Abstract
Managing estrogen receptor-positive breast cancer in young women (<40 years) requires a multidisciplinary/personalized approach, covering both clinical and psychosocial aspects. Five years of tamoxifen has been the standard adjuvant endocrine therapy for many years. Recent data from the adjuvant randomized trials TEXT–SOFT show that the aromatase inhibitor exemestane plus ovarian suppression significantly reduces recurrences as compared with tamoxifen plus ovarian suppression. The ATLAS and aTToM trials represent the first evidence of a beneficial effect of extended endocrine therapy with tamoxifen in premenopausal women. Outside of a clinical trial, no data support neoadjuvant endocrine therapy in young women. In the metastatic setting, tamoxifen or aromatase inhibitors, both with ovarian suppression/ablation, should be the preferred choice, unless rapid tumor shrinkage is needed. No data are available with fulvestrant in young patients.
Keywords
The current definition of ‘young women’ as women under the age of 40 is arbitrary, as biology and the endocrine milieu are a continuum but this group of patients has specific issues such as fertility preservation, induction of treatment-related early menopause, that deserve a different approach and management from slightly older premenopausal and perimenopausal women.
Approximately 11,000 cases of invasive breast cancer (BC) were expected among young US women (<40 years) in 2013, which represent about 5% of all new diagnoses [1]. The same proportion is seen in European countries [2].
Whether young age is associated with unique cancer biology remains controversial. Several studies report an inverse correlation between estrogen receptor (ER) status and age. In a large cohort of Danish women (26,944) diagnosed with primary BC from 1989 to 2004, the proportion of ER+ patients increased with age (from 51.4% in patients <35 years to 67.6% in women 40–44 years) [3]. ER positivity by immunohistochemistry was also lower in 200 patients <45 years from five North American public data sets, as compared with their older counterparts (211 patients ≥65 years; positive vs negative; 71% vs 80%, respectively; p = 0.027) [4]. In a cohort of 399 women <40 years diagnosed at Dana-Farber/Harvard Cancer Center, 68% had ER+ disease [5]. In addition, Anders and colleagues initially reported an age-related expression of key BC-associated genes [4] but when they corrected for subtype and other significant clinicopathologic features (i.e., ER status and histologic grade) no gene differences were retained between age-defined groups (<45 years and ≥65 years) [6]. In a series of 873 patients aged <45 years from 20 publically available data sets, the proliferation gene signatures showed no significant interaction with age in ER+/HER2-tumors [7].
The choice of systemic treatment for invasive BC should not be based on patient's age but only on the biological characteristics of the tumor and patient's comorbidities [8].
Adjuvant endocrine therapy
The most effective adjuvant endocrine therapy (ET) for premenopausal women with ER+ BC is still a matter of debate.
Tamoxifen for at least 5 years is considered standard of care [8–11]. The Early Breast Cancer Trialists' Collaborative Group metaanalyses repeatedly reported the benefits of adjuvant tamoxifen, in premenopausal and postmenopausal women with ER+ BC regardless of age, the use of chemotherapy and nodal status. In the 2011 overview [12], 5 years of tamoxifen compared with no ET were associated with a 15-year risk reduction of 39% in BC recurrence and of 30% in BC mortality. The sustained reduction in BC mortality well beyond year 10 is of particular interest in young women. Efficacy was evident also in disease only weakly ER+ positive and independent of progesterone receptor status. HER-2 overexpression has been associated with tamoxifen resistance [13]: data in younger women are limited, but in the presence of oophorectomy, the impact of adjuvant tamoxifen on outcome is comparable in patients with HER2-positive and HER2-negative tumors [14]. An association between CYP2D6 genotype and tamoxifen metabolism influencing antitumor activity was investigated in >20 published studies with highly inconsistent results [15]. At present, CYP2D6 pharmacogenetic-driven treatment decisions cannot be recommended outside clinical studies. Genetic polymorphisms in premenopausal women will be specifically investigated in Tamoxifen and Exemestane Trial (TEXT) and Suppression of Ovarian Function Trial (SOFT) patients.
The duration of ET has not been adequately studied in young women: in the past, small trials did not suggest any benefit to extend tamoxifen beyond over five years [14,16]. Recent data from the ATLAS and aTTom studies showed that continuing tamoxifen to 10 years gives a further reduction in recurrence and mortality [17,18].
