Abstract
We have developed novel proteinase-modulated contrast agents (PCAs) to detect the activity of proteinases in vivo using magnetic resonance imaging. The PCAs are based on the concept of a solubility switch, from hydrophilic to hydrophobic, that significantly modifies the pharmacokinetic properties of the agent as revealed by the slow efflux kinetics from the activity site. Our compound PCA7-switch detects the activity of the secreted matrix-degrading proteinase matrix-metalloproteinase 7 (MMP-7) in living, tumor-bearing mice. Control experiments were performed using an agent that was not cleaved by MMP-7 (PCA7-scrambled), an agent that could be cleaved by MMP-7 but lacked the solubility switch (PCA7-B), and a standard contrast agent (gadolinium–diethylenetriaminepentaacetic acid). PCA7-switch detected a reduction in MMP-7 activity in tumor-bearing mice treated with a synthetic MMP inhibitor, demonstrating its effectiveness in noninvasive functional imaging of proteolytic activity in vivo.
The association of MMP activity with tumor invasion and metastasis resulted in the development of smallmolecular-weight matrix metalloproteinase inhibitors (MMPIs) that were tested as cancer therapeutics. Despite multiple clinical trials with several different MMPIs, these agents failed to demonstrate a clinical benefit to cancer patients. 2 Although a number of reasons for this failure have been proposed, 2 one overriding issue is the inability to determine if the dose of drug delivered was effective in inhibiting tumor-associated MMP activity. It became clear from these studies that noninvasive assays for modulation of MMP activity in vivo would greatly improve the further development of MMPIs. In addition, in vivo assessment of MMP activity has potential utility in the detection, diagnosis, and prognosis of and assessing response to therapy in many diseases, including cancer. 3
Recently, an MMP-2 inhibitor was radiolabeled with 64Cu to detect the presence of MMP-2 using positron emission tomography. 4 These authors concluded that their probe was inadequate for in vivo tumor evaluation owing to low affinity for MMP-2 and degradation of the compound in vivo.
Magnetic resonance imaging (MRI) has become standard technology, and the required instrumentation is widely available in hospitals and clinical centers throughout the world. The majority of existing contrast agents for MRI are gadolinium (Gd) based and are distributed according to the transcapillary transfer rate and the extracellular volume. Such contrast agents are commonly used in both clinical MRI to detect tumors 5 and in research to assess the physical characteristics of tumor tissue.6–11 Gd is the paramagnetic ion of choice because of its high relaxivity, that is, it efficiently decreases the water spin-lattice relaxation time constant, T1. 12 Low-molecular-weight Gd-chelated agents currently in use, such as Gd–diethylenetriaminepentaacetic acid (DTPA) and Gd-tetraazacyclododecane tetraacetic acid (Gd-DOTA), can readily travel within the microvasculature of tumors, 8 and they exhibit rapid distribution throughout the vasculature followed by a relatively fast decline owing to urinary excretion. 7
These conventional contrast agents cannot provide information about particular gene expression and/or biochemical activity. A number of contrast agents have recently been developed to incorporate bioactive ligands to target specific overexpressed cell receptors.13,14 A family of bioactivatable contrast agent was pioneered by Meade and colleagues based on a “cap-on to cap-off” concept whereby the relaxivity of the contrast agent is modulated by the access of water to the paramagnetic Gd atom, directly affecting the signal intensity of T1-weighted images.15,16 In the prototype reagent, a chemical moiety initially blocking access of water to Gd is released following cleavage by intracellular β-galactosidase, 15 which results in an increased relaxivity. In more recent compounds, a decrease in pH resulted in a decreased relaxivity. 16 We hypothesized that activation of MRI contrast agents by extracellular proteinases is a promising strategy to detect and image biochemical processes and the activity of specific genes associated with tumors and/or metastatic disease. Unlike receptor-based approaches, MRI contrast agents responsive to extracellular proteinases have the potential for enhanced response owing to catalytic activity and substrate turnover at the enzyme target, a form of signal amplification not inherent in receptor-based strategies. Furthermore, proteinases secreted into the extracellular matrix have the potential to be present at markedly higher concentrations than cellular receptors, and drug delivery challenges such as intracellular uptake of the agents are obviated.
Here we present studies with a novel MRI contrast agent designed to display reduced solubility after cleavage by the MMP family member MMP-7 (3.4.24.23, also known as matrilysin or putative metalloproteinase [PUMP]-1). The MMP-7 protein is distinct from most of the other MMP family members for several reasons reviewed in Wilson and Matrisian. 17 It is one of only two family members that contain only the catalytic domain in its active form. Thus, the likelihood that MMP-7 activity is modified by activities inherent in the additional domains found in other MMP family members is decreased, as is the likelihood that the biologic role of MMP-7 is due to activities other than its proteolytic capability. MMP-7 has an unusually shallow catalytic pocket, resulting in distinct characteristics of the substrates recognized by this enzyme. 18 Importantly, MMP-7 expression is predominantly localized to cells of epithelial origin, in contrast to the expression of most MMP family members in cells of connective tissue origin. Thus, in adenocarcinomas (the most common adult tumor types that arise from cells of epithelial origin), MMP-7 is expressed in the malignant epithelial component of the tumor rather than in the surrounding stromal tissue. From a functional imaging perspective, this provides a focused source of an abundantly expressed enzyme to facilitate in vivo detection.
