Abstract
Inflammation in atherosclerotic plaques causes plaque vulnerability and rupture, leading to thromboembolic complications. Cathepsin B (CatB) proteases secreted by macrophages play a major role in plaque inflammation. We used a CatB-activatable near-infrared fluorescence (NIRF) imaging agent to demonstrate the inflammatory component in mice atheromata and the atherosclerosis-modulating effects of atorvastatin or glucosamine treatments. Apolipoprotein E knockout mice (n = 35) were fed normal chow, a Western diet, a Western diet + atorvastatin, a Western diet + glucosamine, or a Western diet + atorvastatin + glucosamine for 14 weeks. Twenty-four hours after the intravenous injection of a CatB-activatable probe, ex vivo NIRF imaging of the aortas and brains was performed, followed by histology. The CatB-related signal, observed in the aortas but not in the cerebral arteries, correlated very well with protease activity and the presence of macrophages on histology. Animals on Western diets could be distinguished from animals on a normal diet. The antiatherosclerotic effects of atorvastatin and glucosamine could be demonstrated, with reduced CatB-related signal compared with untreated animals. Plaque populations were heterogeneous within individuals, with some plaques showing a high and others a lower CatB-related signal. These differences in signal intensity could not be predicted by visual inspection of the plaques but did correlate with histologic evidence of inflammation in every case. This suggests that vulnerable inflamed plaques can be identified by optical molecular imaging.
CHOOSING AN OPTIMAL THERAPEUTIC STRATEGY for carotid or intracranial atherosclerosis, a leading cause of stroke, is not an easy task. Current practice guidelines for the management of carotid stenosis rely heavily on anatomic information, particularly the degree of stenosis. 1 However, clinicians often see a plaque causing high-grade carotid stenosis but no stroke or symptoms. Conversely, it is not unusual to see a carotid plaque causing low-grade stenosis being identified as the culprit lesion for a thromboembolic stroke, probably associated with acute plaque rupture.
The risk of plaque rupture or plaque instability is the key clinical information needed, but current anatomy-based imaging modalities such as carotid duplex ultrasonography, magnetic resonance angiography, and computed tomographic angiography have significant limitations in discriminating rupture-prone, high-risk atherosclerotic plaques from stable ones. 2 Accordingly, there has been a high expectation for molecular imaging to provide information on plaque stability in a manner that could complement or replace conventional imaging.3–5
Activatable near-infrared fluorescent (NIRF) imaging agents 6 have fluorochromes placed in close proximity to each other so that they self-quench at baseline, producing no fluorescent signal and thus minimizing background noise. The cleavable linkers of activatable agents can be chosen to be sensitive to inflammatory proteolytic enzymes such as matrix metalloproteinases or cathepsins, which recognize and cleave specific peptide sequences, to release fluorochromes. Once released, these fluorochromes are dequenched and become brightly fluorescent. Thus, the pathophysiology of vulnerable and rupture-prone atherosclerotic plaques, infiltrated by macrophages and rich in matrix-disorganizing proteolytic enzymes, could be exploited by using protease-sensing molecular optical imaging. The emerging technology of protease-sensing molecular optical imaging, when combined with a noninvasive tomography system 7 or intravascular NIRF catheters 8 during interventional angioplasty, could potentially be used for assessing in vivo protease activity to reflect inflammation and the vulnerability of human carotid or cerebral arteries.
As a step toward clinical translation of molecular optical imaging technology from laboratory to clinic, we investigated if cathepsin B (CatB) NIRF imaging could reliably reflect the antiatherosclerotic effect of atorvastatin and the proatherosclerotic effect of a high-cholesterol diet. This work builds on and extends on previous imaging studies9–11 and uses rigorous imaging and histologic correlations to illuminate imaging findings and make correlations between parameters such as plaque size and inflammation-related protease activity. Moreover, we used the NIRF CatB imaging to determine whether glucosamine—a widely used nutritional supplement that might have a proatherosclerotic effect 12 or an antiatherosclerotic effect 13 —could affect plaque size or inflammation. Studies on glucosamine and atherosclerosis have received great attention because glucosamine is frequently used for the treatment of arthritis, particularly in the elderly, who are also prone to the development of atherosclerosis and stroke.
