.
These results show a targeted accumulation of antibody-coupled probes at the inflamed region. Low enrichment in organs not involved in the inflammatory process is promising for the development of diagnostically applicable contrast agents as it leads to high signal to noise ratios. Based on these findings, it is evident that for the use of antibodies as contrast agents for molecular imaging, interfering effects of leukocyte recruitment have to be excluded. As mentioned, unspecific accumulation of probes in areas of inflammation to a certain degree is a general problem for detection of these diseases, which can also be observed in a study by Klohs and colleagues. 28 Detection of CD40 with a Cy5.5-coupled monoclonal antibody in a cerebral ischemia model was highly specific; nevertheless, a certain background accumulation of the antibody was observed in CD40−/– mice and by detection with an unspecific Cy5.5-coupled IgG antibody but not in sham-treated mice without ischemia. This leads to the conclusion that inflammation itself leads to probe accumulation to a certain extent. This is supported by the fact that studies tracking macrophages also revealed distinct probe accumulation in inflamed regions.5,6,29
The applicability of these probes—as is generally true for optical imaging—is limited by the fact that tissue penetration of NIRF achieves only a few centimeters. Therefore, only regions lying close to the surface of the organism can be examined. In clinical application, this means that mainly inflammation of joints in extremities can be detected by planar optical imaging. Alternatively, optical tomography needs to be conducted to achieve higher tissue penetration. Another important parameter to ensure correct and effective signaling is the choice of the applied probe dose as a high density of fluorophore molecules might lead to quenching effects, which would lead to underestimation of the measured signal intensity.
All in all, we showed that specific targeting of CysLT1R with a NIR fluorophore-coupled antibody seems to be a promising method to detect this receptor involved in inflammatory processes. As CysLT1R is involved in early regulation of inflammatory processes, our probes might be particularly helpful for the detection of early stages of inflammation, thus improving therapeutical potential.
Footnotes
Acknowledgments
We thank Yvonne Heyne for excellent technical assistance.
Financial disclosure of authors: These investigations were supported by the German Research Foundation (DFG) within program Hi 689/6-1. Dr. Matthias Wenzel is an employee and shareholder of Dyomics GmbH.
Financial disclosure of reviewers: None reported.