Methods
Mouse Model
The human colon cancer cell line SW480, which expresses undetectable levels of MMP-7 protein in the conditioned medium, was engineered to express moderate levels of human pro-MMP-7 messenger ribonucleic acid (mRNA) under the control of a strong viral promoter. 19 The MMP-7-expressing variants (SW480Mat) have increased tumorigenicity when injected into the cecum of immunodeficient mice, suggesting that the pro-MMP-7 protein that is expressed becomes catalytically active and functional. 19 Cells were maintained in Dulbecco's Modified Eagle's Medium containing 10% fetal calf serum under standard cell culture conditions.
Athymic nude mice were injected subcutaneously on the left hindlimb with 1.5 × 106 parental control SW480 colon cancer cells and 1.0 × 106 SW480Mat cells on the right hindlimb. These cells grow to approximately 1.5 cm3 tumors within 4 weeks; 50% more parental SW480 cells were used to compensate, in part, for the slower growth of these tumors. For systemic treatment with a broad-spectrum MMPI, mice were injected intraperitoneally with 100 mg/kg/d of GM6001 (Calbiochem, EMC Chemicals, San Diego, CA) for 3 days, with final treatment 2 hours prior to imaging. Animal experiments were approved by the Institutional Animal Care and Use Committee of Vanderbilt University.
Synthesis of Contrast Agents
The PCA7-switch free ligand was synthesized by Biopeptide (San Diego, CA) using standard methods of solid-phase peptide synthesis followed by sequential derivatization of the terminal Gly residue with DOTA tri(t-butyl ester) and the adjacent Lys residue with noctanoic acid prior to hydrolysis from the resin. After purification by preparative high-performance liquid chromatography (HPLC), the ligand was metallated with 1.05 molar equivalents GdCl3 (Spectrum, Gardena, CA) in water at pH 6.5 to 7.0 for 24 hours. The resulting compound was purified by preparative HPLC over a YMC Pack Pro C18 column (100 × 20 mm) using a water-acetonitrile mixed solvent system at 12 mL/min. Fractions containing the target compound were combined and lyophilized to afford PCA7-switch in 61% yield. The identity and purity of each compound were confirmed by high performance liquid chromatography with electrospray ionization mass spectrometry (LC-[ESI]-MS) using a PolymerX (Phenomenex, Torrance, CA) (150 × 4.6 mm) column with a water-acetonitrile (35%) system operating at 1 mL/min.
Compounds were formulated in phosphate-buffered saline (PBS) at 10 mM concentration and filter sterilized with a 0.2 micron membrane filter (Millipore, Billerica MA, USA) prior to injection in animals. In some cases, 1% ethanol was added to enhance solubility.
In Vitro Studies of Contrast Agent Activation by MMP-7
In kinetic experiments, mixtures containing MMP-7 and a contrast agent were incubated in a thermostatically controlled autosampler at 37°C and aliquots were analyzed at various time intervals with the use of high performance liquid chromatography (HPLC) and HPLC with mass spectrometry (HPLC-MS). The time course of hydrolysis was determined by the decrease in the chromatographic peak corresponding to the initial contrast agent. The identities of the observed peaks (contrast agent and cleavage products) were inferred based on mass spectrometry (MS).
For the HPLC studies, a gradient method was used (30% to 5 minutes, 55% to 7 minutes, 100% at 12 minutes, 35% at 22 minutes, with 5 minutes again on PolymerX 150 × 4.6 mm operating at 1 mL/min at room [ambient] temperature [RT]). Rate constants were calculated by fitting experimental data to the integrated first-order rate equation using Sigma Plot 7.1 (Jandel Scientific, San Rafael, CA). The first order of the reactions was confirmed by the independence of rate constants on the substrate concentration. Substrate concentrations as low as 1.7 μM provide sufficient signal intensity with an injection volume 200 μL and a column size of 150 × 4.6 mm.
Reverse-phase HPLC was used to determine the retention time of the cleavage products using conditions (25% rather than 35% acetonitrile) that allowed separation of the intact compound and the cleaved products in the same experiment within a reasonable time.
Initial T1 measurements (inversion recovery) and T2 measurements (Carr-Purcell-Meiboom-Gill spin echo) were performed using a benchtop nuclear magnetic resonance (NMR) spectrometer (Bruker Optics Inc., Minispec, 60 MHz, The Woodlands, TX).
