Abstract
To develop new radiopharmaceuticals for interventional radionuclide therapy of locally recurrent prostate cancer, poly[N-(3-aminopropyl)methacrylamide] [poly(APMA)] polymers were synthesized by free radical precipitation polymerization in acetonedimethylsulfoxide using N,N‘-azobis(isobutyronitrile) as the initiator. The polymers were characterized with nuclear magnetic resonance, size exclusion chromatography, and dynamic light scattering (Mn 5 2.40 × 104, Mw/Mn = 1.87). Subsequently, poly[APMA] was coupled with 1,4,7,10-tetraazacyclododecane-1,4,7,10-tetraacetic acid (DOTA) using 1-(3-dimethylaminopropyl)-3-ethylcarbodiimide hydrochloride as an activator, followed by conjugation with 64Cu radionuclide. Prolonged retention of poly[APMA]-DOTA-64Cu conjugates within the tumor tissues was demonstrated by micro–positron emission tomography at 24 hours following intra-tumoral injection of the conjugates to human prostate xenografts in mice. The data suggest that the poly[APMA]-DOTA-64Cu conjugates might be useful for interventional radionuclide therapy of locally recurrent prostate cancer in humans.
PROSTATE CANCER is the second leading cause of cancer deaths in men, after lung cancer.1,2 Patients diagnosed with a localized primary tumor are usually treated with radical prostectomy or external beam radiotherapy (EBRT). 3 Despite a good initial response to EBRT, some patients may suffer local recurrence of the tumor.
Various approaches, such as gene therapy 4 and radionuclide therapy, 5 have been investigated for treatment of locally recurrent prostate cancer refractory to conventional treatment. Targeted delivery of a large dose of radionuclide to tumors is most critical for the success of radionuclide cancer therapy. Different from systemic radionuclide therapy, interventional radionuclide therapy treats nonresectable solid tumors by direct injection of a therapeutic dose of radionuclide to tumor tissues.6,7 In comparison with systemic radionuclide therapy, interventional radionuclide therapy may achieve delivery of a larger dose of radionuclide to tumors locally while reducing nonspecific radiation exposure to normal organs or tissues. Various materials have been investigated as a delivery vehicle for interventional radionuclide therapy, such as glass microspheres. 8 Recently, synthetic polymers emerged as a promising carrier for radionuclide therapy of cancers using 188Re, 166Ho, or 90Y radionuclides.9–11 Among several copper isotopes, 64Cu is well suited for targeted radiotherapy of cancer because (1) 64Cu has a half-life of 12.7 hours and emits both β− (40.0%) and β+ (17.4%) particles, which allow use of positron emission tomography (PET) to monitor its biodistribution and localization to the tumors in vivo 12 ; (2) it was reported that the effectiveness of 64Cu for radiotherapy was comparable to that of 67Cu, a pure β-emitter.13,14 1,4,7,10-Tetraazacyclododecane-1,4,7,10-tetraacetic acid (DOTA) is a commercially available metal chelator and has been widely used to prepare radiopharmaceuticals for experimental cancer imaging and radionuclide therapy, although it was reported that copper complexes formed by DOTA were less stable than those formed by 4,11-bis(carboxymethyl)-1,4,8,11-tetraazabicyclo[6.6.2]hexadecane (CB-TE2A) and 4,10-bis(carboxymethyl)-1,4,7,10-tetraazabicyclo[5.5.2]tetradecane (CB-DO2A).15,16 In this study, we synthesized cationic poly[N-(3-aminopropyl)-methacrylamide (APMA)]-DOTA copolymers by coupling multiple DOTA molecules on a single APMA polymer chain (Figure 1), which might be used to deliver a high dose of radiation to tumors by direct injection of the poly[APMA]-DOTA-Cu conjugates loaded with a high payload of 64Cu or 67Cu radionuclide. Taking advantage of the positron-emitting capability of 64Cu radioisotope, the distribution and retention of the conjugates within tumor tissues were monitored by microPET after intratumoral injection of the conjugates.
