Abstract
Bioluminescence, positron emission tomography (PET), and fluorescence modalities are currently available for noninvasive imaging in vivo, each with its own merits. To exploit the combined strengths of each and facilitate multimodality imaging, we engineered a dual-reporter construct in which firefly luciferase (FLuc) and a 12–amino acid nonstructural linker were fused in frame to the N-terminus of a mutant herpes simplex virus thymidine kinase (mNLS-SR39TK) kinetically enhanced for positron emission tomography (PET). Furthermore, a triple-reporter construct was developed in which monster green fluorescent protein (MGFP), a recently available enhanced fluorescent protein, was introduced into the fusion vector downstream of an internal ribosome entry site (IRES) to allow analysis by fluorescence microscopy or flow cytometry without compromising the specific activities of the upstream fusion components. FLuc bioluminescence was measured with a cooled charge-coupled device camera and mNLS-SR39TK activity by 9-[4-[18F]fluoro-3-(hydroxymethyl) butyl guanine (18F-FHBG) microPET or 3H-penciclovir net accumulation. Importantly, HeLa cells transiently transfected with the FLuc-mNLS-SR39TK-IRES-MGFP triple reporter retained the same specific activities of the FLuc-mNLS-SR39TK heteroenzyme and the individual unfused enzymes with no change in protein half-lives. The presence of the IRES-MGFP modestly decreased upstream heteroprotein expression. In living mice, somatic gene transfer of a ubiquitin promoter-driven FLuc-mNLS-SR39TK-IRES-MGFP plasmid showed a > 1,000-fold increase in liver photon flux and a > 2-fold increase in liver retention of 18F-FHBG by microPET compared with mice treated with control plasmid. Multifocal hepatocellular fluorescence was readily observed by standard confocal microscopy. This second-generation triple reporter incorporating enhanced components enables bioluminescence, PET, and fluorescence imaging of cells and living animals.
MOLECULAR IMAGING incorporates many different imaging modalities and related technologies, each with its own set of strengths and weaknesses.1–3 Radiotracer-based imaging techniques, such as positron emission tomography (PET) and single-photon emission computed tomography (SPECT), used advantageously for basic research and clinical applications, for example, offer high sensitivity, quantitative data sets, and tomographic display.
They depend, however, on determination of the whole body localization of injected radiation sources (radiopharmaceuticals) and thus are critically dependent on tracer pharmacokinetics. In addition, both PET and SPECT tracers are economically demanding and restricted to facilities equipped with state-of-the-art radiopharmacies.
Bioluminescence imaging, by contrast, involves measuring the light output from a reporter enzyme such as a luciferase on the addition of substrate. Firefly luciferase (FLuc), for example, produces light when it reacts with
Fluorescence imaging has the unique advantage of being able to visualize and localize subcellular events in cellulo and ex vivo on excitation of the reporter by external light sources without the need for an injectable substrate. In contrast to enzyme-based imaging modalities, fluorescence can also detect the presence of fluorescent protein in dead cells. Although deconvolution strategies can compensate in some measure for wavelength-dependent autofluorescence, 4 fluorescence imaging remains limited by the challenges of quantitatively comparing photon output between experiments, especially those conducted in vivo, wherein fluorescence is highly susceptible to photon attenuation, reflectance, scattering, surface geometry, and questions of depth resolution.
As each of these modalities has its own advantages and disadvantages, combining modalities has been an effective way to maximize the information content in a single study. Clinically, combining PET and computed tomography (CT) into a single scanner has been a useful tool in oncology, allowing anatomic localization by CT of biochemical and functional differences as measured by PET. 5 Multimodality studies in vivo that incorporate microPET, fluorescence, and bioluminescence imaging have been enhanced by several fusion reporters, including fusions of herpes simplex virus 1 thymidine kinase (HSV1-TK) and green fluorescent protein (GFP),6,7 Renilla luciferase (RLuc) and GFP, 8 mNLS-SR39TK and enhanced green fluorescent protein (EGFP), 9 and EGFP and HSV1-TK. 10 More recently, HSV1-TK–RLuc, RLuc–monomeric red fluorescent protein (mRFP)–truncated herpes simplex virus 1-SR39TK (tTK), and HSV1-TK–GFP/FLuc constructs have been published.11–13 One common difficulty with various fusion reporters has been a loss of specific activity on generation of fusion enzyme heteroproteins, resulting in diminished sensitivity of the reporter. In this study, we describe the construction and validation in both cell culture and living animals of a second-generation triple reporter that retains enzyme-specific activities for bioluminescence, microPET, and fluorescence imaging.