Magnetic Resonance Imaging
MRI studies were conducted at the Vanderbilt University Institute of Imaging Science on a small animal 7 T animal scanner (Varian Inc., Palo Alto, CA) with a 160 mm bore and a 38 mm volume radiofrequency coil. Studies included the acquisition of a precontrast T1 map using a multiple flip angle approach (α = 12, 24, 36, 48, and 60° or α = 10, 20, 25, 35, and 50°). A gradient-echo sequence was used with the following parameters, unless otherwise specified: repetition time 5 200 or 100 milliseconds, echo time = 3.2 milliseconds, matrix = 128 × 128, field of view = 35 × 35 mm2, number of averages (NA) = 4, and 10 slices 1.5 mm thick. The dynamic contrast-enhanced (DCE)-MRI study used the same sequence repeated a number of times with α = 30° to register T1-weighted images dynamically before, during, and after injection of contrast agent.
Injection of contrast agent was realized through a jugular vein catheter surgically implanted at least 7 days before imaging. The quantity of compound, detailed for each animal in the figure legends, was generally 3.7, 2.3, and 10 μmol Gd for PCA7-switch, PCA7-scrambled, and Gd-DTPA, respectively. During imaging, animals were anesthetized with 3% (initiation) and 1.5% (stabilization) isoflurane in air. A feedback-controlled animal warm-air heater system was used to keep the animal body temperature at physiologic levels, and the respiration rate was continuously monitored (SA Instruments Inc., Stony Brook, NY). The period of time for which the kinetics was recorded was minimized for each compound to minimize the period of anesthesia of the animals since these were imaged typically two or three times in a span of 7 days.
Data analysis was performed using a modified in-house Matlab image viewer (The Mathworks, Natick, MA). 20 Regions of interest (ROI) were manually drawn over the tumors to segment the “rim” and the “core.” This analysis allows separating the intrinsic different pharmacokinetic properties of these two regions of a tumor. This analysis accounts for the known vascular difference between these regions but is still empiric. It is likely that a gradient of vascularity exists within tumors, going from highly vascularized regions at the rim to poorly vascularized regions in the core. Although imperfect, this analysis has the potential to demonstrate this difference between the core and the rim of tumors. For every imaging session, data from the ROI of a given tumor were averaged.
The plots of the scaled signal enhancement patterns of the rim and the core for a given animal for the injection of a given compound are presented. The signal intensity for different sessions can be different since hardware factors such as the tuning of the volume coil, the calibration of the flip angle, and the receiver gain can all affect the signal intensity for a given imaging session. These factors are expected to be constant during the course of an imaging session. Given that only the trends of the enhancement patterns are compared, these considerations will not affect the data analysis, which was performed on individual experiments in which magnetic resonance images over time were collected simultaneously from both the MMP-7-positive and MMP-7-negative tumors. In addition, a ROI was drawn in a muscle, and the data are shown for comparison. The magnitude of the signal intensity was scaled with a constant factor for each imaging session.
Statistical Analysis
A linear fit was applied to the data points from 40 minutes onward for each experiment with a proteinase-modulated contrast agent (PCA) compound (Origin 6.0, Microcal, Northampton, MA), yielding the slope and the standard deviation on the slope. The data for the negative and the positive tumor were compared for each experiment using a standard two-tailed t-test implemented in Matlab to assess the equality of their slopes. An underlying assumption is that the data curves are well described by a linear regression model. The many experimental points for each tumor lead to relatively small standard deviations. For this reason, we considered that the slopes were different when the p value reached the highly significant threshold at 99% (p < .01).
Results
To noninvasively detect activity in vivo using MRI, we generated a series of PCAs. In this design, Gd-DOTA is made hydrophobic by attaching insoluble moieties such as alkyl chains or aromatic rings. The overall solubility of the compound is restored by adding hydrophilic poly(ethyleneglycol) (PEG) chains via a proteinase-sensitive peptide linker. As a result of the cleavage of the peptide sequence, the PEG chains are separated from Gd-DOTA, making this product more hydrophobic.
To generate an MMP-7-selective PCA7-switch, the peptide sequence was a specific substrate that is efficiently cleaved by MMP-7 (PLALKRDR). 21 A different peptide sequence was used previously on an MMP-7-activated fluorescent probe (RPLALWRS) 22 ; it was confirmed that both peptides are cleaved between A and L. Figure 1A shows the chemical formulation of PCA7-switch with a molecular weight of 2,418.92 Da. Alternative compounds with longer alkyl side chains were also explored, but their poor water solubility made them difficult to handle during the synthesis. Two control compounds were synthesized and tested. To generate a control PCA with the same characteristics as PCA7-switch but that is resistant to MMP-7 cleavage, PCA7-scrambled was generated with the same initial solubility features but a scrambled sequence of the MMP-7 selective peptide (RAKDRLLP; Figure 1B). In addition, we generated a control PCA referred to as PCA7-B (Mw = 2,314 Da) that is related to PCA7-switch but that is more hydrophobic such that proteolytic cleavage to release the soluble end did not appreciably change the solubility of the compound (Figure 1C).
