Abstract
Integrins, particularly the αvβ3 heterodimers, play important roles in tumor-induced angiogenesis and invasiveness. To image the expression pattern of the αvβ3 integrin in tumors through a multimodality imaging paradigm, we prepared a cyclic RGDyK peptide analogue (LS308) bearing a tetraazamacrocycle 1,4,7,10-tetraazacyclododecane-N, N′, N″, N‴-tetraacetic acid (DOTA) and a lipophilic near-infrared (NIR) fluorescent dye cypate. The αvβ3 integrin binding affinity and the internalization properties of LS308 mediated by the αvβ3 integrin in 4t1luc cells were investigated by receptor binding assay and fluorescence microscopy, respectively. The in vivo distribution of 111In-labeled LS308 in a 4t1luc tumor-bearing mouse model was studied by fluorescence, bioluminescence, planar gamma, and single-photon emission computed tomography (SPECT). The results show that LS308 has high affinity for αvβ3 integrin and internalized preferentially via the αvβ3 integrin-mediated endocytosis in 4t1luc cells. We also found that LS308 selectively accumulated in αvβ3-positve tumors in a receptor-specific manner and was visualized by the four imaging methods. Whereas the endogenous bioluminescence imaging identified the ensemble of the tumor tissue, the fluorescence and SPECT methods with the exogenous contrast agent LS308 reported the local expression of αvβ3 integrin. Thus, the multimodal imaging approach could provide important complementary diagnostic information for monitoring the efficacy of new antiangiogenic drugs.
INTEGRINS ARE A FAMILY of heterodimeric transmembrane proteins consisting of alpha and beta sub-units. The combination of an alpha and a beta subunit confers the unique binding properties of that integrin. The alpha and beta subunits are noncovalently linked, and each subunit has a large extracellular domain, a single membrane-spanning domain, and a relatively small cytoplasmic domain. 1 The normal function of this family of proteins is mediation of cell-cell contacts as well as cell-extracellular matrix interactions. Additionally, there is considerable evidence for their mediation of tumor progression and metastasis. 2 For example, αvβ3 integrin is strongly expressed on the invasive front of malignant melanoma cells but weakly expressed on preneoplastic melanomas. 3 Integrins, particularly αvβ3, also play important roles in tumor angiogenesis.
Angiogenesis, the formation of new blood vessels, is essential for tumor growth. The recruitment of endothelial cells by developing tumors to form new blood vessels is a complex process, involving the release of growth factors by the developing tumor with support from stromal cells. In such a complex process, there are numerous ways of intervening in tumorigenesis. One of these ways is by inhibition of angiogenesis. Among the biomarkers for angiogenesis that has emerged is the αvβ3 integrin. αvβ3 integrin is highly expressed on the endothelial cells of angiogenic blood vessels but has very low expression in normal blood vessels. 4 Furthermore, antagonism of αvβ3 integrin with antibodies blocks tumor progression through inhibition of angiogenesis. 5 Several of these agents are in clinical trials as cancer therapeutics. 2
Based on the aforementioned functions of αvβ3 integrin as a surrogate biomarker for tumor invasiveness or angiogenesis, noninvasive imaging of αvβ3 expression in diseased tissue would be of great benefit in directing adjuvant therapy for cancer patients. To this end, a number of radiolabeled peptides bearing the RGD motif have been radiolabeled and investigated in tumor-bearing animal models. The peptides are mostly based on the structure of c(RGDfV), a potent and selective binder of the αvβ3 integrin. 6 The binding conformation is mediated by the cyclic structure and by the amino acids flanking the RGD peptide sequence. 7 For imaging applications, the cyclic peptides are typically labeled with radiohalogens, 18F and 125I, as well as radiometals, 99mTc, 64Cu, and 90Y, incorporated into the peptide structure with chelators.8–12 These radiolabeled RGD-based analogues were intended for either single-photon emission computed tomography (SPECT) or positron emission tomography (PET). Both of these imaging modalities are effective for molecular imaging owing to their inherent high sensitivity and their ability to image deep tissues.
As an alternative to these means of imaging with radioisotopes, optical imaging with near-infrared (NIR) fluorescent dyes is highly promising. Optical imaging is a highly sensitive technique that can detect, in vitro and in vivo, minute quantities of light-emitting materials. Additionally, it uses low-energy radiation in the visible or NIR region of light to assess biologic processes. For these reasons, cyclic RGD peptides have been conjugated to NIR fluorescent dyes for optical imaging of subcutaneous tumor xenografts. The fluorescent dye-labeled peptides were shown to be effective for receptor-specific visualization of the αvβ3 integrin-positive tumors.13,14 Although algorithms can deconvolute light propagation through tissues, development of accurate models of the complex photon migration in deep heterogeneous biologic systems remains a major challenge. This limitation would benefit from pairing optical imaging with another imaging modality, such as SPECT. By coupling an optical and a radionuclear method, the location of a target tissue could first be confirmed by SPECT and then monitored longitudinally by optical imaging to report such changes as tumor response to therapy.