Materials and Methods
Plasmids
FLuc was polymerase chain reaction (PCR) amplified from the pGL3 control plasmid (Promega, Madison, WI) without or with a sequence encoding for a 12–amino acid linker (ASIGGSSSGGLA) downstream of FLuc. 14 The SR39 variant herpes simplex virus 1 thymidine kinase containing a mutated nuclear localization sequence (mNLS-SR39TK) was PCR amplified from the Gal4-mNLS-SR39TK-EGFP plasmid. 9 All resulting PCR products were ligated into a TOPO TA vector (Invitrogen, Carlsbad, CA). The constructs were then ligated into the pUB6 control vector (Invitrogen) to generate pUB6-FLuc, pUB6-mNLS-SR39TK, and pUB6-FLuc-mNLS-SR39TK. To make pUB6-FLuc-mNLS-SR39TK-IRES-MGFP, the encephalomyocarditis virus internal ribosome entry site (IRES) sequence was PCR amplified from the pIREShyg2 vector (Clontech, Mountain View, CA) and the monster green fluorescent protein (MGFP) was PCR amplified from the humanized monster green fluorescent protein (phMGFP) vector (Promega); both components were subsequently ligated into the TOPO TA vector and then transferred into pUB6-FLuc-mNLS-SR39TK. All plasmids encoded reporters driven by the same constitutive ubiquitin promoter sequence.
Cell Lines and Transfections
HeLa and 293T cells were cultured at 37°C under 5% CO2 in Dulbecco's Modified Eagle's Medium (DMEM) supplemented with 10% heat-inactivated fetal bovine serum, 1% glutamine, and 0.1% penicillin-streptomycin solution. Cells were seeded in six-well plates for transient transfections (0.5 μg total deoxyribonucleic acid [DNA]/100,000 cells) using Fugene 6 as recommended (Roche Applied Science, Indianapolis, IN).
Bioluminescence Imaging in Cell Culture
Twenty-four hours post-transfection, cells were washed once with phosphate buffered saline (PBS) and incubated for 10 minutes with 150 μg/mL
3H-Penciclovir Uptake in Cell Culture
Twenty-four hours post-transfection, cells were washed once with DMEM and incubated for 1 hour at 37°C with 0.2 μCi/mL 3H-penciclovir (Moravek Biochemicals, Brea, CA; 22 Ci/mmol) to allow uptake of the nucleoside, followed by incubation for 1 hour at 37°C in isotope-free DMEM for washout of untrapped nonphosphorylated nucleoside. Cells were rinsed three times with 4°C PBS to clear extracellular spaces and then lysed in 10 mM sodium borate + 1% sodium dodecyl sulfate solution for 30 minutes. An aliquot of cell lysate was assayed for β-galactosidase enzyme activity or protein content, the latter by bicinchoninic acid protein assay (Pierce, Rockford, IL). Cell lysates (750 μL) were added to 5 mL of scintillation fluid (Beckman Coulter, Fullerton, CA) and counted on a Beckman Coulter LS6500 Scintillation Counter. Data were normalized and presented as μCi/β-galactosidase or μCi/mg protein.
Western Blots
FLuc was detected in whole-cell lysates derived from transiently transfected HeLa cells 24 hours post-transfection using a polyclonal anti-FLuc antibody (Promega) at 1:1,000. Blots were probed with antirabbit secondary antibody (Amersham Biosciences, Piscataway, NJ) at 1:1,000, developed with the ECL Western Blotting reagent (Amersham Biosciences) and imaged using the IVIS 100 imaging system (exposure time 1 minute; binning 4; no filter; f/stop 1; FOV 15 cm). Blots were stripped and reprobed with antiactin antibody (Sigma) at 1:1,000 to confirm uniform loading.