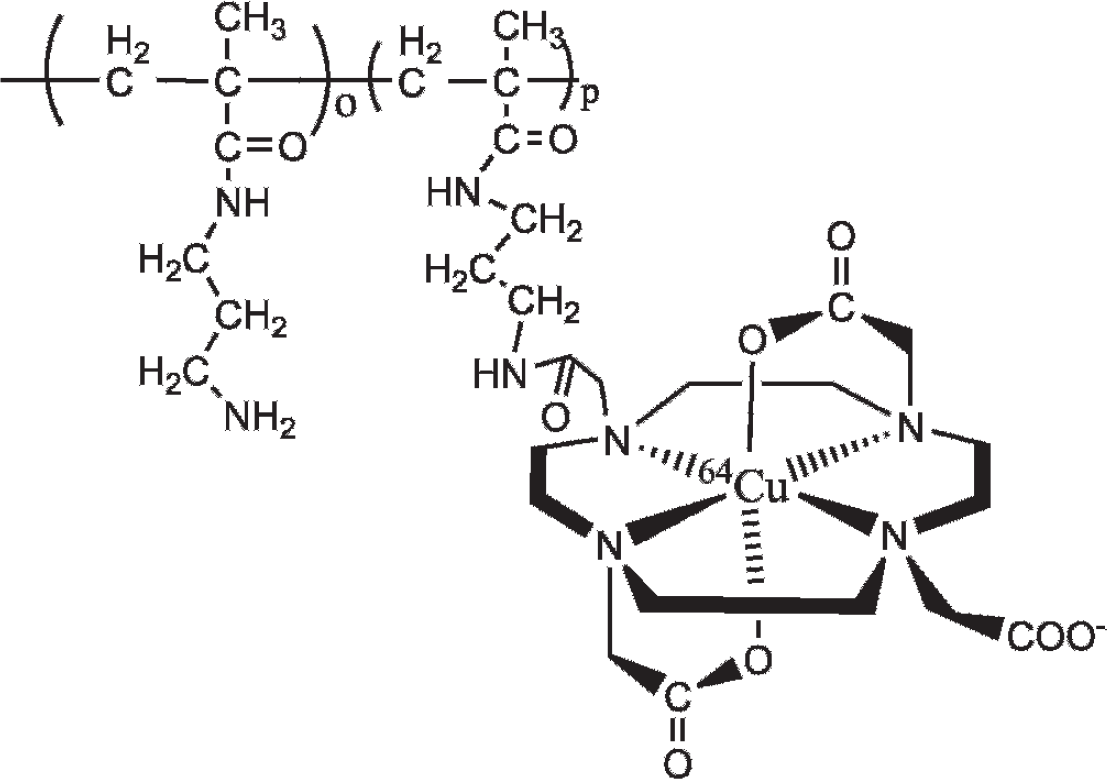
Structure of the poly[APMA]-DOTA-64Cu conjugate.
Materials and Methods
Chemicals
APMA was purchased from Polyscience Inc (Warrington, PA). N-Hydroxysulfonosuccinimide (SNHS) was purchased from Pierce Biotechnology Inc (Rockford, IL). 2,2‘-Azobisisobutyronitrile (AIBN), acetone, methanol, and dimethylsulfoxide (DMSO) were purchased from Sigma-Aldrich (St. Louis, MO). DOTA and 1-(3-dimethylaminopropyl)-3-ethylcarbodiimide hydrochloride (EDC) were purchased from Fluka (Sigma-Aldrich). Radioactive copper 64 chlorides (64CuCl2) were obtained from Mallinckrodt Institute of Radiology, School of Medicine, Washington University (St. Louis, MO).