There has already been a report of αvβ3 integrin imaging in a mouse model where a cyclic RGD analogue bore a dual label of 111In-diethylenetriaminepentaacetic acid (DTPA) and the NIR dye IRDye800. 15 The dual-labeled αvβ3 probe demonstrated receptor-specific localization to a subcutaneous tumor xenograft and allowed for visualization of the tumor both by NIR reflectance imaging and planar gamma scintigraphy. In the previous study, the labeled agent was of relatively low specific activity, and the αvβ3 integrin binding affinity was not established. We prepared a cyclic RGDyK peptide analogue (LS308) bearing a tetraazamacrocycle 1,4,7,10-tetraazacyclododecane-N, N′, N″, N‴-tetraacetic acid (DOTA) and a lipophilic NIR fluorescent dye cypate. The αvβ3 binding affinity and the internalization properties of LS308 mediated by the αvβ3 integrin in 4t1luc cells were investigated by solid-phase receptor binding assay and fluorescence microscopy, respectively. Finally, we imaged the distribution of 111In-labeled LS308 in a tumor-bearing mouse model by NIR fluorescence, bioluminescence, planar gamma, and SPECT imaging.
Materials and Methods
Materials
LS308 and the control LS340, as well as a cypate-labeled cyclic RGD peptide (LS167), were synthesized, purified, and characterized by the methods described previously.16–18 The structures of the compounds are shown in Figure 1. Reagents used in this study were purchased from Sigma-Aldrich (St. Louis, MO).
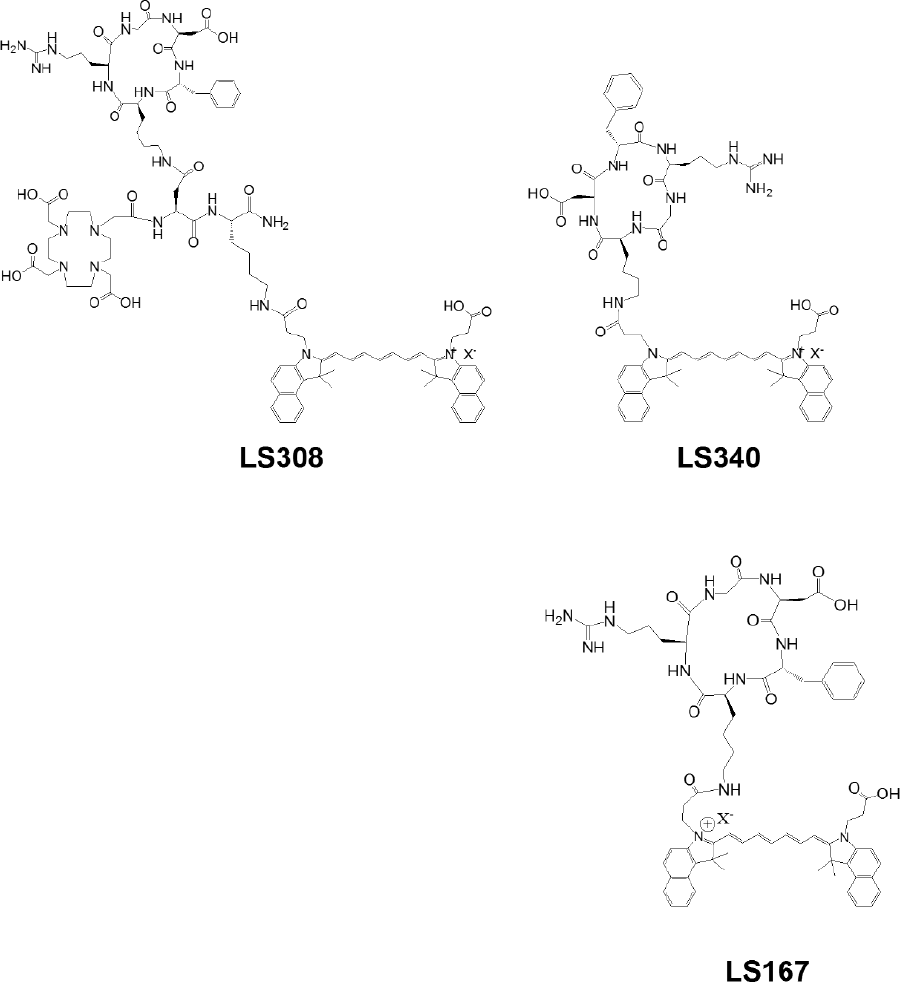
Structures of LS308, LS340, and LS167.
