Abstract
Our group has developed a new molecular tool based on the use of a regioselectively addressable, functionalized template (RAFT) scaffold, where four cyclic (Arg-Gly-Asp) (cRGD) peptide motifs were grafted. The aim of this study was to determine whether RAFT-c(-RGDfK-)4 combined with optical imaging could allow noninvasive detection of deep ovarian metastases. Human ovarian adenocarcinoma IGROV1 cells expressing low levels of integrin αvβ3 (the main receptor for the cRGD peptide) were used for in vitro and in vivo assays in combination with Cy5-labeled RAFT-c(-RGDfK-)4, cRGD, or RAFT-c(-RβADfK-)4. In vivo fluorescence imaging was performed on subcutaneous (SC) tumors and intraperitoneal IGROV1 metastases in nude mice. The accumulation of RGD-Cy5 conjugates in cultured cells or in tumor tissues was examined using confocal laser scanning microscopy. RAFT-c(-RGDfK-)4 exhibited stronger staining in vitro, enhanced tumor-to-background ratio for SC tumors, and allowed early detection of 1- to 5-mm large intraabdominal nodules using noninvasive optical imaging. Histological study revealed that RAFT-c(-RGDfK-)4 accumulated into tumor neovasculature but also into tumor cells. Our data demonstrate that a Cy5-labeled RAFT-c(-RGDfK-)4 is an efficient optical probe for early and noninvasive tumor detection.
Introduction
Recent developments in molecular imaging have shown great potential for early, noninvasive, and real-time detection of deep tumors in small animals [1,2]. The tracers currently used can be radionuclides, paramagnetic agents, or fluorescent agents. The choice of tracer depends mainly on the type of application and the expected resolution and sensitivity, but other parameters, such as cost, required expertise, and risk of exposure, must also be considered. In any case, these tracers have to be attached to a specific ligand, which provides affinity and tissue specificity as well as the biological and pharmacodynamic properties. For these purposes, highly specific Arg-Gly-Asp (RGD) peptides were selected and widely used for targeting angiogenesis and metastasis [3,4]. Since these original studies were performed using SPECT or PET imaging, numerous applications of the RGD peptides were reported as summarized by Kwon et al. [5]. Altogether, these studies emphasized that improving the biophysical and pharmacodynamic properties of RGD-containing peptides greatly augments their performance as imaging probes.
Multivalent presentation of ligands significantly improves the targeting [6]. Thus, multivalent ligands containing polymers have been described, using a backbone composed, for example, of dendrimers [7], antibodies [8], polylysine [9], or nanoparticles [10]. Although highly efficient, these molecules have some drawbacks. First, their chemical formulation is not precisely defined, and thus the number of ligand motifs being added is random and cannot be controlled. Second, the conformation of these molecules is not constrained. It is thus impossible to separate spatially the different biological functions presented by a single molecule nor is it possible to know its exact structure. To overcome these problems, we generated a molecular platform called regioselectively addressable functionalized template (RAFT) [11,12]. RAFT is a cyclic decapeptide forming a ring with two faces. On the upper face, four copies of the c(RGDfK) peptide were grafted for recognition of the αvβ3 integrin receptor overexpressed on the tumor vasculature [13,14] and on the surface of tumor cells [15–18]. The bottom face of the RAFT can be used to link one or two biologically active drugs (plasmids, oligonucleotides, peptides, tags, or chemicals) or contrasting agents for in vivo molecular imaging or a combination of both. The required chemistry being regio- and chemoselective, the synthesis and purification of the final molecules are perfectly controlled even at gram scale. In addition, the RAFT architecture allows a spatial separation between the targeting and “drug-delivery” domains. Moreover, RAFT is also interesting because it allows a presentation of four RGD motifs on an area of ± 0.7 nm2 [19], which represents a very high density of ligands on such a small surface.
The aim of the present study was to compare the tumor-targeting specificity of Cy5-labeled RAFT-c(-RGDfK-)4 ligand to that of its monomeric analog, Cy5-cRGD, using both subcutaneously and intraperitoneally implanted αvβ3-positive tumor models by optical imaging.