We studied both the aorta and cerebral vessels and demonstrated a translational methodology that will likely be applicable in a variety of preclinical and clinical antiatherosclerotic interventions.
Materials and Methods
This study was performed in accordance with National Institutes of Health guidelines for the care and use of laboratory animals.
Animals
We used apolipoprotein E (ApoE) knockout (ApoE−/−) mice (n = 35; Japan SLC, Shizuoka, Japan) fed on a normal chow diet (protein 20.0%, fat 4.5%, fiber 6.0%, ash 7.3%; n = 7), a Western diet (sucrose 34.1%, milk fat 21%, casein 19.5%, cellulose 5%, comstarch 15%, cholesterol 0.2%; n = 7), a Western diet mixed with statin (0.01% wt/wt of atorvastatin; n = 7), a Western diet mixed with glucosamine (0.46% wt/wt of glucosamine; n = 7), or a Western diet mixed with atorvastatin and glucosamine (n = 7) for 14 weeks, which started 8 weeks after they were born. The animals were anesthetized with 2% isoflurane in a mixture of 30% oxygen and 70% nitrogen, and a 2 nmol CatB probe (ProSense-680, Visen Medical, Woburn, MA) in 150 μL Dulbecco's Modified Eagle's Medium (DMEM) (Gibco, NY) was intravenously injected. Twenty-four hours after the tail vein injection of the probe, blood was collected from the retro-orbital plexus and was centrifuged to measure lipid and glucose levels (Laboratory Animal Clinical Pathology Services, Uijeongbu, Korea). The animals were euthanized, and the aortas, after being photographed with a digital camera attached to a surgical microscope (S6E-zoom-stereomicroscope, Leica Microsystems, Heerbrugg, Switzerland), were carefully excised. After NIRF reflectance imaging, the aortas were used for immediate oil red O staining (n = 4 animals, selected randomly in each group) or embedded in paraffin for histology.
NIRF Imaging and Lesion Assessment
NIRF imaging and lesion quantification were performed as published previously.11,14 The excised brains and aortas were washed with DMEM three times and imaged ex vivo by using a NIRF imaging machine with a charge-coupled device camera (CoolSnap EZ, Roper Scientific, Tucson, AZ). White light and Cy5.5 NIRF images (1-second acquisition) were acquired. Six subdivisions of the aortic tissues were defined as regions of interest, including the aortic arch, brachiocephalic artery, left common carotid artery, left subclavian artery, and upper and lower segments of the descending thoracic aorta. After normalization, the median NIRF signal intensities of the entire tissues and the regions of interest were measured using the histogram function of Adobe Photoshop (Adobe Photoshop CS-3 Extended, Adobe Systems, San Jose, CA).
Quantification of Aortic Atherosclerotic Lesions
Atherosclerotic lesion size was determined using the en face method after oil red O staining. After capturing the pinned-open aortas with a digital gross photography system (L-800X, HuminTec, Suwon, Korea), the amount of oil red O staining was quantified with color thresholding to delimit areas of staining and analyzed with the Adobe Photoshop histogram function. Data are reported as a percentage of the aortic surface covered by atherosclerotic lesions divided by the total surface area of the aorta. Inter- and intrauser reproducibility for the measurement was excellent (data not shown).
Histology
Longitudinal aortic sections (5 μm) or sagittal brain sections (4 μm) of the paraffin blocks were used for Masson trichrome staining or immunohistochemistry. Immunohistochemistry was performed using the avidinbiotin-peroxidase method. Briefly, sections treated with 0.3% of hydrogen peroxide were incubated for 60 minutes with primary antibodies, followed by biotinylated secondary antibodies (EnVision, DAKO, Glostrup, Denmark). The reaction was visualized with DAB substrate (DAKO, Glostrup, Denmark) and counterstained with Harris hematoxylin solution. Cells were identified with anti-Mac3 antibody (1:75; BD Bioscience, San Jose, CA) for macrophages. Staining for CatB was done using rabbit polyclonal antibody (1:75; Biovisions, Mountain View, CA). TG-2 staining was performed by using mouse monoclonal antibody (1:1,000; Lab Vision, Fremount, CA).