Radiochemistry
LS308 was radiolabeled with 111In as previously described. 19 Briefly, 111In was added to labeling buffer (ammonium acetate, 100 mM, pH 5.5) followed by LS308 in dimethyl sulfoxide (DMSO) (final concentration of 20% by volume). The mixture was heated at 80°C for 1 hour. Radiochemical purity was determined by reverse-phase high-performance liquid chromatography (A = water, 0.1% trifluoroacetic acid [TFA]; B = acetonitrile 0.1% TFA; Vydac, C-18, 201HS5415, 4.6 × 150 mm, linear gradient 30 to 70% B in 10 minutes, 1 mL/min) with a scintillation detector.
Solid-Phase Receptor Binding Assay
The receptor binding assays were carried out using purified human αvβ3 integrin (Chemicon International, Inc., Temecula, CA) in a 96-well White Polystyrene Medium Bind Stripwell Microplate (Corning Headquarters, Corning, NY). The microplates were coated with the purified αvβ3 integrin (500 ng/mL; 100 μL/well) in coating buffer” (25 mM Tris-HCl, pH 7.4, 150 mM NaCl, 1 mM CaCl2, 1 mM MgCl2, and 1 mM MnCl2) for 16 hours at 4°C. The wells were then blocked for 2 hours with 150 μL of binding buffer (coating buffer with 0.5% bovine serum albumin). After washing with binding buffer (twice), the plates were incubated with 125I-c(RGDyK) (2 nM) in the presence of various concentrations of ligand ranging from 1 pM to 10 μM (n = 3) for 16 hours at room temperature, followed by washing with binding buffer (three times). Less than 10% of the added 125I-c(RGDyK) was bound to the protein, justifying the use of the Cheng-Prusoff equation to calculate affinity constant (Ki) values. Bound radioactivity to αvβ3 integrin was counted by a Packard Cobra II Auto-Gamma Counter (Meriden, CT). Nonspecific binding of 125I-c(RGDyK) was < 10% of the total binding. The 50% inhibitory concentrations (IC50) and Ki were calculated by nonlinear regression using GraphPad Prism 4 software (GraphPad Software, Inc., San Diego, CA). Data were fit to a sigmoidal dose response with a variable Hill slope parameter (Table 1). Previously determined values for 125I-c(RGDyK) were used to calculate the Ki values (dissociation constant KD = 0.4 nM).
Solid-Phase Heterologous Competitive Binding

CI = confidence interval; IC50 = 50% inhibitory concentration; Ki = affinity constant.
Fluorescence Microscopy
The 4t1Luc cells were purchased from Sibtech (Brookfield, CT) and maintained in a humidified atmosphere containing 5% CO2 at 37°C in Dulbecco's Modified Eagle's Medium (DMEM) supplemented with 10% fetal calf serum, 100 U/mL penicillin, and 100 Us/mL streptomycin. The cells were grown on Lab-Tek slides and treated with 1 μM LS308 or 1 μM LS340 for 1 hour. After treatment, the cells were rinsed with phosphate-buffered saline (PBS) containing 1 mM CaCl2, mounted with ProLong Gold mounting medium (Invitrogen, Carlsbad, CA), and cover-slipped. The cells were visualized with a NIR confocal microscope using a water immersion objective (60×/1.20 M, 0.13–0.21 NA) and a 780 nm laser. Fluorescence emission was captured at 805 to 830 nm. For the blocking studies, the cells were treated (30 minutes, ambient temperature) with either monoclonal antibody (Mab)-anti-αvβ3 (60 nM, Chemicon) or c(RGDyK) (100 nM) prior to the addition of dye-conjugated peptides.
Immunocytochemistry
4T1 and 4t1luc cells were seeded on Lab-Tek slides and grown in DMEM with 10% fetal bovine serum (Gibco BRL, Grand Island, NY) and 100 U/mL penicillin at 37°C in a humidified 5% CO2 incubator. The cells were fixed (3% paraformaldehyde in PBS) for 30 minutes. After rinsing (three times, 5 minutes each, PBS with 1 mM CaCl2 [PBS-Ca]), the cells were treated with 3% H2O2 (15 minutes) to reduce endogenous peroxidase activity. Nonspecific binding sites were blocked by PBS-Ca containing 1% normal rabbit serum (blocking solution) for 1 hour. For immunoperoxidase staining, the cells were incubated overnight at 4°C with anti-integrin αv antibody or anti-integrin β3 antibody (goat; Santa Cruz Biotechnology, Inc., Santa Cruz, CA). The antibodies were diluted by 1:250 (v/v) in the blocking solution. After rinsing (three times, 5 minutes each, PBS-Ca), the cells were incubated at room temperature (1 hour) with biotinylated antigoat IgG antibody (Vector, Burlingame, CA) in the blocking solution. Bound antibodies were detected with avidin-horseradish peroxidase conjugate and diaminobenzidine. The slides were counterstained with Mayer hematoxylin, and the coverslips were mounted for light microscopy.