Synthesis and Characterization of Poly[APMA]
Poly[APMA] was synthesized by free radical precipitation polymerization of APMA in an acetone-DMSO mixture using AIBN as the initiator. The reaction mixture was sealed in an ampule under vacuum after deoxygenation by three cycles of liquid nitrogen/vacuum in a dry ice bath. The polymerization was then carried out at 55°C for 24 hours. The precipitated polymer was recovered by filtration. The crude product was dissolved in a small volume of methanol and precipitated by slowly adding into a 20-fold volume excess of acetone. The polymer was dried in a vacuum oven at room temperature until constant weight was obtained. The number average molecular weight (Mn) and molecular weight polydispersity index (PDI = Mw/Mn) were estimated by size exclusion chromatography (SEC) using a 4.6 × 250 mm Synchropak CATSEC-300 column (Eprogen Inc, Darien, IL). The mobile phase was 0.3 M NaCl, 0.1% trifluoroacetic acid (TFA) buffer (pH 2.5). The system was equipped with a seven-angle BIMwA multiangle static light scattering detector and BIDNDC differential refractometer (Brookhaven Instruments, Holtsville, NY). The BIMwA detector was equipped with a 30 mW vertically polarized solid-state laser (660 nm) as a light source. SEC data were analyzed using PSS WinGPC Unity software (Polymer Standards Services, Mainz, Germany). The hydrodynamic diameter of the synthesized polymers in water (2 mg/mL) was estimated by dynamic light scattering using the 90Plus particle size analyzer (Brookhaven Instruments). 1H-Nuclear magnetic resonance (NMR) spectra were recorded in D2O solution using a Varian AC400 NMR spectrometer (Varian NMR Systems, Palo Alto, CA): D2O (δ) 3.1 (b, 2H, NHCH2CH2CH2NH2), 2.9 (b, 2H, side chain NHCH2CH2CH2NH2), 1.82 to 1.51 (m, 4H, NHCH2-CH2CH2NH2 and CH2 of backbone), and 1.01 to 0.71 (m, 3H, CH3).
Synthesis of Poly[APMA]-DOTA Conjugates
To prepare poly(APMA)-DOTA conjugates, DOTA was first activated by EDC using molar ratio of DOTA: EDC:SNHS = 10:5:4. 17 Typically, 24 mg (48 μmol) of DOTA was dissolved in 1 mL water and mixed with 4.6 mg (24 μmol) of EDC in 0.26 mL water at room temperature. The pH of the reaction mixture was adjusted to 5.0 using 0.1 M NaOH. SNHS (4.2 mg, 19.2 μmol) was then added to the stirred solution in the ice bath, and the pH of the reaction mixture was adjusted to 5.5 using 0.1 M NaOH. The activation reaction was conducted for 40 minutes at 4°C. Poly[APMA] (12 mg, 68 μmol of monomer units, in 0.6 mL water) was cooled to 4°C and added to the reaction mixture. The pH was adjusted to 8.5 with 0.1 M NaOH, and the reaction was allowed to incubate overnight. The APMA polymer-DOTA conjugates were purified from low molecular weight impurities by centrifuge filtration using a Vivaspin 2 spin column (MWCO 10 kDa, Vivascience, Hannover, Germany). After purification, the residual water was removed by freeze-drying. The content of DOTA in the poly[APMA]-DOTA conjugate was determined from the integral intensities of DOTA and APMA in 1H NMR. The hydrodynamic diameter and zeta potential of the conjugates in water were estimated using the 90Plus particle and zeta potential analyzer. 1H NMR: D2O (δ) 3.45 (b, CH2COOH and CH2CONH of DOTA), 3.25 to 2.80 (m, NHCH2CH2CH2NH of APMA, NCH2CH2N of DOTA), 1.85 to 1.49 (m, CH2 of APMA backbone, NHCH2CH2CH2NH), 1.01–0.69 (m, CH3).