Cathepsin B (CatB) near-infrared fluorescence (NIRF) imaging to identify and characterize atherosclerotic lesions. On the light image (A, D), a representative 22-week-old ApoE knockout mouse on a Western diet has multiple atherosclerotic plaques in the aorta and its branches. CatB NIRF images and pseudocolor overlay images of the vessels (B, C, E, F) show stronger protease activity signal in some plaques or plaque areas (red arrows) than in others (blue arrows). The plaques with a higher or lower CatB signal correlate precisely with CatB/macrophage (Mac-3)-positive immunohistochemistry data (G, H). Unlike the CatB image, the light image does not predict which plaques will show macrophage infiltration (A, D). Plaque size is not predictive of the CatB activity, and the bigger plaque has weaker CatB activity (blue curved arrows), whereas the smaller plaque has stronger activity (red curved arrows). DAB-immunoreactive areas for CatB and macrophages are located mainly in the plaque caps or shoulder regions (G, H, and insets G*, H*). White scale bars and black scale bars (G, H) = 1 mm; black scale bars (G*, H*) = 100 μm.
Statistical Analyses
Data are presented as mean ± standard deviation. One-way analysis of variance (ANOVA) with the Dunnett post hoc test was used for comparison of continuous variables between groups. Bivariate correlations were calculated to see how two variables are related. A p value of less than .05 was considered statistically significant.
Results
The NIRF CatB-related signal in mice atheromata correlates closely with proteolytic enzymes from macrophages in histology sections
Figure 1 shows that the NIRF CatB-related signal correlated very well with the presence of macrophage infiltration in some plaques and thus with the presence of CatB in the interstitium. Our histologic studies confirmed that macrophages and CatB colocalize. This supports the known propensity of these inflammatory cells to secrete the CatB proteolytic enzyme, which could cleave and activate the NIRF imaging probe. CatB activities and infiltration of macrophages were observed mainly in the caps or shoulders of atherosclerotic plaques. The inflammatory activity of each plaque, as determined by the CatB-related imaging signal, corresponded exactly to Mac-3 or CatB immunoreactivity on histologic studies. Notably, some plaques had a stronger CatB imaging signal than others, and these differences in signal intensity could not be predicted by visual inspection of the plaques. For example, bigger plaques did not always have stronger CatB activities than smaller ones. The CatB-related signal gave information about the plaques that could not be obtained with means other than histology, and in every case where imaging predicted an inflammatory component in the plaque, histologic studies confirmed it to be the case.
NIRF CatB imaging of mice atheromata reflects the proatherosclerotic effect of a Western diet and antiatherosclerotic effect of atorvastatin in a quantitative manner
Five-month-old ApoE knockout mice fed on a normal diet, a Western diet, or a Western diet mixed with atorvastatin for about 3 months could clearly be distinguished from each other on the basis of NIRF CatB imaging. The atherogenic effect of the Western diet and the antiatherogenic effect of the atorvastatin were clearly demonstrated (Figure 2). As shown in Figure 3, the mean of the CatB-related signal intensities (arbitrary units [AU]) from the whole aortas was significantly higher in the Western diet group (53.9 ± 22.0 AU) than in the atorvastatin group or the normal-diet group (26.0 ± 10.5 AU, 26.6 ± 19.8 AU; p < .05, one-way ANOVA with the Dunnett post hoc test). The mean of CatB-related signal intensities in the aortic arch, the most proximal portion of the aortic tissue from the heart, was the highest in each group, and the values did not show significant differences. In the brachiocephalic and left common carotid arteries, the second-most proximal portion, the mean CatB activities were lower in the normal-diet group than in the other groups, which did not reach statistical significance either. In the remaining more distal portions, that is, the left subclavian artery and the descending thoracic aorta, the mean CatB activities were higher in the Western diet group than in the atorvastatin group and the normal-diet group. Therefore, the intergroup differences of the protease-related signal in the whole aorta could be attributed to differences in the left subclavian artery and thoracic aorta, particularly the lower segment of the thoracic aorta, with statistical significance (p < .05, one-way ANOVA with Dunnett post hoc test).