Optical and Planar Gamma Imaging
Animal studies were conducted in accordance with our animal protocol, approved by the Washington University Animal Studies Committee. Syngenic tumors were prepared by subcutaneous implantation of 4t1luc cells (2 × 105 cells, 100 μL PBS) in the shoulders of 4-week-old male NCR nude mice (Taconic, Hudson, NY). Imaging studies were conducted when tumors reached 5 to 10 mm maximum diameter (≈10 days). For in vivo imaging, the mice were anesthetized with a ketamine-xylazine cocktail. Imaging agents were administered by intravenous injection via the lateral tail vein. The mice received 0.3 nmol imaging agent in 20% DMSO (100 μL), as determined by ultraviolet absorption of cypate (ε780nm = 2.22 × 105 M−1cm−1). For NIR reflectance imaging, mice bearing 4t1luc tumors were injected with LS340 (n=3), LS308, or a mixture of LS308 with 0.5 mg c(RGDyK) (18 mg/kg) for blocking. For multimodal imaging, mice received 111In-LS308 (164 μCi/μg, 40 μCi, 0.14 nmol, n = 2, or 138 μCi/μg, 200 μCi, 0.80 nmol, n = 2). The mice were imaged with a Kodak IS4000MM multimodality imaging system at 1, 4, 8, 24, and 72 hours after injection for both NIR reflectance imaging and gamma scintigraphy. Planar fluorescence, bioluminescence, x-ray, and scintigraphic imaging were performed without repositioning the animals. The camera field of view was 80 mm, with an f-stop of 2.8 and 2× binning for all modalities except bioluminescence. Bioluminescence imaging was conducted after intraperitoneal injection of 150 mg/kg k-luciferin in 50 μL saline (Gold Biotechnology, St. Louis, MO). Acquisition time was 5 minutes, with 8× binning. For NIR fluorescence imaging, broadband illumination from a 150 W halogen lamp was filtered by 755/35 nm optical bandpass filter (Eastman Kodak, New Haven, CT) and uniformly illuminated the subject, and the emitted light was captured via a 12-bit cooled charge-coupled device camera after 830 wide-angle longpass filtering (e830WA, Eastman Kodak). Gamma detection was accomplished by sliding the phosphor screen directly underneath the subject, which converts incident x-rays and radioactivity to white light. Regions of interest (ROIs) were drawn within the areas related to the liver, kidneys, tumor, and a control region on the opposite flank for comparison of relative in vivo fluorescence distribution at each time point. Mean intensities for ROIs were plotted with respect to time for comparison between groups.
SPECT and CT and Biodistribution
In addition to NIR-reflectance imaging and planar scintigraphy, mice were also imaged using a dual-detector nano-SPECT instrument (Bioscan, Washington, DC). The mice were placed in a bed fitted with tubing to deliver anesthesia and computed tomography (CT), followed by SPECT. CT scans were performed using a tube voltage of 45 kVp and a scan time of 1.5 seconds per projection. SPECT data were collected by monitoring 111In decay at 171 and 245 ±10% keV in a helical scan at 16 projections and 60 seconds per projection using a nine-pinhole low-energy collimator. The mice were imaged with the nano-SPECT instrument at 4 and 24 hours postinjection. 20 The CT and SPECT images were reconstructed using Interview XP and HiSPECT (Bioscan, Washington, DC), respectively, and coregistered and analyzed using PMOD (PMOD Technologies Ltd., Zurich, Switzerland). Volumes of Interest (VOIs) were constructed in PMOD. After the last imaging time point, the mice were sacrificed and specific tissues were removed and placed on a flat plate for ex vivo NIR fluorescence imaging. The tissues were placed in preweighed vials and weighed for subsequent measurement of radioactivity by a gamma counter. The uptake of the probes in tissues was expressed as the percent injected dose per gram tissue (%ID/g).