The effectiveness of the solubility switch was verified in vitro. The HPLC retention time of PCA7-switch was 1.9 minutes, whereas that of the cleaved product after treatment with MMP-7 increased to 13.2 minutes. Note that these times are different from the elution times during purification in which different elution solvents were used. The changes in relaxivity as determined with a benchtop NMR spectrometer were 0 for ΔT1 and 10% for ΔT2 for PCA7-switch after cleavage by MMP-7 and were found not to be statistically different. The HPLC retention time of the intact and the cleaved control PCA7-B compound was similar at 6.9 minutes and 8.0 minutes, whereas the r1 relaxivity of the cleaved product increased by 37% (35% for ΔT1 and 75% for ΔT2), possibly through aggregation after cleavage, leading to longer rotational correlation times. It is not known whether such aggregation and relaxivity increase for PCA7-B might also occur in vivo. Thus, PCA7-switch displayed the anticipated change in solubility following MMP-7 cleavage, and PCA7-B represents a control compound in which MMP-7 cleaved the agent with minimal change in solubility.
Figure 2 displays representative axial images of a mouse bearing two subcutaneous xenografts, one of SW480 cells, an MMP-7-negative human colorectal cancer cell line, and the second of the same cell line engineered to express MMP-7 (MMP-7 positive) and imaged shortly after intravenous administration of PCA7-switch. The MRI intensity in the bladder of animals injected with the PCA7 compounds increased rapidly, indicating an efficient filtration by the kidneys (data not shown). The signal intensity in both tumors was enhanced after injection of PCA7-switch. It is noted that the signal intensity in the rim of the tumors was, in general, enhanced more rapidly than the core (see center panel), which is usual for subcutaneous tumor xenografts.23–26 Histologic examination revealed tumor heterogeneity (see Figure 2B) that was similar in both control and MMP-7-positive tumors. Immunohistochemical analysis with an MMP-7-specific antibody showed enhanced tumor-cell reactivity in MMP-7-positive versus MMP-7-negative xenografts, although both tumors showed some background reactivity in necrotic areas (data not shown).

Chemical formulation of compounds used in this study. A, PCA7-switch; B, PCA7-scrambled; and C, PCA7-B.
The in vivo response to the MMP-7-cleavable contrast agent is better visualized by measuring the signal enhancement patterns over time since the strategy is to modulate its pharmacokinetic properties following cleavage in the tumor microenvironment. A mouse bearing control and MMP-7-expressing tumors was injected on different days with first PCA7-switch followed 2 days later by the uncleavable variant PCA7-scrambled (Figure 3). Different enhancement patterns were obtained in the core of MMP-7-positive and -negative tumors for the injection of PCA7-switch, but not after injection of PCA7-scrambled. For PCA7-switch (see Figure 3A), the MRI signal increased in the MMP-7-expressing tumor throughout the 160-minute time course, whereas the signal in the control tumor started to show a gradual decline after ≈60 minutes. The slope of linear fits applied from 40 minutes onward was 20.6 ± 0.9 for the positive tumor but only −0.9 ± 0.3 for the negative tumor. The difference in slopes is highly significant (p < .01). The values of the slopes, standard deviations, and significance level are tabulated for five different mice tested with PCA7-switch (Table 1). The steady signal increase in the core of the MMP-7-expressing tumor, whereas the rim of the same tumor showed a marked clearance, indicates that a mechanism that favors the accumulation of PCA7-switch product in the core is active. All five mice tested with PCA7-switch showed a significant (p < .05) difference in response between the MMP-7-positive and MMP-7-negative tumors (Figure 4), with four being different at high significance (p < .01); the somewhat lower significance in the data from the fifth animal may be due to the shorter period of data acquisition for this mouse (see Figure 4A). Data from a sixth mouse (not shown) were qualitatively similar, but motion-related artifacts precluded temporal analysis of those data.

A, Representative axial images from a mouse. The matrix metalloproteinase-7 (MMP-7)-positive and -negative subcutaneous tumors are indicated. B, Histology of a representative SW480mat tumor stained with hematoxlin and eosin showing typical heterogeneity with regions of vascularized tumor (dark) and necrosis (pink). Scale bar is 0.5 mm.