Radiolabeling of Poly[APMA]-DOTA Conjugates with 64Cu Radionuclide
Poly[APMA]-DOTA conjugates were radiolabeled with 64Cu radionuclide by adding 50 μCi of 64CuCl2 into a solution of 0.2 mg of poly[APMA]-DOTA in 0.5 mL of 0.1 M sodium acetate buffer (pH 5.5) and incubating at 55°C for 50 minutes. Subsequently, poly[APMA]-DOTA-64Cu conjugates were purified by centrifuge filtration using Vivaspin 2 to separate the conjugates from free 64CuCl2 using distilled water as the solvent. Specific activity (μCi/mg) of the poly[APMA]-DOTA-64Cu conjugates was determined by measuring the radioactivity of the conjugates purified with the Vivaspin column. Radiolabeling efficiency was determined by dividing the radioactivity of the purified radiolabeled conjugates by the total radioactivity of the conjugates and the filtrates containing nonconjugated free copper radionuclide and then multiplying by 100. To assess nonspecific binding of the 64Cu radionuclide to the APMA polymer chain, the poly[APMA] polymers were radiolabeled with 64CuCl2 under the same conditions as those used for radiolabeling of the poly[APMA]-DOTA conjugates, followed by counting radioactivity of the poly[APMA]-64Cu purified with a Vivaspin column. The stability of the radiolabeled conjugates was assessed by incubation of the poly[APMA]-DOTA-64Cu conjugates in bovine serum at 37°C for 1 hour, followed by counting radioactivity of the conjugate and the serum separated from the conjugates by the Vivaspin. Finally, the radiolabeled conjugates dissolved in normal saline were sterilized by filtration with a 0.22 μm Millipore filter prior to use in animal studies.
Implantation of Human Prostate Cancer Xenografts in Mice
Animal studies were performed according to a protocol approved by the Animal Investigation Committee, Wayne State University. PC-3 human prostate cancer cells (American Type Culture Collection, Manassas, VA) were cultured in RPMI1640 medium supplemented with 10% fetal bovine serum, 100 U/mL penicillin, and 100 mg/mL streptomycin (Biosource International, Camarillo, CA). To establish a human prostate cancer xenograft in mice, PC-3 cells (5 × 106/injection site) were injected to a right flank of an immunocompromized athymic mice (male, 4–5 weeks old) purchased from Harlan Laboratory (Indianapolis, IN). When the tumor xenografts reached about 0.8 × 0.8 cm2 in size, the tumor-bearing mice were subjected to microPET study after intratumoral injection of the radiolabeled conjugates.
MicroPET Study
MicroPET studies were performed in a method similar to that described before, 18 using a microPET R4 tomograph (Concorde Microsystems, Knoxville, TN). Briefly, the tumor-bearing mice were anesthetized with a mixture of ketamine (100 mg/kg) and xylazine (7 mg/kg) and positioned in a spread-supine position on the microPET bed. Following a transmission scan, the poly[APMA]-DOTA-64Cu conjugates (10–15 μCi in 0.1 mL normal saline) were directly injected to the tumor xenograft tissues. Static data acquisition was performed at 1 and 24 hours postinjection, respectively, which consisted of two scan frames at two different bed positions (15 minutes duration for each bed) with a 2 cm overlap. The attenuation-corrected whole-body images were reconstructed using an iterative OSEM-2D algorithm. 19 The whole-body microPET images were visually examined for retention and biodistribution of radioactivity of the conjugates in the tumor regions.
To assess intratumoral retention of the conjugates quantitatively, the image data were processed using the ASIpro microPET data analysis software (Concorde Microsystems) with measured attenuation, scatter correction, and the OSEM2D iterative algorithm. A predetermined calibration factor from a scan of a phantom containing 64CuCl2 was used to convert counts/pixel/min to KBq (μCi)/cc for the 64Cu radioactivity. Regions of interests were drawn over the tumors, and the average concentration of the radionuclide KBq (μCi)/cc for the defined area was obtained as a weighted (by area) average of the radioactivity from all image planes of the defined tumor region. Finally, the decay-corrected percentage of injected dose per gram tissue (%ID/g) for the tumors was calculated by dividing the obtained average tracer concentration (μCi/cc) in the tumor region by the injected activity (μCi) over tumor weight (assuming 1 g = 1 cc).