Materials and Methods
RGD Peptide Synthesis and Labeling
RAFT is a cyclic decapeptide [c(-Lys(Boc)-Lys(Alloc)-Lys(Boc)-Pro-Gly-Lys(Boc)-Lys(Alloc)-Lys(Boc)-Pro-Gly-)] with up to six lysine residues (Figure 1A). Protection of the lysine in position 1, 3, 6, or 8 and of the two in positions 2 and 7 results in RAFT molecules having two orthogonally addressable domains pointing on either side of the cyclopeptide backbone. On the upper face, four copies of the cyclo (RGD-D-Phe-Lys) [c(RGDfK)] peptide were grafted for recognition of the integrin. A negative control molecule [(Cy5)RAFT(c[-RβADfK-])4] was also synthesized. Because of the presence of a βA residue in place of the G, this peptide does not recognize the integrin. The bottom face can be used to link one or two drugs (plasmids, oligonucleotides, peptides, tags, chemicals) and/or contrasting agents for molecular imaging. The four c(RGDfK) motifs were linked on the RAFT using an oxime bond (R1-O-N=C-R2). This reduces the risk that one domain sterically affects the conformation and accessibility of the other as is observed when cRGD is linked directly to another function. Cy5 N-hydroxysuccinimide (NHS) ester (Amersham Biosciences, Uppsala, Sweden) was added as described previously [11,12].
Cell Lines and Culture Conditions
HEK293(β3) and HEK293(β1) cells, stable transfectants of human β3 and β1 subunits, respectively, from the human embryonic kidney cell line (kindly provided by J.-F. Gourvest, Aventis, France) were cultured in DMEM enriched with 4.5 g.L−1 glucose and supplemented with 1% glutamine, 10% fetal bovine serum (FBS), 50 U/mL penicillin, 50 μg/mL streptomycin, and 700 μg/mL Geneticin (G418 sulfate, Gibco, Paisley, UK). Human ovarian adenocarcinoma IGROV1 cells were kindly provided by Dr. Laurent Poulain (Centre de Lutte Contre le Cancer François Baclesse, Caen, France) and maintained in RPMI 1640 supplemented with 10% FBS and antibiotics mentioned above. All cell lines were cultured in 75-cm2 flasks with 15 mL of growth medium and kept at 37°C in a humidified 95% air/5% CO2 atmosphere.
Stable Transfection of IGROV1 Cells with Luciferase Expression Vector
IGROV1 cells were cotransfected with a modified firefly luciferase reporter gene, pGL3-Luc plasmid (Promega, Charbonnière, France), and an ampicillin resistance gene, pcDNA, using jetPEI™ (PolyPlus-Transfection, Illkirch, France) according to the manufacturer's recommendations. Selection of stable transfectants was performed by adding G418 to the culture medium at a final concentration of 800 μg/mL. The expression of luciferase was confirmed by using the Luciferase assay system (Promega). Colonies of cells expressing luciferase, referred to as IGROV1/pGL3, were grown in the culture medium supplemented with G418 (800 μg/mL).

Animals and Tumor Models
Animal procedures were in agreement with the EEC guidelines. Female athymic Swiss nude mice, purchased from IFFA-Credo (Marcy l'Etoile, France) at 6–8 weeks of age, were used and maintained under specific pathogen-free conditions. Mice were injected subcutaneously (SC) in the right flank with 5 × 106 IGROV1 cells resuspended in 200 μL of PBS (the number of animals is indicated in the figure legends). When the tumors reached 6–8 mm in diameter (14-21 days after implantation), the mice were used for imaging experiments. They usually received 10 nmol of the indicated peptides intravenously (IV), unless otherwise indicated in the legend.
For fluorescence reflectance imaging (FRI) of peritoneal carcinomatosis, mice were injected intraperitoneally (IP) with 2 × 107 IGROV1/pGL3 cells resuspended in 200 μL of PBS. After 3 weeks, the small nodules disseminated in the abdomen cavity were visualized using bioluminescence imaging (BLI). This IGROV1/pGL3 subclone of the IGROV1 cell line stably expressing luciferase produced routinely one to three nodules per mouse with diameters ranging between 1 and 5 mm at time of sacrifice. Three groups of mice (
Analysis of Integrin αvβ3 Expression by Flow Cytometry
Adherent cells were harvested by trypsin/EDTA and washed with cold PBS. Cell pellets (106 cells) were incubated with R-phycoerythrin-conjugated 23C6 monoclonal antibody specific to the human αvβ3 integrin only (MAb 23C6-RPE; Becton Dickinson Biosciences, San Jose, CA), diluted 1:100 in 1% BSA in PBS containing 1 mM CaCl2 and 1 mM MgCl2 (PBS/Ca2+/Mg2+) or the same volume of PBS/Ca2+/Mg2+ with 1% BSA as negative control, on ice in the dark for 30 min. After washing, the labeled cells were resuspended in cold PBS/Ca2+/Mg2+ with 1% BSA and analyzed with a FACScan system (Becton Dickinson, France). The results are reported as phycoerythrin fluorescence (FL2) histogram counts.