Bioluminescence Imaging
Animal protocols were approved by the Animal Studies Committee at Washington University School of Medicine. In vivo transfection of mouse hepatocytes was performed using the hydrodynamic method.16,17 Briefly, plasmid DNA (pUB6 control 3 mg or pUB6-FLuc-mNLS-SR39TK-IRES-MGFP 3 mg) was diluted in phosphate buffered saline (without Mg2+, Ca2+) at a volume of 3 mL and rapidly (within 5–7 seconds) injected into the tail veins of mice (FVB; 8-week-old males; n = 4) using a 3 mL syringe fitted with a 27-gauge needle. Twenty-four hours later, mice were anesthetized using isoflurane (2.5% vaporized in O2) and imaged for liver FLuc expression (after intraperitoneal injection of 150 μg/g body weight [BW]
18F-FHBG Synthesis and MicroPET
18F-FHBG was synthesized as previously published. 18 Twenty-five hours following in vivo transfection of mouse hepatocytes and 1 hour after bioluminescence imaging, mice were injected via the tail vein with 18F-FHBG (120 μCi). One hour later, microPET was carried out at the Small Animal Imaging Core Facility in an F120 microPET scanner (Concorde Microsystems, Nashville, TN). Images were reconstructed and corrected for radio-isotopic decay but not for tissue attenuation. Regions of interest were selected manually and analyzed for radioactivity content (μCi/g tissue/μCi injected dose/g BW; standard uptake value [SUV]) using the Analyze software system (Mayo Clinic Biomedical Imaging Resource, Rochester, MN).
3H-Penciclovir Biodistribution In Vivo
Forty-six hours following in vivo transfection of mouse hepatocytes, mice (n = 3) were injected via the tail vein with 3H-penciclovir (2 μCi) in 100 μL saline. One hour postinjection, mice were sacrificed and organ samples were collected and weighed. Sliced samples were stored at −80°C overnight, thawed, and then incubated at 65°C for 18 hours in 1 mL hyamine hydroxide. After addition of 200 μL 30% H2O2 and 50 μL glacial acetic acid, samples were incubated at 65°C for 30 minutes. After cooling, samples were diluted in 15 mL Cytoscint scintillation fluid (MP Biomedicals, Solon, OH), counted on a Beckman Coulter LS6500 Scintillation Counter, and presented as SUV (μCi/g tissue/μCi injected dose/g BW).
Fluorescence Imaging
Immediately following microPET, animals were sacrificed and liver specimens were collected. The specimens were frozen in optimal cutting temperature media and cryosectioned (5 μm). MGFP expression was detected using an inverted Zeiss Axiovert 200 laser scanning confocal microscope coupled to a Zeiss LSM 5 PASCAL Vario Two RBG system (Carl Zeiss Microimaging GmbH, Oberkochen, Germany) fitted with a 488 nm Ar laser. The same instrument gain and power were used to image liver tissues transfected with either pUB6 control or the pUB6-FLuc-mNLS-SR39TK-IRES-MGFP.
Analysis
Data were reported as mean values ± standard error of the mean. Pairs were compared with the Student's t-test, and values of p < .05 were considered significant.
Results
Construction of a Triple Reporter and Validation in Cell Culture
As a first step toward generation of a multimodality imaging reporter, we created a FLuc-mNLS-SR39TK fusion protein with a 12–amino acid glycine- and serine-rich linker between the two proteins, leaving the C-terminus of thymidine kinase free (Figure 1). This linker length was chosen because 11 or more amino acids are generally required to separate proteins sterically and retain biologic activity.14,19 This fusion was placed downstream of a mammalian ubiquitin promoter that allows for stable expression across a broad range of tissues. 20 Other promoter choices are available, such as the cytomegalovirus (CMV) promoter, which is widely used and thought to provide strong unregulated transcriptional activity in vitro. Its tissue-specific expression, however, may be variable in transgenic animals21,22 and prone to inactivation over time.23,24 Activity of the CMV promoter also depends on other key regulatory genes, including nuclear factor kB, which activates the CMV promoter following liver transfection, 25 and tp53, which down-regulates the promoter. 26 Furthermore, intravenous administration of an adenoviral vector expressing herpes simplex virus thymidine kinase under the control of CMV or respiratory syncytial virus promoters led to greater abnormalities in liver function and liver apoptosis compared with other mammalian promoters, 27 suggesting a possible synergistic toxicity of viral promoters and thymidine kinase.

Schematic of pUB6-FLuc-mNLS-SR39TK.
To test the specific activity of both components of the fusion protein, pUB6-FLuc-mNLS-SR39TK was transiently transfected into HeLa and 293 cells in parallel with unfused pUB6-FLuc and pUB6-mNLS-SR39TK, all under the control of the same ubiquitin promoter. FLuc activity was quantified as normalized bioluminescence output (photons/s/β-galactosidase) and mNLS-SR39TK activity was quantified as normalized net uptake of 3H-penciclovir (μCi/β-galactosidase). In both cell lines, the activity of the FLuc and mNLS-SR39TK components of the fusion protein fully recapitulated the specific activity of each respective unfused reporter protein (Figure 2).