Results
Receptor Binding and Fluorescence Microscopy
All peptides were labeled with cypate for optical imaging (see Figure 1). LS167 is a cyclic peptide analogue of c(RGDyK) and does not have DOTA conjugate. Therefore, it served as a positive control to assess the effect of DOTA incorporation (LS308) on the biologic properties of cypate-labeled c(RGDyK). LS340 was designed to serve as a negative control for this study. It has the same amino acids as LS167 and LS308 in a scrambled order (see Figure 1) to minimize binding to αvβ3 integrin. 125I-c(RGDyK) was used as the tracer for the competitive receptor binding assay. The results show that c(RGDyK), LS308, and LS167 all inhibited the binding of 125I-c(RGDyK) to immobilized αvβ3 in a log dose-dependent manner (Figure 2). In contrast, the nonbinding control, LS340, did not displace 125I-c(RGDyK), even at its highest concentration (10 μM; data not shown). For LS308 and LS167, there was significant deviation of the Hill slope from unity, whereas the deviation for c(RGDyK) was less pronounced (see Table 1). To determine the αvβ3 receptor-mediated uptake of LS308 by αvβ3 receptor-positive 4t1luc cells, we investigated binding and accumulation of LS308 in these cells by NIR fluorescence microscopy. The results showed that LS308 internalized in 4t1luc cells, but the internalization was inhibited by pretreatment with an inhibitory peptide c(RGDyK) and a function-blocking antibody (Mab-anti-αvβ3). As expected, LS340 did not significantly accumulate in the αvβ3 receptor-positive cells (Figure 3), confirming the role of αvβ3 receptor-mediated endocytosis of LS308 in 4t1luc cells.

Solid-phase heterologous receptor binding assay to assess the affinity of dye-labeled conjugates.

Fluorescence microscopy of 4t1luc cells treated with LS308 (1 μM, A-C) or LS340 (1 μM, D). 4t1luc cells pretreated with c(RGDyK) (100 nM, B) or a function blocking Mab-anti-αvβ3 (60 nM, C).
Optical Imaging and Biodistribution
The tumor tissue was identified by bioluminescence imaging after administration of luciferin (Figure 4). The tumor was also visualized by fluorescence imaging within 6 hours after injection of LS308. The contrast between the tumor and the surrounding normal tissues was maintained for the duration of the study. Coregistration of the bioluminescence and fluorescence images demonstrated a heterogeneous uptake of LS308 in the tumor (see Figure 4). Tumor and non-target tissue accumulation was monitored over time by ROI analysis of the NIR fluorescence imaging data (Figure 5). Tumor to tissue ratios, monitored by fluorescence imaging, revealed different contrast ratios for a relatively low (0.14 nmol) and high (0.80 nmol) quantity of LS308 (Figure 6). A good agreement was observed between the tumor to tissue ratios from representative in vivo and ex vivo tissues at 24 and 72 hours (see Figure 5 and Figure 6). Kidney and liver accumulation was high, which is similar to the clearance pathways observed for an αvβ3 targeted Cy5.5-(RGDyk) conjugate.14 Receptor-specific uptake of LS308 was evaluated in relation to the LS340. The results show a significantly reduced uptake of LS340 in tumor relative to LS308 4 hours postinjection (Figure 7).

In vivo coregistered planar bioluminescence (hot; behind) and near-infrared fluorescence (rainbow; front) false color imaging (left) and noncollimated scintigraphy (right) of 4t1luc tumors captured 72 hours after injection of 111In-LS308 (200 μCi). Subcutaneous 4t1luc tumors are indicated by red circles obtained from luminescence imaging. Liver (L) and kidneys (K) are indicated by arrows.

Comparison of tumor accumulations in dissected tissues: (A) mice were sacrificed 24 hours postinjection (40 μCi 111In-LS308, 0.14 nmol LS308); (B) mice were sacrificed 72 hours postinjection (200 μCi 111In-LS308, 0.80 nmol LS308).
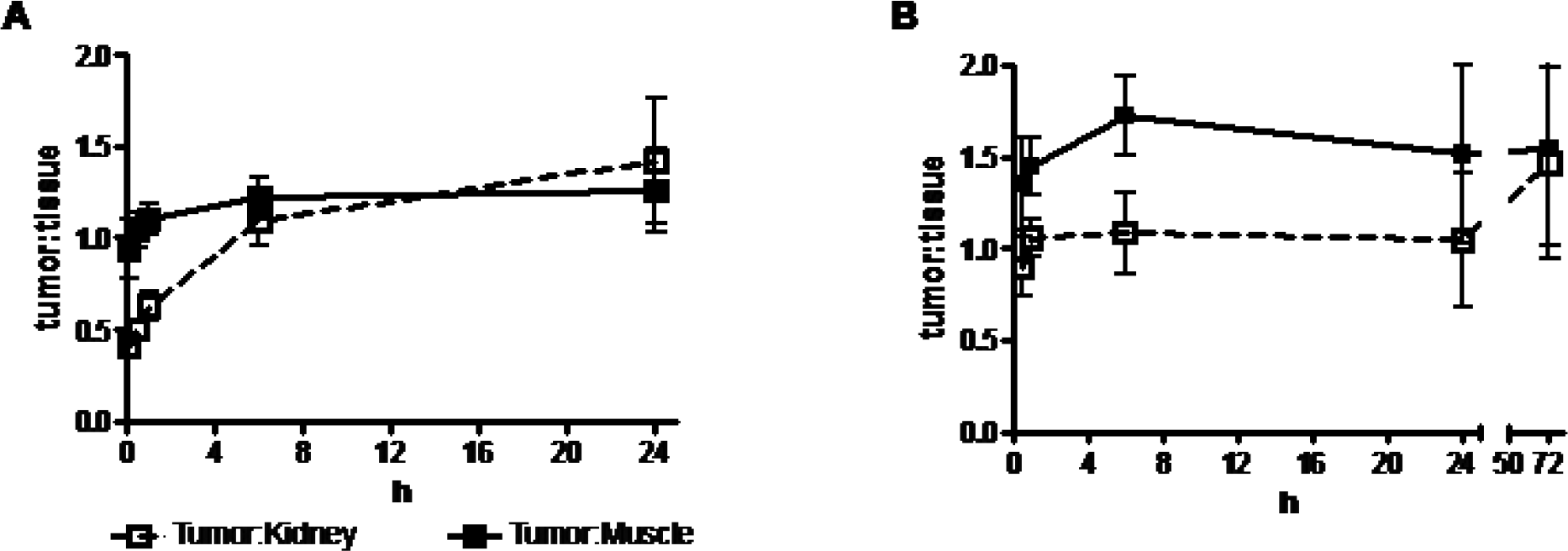
Comparison of tumor to tissue fluorescence intensity ratios: (A) 40 μCi 111In-LS308 (0.14 nmol LS308) and (B) 200 μCi 111In-LS308 (0.80 nmol LS308).