Cathepsin B (CatB) near-infrared fluorescence (NIRF) imaging to demonstrate the proatherosclerotic effect of a high-cholesterol diet and the antiatherosclerotic effect of atorvastatin or glucosamine treatment. Ex vivo NIRF CatB imaging was performed in 22-week-old ApoE knockout mice (n = 35) fed a normal diet, a Western diet, a Western diet mixed with atorvastatin, a Western diet mixed with glucosamine (GL), or a Western diet mixed with atorvastatin + GL for 14 weeks. In the representative images, the protease-related signal is stronger in the mouse on a Western diet than in the mouse on a normal diet. In the animal on a Western diet mixed with atorvastatin and/or glucosamine, the CatB-related signal is weaker than in the animal on a Western diet alone. Light images and oil red O staining results corroborate the pro- and antiatherosclerotic effects. Scale bars = 2 mm.
Glucosamine tends to attenuate the CatB-related signal in the atherosclerotic aortas of ApoE knockout mice on a Western diet
Figure 2 and Figure 3 show that the mean of CatB-related signal intensities from the whole aortas was lower, with marginal significance in the glucosamine group (30.6 ± 23.9 AU, p = .07) and the atorvastatin + glucosamine group (32.1 ± 7.3 AU, p = .098) than in the Western diet group (53.9 ± 22.0 AU) (ANOVA with Dunnett post hoc test). The possible antiatherosclerotic effect of atorvastatin + glucosamine combination therapy was not different from that of either atorvastatin monotherapy or glucosamine monotherapy. Oil red O staining of the aortas for the four randomly selected animals in each group (Figure 2 and Figure 4) showed that atorvastatin treatment could reduce the plaque size of the ApoE knockout mice on a Western diet (from 48.5 ± 12.8% to 24.8 ± 7.8%, p < .05, one-way ANOVA with Dunnett post hoc test). Glucosamine appeared to have a similar effect (to 32.3 ± 8.2%; see Figure 2), which did not reach statistical significance (p = .14, Dunnett post hoc test).

Quantification of cathepsin B (CatB) near-infrared fluorescence (NIRF) imaging signal intensity and oil red O staining–positive atherosclerotic lesions. A, The CatB-related signals (arbitrary units [AU]) from the whole aortas (n = 35) are significantly higher (p = .04, ANOVA) in the Western diet group compared with the other groups, including the normal-diet group (p = .03), the atorvastatin group (p = .02), the glucosamine group (p = .07), and the atorvastatin + glucosamine group (p = .098) (Dunnett post hoc test). Subgroup analyses indicate that much of the differences in the whole aortas could be attributed to signal differences in the left subclavian arteries and the lower thoracic aortas from among six subdivisions: the aortic arch (AA); brachiocephalic artery (BC); left common carotid artery (LC); left subclavian artery (LS); upper thoracic aorta (UT); and lower thoracic aorta (LT). B, The extent of atheromata, measured by calculating the percentage of oil red O staining of the total aortic surface area (n = 4 in each group), was smaller in the atorvastatin-treated groups than in the Western diet group (p < .05, ANOVA with Dunnett post hoc test). Glucosamine-only treatment tended to have a similar effect, which did not reach statistical significance (p = .14, Dunnett post hoc test). C, The CatB activities from the individual whole aortic tissues were weakly correlated with the plaque extents (plaque area %) of the corresponding aortas, which showed a marginal significance (p = .07, Pearson correlation). * p < .05; #p < .1.
Glucosamine treatment does not completely suppress TG-2 expression of atherosclerotic plaques
Recently, glucosamine was shown to inhibit TG-2 activity 15 that could contribute to various inflammatory disease pathologies, including atherosclerosis. 16 In line with the decrease in the plaque burden, TG-2 expression of the entire aorta was less abundant in the glucosamine group than in the Western diet group (see Figure 4). However, TG-2 expression of an individual plaque did not show any noticeable intergroup differences or treatment group–specific patterns.
The antiatherosclerotic effect of atorvastatin and/or glucosamine was not associated with cholesterol lowering
The antiatherosclerotic effect of atorvastatin was not associated with lower cholesterol serum levels (Table 1). In fact, total cholesterol levels were nonsignificantly higher in the atorvastatin group (1,163 ± 105 mg/dL) than in the Western diet group (896 ± 336 mg/dL, p = .12). They were significantly lower in the normal-diet group (460 ± 54 mg/dL) than in the Western diet group (p = .01) or the Western diet + atorvastatin groups, including the atorvastatin group (p = .00) and the atorvastatin + glucosamine group (790 ± 201 mg/dL; p = .04). They were nonsignificantly lower in the normal-diet group than in the Western diet + glucosamine group (718 ± 247 mg/dL; p = .14). Between the Western diet group and the Western diet + glucosamine group, there was no difference (p = .42). It is notable, however, that the paradoxical increase in the cholesterol levels by atorvastatin could be prevented by the concomitant treatment of glucosamine (p = .02). Intergroup differences of low-density lipoprotein (LDL) cholesterol levels show trends similar to those of the total cholesterol levels. High-density lipoprotein cholesterol, triglyceride, and glucose levels did not differ between the groups (one-way ANOVA with Dunnett post hoc tests).