In Vitro Binding Studies
Cells were seeded on sterilized 18-mm-diameter glass coverslips in 12-well plates (3 × 105 cells per well), and incubated overnight at 37°C. Afterwards, the cells were washed with PBS and incubated at 37°C in the presence of Cy5-labeled peptide [RAFT-c(-RGDfK-)4, cRGD, or RAFT-c(-RβADfK-)4] at a final concentration of 0.1 or 0.5 μM for 30 min. They were then washed with PBS and fixed with 2% paraformaldehyde at room temperature for 10 min. The nuclei were stained with 5 μM Hoechst33342, and the coverslips were inverted onto glass slides using Moewiol (Calbiochem, San Diego, CA) as a mounting medium. The slides were observed by confocal laser scanning microscopy (CLSM; LSM510, Zeiss, France).
Immunohistochemistry
Frozen tumor sections (7-10 μm thickness) were stained with a mouse anti-human integrin αvβ3 monoclonal antibody, LM609 (1:100; Chemicon, Temecula, CA), using avidin-biotin immunoperoxidase technique.
In Vivo Optical Imaging
A Hamamatsu imaging system (Hamamatsu Photonics, Hamamatsu City, Japan) was used, which allows detection of both fluorescent and bioluminescent signal as described previously [12]. Mice were anesthetized with warmed isoflurane in order to precisely control the body temperature (variation of less than 1°C) (Minerve, Esternay, France) and illuminated with a monochromatic 633-nm light (50 μW cm−2). The reemitted fluorescence was filtered by using a colored glass filter RG 665 (optical density >5 at the excitation wavelength 633 nm) and collected with a 16-bit cooled charge-coupled device (CCD) camera ORCAII-BT-51 (Hamamatsu). This highly sensitive, very low noise camera can be cooled to −80°C. For FRI, exposure time typically ranged from 20 to 100 msec, which provided a dynamic range of up to 65,535 gray levels. Unless mentioned, the time of 100 msec was used for all conditions. Image processing, including pseudocoloring and superimposing images, measurement of the fluorescence intensity (number of photons per pixel) for each region of interest (ROI), were performed using the Wasabi software (Hamamatsu). For determination of tumor contrast, the mean fluorescence intensity of the tumor area (
In Vivo Distribution of Cy5-labeled RGD Peptides in Tumor-bearing Mice
Tumor samples were frozen in liquid nitrogen and stored at −80°C. Sections of 20- to 30-μm thickness were fixed with 2% paraformaldehyde at room temperature for 10 min. Afterwards, the nuclei were stained with 5 μM Hoechst33342, the coverslips were mounted using Moewiol and kept at 4°C in the dark until observation using CLSM.
Statistical Analysis
All data are given as mean ±
Results
Expression of αvβ3 Integrin in IGROV1 Cells
When compared to the strong αvβ3 expression in HEK293(β3) (Figure 1B, B1), IGROV1 cells were found to express low to moderate levels of the integrin (Figure 1B, B3), whereas HEK293(β1) cells were found negative as expected (Figure 1B, B2). We then verified that IGROV1 tumors remained αvβ3-positive after inoculation and proliferation in nude mice. Immunoperoxidase labeling of tumor sections (Figure 1B, B4) showed positive αvβ3 staining in the periphery of tumor nodules and especially on the basal membrane of tumor cells in contact with the extracellular matrix.
In Vitro Binding Studies
Figure 2 compares fluorescence images of IGROV1 cells incubated with Cy5-cRGD, Cy5-RAFT-c(-RGDfK-)4, or Cy5-RAFT-c(-RβADfK-)4 at a final concentration of 0.5 μM during 30 min at 37°C. Negligible signal from Cy5 was detected when cells were incubated with RAFT-c(-RβADfK-)4.