Comparison of tumor accumulations of LS308 (0.3 nmol, n = 3) and a nonbinding control LS340 (0.3 nmol). n = 3;*p < .05 relative to LS308.
Radiochemistry, SPECT and CT, and Radiochemical Biodistribution
111In-LS308 was prepared in two batches of comparable specific activities (164 μCi/μg, 300 μCi/nmol or 138 μCi/μg, 253 μCi/nmol). In both cases, radiochemical purity was > 95%. To investigate whether LS308 would localize to tumors expressing the αvβ3 receptor, it was radiolabeled with 111In and administered to 4t1luc-bearing mice. In vivo tumor imaging was performed by gamma scintigraphy (see Figure 4) and SPECT (Figure 8), with both methods demonstrating uptake in the target tissue. NIR fluorescence intensity, and gamma counting of dissected tissues revealed a similar biodistribution trend in terms of target and non-target tissue uptake at both the 24- and 72-hour time points for 111In-LS308 (Table 2). For both time points, the kidneys and liver received the majority of radioactivity and produced high levels of NIR fluorescence. At 24 and 72 hours, tumor uptake was 7.59 ± 0.45% ID/g and 2.48 ± 0.88 %ID/g, respectively (see Figure 5).

SPECT images of LS308 (4 hours postinjection, 0.14 nmol, 0.26 μg, 40 μCi 111In); 4t1luc subcutaneous homografts (orange arrowheads) implanted bilaterally in the shoulder are clearly visualized.
Comparison of Tumor to Tissue Ratios of Dissected Tissues (Mean ± SD)

NIR = near-infrared.
Discussion
The goal of this research was to evaluate the affinity, receptor specificity, and tumor localization properties of a multimodal imaging agent targeted to the αvβ3 integrin. The structure of the αvβ3 targeted peptide (LS308) contains the cyclic lactam c(RGDyK) peptide sequence, known to possess high affinity and selectivity for the αvβ3 integrin. The conjugation of a GK sequence to the e-amino group of the lysine residue of c(RGDyK) provides a means of attaching the DOTA and cypate to the same peptide. DOTA was chosen for its ability to form complexes with a variety of radiometals, such as 111In, 64Cu, and 177Lu, for radioimaging and radiotherapy. Cypate, when conjugated to an αvβ3-targeted peptide such as c(RGDfV), is capable of receptor-specific imaging of αvβ3-positive xenografted tumor in mice.17,21 For tumor imaging, we chose the 4t1luc mouse mammary tumor that matures rapidly in mice. These cells have been stably transfected with luciferase for generation of bioluminescence after intravenous administration of luciferin. This provides the added benefit of identifying the tumor tissue and colocalizing bioluminescence with fluorescence imaging and radioactivity from tumor-homing peptides.
Affinity of the targeting ligand is of the utmost importance in determining uptake in the target site. To avoid a loss of affinity, the DOTA and cypate should not interfere with the RGD binding motif. This was achieved by sequentially conjugating DOTA and cypate to the α- and ε-amino groups of Lys, as shown in Figure 1. The results from the solid-phase heterologous binding assay show that the affinity of LS308 was maintained relative to c(RGDyK) (see Table 1). The deviation of Hill slope values for LS308 and LS167 from unity suggests overestimation of the Ki values of these peptides. Shallow values in Hill slope can indicate, among other possibilities, cooperativity or a heterogeneous population of receptors. This phenomenon has been previously observed by others in similar assays. 22 In spite of this discrepancy, the potent IC50 value of LS308 indicates a high affinity for the αvβ3 integrin.