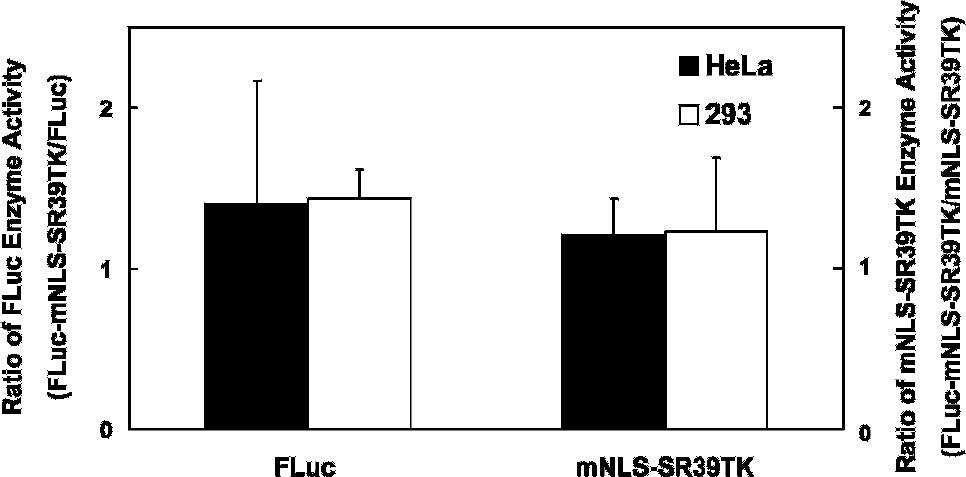
Preservation of native enzyme-specific activities in a fusion reporter in cellulo. Both HeLa and 293 cells were transfected with either pUB6-FLuc, pUB6-mNLS-SR39TK, or pUB6-FLuc-mNLS-SR39TK and normalized to transfection efficiency with β-galactosidase. Normalized FLuc enzyme activity was expressed as photons/s/β-galactosidase, whereas normalized mNLS-SR39TK activity was expressed as trapped μCi/β-galactosidase. Ratios of activity of the fusion reporter FLuc-mNLS-SR39TK over unfused FLuc or mNLS-SR39TK are shown for each cell line. There are no significant differences between cell lines.

Schematic of pUB6-FLuc-mNLS-SR39TK-IRES-MGFP.
A third imaging modality, fluorescence, was added to the reporter by cloning an IRES-coupled MGFP downstream of the FLuc-mNLS-SR39TK fusion sequence (Figure 3). Although the double- and triple-reporter constructs displayed similar overall transfection efficiencies as determined by cotransfection with pRL-CMV (Figure 4A), less photon flux was detected from the FLuc-mNLS-SR39TK fusion protein in the presence of the IRES (see Figure 4, A and B). A similar decrease was seen in the nucleoside trapping activity of the mNLS-SR39TK component of the fusion protein (see Figure 4B), implying that the enzyme activities of both components of the fusion reporter were affected equally. To test the hypothesis that addition of the IRES-MGFP in pUB6-FLuc-mNLS-SR39TK-IRES-MGFP decreased protein expression levels of the FLuc-mNLS-SR39TK fusion protein compared with expression levels produced by pUB6-FLuc-mNLS-SR39TK, Western blots were performed following transfection of Hela cells with each respective reporter (Figure 4C). When normalized for differences in loading, the net reduction in bioluminescence of the fusion reporter component could be attributed to a decrease in expression levels of the fusion protein. Thus, although the specific activities of the triple reporter were retained, the presence of the IRES acted to reduce expression levels of the heteroprotein reporter by ≈50%.