In contrast, positive Cy5 signals were observed in both RAFT-c(-RGDfK-)4- and cRGD-treated cells, but RAFT-c(-RGDfK-)4-treated cells were more strongly labeled. When Cy5-RAFT-c(-RGDfK-)4 was used at a lower concentration (0.1 μM), it still produced a stronger signal (data not shown) than its monomeric analog.
Dose Escalation and Administration Routes of Cy5-labeled RAFT-c(-RGDfK-)4 for In Vivo Imaging
Figure 3A shows typical FRI images of a Swiss nude mouse bearing SC IGROV1 tumor at different time points after IV injection of 10 nmol Cy5-RAFT-c(-RGDfK-)4. The probe accumulates strongly in the tumor within 5 min, but the whole body is also strongly fluorescent due to the presence of unbound, circulating molecules. Two hours postinjection, the tumor/skin ratio augments significantly, reaches its maximum between 3 and 6 hr, slowly decreases at 24 hr, and still allows visualizing of the tumor at 48 hr. Strong signal is also observed in the kidneys and bladder (image not shown) during the first 6 hr, reflecting the prominent and fast renal excretion of the RAFT-c(-RGDfK-)4 molecule.
To determine whether RAFT-c(-RGDfK-)4 accumulates in the tumor in a dose-dependent manner, 0.2, 2, or 10 nmol of Cy5-RAFT-c(-RGDfK-)4 per mouse were injected IV. Figure 3B shows that indeed the tumor signal is dose dependent with 10 nmol producing the strongest signal as well as the highest contrast. Nonetheless, injection of 2 nmol Cy5-RAFT-c(-RGDfK-)4 was sufficient for visualizing the tumors with an acceptable contrast of more than 2 obtained 3 hr postinjection.