In addition to high affinity, a tracer for molecular imaging should have high specificity for the targeted receptor. Because the receptor binding assays were carried out with purified proteins, fluorescence microscopy experiments were performed with whole cells that could equally express other integrins besides the αvβ3 heterodimer. Pretreating 4t1luc cells with c(RGDyK) inhibited LS308 binding and subsequent internalization, which is indicative of a receptor-mediated endocytosis. Furthermore, pretreatment with a function-blocking Mab-anti-αvβ3 also inhibited the binding of LS308, confirming the specificity of this peptide for the αvβ3 integrin.
An important consideration in performing an imaging experiment with a radiotracer bearing two reporter moieties for different imaging modalities is the specific radioactivity. In this study, the two reporter moieties are a radioisotope, 111In, for nuclear imaging and cypate for NIR optical imaging. If the specific activity is too low, then the unlabeled peptide-dye conjugate can inhibit the accumulation of the radiolabeled peptide in vivo. If the specific activity is too high, even though the radionuclear method could clearly visualize the tumor, visualization by optical imaging could be hindered owing to a low concentration of the peptide-dye conjugate. Of course, this can be overcome by administering large doses of the radiolabeled peptide. In this study, the specific activities were in excess of 10-fold than previously reported for 111In-labeled peptides.15,23 The ramification for this finding is that the high specific activity 111In-labeled peptides bearing an NIR fluorescent dye can be used for in vivo molecular imaging.
To demonstrate that LS308 accumulation in the tumor was receptor specific, equal quantities of LS308 and LS340 were administered to 4t1luc tumor-bearing mice. At 4, 8, and 24 hours, there was a clear reduction in 4t1luc tumor accumulation of the nonbinding control relative to LS308 (see Figure 7), thus demonstrating that the observed in vivo tumor uptake of LS308 is αvβ3 mediated (see Figure 4 and Figure 8). In contrast to the cell microscopy study, however, we found that coadministration of a competitive dose of c(RGDyK) with LS308 was ineffective in blocking the uptake of LS308 in the tumor in vivo. The chosen quantity of c(RGDyK) was based on previous work that showed that 18 mg/kg of c(RGDfK) was sufficient to competitively inhibit the accumulation of 18F-labeled RGD peptide analogue in an αvβ3-positive M21 xenograft. Interestingly, we did not observe any inhibition of LS308 accumulation at this dosing level, in spite of the fact that 18 mg/kg of c(RGDyK) is in 1,000-fold excess of the administered dose of LS308. Based on the efficient inhibition of LS308 accumulation in 4t1luc cells in vitro by both c(RGDyK) and a function-blocking anti-?αvβ3- monoclonal antibody, we hypothesized that the lack of competitive inhibition is due to either differing clearance properties between LS308 and c(RGDyK), extremely rapid αvβ3 recycling after c(RGDyK)-mediated internalization, or both. 24
Nuclear imaging of 111In-LS308 was first performed by planar gamma scintigraphy. The imaging capabilities of the scintigraphic detector relative to the NIR filters show that the scintigraphic image is more diffuse than the NIR image (see Figure 4). In spite of the diffusivity generated by this detection scheme, 111In-LS308 tumor accumulation was observed in the scintigraphic image, aided by coregistration with bioluminescence. There is a good correlation between the observed kidney and liver accumulation by gamma scintigraphy and the ex vivo NIR fluorescence intensity measurement. The scintigraphy obtained is comparable to a previously published image of 111In-DTPA-IRDye800-labeled RGD peptide analogue generated with a dedicated gamma camera equipped with collimators. 15 Both cameras generated diffuse images of the tumor with barely discernible tumor margins. However, neither specific activity nor injected dose was optimized for the gamma scintigraphy to improve tumor visualization.
In addition to gamma scintigraphy, SPECT was performed on mice bearing the 4t1luc tumors to determine whether tumor accumulation is sufficient for visualization with this imaging modality. The high liver accumulation observed in the SPECT images is consistent with the ex vivo fluorescence and radioactivity biodistribution data. The SPECT images suggest a nonuniform distribution within the liver. The 4t1luc tumors are clearly visible but display the same nonuniform distribution of radioactivity as that observed in the liver.
Interestingly, the pattern of exogenous LS308 distribution within the tumor as reported by SPECT and NIR fluorescence imaging was different from the endogenous bioluminescence tumor imaging. Both fluorescence and SPECT images showed the highest uptake in specific regions in the tumor relative to the entire tumor tissue identified by bioluminescence imaging (see Figure 4 and Figure 8). This finding clearly illustrates the complementary nature of the contrast mechanisms. Whereas bioluminescence reports the expression of transfected luciferase enzyme in 4t1luc cells and identifies virtually all of the tumor cells, the fluorescence and SPECT images indicate that LS308 accumulated in areas of high αvβ3 protein density in the tumor. The result suggests that not all of the 4t1luc cells express both luciferase and αvβ3 proteins simultaneously.