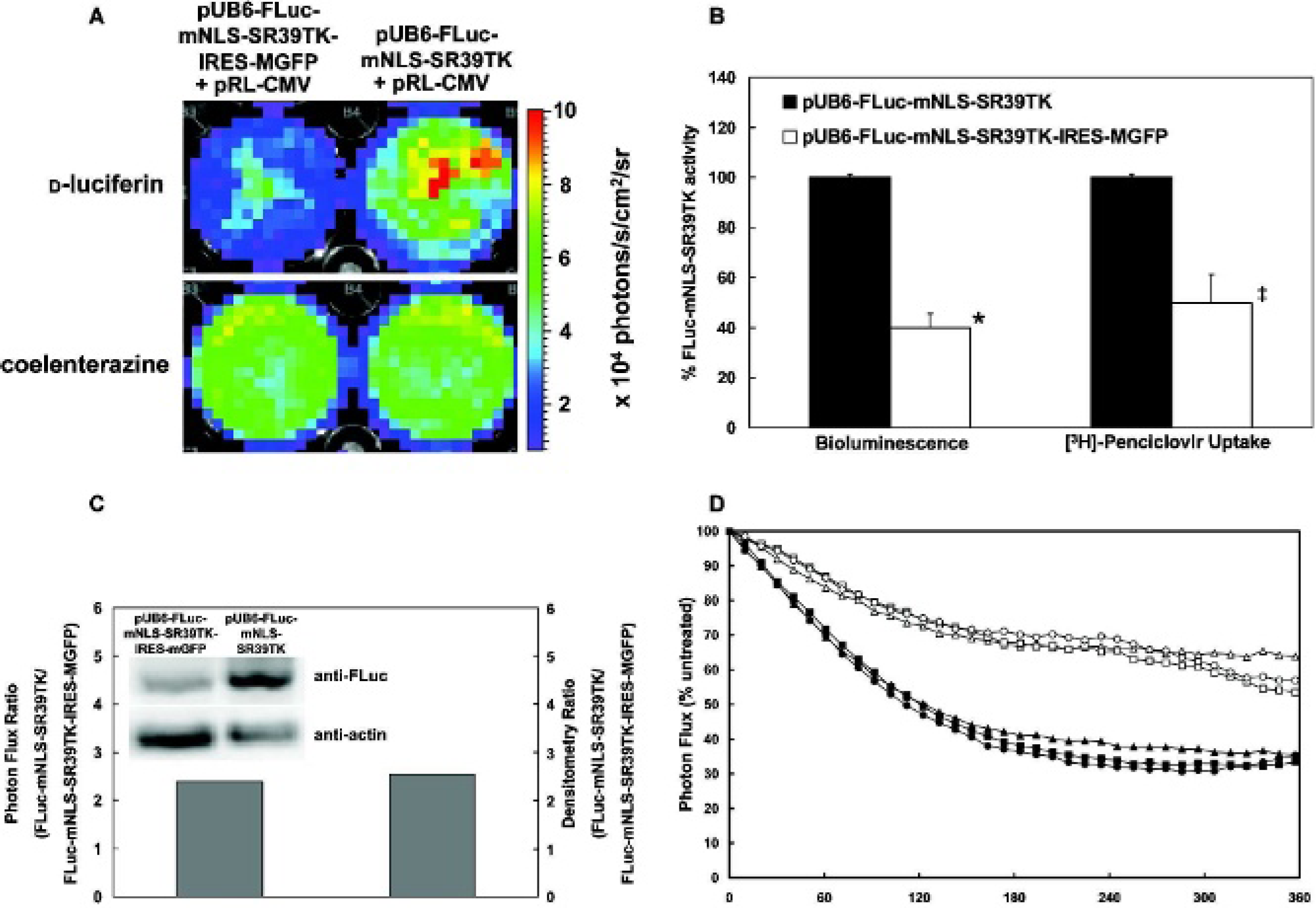
Effect of addition of an internal ribosome entry site (IRES) on expression levels of the FLuc-mNLS-SR39TK fusion reporter protein in cellulo. A, HeLa cells were transfected with pUB6-FLuc-mNLS-SR39TK-IRES-MGFP (left) or pUB6-FLuc-mNLS-SR39TK (right), as well as pRL-CMV, to normalize for transfection efficiency. Bioluminescence images of the same HeLa wells with
To determine if the IRES-induced decrease in net expression of the triple reporter could be attributed to differences in the half-life of the fusion protein, HeLa cells were transiently transfected with either pUB6-FLuc-mNLS-SR39TK or pUB6-FLuc-mNLS-SR39TK-IRES-MGFP and bioluminescence output was determined over time in the absence or presence of either actinomycin D (1 μg/mL) or cycloheximide (100 ng/mL). As shown in Figure 4D, there was no difference in protein stability in the presence of the IRES-MGFP on blockade of either protein transcription or translation. Thus, the modest IRES-induced reduction in fusion reporter expression levels must be attributed to an upstream cis-acting transcriptional inhibitory activity of the IRES.