We then investigated whether tumor labeling was dependent on the route of administration. Ten nanomoles Cy5-RAFT-c(-RGDfK-)4 was administered IP or IV, and the kinetics of accumulation in the tumors, contrast, and intensity levels were measured as presented in Figure 3C. No significant difference was observed when the peptides were injected IP, indicating that Cy5-RAFT-c(-RGDfK-)4 is still small enough to be efficiently delivered to the remote tumors after IP injection.


RAFT-c(-RGDfK-)4 versus cRGD
In order to confirm whether the grafting of multiple cRGD motifs to RAFT could enhance the tumor-to-background ratio, time course analysis of fluorescence imaging was performed using IGROV1 SC tumor-bearing mice after IV injection of 10 nmol Cy5-labeled cRGD, RAFT-c(-RGDfK-)4, and the control RAFT-c(-RβADfK-)4. Administration of cRGD and RAFT-c(-RGDfK-)4, but not RAFT-c(-RβADfK-)4, produced strong signals in the tumors (Figure 4A), but Cy5-cRGD injection was also associated with an important diffuse signal in the skin. Altogether, the contrast (tumor-to-skin ratio) was found to be statistically better with RAFT-c(-RGDfK-)4 (Figure 4A). At 6 hr after IV injection, Cy5-RAFT-c(-RGDfK-)4 provided the expected contrast (3.24 ± 0.772), significantly higher than those of the control Cy5-RAFT-c(-RβADfK-)4 (1.44 ± 0.299;
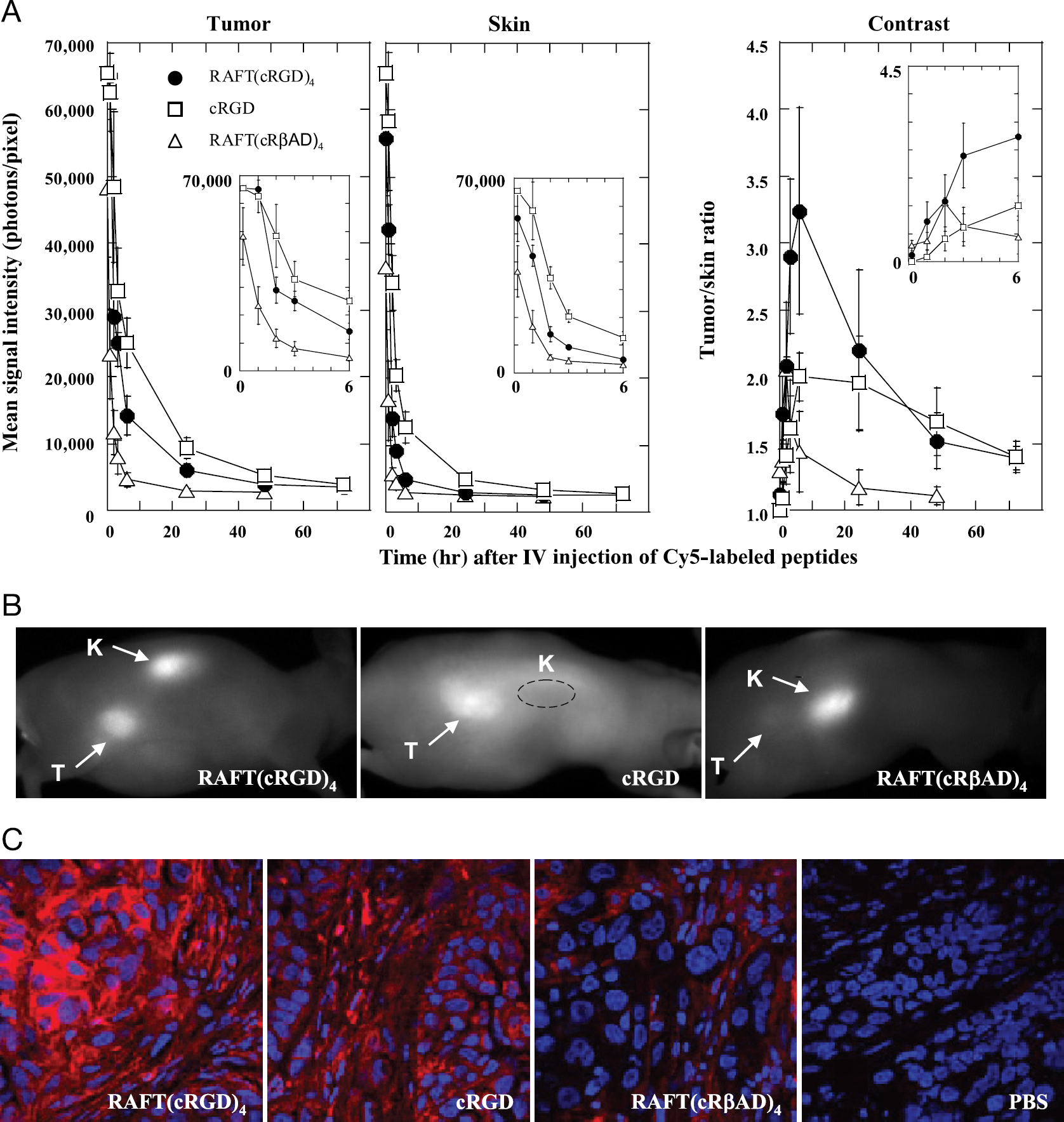
Cy5-RAFT-c(-RGDfK-)4 Allows Noninvasive Detection of Abdominal Metastases
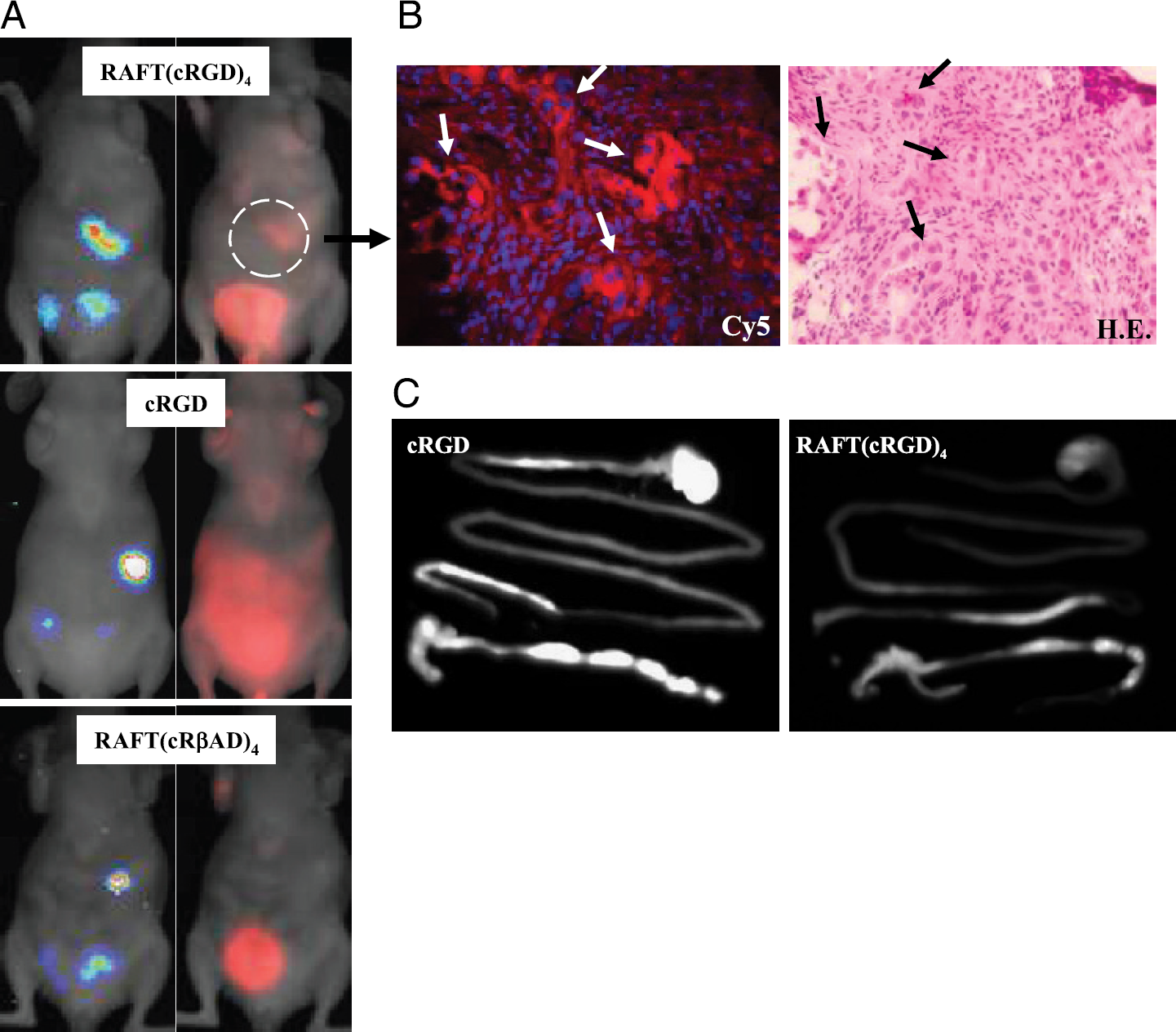
We finally tried to determine whether Cy5-RAFT-c(-RGDfK-)4 or Cy5-cRGD could allow a noninvasive detection of deep, small IGROV1/pGL3 peritoneal tumors. As expected, luciferase-positive nodules located in the abdominal region were identified in each mouse. In three out of