The ATLAS trial included 15.244 premenopausal and postmenopausal women [17] randomized to ten versus 5 years of adjuvant tamoxifen. With a median follow-up of 7.6 years, continuing tamoxifen reduced the risk of BC recurrence, compared with a 5-year treatment course (18% vs 21%; RR: 0.84; 95% CI: 0.76–0.94). A persistent and more significant effect was found after year 10 (RR: 0.90; 95% CI: 0.79–1.02 during years 5–9 and 0.70; 95% CI: 0.62–0.90 during subsequent years). There was a 29% reduction in the risk of BC mortality after year 10 (RR: 0.71; 95% CI: 0.58–0.88). The effect was independent of age (10% of patients were premenopausal at study entry) and nodal status (41% of patients were node-positive at diagnosis): despite being only a small subgroup, the proportional risk reduction was the same in premenopausal patients as compared with postmenopausal patients (RR: 0.81 and 0.85, respectively). The study group had an increased risk for pulmonary embolism (RR: 1.87; 95% CI: 1.13–3.07, p = 0.01), and endometrial cancer (RR: 1.74; 95% CI: 1.30–2.34; p = 0.0002) with lower risk in premenopausal women, but no increase in the incidence of stroke (RR: 1.06; 95% CI: 0.83–1.36), and a decrease in the incidence of ischemic heart disease (RR: 0.76; 95% CI: 0.60–0.95; p = 0.02). The aTTom trial was conducted on 7000 women, most with unknown ER status, randomized to 10 versus 5 years of adjuvant tamoxifen: the reported findings confirm the ATLAS reduction in recurrence and death from BC [18]. Extended tamoxifen therapy is therefore an option that should be considered in premenopausal women at high risk for late relapse (i.e., pathologically involved nodes, larger tumors, high tumor grade) taking into account quality-of-life (QoL) issues.
The NCIC-CTG MA.17/BIG 1–97 trial reported a significant benefit in postmenopausal women with ER+ tumors with 5 years of letrozole after 5 years of tamoxifen [19]. The benefit, in terms of disease-free survival (DFS), was higher (hazard ratio [HR]: 0.25; 95% CI: 0.12–0.51) in premenopausal women at diagnosis who became definitively postmenopausal at the time of randomization after 5 years of adjuvant tamoxifen [20].
Extended ET with an aromatase inhibitor (AI) after tamoxifen has never been evaluated in premenopausal BC survivors. AIs alone are contraindicated in premenopausal women because the suppression of peripheral aromatase results in reduced feedback to the hypothalamus and consequent ovarian stimulation [21]. A Phase II single-arm trial in women who remained premenopausal after at least 4.5 years of adjuvant tamoxifen evaluated 2 years of ovarian function suppression (either the gonadotropin-releasing-hormone agonist (GnRHa) leuprolide 7.5 mg monthly or 22.5 mg every 3 months, or bilateral salpingo-oophorectomy [BSO]) [22]. After six to 8 weeks on ovarian function suppression (OFS), participants began the AI letrozole 2.5 mg orally daily for 2 years. Due to unsufficient accrual over 3.5 years the study closed after only 16 patients enrolled. Four patients stopped treatment before 1 year, owing to toxicity; five completed 2 years of protocol-directed therapy; and seven remained on treatment as of study closure. Hot flashes, vaginal dryness and joint/muscle pain were common. This study's poor accrual may suggest young women may not be highly motivated to pursue extended ET and future studies of this strategy may be challenging. Despite the different toxicity profile of extended tamoxifen, it will be important to monitor what proportion of young women will choose to continue ET beyond 5 years and their motivations.
Considering the efficacy shown in postmenopausal women [23], adjuvant AIs in combination with OFS have been first investigated in premenopausal patients with early BC in the Austrian Breast and Colorectal Cancer Study Group (ABCSG)-12 trial (Figure 1) which still shows, with a median follow-up of 94.4 months, that 3 years of adjuvant therapy with the AI anastrozole plus the GnRHa goserelin provide a comparable DFS (HR: 1.13; 95% CI: 0.88–1.45; p = 0.335), to that associated with tamoxifen plus goserelin [24]. The overall survival (OS) was not significantly worse with anastrozole than with tamoxifen (HR: 1.63; 95% CI: 1.05–1.45; p = 0.030) but after disease recurrence, women receiving tamoxifen were more likely to be switched to an AI than those in the anastrozole group (61% vs 41%, respectively) who were switched to second line, non-AIs, ET [24]. In overweight patients (BMI ≥25 kg/m2) treated with anastrozole the risks of recurrence and death were significantly higher (HR: 1.49; 95% CI: 0.93–2.38; p = 0.08 and HR: 3.03; 95% CI: 1.35–6.82; p = 0.004, respectively) than in patients treated with tamoxifen. The increased total body aromatization in the fat tissue may in fact negatively affect AIs efficacy, inducing incomplete suppression of estrogen production in peripheral body fat.