For PCA7-scrambled (see Figure 3B), the slopes were 16.0 ± 1.3 min−1 for the positive tumor and 9.5 ± 0.9 min−1 for the negative tumor. Although these slopes for the PCA7-scrambled control are also significantly different (p < .01) (see Table 1), it can be appreciated from the figure that the difference in slopes for PCA-scrambled (see Figure 3B) is much less pronounced than for PCA7-switch (see Figure 3A) in the same animal. For two additional animals tested with PCA7-scrambled, motion-associated noise in the data precluded a quantitative temporal analysis, although these animals also showed only small differences, if any, in the response between the MMP-7-positive and MMP-7-negative tumors with this control compound.
The same mouse was injected with the standard contrast agent Gd-DTPA (Magnevist, Berlex, Wayne, NJ). Rapid uptake and clearance of all contrast agents were apparent in the muscle (see Figure 3, A-C). The Gd-DTPA images demonstrated that the outer rim of both tumors displayed a very rapid increase in signal within the first 20 minutes (see Figure 3F) followed by a clearing, indicating that the tumor rims are well vascularized by leaky neovessels. The signal in the core was retained longer, suggesting that it is not as well vascularized and signal enhancement occurs by direct transport of contrast agent in the microvasculature and/or by diffusion of contrast agent that first extravasated in the rim. In each of the mice examined, the temporal response to Gd-DTPA was similar in both tumors and with generally a somewhat slower efflux from the core compared with the rim (data not shown). Given that the molecular weight of GdDTPA (938 Da) is much smaller than that of PCA7-switch (2.4 kDa), it is expected to leak out of the tumor neovasculature and to diffuse more rapidly into and out of the core of tumors.
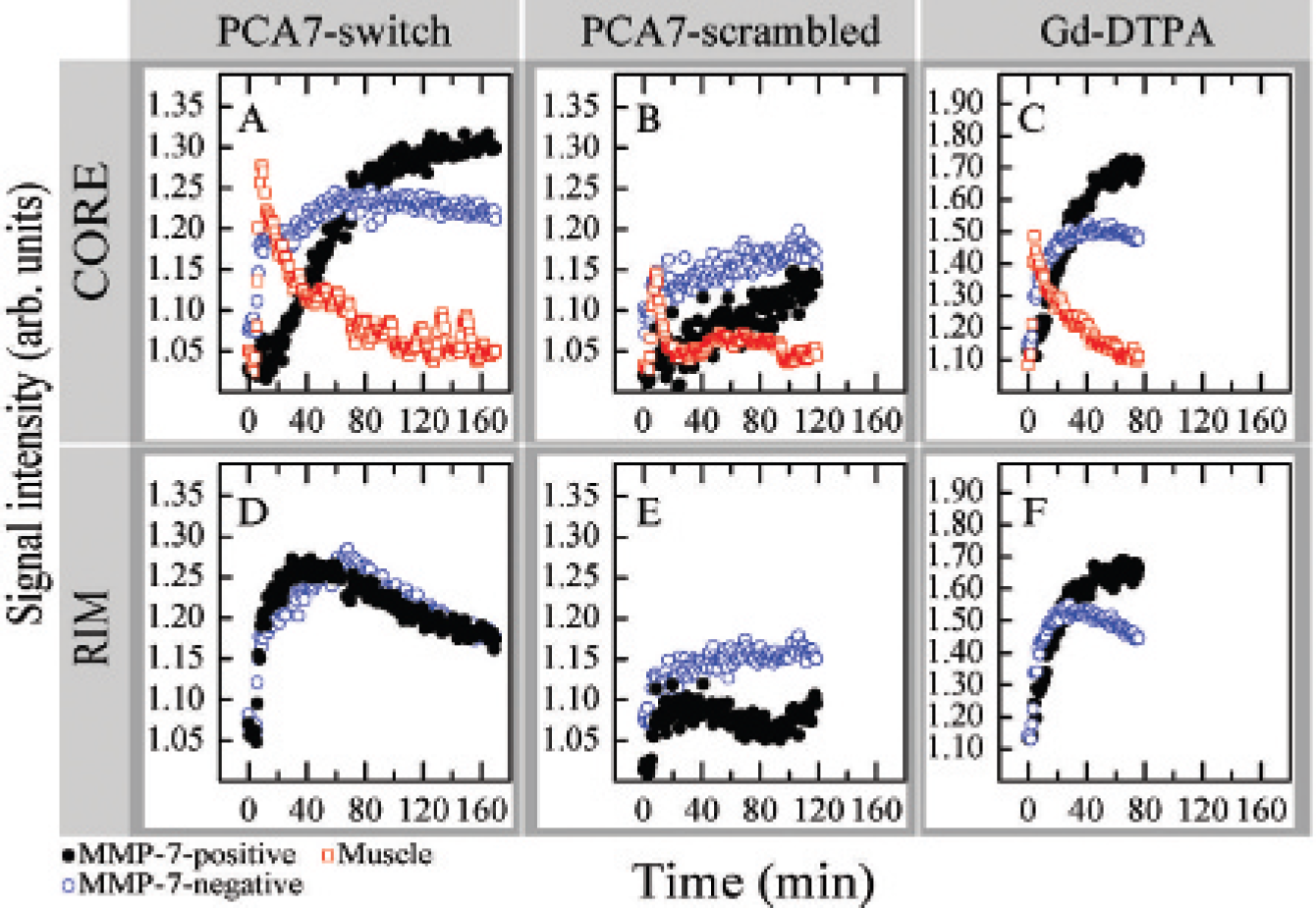
Comparison of PCA7-switch, PCA7-scrambled, and Gd-DTPA in matrix metalloproteinase-7 (MMP-7)-positive and -negative tumors. The enhancement patterns in the core (top row) and rim (bottom row) of MMP-7-positive (black circles, 111 mm3) and -negative (blue circles, 224 mm3) SW480 tumors and dorsal muscle (red squares). The same mouse was injected with PCA7-switch (3.7 μmol Gd; A and D) and PCA7-scrambled (2.3 μmol Gd; B and E) 2 days apart, 4 days after Gd-DTPA (10.0 μmol Gd; C and F).