Median NIRF CatB signal intensities of the whole aortic tissues correlate “weakly” with the oil red O–positive area percentages of the total surface areas
There was only a weak correlation between the CatB activity of each aortic tissue and the oil red O staining–positive lesional extent in the corresponding aorta, expressed as a percentage of the total surface area (p = .07, r2 = .18, Pearson correlation; see Figure 3C).

Glucosamine treatment and transglutaminase-2 (TG) expression in atherosclerosis. In line with an overall decreased plaque burden, TG-2 expression of the entire aorta is less abundant in the glucosamine-treated group than in the Western diet group. However, TG-2 expression of an individual plaque does not show intergroup differences or treatment group–specific patterns. TG-2 expression is observed in and around the plaque caps and underlying necrotic cores (black arrowheads and blue arrow). On the higher-magnification view, TG-2 expression is observed in the macrophages (blue and red arrows). In the Western diet group, the macrophage staining for a nearby section (Mac-3) reveals clustered macrophages. M-T = Masson trichrome staining. Black scale bars = 1 mm; red scale bars = 50 μm.
Cholesterol and Glucose Levels (Mean ± SD mg/dL) of the Apolipoprotein E Knockout Mice (n = 6 in Each Group)
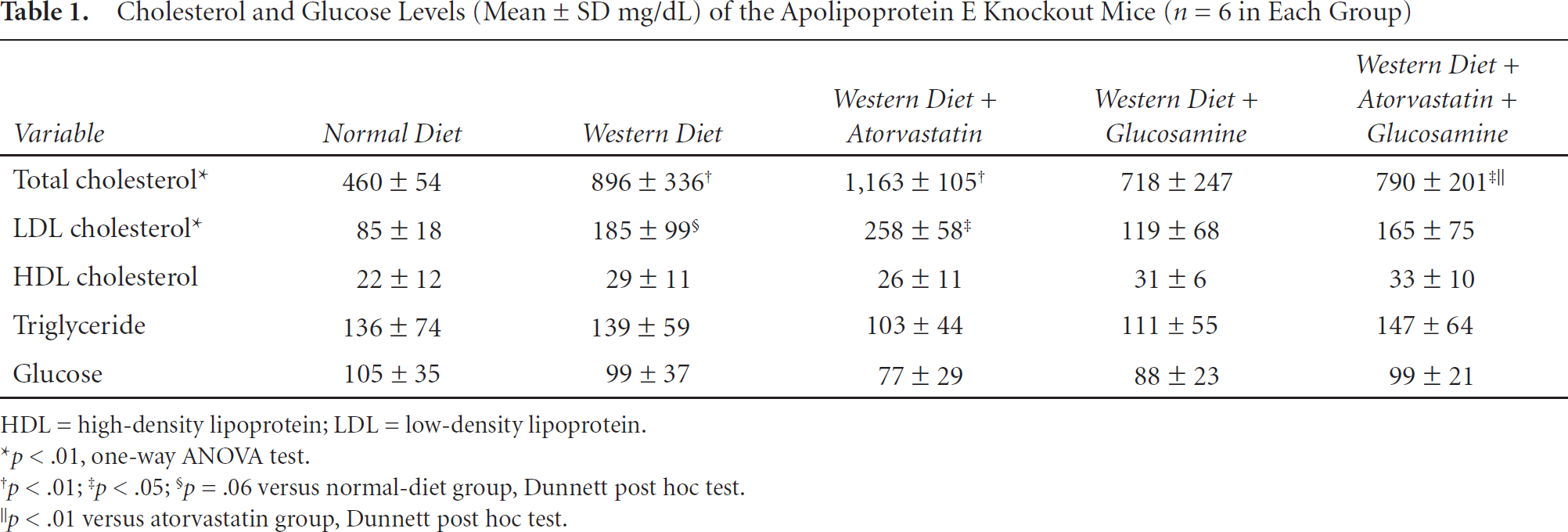
HDL = high-density lipoprotein; LDL = low-density lipoprotein.
p < .01, one-way ANOVA test.
p < .01
p < .05
p = .06 versus normal-diet group, Dunnett post hoc test.
p < .01 versus atorvastatin group, Dunnett post hoc test.