Tissue Radioactivity Assay Ex Vivo
On completion of the microPET studies at 24 hours postinjection, the tumor-bearing mice were euthanized under anesthesia and postmortem tissues were harvested, weighted, and counted for radioactivity with a Packard Cobra II Gamma Counter (Perkin-Elmer, Wellesley, MA). Tissue radioactivity was calculated and expressed as a decay-corrected percentage of injected dose per gram tissue.
Results
Synthesis and Characterization of Poly[APMA]-DOTA-64Cu Conjugates
The synthetic pathway for the poly(APMA)-DOTA-64Cu conjugates is shown in Figure 2. The polymer was first synthesized by a conventional free radical precipitation polymerization, and the polymerization conditions, such as initiator concentration, were optimized to obtain polymers with the desired molecular weight. 20 Poly[APMA] is not a biodegradable polymer, so the target molecular weight (20,000–40,000 g/mol) was selected to ensure ultimate renal elimination. 21 To conjugate DOTA to poly[APMA], an active ester of DOTA was prepared by a reaction with EDC and SNHS (see Figure 2). The use of SNHS stabilizes the amine-reactive intermediate and increases the efficiency of the EDC-mediated coupling reaction. The amine-reactive SNHS ester of DOTA had sufficient stability to permit efficient conjugation by forming a stable amide bond with poly[APMA]. Because of its multivalent nature, a large excess of DOTA was used to ensure that only one carboxylic acid group is activated and that no undesired crosslinking takes place in the reaction with poly[APMA]. Simple centrifuge filtration was sufficient to purify the poly[APMA]-DOTA conjugates from unreacted DOTA and other low molecular weight byproducts. The purified poly[APMA]-DOTA conjugates were first characterized by 1H-NMR to determine the content of DOTA groups in the conjugates (Figure 3). The conjugation of DOTA to poly[APMA] resulted in broadening of its NMR signals. We have used the integral intensities of the CH2-COOH and CH2-CONH protons of DOTA (≈3.4–3.5 ppm) and the integral intensity of the CH3-C in the poly[APMA] backbone (0.8–1.0 ppm) to calculate the content of DOTA in the conjugates. The data show that, on average, the poly[APMA]-DOTA conjugates contained 16.6 mol% of the monomer units modified with DOTA. Assuming the molecular weight of poly[APMA] to be 24,000 daltons, that translates into 28 DOTA groups per average polymer chain. The hydrodynamic diameter of poly[APMA] was 58 ± 2 nm. The diameter increased after DOTA conjugation to 87 ± 6 nm, indicating only a negligible extent of crosslinking owing to the reaction with DOTA. The observed hydrodynamic diameter of both the poly[APMA] and poly[APMA]-DOTA is higher than expected for a polymer with Mn ≈ 24,000. The apparent discrepancy can be partly explained by the fact that the hydrodynamic diameter is a z-average, whereas the molecular weight is a number average. Polyelectrolyte effects in solutions of poly[APMA] further complicate a precise analysis of the molecular size. Lastly, the polymers might contain a small amount of high molecular weight molecules that distort the measurement. The cationic nature of the used polymer is a crucial feature necessary for effective retention in the tumor tissue after intratumoral administration. The high content of negatively charged DOTA groups in the conjugates will reduce the overall positive charge. To confirm the cationic nature of poly[APMA]-DOTA conjugates, we have measured their zeta potential. The positive value of 4.7 mV confirms that despite the high degree of modification, the conjugates overall remain positively charged and should be suitable for the intended use. Finally, the poly[APMA]-DOTA conjugates were radiolabeled with 64Cu radionuclide by incubation of the conjugates with 50 μCi of 64CuCl2 in 0.5 mL of acetate buffer (pH 5.5) at 55°C for 50 minutes. Specific activity of the poly[APMA]-DOTA-64Cu conjugates was estimated at 200 to 250 μCi/mg, whereas the radiolabeling efficiency was estimated at 16.7 ± 5.0%. In the control, a small amount of 64Cu radionuclide (4.0–4.5 μCi) was found to be nonspecifically bound to the poly[APMA] polymers after incubation of the polymers with 50 μCi of 64CuCl2 under the same conditions used for radiolabeling the poly[APMA]-DOTA conjugates. Additionally, a fraction of radioactivity (≈2.0–2.5 μCi) was found in the serum free of the conjugates after incubation of the purified poly[APMA]-DOTA-64Cu conjugates (10 μCi) in 0.5 mL of bovine serum at 37°C for 1 hour.