Slopes of the Linear Curves Fitted to the Enhancement Patterns after 40 Minutes Postinjection of a PCA7 Compound

NS = not significant (p > .05).
Animals are assigned numbers in the table such that the reader can track the results obtained from single individuals sequentially injected with different compounds.
Linear fit from 25 minutes postinjection to include more data points to improve the statistics, but a difference was still not found. The acquisition was stopped after 50 minutes.

Comparison of PCA7-switch, before and after treatment with matrix metalloproteinase inhibitor (MMPI), and PCA7-B in MMP-7-positive and -negative tumors. Enhancement patterns for the core (top row) and the rim (bottom row) of MMP-7-positive (closed circles, 47 mm3) and -negative (open circles, 27 mm3) tumors from a dorsal muscle (open squares) corresponding to the injection of PCA7-switch (1.0 μmol Gd; A and D) following treatment with the MMPI GM6001 (1.0 μmol Gd; B and E) and of PCA7-B (2.0 μmole Gd; C and F).
The ability of PCA7-switch to reflect inhibition of tumor-associated MMP activity by a systemically administered MMP inhibitor was tested. A mouse bearing control and MMP-7-expressing tumors was injected with PCA7-switch and imaged. After a delay of 24 hours, the mouse was treated with GM6001, injected intraperitoneally at 100 mg/kg/d. Two days later, starting at about 2 hours following the third administration of GM6001, the same mouse was imaged again using the same PCA7-switch contrast agent. Before treatment with the MMPI, the PCA7-switch contrast enhancement again distinguished between the cores of the MMP-7-expressing and control tumors in that the signal in the MMP-7-expressing tumor increased over the time course of the experiment but saturated at approximately 20 minutes in the MMP-7-negative tumor (see Figure 4A). Before treatment, the slope for the positive tumor (1.9 ± 0.7 min−1) was significantly different (p < .05) from the slope in the negative tumor (−0.4 ± 0.4 min−1) despite the short data acquisition period for this particular animal. Interestingly, in this mouse, the rim of the tumor showed a similar discrimination between MMP-7-expressing and control tumors (see Figure 4D). In contrast, treatment of the same mouse with the synthetic MMPI eliminated the difference between the response in the two tumors, and both control and MMP-7-positive tumors showed similar kinetics of signal enhancement (see Figure 4, B and E), both showing washout of contrast (negative slope) after 60 minutes. After MMPI treatment, the washout was somewhat faster from the MMP-7-positive (−7.4 ± 0.8 min−1) versus the -negative tumor (−4.7 ± 0.8 min-1). This inhibition of contrast accumulation by treatment with MMPI was reproduced in a second animal (see Table 1). This animal (mouse 5) before treatment showed accumulation of PCA7-switch contrast in the MMP-7-positive tumor (slope 4.8 ± 0.8 min−1) and a highly significant difference in slope (6.8 min−1, p < .01) between positive and negative tumors, but after treatment with MMPI, there was washout of contrast from both tumors (negative slope), with no significant difference between the tumors (see Table 1). We note that for mouse 6 after MMPI treatment, the slope for the negative tumor is larger than for the positive tumor, that is, somewhat faster washout from the positive versus negative tumors, a trend not observed in any of the seven untreated animals injected with PCA7-switch.