NIRF CatB-related signals are observed in the aortas but not in the cerebral arteries of the ApoE knockout mice
Unlike in the aortic tissues with advanced atherosclerotic changes and a high CatB-related signal, a high protease-related signal was never observed in any cerebral arteries or brain cortices of the ApoE knockout mice (n = 35; Figure 5). Cerebral artery atherosclerosis or CatB immunoreactive areas were not observed on the histology either (data not shown).
Discussion
CatB-based protease-sensing NIRF imaging allowed us to clearly demonstrate plaques with an inflammatory component and reliably distinguish them from noninflamed plaques. This allowed (a) a clear distinction to be made between animals on normal and Western (proatherogenic) diets (b) and an assessment of antiatherosclerotic interventions such as atorvastatin and glucosamine. CatB-based imaging gave molecular information about the plaques that could not be obtained with means other than histology. This suggests that CatB-based NIRF imaging could complement anatomically based conventional imaging by providing information on plaque inflammation and stability through imaging the activity of proteolytic macrophage enzymes.3–5 Similarly, NIRF CatB imaging could complement current preclinical tools for translational atherosclerosis research in demonstrating the molecular effects of antiatherosclerotic interventions in a quantitative manner. For example, we have shown in our present study how imaging could demonstrate the atherosclerosis-modulating effect of both atorvastatin and glucosamine.

Cathepsin B–related near-infrared fluorescence molecular optical imaging to reveal significant inflammatory protease-related activity in the aorta (A, D) but not in the cerebral arteries (B–C, E and F) of the ApoE knockout mice fed on a normal diet or a Western diet. Scale bars = 2 mm.
CatB imaging shows protease activity in atherosclerotic plaques with high precision, as demonstrated with imaging-histology correlations (see Figure 1). The CatB activity signal on molecular imaging colocalized with protease immunoreactivity on the histology. It is important to note that atherosclerosis is a systemic disease, and unlike medical therapy, endarterectomy and endovascular intervention are localized treatments. In this context, it is important to note that some plaques had a strong CatB signal on the molecular imaging, whereas others had a relatively weak or only faint signal. In the clinical setting, the former plaques could be classified as being more vulnerable to rupture and thus requiring more aggressive treatments than the latter ones. A high CatB-related signal with dense macrophage infiltration was mainly observed in the rupture-prone areas of plaques, such as the shoulder regions or plaque cap surface of atheromata. This brightens clinical prospects for surface-weighted reflectance fluorescence imaging techniques, combined with fluorescence-sensing intravascular catheters, 8 to detect high-risk plaques in the atherosclerosis clinic.