To verify this hypothesis, we compared the αvβ3 integrin expression in both 4T1 and 4t1luc tumor cells by immunocytochemical assay (Figure 9). The result shows a heterogeneous expression of the target proteins in these cells and could reflect the observed punctuate accumulation of LS308 in the tumor tissue in vivo. Alternatively, LS308 could be targeting tumor-induced angiogenic blood vessels, which have a high level of αvβ3 integrin. Correlating the αvβ3 expression pattern by histology and noninvasive imaging will be the subject of a future study.
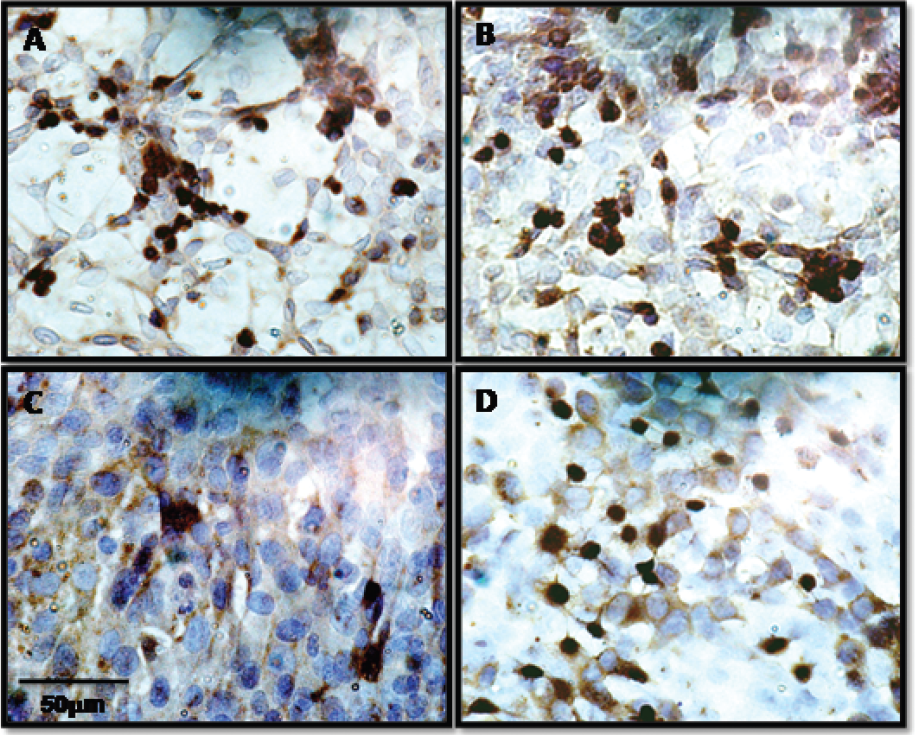
Immunocytochemical staining of integrin αv (A and C) and integrin β3 (B and D) expression in 4T1 (A and B) and 4t1luc cells (C and D). Anti-integrin αv and anti-integrin β3 antibodies were used to identify the respective proteins (scale bar A-D = 50 μm).
The excellent agreement between the optical and nuclear methods indicates that both labels are not metabolized during or after tumor localization. For example, both methods show that clearance is predominantly through the liver. This observation is consistent with previous clearance properties of a radiolabeled cypate-conjugated peptide. 18 The tumor accumulation of 111In-LS308 in the tumor was higher than that generally observed with the M21- and the MDA-MB-435-αvβ3 positive cell lines using other radiolabeled cyclic RGD peptides that do not bear a dye for optical imaging.25,26 Moreover, the tumor retention of LS308 persists strongly up to 72 hours.
Conclusions
We have demonstrated that LS308 has high affinity for the αvβ3 integrin and internalizes preferentially via the αvβ3 integrin-mediated endocytosis in 4t1luc cells. Moreover, LS308 can indeed localize to αvβ3-positive tumors in a receptor-specific manner. The tumors were visualized by both optical and nuclear imaging methods. Whereas the endogenous bioluminescence imaging was used to identify the tumor mass, the fluorescence and SPECT methods with the exogenous LS308 contrast agent reported the local expression of αvβ3 expression. Thus, the multimodal imaging approach could provide important complementary diagnostic information for improving patient management.
Footnotes
Acknowledgment
Financial disclosure of authors: This study was supported in part by the National Institutes of Health (R01 CA109754, R33 CA100972, U54CA136398, and U54 CA119342).
Financial disclosure of reviewers: None reported.