The PCA7-B contrast agent that does not exhibit a solubility switch showed similar enhancement patterns for both tumors following intravenous administration (see Figure 4, C and F). For this reagent, the curves were almost constant in time with slopes of 0.5 ± 0.3 min−1 for the positive tumor and −0.4 ± 0.3 min−1 for the negative tumor. Although these slopes may be somewhat different (p < .05) in this mouse, no difference was found in a second animal injected with PCA7-B (p > .05). The lack of a difference in response of the MMP-7-positive versus negative tumors to PCA7-B is analogous to that seen with the PCA7-switch after treatment of the animals with MMPI. Although a plot of amplitude of the difference in the response of the MMP-7-positive versus MMP-7-negative tumors with PCA7-switch, measured from the slopes of the MRI intensities starting 40 minutes following injection of the contrast agent, shows some mouse-to-mouse and/or experiment-to-experiment variation in response to this contrast reagent, the various controls (PCA7-scrambled, PCA7-B, and PCA7-switch after MMPI treatment) show either no difference between the tumors or a much smaller difference, particularly when compared within the same animal (Figure 5). These observations are consistent with the premise that PCAs containing a solubility switch can be used to detect both MMP proteolysis in a tumor and the efficacy of MMPI within tumors.
Discussion
We have developed MRI contrast agents that monitor proteolytic activity in vivo by virtue of an alteration in agent solubility and enhanced retention in tissues expressing a particular proteinase activity. Using a murine model system in which the activity of a single proteinase was genetically altered in one of two tumors implanted in the hindlimbs of the same mouse, we controlled variables related to animal physiology and assessed the specificity of the response. Control agents of similar molecular weight lacking either substrate recognition or a change in solubility on cleavage provided additional validation of the solubility switch as a functional imaging approach. The PCA7-switch reagent exhibits a significant change in solubility on cleavage by MMP-7 and detects tumor-associated MMP-7 activity in vivo, a response not afforded by the control reagent, PCA7-B, which exhibits in vitro a small change in relaxivity on proteolytic cleavage but lacks the solubility switch.

Difference in slopes of the magnetic resonance imaging (MRI) intensity versus time plots between matrix metalloproteinase 7 (MMP-7)-positive and -negative tumors obtained with either PCA7-switch (left side, open circles) or various controls (right side), PCA7-switch after MMPI (diamonds), PCA7-scrambled (square), or PCA7-B (triangles). Data are calculated from slopes in Table 1, and the results obtained in the same animals are identified by numbers (1, 2, and 3; see Table 1).
The agents will be rapidly and nonspecifically distributed in the microvasculature of both positive and negative tumors. In our hypothesis, PCA7-switch will be cleaved and specifically retained in the positive tumor. This difference between nonspecific and specific signal enhancement depends on the level of MMP-7 activity in the tumors and is thus expected to vary from animal to animal. Visible differences in the trend of the signal enhancement pattern are observed around 40 minutes after injection of PCA7-switch (see Figures 3 and 4). Thus, the curves were fitted assuming a linear change in signal from 40 minutes after injection of the agent. The differential response of PCA7-switch in the MMP-7-positive versus MMP-7-negative tumors is consistent with our previous finding of a selective response of these kinds of xenograft tumors to an MMP-7-targeted optical proteolytic beacon, 22 an observation consistent with enhanced MMP-7 immunoreactivity in the MMP-7-positive tumors.
The concentration of agent in the vasculature depends on many factors. This includes the molecular weight, the solubility, and the net electric charge of the compounds, as well as the heart rate and the renal status of the animal. The mice used here do not exhibit any sign of kidney dysfunction. The MRI signal from the bladder of animals displays a rapid and intense increase after injection of every compound used in this study. Moreover, kidney dysfunction, if it were to occur, would cause animal-to-animal variations but could not be invoked to explain signal variations between two tumors on the same animal for a given experiment. Similarly, the heart rate can cause animal-to-animal and experiment-to-experiment variations but could not explain differences between two tumors on the same animal during the same experiment. In mice, it is difficult to measure accurately the concentration of contrast agent in the blood pool, so we instead compared the concentration in muscle tissues around the spine of animals. The enhancement patterns for all animals and for all compounds were very similar, except for Gd-DTPA, which exhibited a much faster clearance. We conclude from these observations that neither the heart rate nor a hypothetical kidney dysfunction could affect a given experiment, although such differences could account for variations from one animal to another.
Many physicochemical factors can influence the localization of contrast agent in a tumor. Well-vascularized areas are expected to show rapid uptake and clearance characteristics. Diffusion of small molecules occurs in every tissue and is certainly expected to be present in tumors. Thus, poorly vascularized areas may still exhibit a signal enhancement from contrast agent that first leaked out of the vasculature from a well-vascularized area and that subsequently diffused in the tissue. This enhancement by diffusion must therefore be slower than that in a well-vascularized area. In addition, for a given extracellular matrix arrangement, molecular weight and charge are expected to modulate the diffusion of a molecule. The molecular weights of our three compounds were similar at 2,418 Da (PCA7-switch), 2,418 Da (PCA7-scrambled), and 2,314 Da (PCA7-B). For comparison, the molecular weight of Gd-DTPA (Magnevist, Berlex) is 938 Da. The same amino acids were used for PCA7-switch and PCA7-scrambled but in a different order, where the other constituents of the molecules are identical. Thus, the net charge of these two compounds is expected to be identical. Based solely on diffusion, the core of similar tumors of animals injected with any of the three PCA compounds should be similar. Thus, the increased retention of PCA7-switch in MMP-7-positive tumors cannot be explained simply by differences in the diffusion of this reagent.