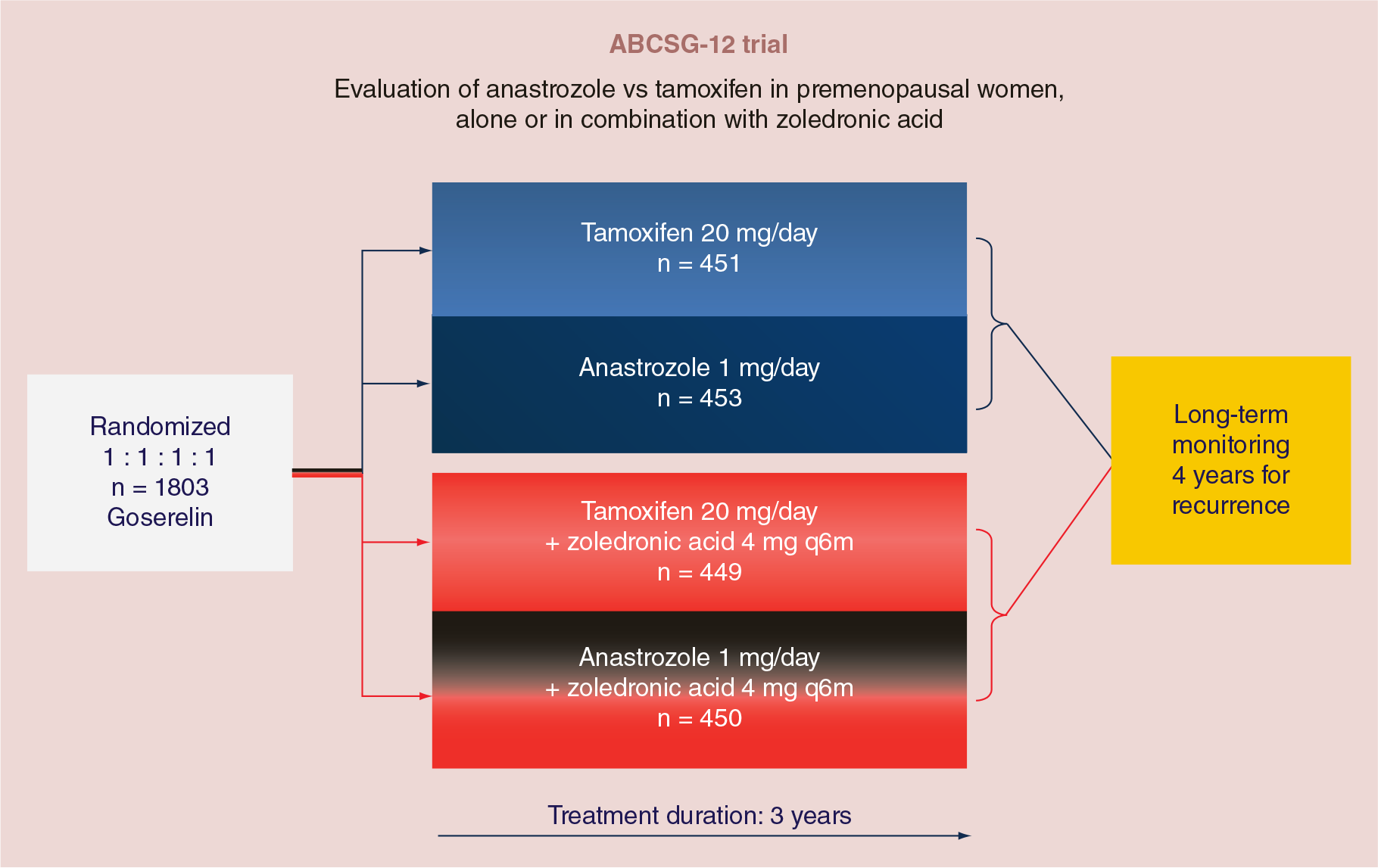
Austrian Breast and Colorectal Cancer Study Group-12 trial design.
The TEXT and SOFT trials were designed (Figure 2) to evaluate 5 years of therapy with the AI exemestane plus the GnRHa triptorelin versus 5 years of tamoxifen plus triptorelin [26]. In addition, the SOFT trial evaluated 5 years of tamoxifen plus triptorelin versus tamoxifen alone in women who were suitable candidates for treatment with adjuvant tamoxifen.