Reporter Imaging In Vivo
Bioluminescence Imaging
To validate the triple-reporter construct in living animals, somatic gene transfer was performed by introduction of plasmid DNA into the livers of FVB mice via high-volume injection through the tail vein. Twenty-four hours postinjection of DNA, mice were imaged in the IVIS imaging system (Figure 5A). Compared with the mouse liver injected with control DNA, > 1,000-fold more photon flux was detected in the liver of the mouse injected with pUB6-FLuc-mNLS-SR39TK-IRES-MGFP (Figure 5B).
MicroPET Imaging
Following bioluminescence imaging, the mice were injected with 18F-FHBG (120 μCi) and imaged by microPET (Figure 6A). Quantitative analysis of the liver of the mouse injected with pUB6-FLuc-mNLS-SR39TK-IRES-MGFP plasmid showed 2.2-fold greater 18F-FHBG retention than the mouse injected with control plasmid (Figure 6B). Similarly, liver to muscle SUV ratios of radiotracer were 2-fold higher for the triple reporter compared with the control (Figure 6B, inset). In addition, 3H-penciclovir biodistribution analysis of mice showed a comparable 2-fold increase in net retention of tracer in the liver of the mice injected with pUB6-FLuc-mNLS-SR39TK-IRES-MGFP compared with pUB6 control (Figure 6C).

Bioluminescence imaging in vivo. A, Bioluminescence images obtained 24 hours following hydrodynamic delivery of control pUB6 or pUB6-FLuc-mNLS-SR39TK-IRES-MGFP plasmids. B, Logarithmic plot of photon flux from regions of interest analyzed over the liver from bioluminescence images. Compared with pUB6 control, *p > .05.
Fluorescence Imaging
Mice were sacrificed after microPET and livers were cryosectioned for confocal microscopy to detect expression of MGFP. Scattered cytoplasmic fluorescence was visible throughout foci only in the livers of mice injected with the triple reporter (Figure 7).
Discussion
In this study, we present a second-generation construct that enables multimodality imaging by incorporating reporter genes for bioluminescence, microPET, and fluorescence imaging. Not only does each of these modalities have technical merits and weaknesses, but within each modality, there are many choices of reporter genes.
1
Many different luciferases, for example, are available for bioluminescence imaging, including FLuc (Photinus pyralis), RLuc, Gaussia luciferase, and green and red click beetle (Pyrophorus plagiophthalamus) luciferases. Although RLuc has been widely used in bioluminescence imaging, codon humanized FLuc is often preferred to RLuc for biologic applications in vivo because RLuc emits blue light, which is highly attenuated in living tissue, and also exhibits high bursting activity, thereby requiring careful timing of light readout. Furthermore, coelenterazine, the substrate for RLuc, is more expensive than

Micro–positron emission tomography (microPET) in vivo. A, Coronal microPET images obtained 1 hour postinjection of 18F-FHBG in FVB mice 26 hours following hydrodynamic delivery of pUB6 control or pUB6-FLuc-mNLS-SR39TK-IRES-MGFP plasmids. Arrows point to the liver in each image. B, Standard uptake value (SUV) analysis of the liver from the microPET images. Compared with pUB6 control, *p < .01. Inset, SUV of liver normalized to SUV of muscle. Compared with pUB6 control, ‡p < .025. C, 3H-Penciclovir tissue content determined as SUV in two separate lobes of the liver obtained 1 hour postinjection of tracer in FVB mice 47 hours following hydrodynamic delivery of pUB6 control or pUB6-FLuc-mNLS-SR39TK-IRES-MGFP plasmids. In median lobe segments compared with pUB6 control, *p = .05. In right lobe segments compared with pUB6 control, ‡p = .08.