The blood flow in tumors is highly heterogeneous and can lead to substantial regional and temporal variations.27–29 The pharmacokinetics of numerous contrast agents has been revealed by DCE-MRI in the past. A different signal enhancement pattern has been noted between the rim and the core of tumor xenografts on mouse models, revealing a more or less gradual decrease in the perfusion toward the core.23–26 This behavior was modulated by the size of the tumor, where larger tumors have a higher probability of having a necrotic center, which may often have resulted from this decrease in perfusion. The slow-intensity increase in the core of the larger positive tumors in Figure 4 could be explained by slow diffusion. However, a similar trend was seen in Figure 3, where the positive tumor is smaller than the negative tumor. Clearly, diffusion alone cannot explain the enhancement patterns. Examination of the maximum intensity reached for the positive versus negative tumors also argues against differences in diffusion resulting from different-sized tumors. As illustrated in Figures 3 and 4, it is noted that the intensity in the core of the positive tumors was generally larger than in the negative tumor in each of the seven animals injected with PCA7-switch, consistent with diffusion playing a minor role in the response.
We suggest that the mechanism responsible for the switch in the pharmacokinetic properties is a change in the solubility of PCA7-switch following cleavage by MMP-7. By HPLC, the retention time of PCA7-switch is similar to that of PCA7-scrambled, indicating that their solubility is comparable. Thus, the pharmacokinetic properties of these two compounds are expected to be similar. However, a marked difference between the enhancement patterns of PCA7-switch and PCA7-scrambled is observed (see Figure 3). Our interpretation is thus that cleavage of PCA7-switch can take place in the rim and the core of the MMP-7-positive tumors. Strong perfusion in the rim of the tumors can wash out any cleaved compound, even if its solubility has decreased, but we cannot exclude the possibility that some of the compound cleaved in the rim might diffuse to the core. In any case, signal intensity in the core of the positive tumors increases throughout the measurement period (up to 160 minutes) presumably owing to the altered pharmacokinetic properties of the cleaved compound coupled to a smaller perfusion in this area.
The selectivity of the solubility switch is determined by the substrate peptide sequence. The peptide sequence similar to that used for PCA7-switch demonstrates a 56-, 13-, and 30-fold selectivity for MMP-7 compared with MMP-2, −3, and −9, respectively, based on in vitro catalytic efficiency, kcat/Km, determinations 22 and from unpublished results (J. O. McIntyre and L. M. Matrisian, 2006). Our in vivo studies demonstrate that PCA7-switch responds to MMP-7 expression in a background of other proteinases expressed by SW480 cells based on the differences observed between the parental cells and MMP-7-transfected variants. The mRNAs for MMP-9, −13, −14, and −15 are reported to be expressed by SW480 cells in culture. 30 However, we cannot rule out the possibility that PCA7-switch is responsive to proteinases other than MMP-7. More than 550 proteinase genes are reported in the human genome, 31 and even within the metzincin clan of metalloendopeptidases (http://merops.sanger.ac.uk/), 32 comprising the MMPs (also known as matrixins, family M10A) and the adamalysins (also known as a distintegrin and metalloproteinase, or ADAMs, family M12B), there are at least 60 members, most with untested cleavage potential for the peptide substrate used in PCA7-switch. GM6001, the MMPI used in the experiment described in Figure 4, is a hydroxamatebased inhibitor with broad specificity for MMPs and many ADAMs.33,34 Interestingly, the rate of clearance of PCA7-switch from control SW480 tumors lacking MMP-7 is more rapid in the presence of GM6001 compared with the absence, suggesting that there may be GM6001-sensitive proteinases in the parental cells that have some influence on the solubility of PCA7-switch. The limits in the specificity of PCA7-switch, however, do not diminish its utility in monitoring tumors with high MMP-7 expression or in assessing the effect of MMPIs that include MMP-7 as part of their inhibition profile.
In summary, we have developed novel functional imaging agents capable of noninvasive, selective monitoring of enzymatic activity associated with a subset of proteinases in vivo. Although the same principle could be exploited for other imaging modalities or for targeted therapy, the widespread use of MRI in the clinical setting enhances the potential applicability of solubility-switch compounds as biomarkers for tumor-associated proteolytic activity for cancer detection, diagnosis, prognosis, or response to therapy.
Footnotes
Acknowledgment
We wish to acknowledge Sedef Everest for technical assistance.