four mice of the Cy5-RAFT-c(-RGDfK-)4-treated group, the FRI signal colocalized with luciferase-positive nodules (representative images shown in Figure 5A, top). In the fourth mouse, the FRI signal was too weak, certainly because of the very small size of the nodule as shown by the very low BLI intensity. Note that in all four mice, the diameter of the tumor nodules ranged between 1 and 5 mm and were hardly visible under normal light after the abdomen was opened. We thus performed FRI-guided biopsy, and frozen tissue sections were observed by CLSM. As shown in Figure 5B, Cy5-labeled cells were also presenting a typical pattern of malignant cells after hematoxylin-eosin staining (arrows). These data clearly reflect the good sensitivity of the Cy5-RAFT-c(-RGDfK-)4 staining and confirm that of BLI. In contrast, no tumor-specific FRI signal showed up in the four mice injected with Cy5-cRGD (Figure 5A, middle). This was mainly due to the strong background fluorescence in the intestine that eventually masked the specific Cy5-cRGD signal and prevented identification of the positive nodules. Indeed, normalized images of the extracted tissues (stomachs and intestines) from tumor-free mice (Figure 5C) clearly showed that the gastrointestinal uptake was higher for Cy5-cRGD than its multivalent analog. It is important to note that when a similar LUT was used, no significant autofluorescence from mouse diet in the intestine of PBS-injected mice was visible (data not shown). Finally, as expected, none of the four Cy5-RAFT-c(-RβADfK-)4-injected mice showed detectable signal in BLI positive tumors (representative images shown in Figure 5A, bottom).