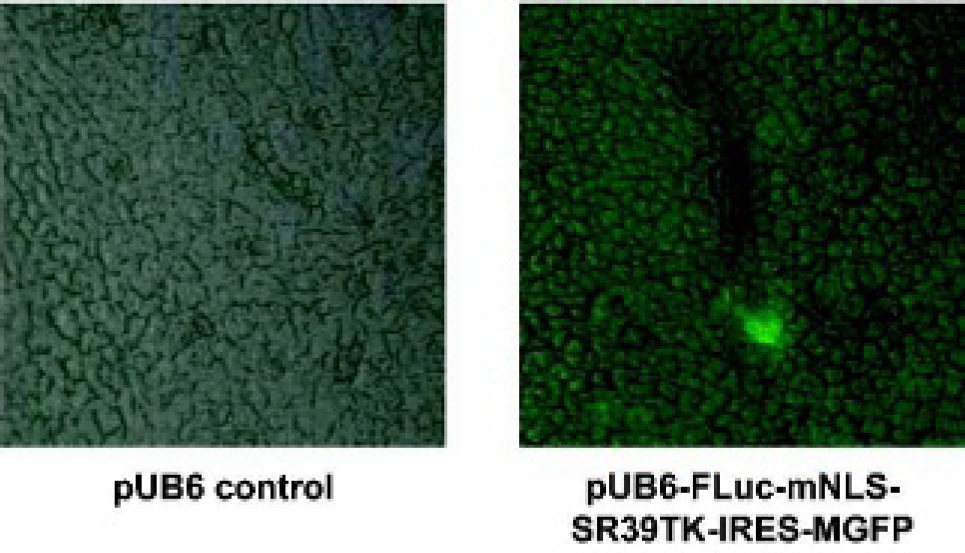
Fluorescence imaging ex vivo. Green fluorescent protein expression levels from the livers of FVB mice collected 28 hours following hydrodynamic injection of pUB6 control (left) or pUB6-FLuc-mNLS-SR39TK-IRES-MGFP (right). Fluorescence images are superimposed over phase contrast images (×40 original magnification).
Similarly, the ability to obtain true tomographic data continues to spur research to improve reporter genes for microPET imaging, such as HSV1-TK.28,29 The reporter mNLS-SR39TK has been optimized for improved sensitivity in vivo, and the herpes simplex virus 1 mutant SR39TK is itself a reporter with improved specificity for purine as opposed to pyrimidine substrates. 30 The latter substrates tend to display higher cross-reactivity with endogenous human thymidine kinase. Further mutations in an N-terminal nuclear localization sequence of SR39TK led to a more even distribution of the protein throughout the nucleus and cytosol and an improvement in purine nucleoside net retention of 12-fold over wild-type HSV1-TK. 9
Finally, the fluorescence reporter gene MGFP was placed in the reporter plasmid. This gene expresses a variant GFP that not only produces a brighter signal but is also more resistant to photobleaching than native GFP; the greater persistence of expression of MGFP perhaps reflects reduced induction of apoptosis linked to expression of many commercially available GFPs. 31 MGFP represents just one of the more recently developed improved fluorescent proteins. Indeed, a number of improved monomeric fluorescent proteins in a wide range of colors created via iterative somatic hypermutation have also been published.32,33 The susceptibility of fluorescence in vivo to attenuation artifacts and depth resolution difficulties can be partially overcome with the use of red fluorescent proteins and spectral unmixing, although red-shifted fluorescent proteins with longer emission wavelengths, such as mPlum, often have decreased quantum yields. 33 A comprehensive summary of important requirements for choosing a fluorescent protein was recently published. 34
Different combinations of similar and other classes of reporter genes have been used to generate a variety of published fusion reporter proteins. Before thymidine kinase, cytosine deaminase (CD) was developed as a reporter gene as it converts the nontoxic antifungal agent 5-fluorocytosine to the highly toxic 5-fluorouracil. Its efficacy as a reporter, however, is diminished as a result of efflux of substrate out of both CD-expressing and control cells. 35 More recently, a fusion CD and wild-type HSV1-TK reporter separated by a polyglycine linker showed strong correlation between the expression of the two component proteins and was used to generate parametric images of CD activity based on net uptake of a radiolabeled pyrimidine analogue by the HSV1-TK, thereby eliminating the need to measure CD activity directly. 36
Aside from HSV1-TK, other reporter genes can be used in nuclear imaging studies, including human somatostation receptor subtype 2A (hSSTr2). 37 A key difference between HSV1-TK and hSSTr2 is that hSSTr2 is a receptor and therefore interacts with its radiolabeled ligand in a 1:1 ratio, as opposed to HSV1-TK's enzymatic activity, which allows a single enzyme to interact with many substrate molecules, thereby amplifying the signal. Using a single adenovirus construct that encoded both hSSTr2 and HSV1-TK, it was demonstrated that thymidine kinase was more efficient at trapping its cognate radiolabeled substrate compared with binding of ligand by hSSTr2. 38
A new reporter for use with gamma cameras is the sodium-iodide symporter (NIS), a transmembrane protein expressed in the thyroid whose function is to transport iodine into cells. 39 Unlike enzyme- or receptor-based reporters with organic or organometallic substrates, NIS produces signal by simple transport of radioiodine into cells and nonspecific retention of the tracer but is, as with CD, susceptible to tracer efflux. The other disadvantage of NIS as a reporter is that there are high background signals in the thyroid and stomach that may confound in vivo imaging studies. In spite of these drawbacks, however, NIS expression is able to be monitored by direct measurement of radioiodine net uptake with widely disseminated gamma cameras and without the requirement of a sophisticated radiopharmacy. The symporter was also recently incorporated into a human NIS-IRES-eGFP dual-reporter gene to combine nuclear and optical imaging modalities. 40