There was only a weak correlation between the total NIRF CatB signal intensities of whole aortas and the quantity of oil red O staining lesions in the same tissue. This reflects discordance between inflammatory activity and lipid accumulation in atherosclerosis. This suggests that lipid accumulation and inflammation can be viewed as separate entities, and although related, they appear to have some measure of independence from each other. The inflammatory component of atherosclerosis is superimposed on preexisting lipid accumulations, and although it is the component of atherosclerosis responsible for the advent of symptoms, regular imaging aimed at anatomic disturbances or lipid accumulation will not detect the more subtle, but likely very important, inflammatory component that predicts symptomatic complications. Plaque inflammation could not be predicted by visual inspection of the atherosclerotic plaques under the surgical microscope, but a CatB-related signal on imaging was highly effective at identifying inflammation when present. Plaque size was not predictive of the inflammatory protease activity either. This is in line with the fact that conventional anatomy-based imaging tools often fail to predict plaque instability and subsequent stroke. Molecular imaging can probe the underlying molecular alterations 17 of vascular inflammation causing plaque rupture, not accessible to conventional imaging. A prospective study to see if molecular imaging could predict subsequent plaque rupture would be interesting; however, reliable animal models of spontaneous plaque rupture are still lacking. 18
We and others have shown that molecular optical imaging can be used for in vivo or ex vivo therapeutic monitoring of antiatherosclerotic treatment,10,19 as was also reported by Graf and colleagues using a targeted probe, an antibody against a splice variant of fibronectin extradomain B linked to a NIRF dye. 20 In the present study using a targeted activatable probe, we demonstrated that CatB-related signals in the aortas were higher in the Western diet group than in the normal-diet group. When animals were fed on a Western diet mixed with atorvastatin, the CatB-related signal was decreased to a level similar to that of the normal-diet group. An intriguing feature of ApoE knockout mice is that statin's cholesterol-lowering effects are not consistently observed.21,22 We revealed nonsignificant increases in cholesterol levels after atorvastatin medication for 2 months. Similarly, another group of researchers reported that simvastatin administration for 2 months elevated serum total cholesterol about 35%. 22 Given that the decreased CatB activity in our study was not associated with cholesterol lowering, it is likely that a direct antiinflammatory pleiotropic effect of statin may have been involved in the antiatherogenesis.21–23 Recently, Kircher and colleagues used a micro-single-photon emission computed tomography small-animal imaging system to show that statins reduced 111indium oxine–labeled monocyte recruitment to existing atherosclerotic lesions in mice by acting on the vessel wall, not on the monocytes. 24 Tang and colleagues showed that ultrasmall superparamagnetic iron oxide (USPIO)-enhanced magnetic resonance imaging could demonstrate reduction of USPIO-defined inflammation in human carotid atheromata through aggressive lipidlowering therapy over a 3-month period. 25 These studies, in line with our results, also support the notion that statin has a direct antiinflammatory effect. Moreover, we found that the paradoxical increase in the cholesterol levels by atorvastatin could be lowered by the concomitant supplementation of glucosamine. However, the mechanism and possible clinical implications need further investigations.
The pro- and antiatherosclerotic effects of a Western diet and atorvastatin on the protease activities were exhibited mainly in the distal portion of the aorta and its branches, including the left subclavian artery and lower thoracic aorta. In the arch, the most proximal portion of the aortic tissue, the mean CatB activities were the highest, but without significant intergroup differences. In the midportion, the brachiocephalic and left common carotid arteries, the mean CatB activities were nonsignificantly lower in the normal-diet group than in the Western diet group. However, there was no difference between the Western diet and atorvastatin groups. It is likely that aortic atherosclerosis progresses along a proximal-to-distal direction in ApoE knockout mice. 26 This could cause ceiling effects in the proximal aortic tissues, which may have sustained early damage too heavy to permit the statin's antiatherosclerotic effect to show.
The literature to date has shown conflicting results about glucosamine and atherosclerosis. Tannock and colleagues reported that glucosamine, when added to the drinking water, had accelerated early atherosclerosis after 5 weeks but not late atherosclerosis after 10 or 20 weeks in low-density lipoprotein receptor-deficient (LDLR−/−) mice. 12 They concluded that despite exacerbated hyperlipidemia, glucosamine appeared to be safe because long-term supplementation of the drug has no effect on the progression of atherosclerosis. On the other hand, Duan and colleagues showed that intraperitoneal injection of glucosamine for 8 weeks had reduced atherosclerotic lesions in aortic root of the ApoE knockout mice at 12 weeks of age without changes in plasma glucose and lipids. 13 As a mechanism for the protective role, the authors suggested increased production of heparan sulfate proteoglycan (HSPG) associated with decreased protein transport across endothelial cell monolayers, decreased monocyte binding to the subendothelial matrix, and decreased vascular smooth muscle cell proliferation. 13 They argued that a decrease in HSPG might be a general inflammatory reaction because lipopolysaccharide, tumor necrosis factor α, homocysteine, lysolecithin, and oxidized LDL could decrease HSPG.