Synthetic scheme of the poly[APMA]-DOTA-64Cu conjugate.

Structures and typical 1H-nuclear magnetic resonance spectra of poly[APMA] and poly[APMA]-DOTA in D2O (n = 169, o = 0.834, p = 0.166).
Intratumoral Retention of the Poly[APMA]-DOTA-64Cu Conjugates Assessed by MicroPET
The poly[APMA]-DOTA-64Cu conjugates were directly injected into the human prostate cancer xenografts in mice (N = 5), followed by microPET at 1 and 24 hours postinjection, respectively, to assess intratumoral retention of the conjugates. Intense radioactivity in the tumor region was visualized on the images obtained at 1 and 24 hours postinjection, indicating prolonged retention of the conjugates in the tumor tissues (Figure 4). Excretory radioactivity of the conjugates was seen in the kidneys and the urinary bladder on the images obtained at 1 hour postinjection but not on the images obtained at 24 hours postinjection (images not shown), indicating renal clearance of the conjugates escaped from the tumor injection sites. PET quantitative analysis showed that large amounts of the poly[APMA]-DOTA-64Cu conjugate (18.16 ± 2.03 %ID/g) were present in the region of the tumors at 1 hour following intratumoral injection. At 24 hours postinjection, a significant amount of radioactivity (5.19 ± 1.57 %ID/g) remained in the tumors, indicating prolonged intratumoral retention of the conjugates. There was only a small amount of radioactivity in the other regions of the mouse body. To further confirm intratumoral retention of the conjugates, postmortem tissues were harvested for ex vivo radioactivity assay on completion of the imaging studies at 24 hours postinjection. Radioactivity of the tumor tissues was found to be 4.87 ± 1.18 ID%/g, which correlated well with the data from PET quantitative analysis. Radioactivity of other tissues was determined as follows: liver, 1.39 ± 0.34 ID%/g; lungs, 1.63 ± 0.82 ID%/g; kidneys, 1.29 ± 0.41 ID%/g; spleen, 0.35 ± 0.21 ID%/g; intestines, 0.66 ± 0.22 ID%/g; muscle, 0.22 ± 0.16 ID%/g; brain, 0.07 ± 0.05 ID%/g; and blood, 0.17 ± 0.07 ID%/g. In contrast, the majority of 64CuCl2 (20 μCi in 0.1 mL normal saline) was rapidly washed out from the tumors after intratumoral injection, with intense radioactivity in the liver by hepatic clearance (images not shown).

Representative micro–positron emission tomographic (microPET) images show prolonged intratumoral retention of the poly[APMA]-DOTA-64Cu conjugates directly injected to tumors. The athymic mice (N = 5) bearing human prostate cancer xenografts were intratumorally injected with the poly[APMA]-DOTA-64Cu conjugates and subjected to microPET at 1 hour (A) or 24 hours (B) postinjection of the conjugates. Intense radioactivity that remained in the region of the tumor indicated prolonged retention of the conjugates in the tumor tissues. Different maximum %ID/% (decay corrected) was used to show the distribution of low radioactivity outside the tumor region. %ID/g = percentage of inject dose per gram. Arrows indicate retention of the conjugates in the tumor tissue.