The idea of combining nuclear and optical imaging techniques has wide appeal because the two modalities have such complementary strengths. An early fusion construct to incorporate the dual modalities of microPET and bioluminescence was a fusion of SR39TK and RLuc, which indicated that the activity of the thymidine kinase protein may be more susceptible to fusion-dependent inhibition of activity than the luciferase protein. 11 Extension of the linker to 20 amino acids from either 18 or 10 amino acids did increase thymidine kinase activity but only to a maximum of 45% of the unfused SR39TK-positive control. A significant improvement relative to control was made by truncating the SR39TK by deletion of the first 135 base pairs to create tTK. A fusion protein consisting of humanized Renilla luciferase (hRLuc), mRFP, and tTK demonstrated enzyme activity equivalent to over 100% of unfused tTK activity but did not fully recapitulate the activity of unfused hRLuc. 12 Another triple-fusion reporter composed of wild-type HSV1-TK, GFP, and FLuc demonstrated greater signal in all three modalities compared with any of the proteins in a dual fusion, although corresponding differences in expression were not resolved by Western blot and comparisons were not made to unfused proteins. 13 The relative merits of the various fusion reporters remain difficult to compare directly, however, given the differences in the strengths of their promoters.
Given that attempts to fuse in-frame three reporter proteins seem to compromise the activity of at least one enzyme, with HSV1-TK being particularly sensitive, and that fluorescence images cannot be quantitatively compared across modalities, we placed MGFP downstream of an IRES as opposed to fused directly onto the C-terminus of mNLS-SR39TK. Indeed, evidence suggests that several key amino acids at the C-terminus of HSV1-TK are required to maintain enzymatic activity, 41 and this should be considered when designing a fusion reporter. 11
The utility of our triple reporter lies in its ability to allow investigators to take advantage of the particular strengths of each imaging modality without being limited by each modality's inherent weaknesses. Importantly, the intervening linker and free C-terminus of thymidine kinase in the FLuc-mNLS-SR39TK fusion component of the reporter maintain the specific activities of each enzyme at the comparably high levels associated with the unfused engineered proteins. The mutation introduced into the N-terminus NLS of SR39TK, the coding region containing cryptic transcriptional initiating sequences in gonadal tissue, is also predicted to obviate the tendency of HSV1-TK to produce sterile males when engineered into transgenic reporter mice.42,43 Although microPET offers true tomography and quantitative analysis of particular regions, organs, or tumors, execution of the studies is time-consuming and expensive.
Bioluminescence imaging, on the other hand, offers a more inexpensive method to screen multiple animal subjects rapidly to obtain semiquantitative data with an improved signal to noise ratio, as seen in a comparison of Figures 5 and 6. This improved signal to noise ratio is a function of several contributing factors to the background signal in microPET that do not exist in bioluminescence imaging. First, uncleared
Ideal uses for this reporter would be to optimize experimental setups, test unknown hypotheses, or screen a parametric high-throughput matrix using bioluminescence imaging as a primary readout and then select a subset of animal subjects for more intensive microPET studies. The MGFP component of the reporter additionally allows improved flow cytometry analysis, confocal fluorescence microscopy, and subcellular localization, as well as in vivo and ex vivo fluorescence analysis of selected tissues, organs, or animals. The ability to readily transition between imaging modalities may render this second-generation triple reporter construct valuable to investigators both within and outside the molecular imaging community.
Footnotes
Acknowledgments
We thank Margaret Morris, Lori Strong, and Jerrell Rutlin for microPET technical assistance and colleagues of the National Cancer Institute Small Animal Imaging Resource Program (R24 CA83060).