Our study showed that high-dose glucosamine could attenuate the activity of CatB, one of the important inflammatory proteases from macrophages, in the atherosclerotic aortas of ApoE knockout mice. We also observed that glucosamine appeared to reduce the plaque size. Recently, glucosamine was revealed to have a TG-2 inhibitory function. 15 It is well known that TG-2 expression is increased in established atherosclerotic lesions. In addition, there has been a notion that TG inhibition may be beneficial in diseases associated with inflammation, 16 which is in line with the results of our study. Aberrant induction of TG-2 activity, which triggers continuous nuclear factor κB activation without I-κBα kinase signaling, 27 contributes to various inflammatory disease pathologies, including atherosclerosis. 16 On the contrary, Boisvert and colleagues showed in lethally irradiated LDLR−/− mice transplanted with TG-2−/− or TG-2+/+ bone marrow that macrophage TG-2 expression promoted both apoptotic cell clearance and ABCA1 protein and mRNA expression in vitro and limited atherosclerotic lesion size in vivo. 28 Previously, transforming growth factor β (TGF-β) receptor expression was reported to be most robust in the fibrous cap, and TGF-β theoretically promotes plaque stability by stimulating collagen synthesis. 29 Taken together, these led Boisvert and colleagues to argue that macrophage TG-2 deficiency might disturb plaque stability by decreased TGF-β production as well as increase plaque size by decreased phagocytosis, 28 which is in opposition to our data. However, transglutaminase is involved not only in phagocytosis but also in macrophage adhesion to and transmigration across the endothelium at sites predisposed to atherosclersosis, one of the earliest detectable cellular responses in atherogenesis leading to the formation of atherosclerotic lesions.30–34 Glucosamine may not destabilize the plaque either if we consider that it decreased the activity of CatB proteolytic enzyme in the atherosclerotic aortas of the ApoE knockout mice in our study. In addition, glucosamine treatment does not seem to completely suppress TG-2 expression, as was shown in the immunohistochemical study. Moreover, some researchers showed that glucosamine could induce TGF-β expression. 35 Aforementioned conflicting results about the roles of glucosamine and/or TG for atherosclerosis could arise from a lot of methodologic differences between the studies. Our study, based on the NIRF CatB imaging of the aortic tissues, could provide specific molecular information related to the inflammatory pathophysiology of atherosclerosis in a quantitative manner, which has not been available in this research field. Although our study was limited to the ex vivo imaging, we and others have previously shown that—despite the low penetration depth of the emitted light—fluorescence-mediated molecular tomography could sense a NIRF signal from the aorta in vivo. 10 Many of the light penetration issues might be obviated in angiographic catheter-based intraluminal detection systems; however, such systems are not currently available for mice.
We tried to apply our technique to the cerebrovascular tree. Although a CatB-related signal was consistently observed in the aorta and its branches, no such signal was ever detected from the cerebral arteries of any ApoE knockout mice in our study, and neither did histology show any evidence of atherosclerosis in these vessles. We believe that this is a meaningful negative result. Kitayama and colleagues also reported that ApoE knockout mice did not have morphologic changes in the cerebral arterioles, although the animals did have cerebral endothelial dysfunction. 36 The human internal carotid artery (ICA) drains directly into the middle cerebral artery (MCA) without flowing a distance through the circle of Willis, but the mouse MCA arises at a significant distance from the site of the ICA, reaching the circle of Willis. Okuyama and colleagues argued that these anatomic distinctions might mitigate the capacitor and equalizer functions of the circle of Willis so much that the resultant hemodynamic changes would render the human MCA, but not the mouse MCA, more likely to undergo cerebrovascular accidents. 37 These hemodynamic changes might be partly related to the lack of atherosclerotic lesions in the mouse cerebral arteries. Given that CatB imaging is a sensitive tool to be able to detect subtle activities of inflammatory proteases, 17 our translational molecular imaging study is not only corroborating previous research, in which the cerebral arteries of ApoE knockout mic or ApoE/LDL double knockout mic were free from established atherosclerosis, 38 but also revealing the lack of early inflammatory changes in the cerebral arteries of ApoE knockout mic.
In summary, our study demonstrates a rationale for the clinical translation of NIRF CatB imaging to humans. CatB imaging is an effective tool for translational preclinical atherosclerosis research by providing molecular information on plaque inflammation.
Footnotes
Acknowledgments
Financial disclosure of authors: This research was supported by a grant from the Korea Healthcare Technology R&D Project (for the Translational Stroke Research Center; A08020, to Dr. Dong-Eog Kim), Ministry of Health & Welfare, Republic of Korea.
Financial disclosure of reviewers: None reported.