Discussion
Recently, Wang et al. reported their successful application of near-infrared (NIR) optical imaging with cRGD-Cy5.5 conjugate for detecting αvβ3 integrin expression in SC human Kaposi's sarcoma and melanoma xenografts in nude mice [20]. A subsequent report from Chen et al. [21], who used NIR optical imaging with cRGD-Cy5.5, shows similar results in mouse SC brain tumor xenografts. Nonetheless, they did not succeed in visualizing deep tumors noninvasively. In addition, nuclear imaging with radionuclide-labeled cRGD showed the unfavorable hepatobiliary excretion of cRGD that may result in high, nonspecific accumulation of cRGD in the liver and intestine, thus limiting the detection of tumor lesions located in the abdomen [22]. Modified F18-galacto-RGD was then shown to present higher metabolic stability and renal elimination and was subsequently used for PET imaging of patients with cancer [23].
As shown by different groups, polymers presenting several cRGD peptides have improved properties when compared to cRGD monomers [24–26], and multimeric structures with up to eight RGD units linked via different spacers have been described [27–29]. Radionuclide-labeled cRGD dimers showed increased binding affinity in vitro and improved tumor accumulation and tumor-to-background contrast in tumor-bearing mice by PET imaging. Moreover, the hepatic and intestinal uptake of dimeric RGD was significantly lower than that of monomeric RGD peptide [28,30]. Finally, Wu et al. demonstrated the advantage of presenting four copies of the RGD motif to improve the integrin binding and tumor uptake using PET in a glioma model [26].
Our molecular platform called RAFT allowed grafting four cRGD motifs on one side of the platform and a marker dye, indocyanine Cy5, on the opposite [11,12]. We used this Cy5-RAFT-c(-RGDfK-)4 and the IGROV1 human ovarian adenocarcinoma cell line expressing a relatively low level of αvβ3 integrin to study the advantage of the multivalent cRGD molecule in vitro and in vivo. In vitro, Cy5-RAFT-c(-RGDfK-)4 accumulated more strongly in IGROV1 cells than the cognate cRGD. In parallel, Cy5-RAFT-c(-RβADfK-)4-treated cells remained completely negative, confirming the good specificity of the RGD-mediated interaction.
It has been suggested that multimeric cRGD peptides could induce integrin clustering and integrin-dependent endocytosis [31]. Furthermore, simultaneous binding of multiple RGD motifs presented by a single platform to one integrin or binding of multiple integrins to a single multimeric RGD peptide could lead to cooperative, entropically enhanced affinity and steric stabilization of the ligand-receptor interaction [24,25]. Our results are in agreement with these observations and support the idea that multimeric presentation of the cRGD motifs may enhance the integrin-dependent internalization.
We then compared the tumor accumulation and targeting specificity of the different molecules after IV injection into nude mice bearing SC IGROV1 tumors. Although cRGD produced a stronger fluorescence signal in the tumor than RAFT-c(-RGDfK-)4, its level in skin was also higher, resulting in a lower tumor/skin ratio. Besides the expected specific RGD-mediated binding, other factors such as molecule perfusion and increased vascular permeability also participate in the accumulation of a probe in a tumor. This is especially important for low molecular weight compounds like Cy5-cRGD [three times smaller than Cy5-RAFT-c(-RGDfK-)4]. In addition, it is known that endocytosis of cRGD may follow a nonspecific pathway, whereas dimeric and even multimeric cRGD may predominantly induce integrin-dependent endocytosis [31]. Hence, the concentration of Cy5-RAFT-c(-RGDfK-)4 molecules in the tumor may reflect more accurately the integrin level. Once more, the specificity of this interaction was demonstrated when Cy5-RAFT-c(-RβADfK-)4 was injected. Indeed, despite a weak signal related to its passive accumulation due to the increased vascular permeability, this negative control molecule did not bind to the tumor. It was previously reported that injection of Cy5.5-c(KRGDf) at a dose higher than 1.5 nmol/mouse was not associated with increased uptake in tumor tissues using a model of Karposi's sarcoma [5] or glioblastoma [21]. Our results obtained using the monomeric or multimeric cRGD molecules did not sustain these results. Indeed, injecting up to 10 nmol/mouse provided increasing signals in the tumors and improved contrasts (signal-to-noise ratio). This discrepancy may be due to the use of different tumor models and also to differences in the mode of acquisition. In our model, the time of exposure was always fixed at 100 msec, whereas Kwon et al. changed it according to the conditions, with exposure times of up to 3000 msec [5]. In the other study, the time of exposure was set up to 120 sec. In these conditions, the background level becomes a real problem, whereas in our case the autofluorescence was negligible.