Discussion
The polymers described in this study offer the advantage of a flexible synthetic methodology that permits easy incorporation of desired functions. The polymers can be prepared at a range of molecular weights by modulating the concentration of the initiator in the polymerization mixture. More importantly, the cationic charge of the poly[APMA] is considered beneficial for enhanced retention of the polymers conjugated with drugs or radionuclide in the tumors following intratumoral injection. 22
Poly[APMA]-DOTA conjugates hold potential as a vehicle or carrier for interventional radionuclide therapy of locally recurrent prostate cancer. Each of the poly[APMA]-DOTA conjugates contains multiple DOTA molecules, which allow delivery of a high payload of copper radionuclide to tumors by injection of a single dose of the conjugates. Prolonged retention of the conjugates within the tumor tissues is desirable for tumor-specific radiation using copper radionuclide. Although intratumoral retention of the conjugates may be related to the molecular size of the conjugates, it is probably more likely to be related to the positive charge of the conjugates, which allow binding of the conjugates to negatively charged macromolecules in the tumor tissues, such as components of cellular membranes. Additional studies are needed to elucidate molecular mechanisms underlying prolonged intratumoral retention of the conjugates, which may provide information for development of even better vehicles for interventional radionuclide therapy.
Physiologically, copper radionuclide disassociated from the copper conjugates will be rapidly cleared by the hepatic clearance pathway and excreted to the intestinal tracts via the bile ducts. The presence of a small quantity of 64Cu radioactivity in the liver suggested that the poly[APMA]-DOTA-64Cu conjugates retained in the tumor tissues were quite stable, which was consistent with the result of the in vitro stability assay.
In this study, no symptoms of acute toxicity or death of the animals were observed after intratumoral injection of the poly[APMA]-DOTA-64Cu conjugates (up to 0.5 mg/mouse, 10–15 mCi/0.2 mg). However, a study of the long-term toxicity of the poly[APMA]-DOTA conjugates, such as chronic renal toxicity, needs to be conducted prior to a potential clinical trial of the poly[APMA]-DOTA-64Cu conjugates for interventional radionuclide therapy of prostate cancer in humans.
Although the lethal effects of 64Cu radionuclide were compatible with those of 67Cu radionuclide, their radiocytotoxicity mechanisms were found to be quite different from each other.13,14 High-energy gamma rays were important for therapeutic use of 67Cu radionuclide, whereas Auger electrons, not gamma rays, might play a major role in radiocytotoxicity by 64Cu radionuclide. Because intracellular uptake of the poly[APMA]-DOTA-64Cu conjugates may be limited by their molecular size, use of 67Cu radionuclide to radiolabel the poly[APMA]-DOTA conjugates may be more desirable for maximum therapeutic efficacy of the conjugates. The utility of poly[APMA]-DOTA-64Cu conjugates for systemic radionuclide cancer therapy will likely be limited by a lack of tumor-targeting capability and fast hepatic clearance owing to its high cationic charge, which, for all practical purposes, makes them incompatible with the passive accumulation in tumors. Recently, a polymer-peptide conjugate of N-(2-hydroxypropyl) methacrylamide (HPMA), RGD4c peptide binding to aVβ3 integrin molecules, and diethylenetriaminepentaacetic acid (DTPA) radiolabeled with 90Y radionuclide (eg, HPMA-RGD-DTPA-90Y polymer-peptide conjugate) was investigated and found to be effective for radionuclide therapy of prostate cancer in an animal model.23,24 It will be interesting to synthesize RGD-Poly(HPMA)-DOTA-67Cu conjugates and compare them with RGD-HPMA-DTPA-90Y polymers for their efficacy for targeted radionuclide therapy of prostate cancer.
Conclusions
Prolonged retention of the poly[APMA]-DOTA-64Cu conjugates in the human prostate cancer xenograft tissues was demonstrated by microPET at 1 and 24 hours following intratumoral injection of the conjugates. The data suggested that the poly[APMA]-DOTA-64Cu/67Cu conjugates might be useful for interventional radionuclide therapy of locally recurrent prostate cancer in humans.
Footnotes
Acknowledgment
We thank Haiyuan Zhang for assistance in the preparation of some of the Poly[APMA]-DOTA-64Cu conjugates and the microPET study.