Kidneys were strongly positive after IV injection of Cy5-RAFT-c(-RGDfK-)4. This indicated its elevated renal uptake. Cy5-cRGD-treated mice did not present such a strong staining of the kidneys, but, both molecules were rapidly evacuated with urine. Compared to Cy5-RAFT-c(-RGDfK-)4, Cy5-cRGD was more strongly absorbed by the intestine, resulting in a stronger diffuse signal in the abdomen than the tetramer. This was further confirmed on dissected organs. A stronger fluorescent signal was always observed in the intestine of Cy5-cRGD-treated animals even after removing the feces by extensive washing. This suggests that Cy5-cRGD shows both hepatobililary and renal excretion pathways, but Cy5-RAFT-c(-RGDfK-)4 is mainly excreted by kidney.
Our optical imaging system can be used in a dual mode for detection of fluorescence and bioluminescence without moving the mice. Appearance of abdominal metastases obtained by IP injection of IGROV1/pGL3 cells, stably expressing luciferase, was monitored using BLI. The high sensitivity of BLI [32] allowed us to identify the presence of deep and small metastases undetectable by palpation. FRI performed 3 hr after IV injection of the Cy5 peptides demonstrated that in three out of four mice, the small metastases could be detected noninvasively solely when using Cy5-RAFT-c(-RGDfK-)4. None of the four Cy5-cRGD or Cy5-RAFT-c(-RβADfK-)4-treated mice presented a positive colocalization of the Cy5 and BLI signals. This emphasizes that Cy5-RAFT-c(-RGDfK-)4 vector is a more potent probe for imaging of cancer due to its lower uptake by the hepatobiliary system and decreased nonspecific binding to the intestine and the skin. Detection of deep metastasis still represents a major challenge for 2-D FRI. In addition, quantification is hazardous using 2-D FRI because the amount of signal will be dramatically affected by the depth of the fluorochrome and background absorption heterogeneity [33]. It is thus important to develop fluorescence-mediated tomography, as demonstrated by Ntziachristos et al. [34].
Confocal laser microscopy of tumors showed a strong labeling of tumor vasculature and of tumor cells by Cy5-RAFT-c(-RGDfK-)4. In tumor stroma, we found the expected signal in endothelia. In addition, the cytoplasm of microscopic nests of αvβ3-positive tumor cells were also strongly stained by Cy5-RAFT-c(-RGDfK-)4 fluorescence. This clearly supported the advantage of using RAFT-c(-RGDfK-)4 to target tumor vasculature and αvβ3-positive tumor cells. This is a very important asset for tumor imaging and future targeted therapy.
In summary, our in vitro and in vivo studies show that the multimeric RAFT-c(-RGDfK-)4 increases the selectivity of cRGD because Cy5-RAFT-c(-RGDfK-)4 targets more specifically SC tumors as well as abdominal metastases. This may mainly be due to its lower hepatobiliary excretion and decreased nonspecific binding to the intestine and skin. In addition, besides its improved targeting properties, the RAFT molecule was also designed to allow an easy and chemoselective covalent addition of one or two biologically active compounds on its second functional face. Altogether this makes RAFT-c(-RGDfK-)4 a very promising vector for future therapeutic and clinical imaging applications.
Footnotes
Abbreviations
Acknowledgments
This work was supported by the Institut National de la Santé et de la Recherche Médicale (INSERM), the Centre National de la Recherche Scientifique (CNRS), the Région Rhône Alpes, the program Interdisciplinaire 2001–2004 “Imagerie du Petit Animal,” the Cancéropole National and the Institut Universitaire de France (IUF), the Ligue Nationale contre le Cancer (EG), and Association pour la Recherche sur le Cancer (ARC). We are grateful to Dr. Laurent Poulain (Centre de Lutte Contre le Cancer Francois Baclesse, Caen, France) for giving us IGROV1 cells and to Dr. J.-F. Gourvest (Aventis, France) for HEK293(β3) and HEK293(β1) cells. We are also very grateful to Philippe Rizo (CEA Leti) for offering the FRI imaging system. We thank Corine Tenaud and Dominique Desplanques for technical assistance and Dr. Alexeï Grichine (IFR73, Institut Albert Bonniot, France) for the guidance in using confocal laser scanning microscopy.