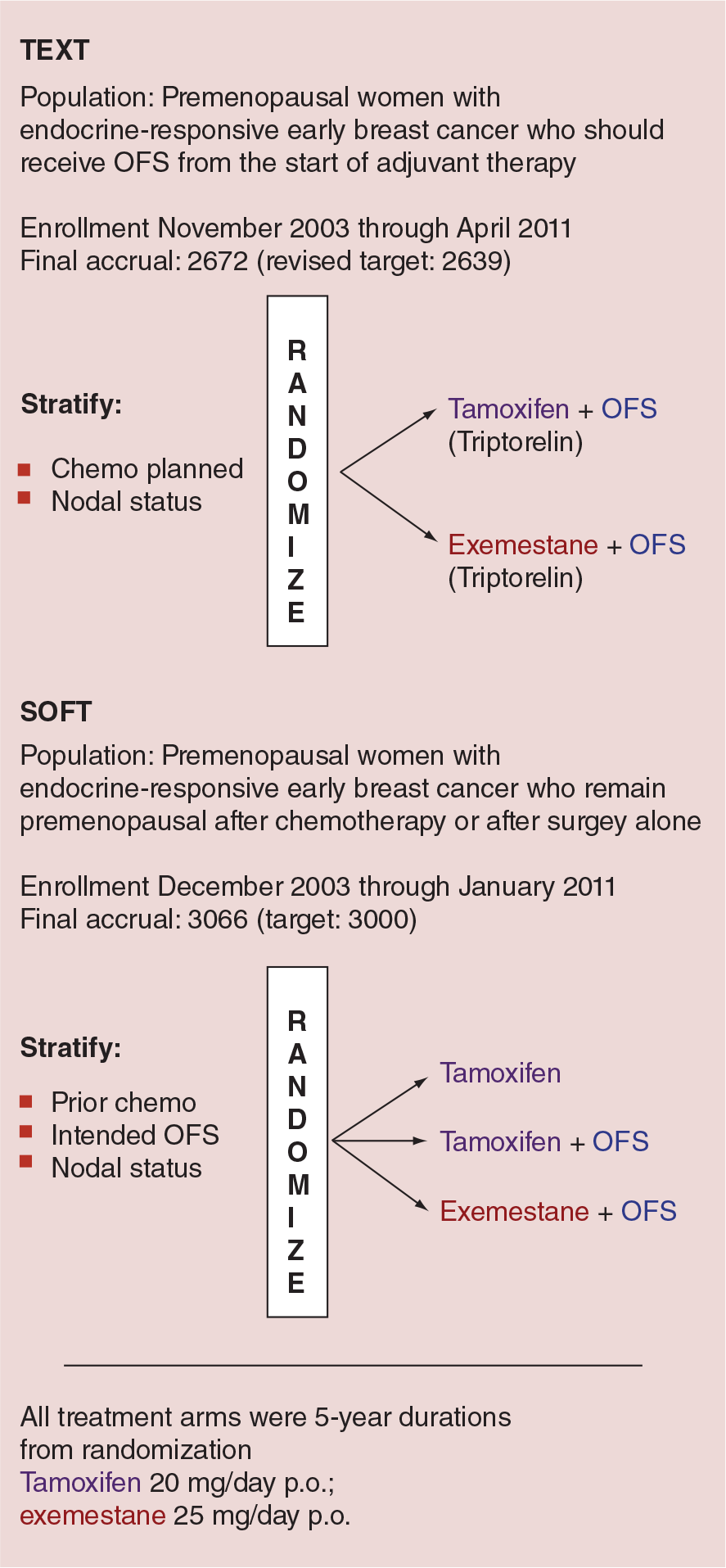
TEXT and SOFT design.
The planned primary combined analysis of TEXT and SOFT comparing adjuvant exemestane plus OFS with adjuvant tamoxifen plus OFS after a median follow-up of 68 months has shown a 5-year DFS of 91.1% among patients who received exemestane plus OFS and of 87.3% among those assigned to tamoxifen plus OFS (HR: 0.72; 95% CI: 0.60–0.85; p < 0.001), in other words a 28% relative reduction in the risk of disease recurrence, second invasive cancer or death. The difference in OS was not significant [27]. The analysis also showed a 34% relative reduction in the risk of BC recurrence. These results compare favorably with the results of randomized trials of adjuvant AIs versus tamoxifen in postmenopausal women [23].
The difference between the results of TEXT and SOFT and of ABCSG-12 trial may relate to greater statistical power in the combined analysis of TEXT and SOFT (three times the number of events of disease recurrence, second invasive cancer or death), to different treatment durations or to both. The shorter duration of the AI in the ABCSG-12 trial may have been insufficient, as compared with 3 years of tamoxifen, which is known to exert a carryover effect [12]. A difference in the AIs efficacy is unlikely, given the absence of a difference reported in postmenopausal women [28]. Of note, >90% of patients in both trials remained disease free without adjuvant chemotherapy, suggesting that combined adjuvant et al. one, in appropriately selected ER+ premenopausal women, can be effective. A word of caution should be raised in very young women, as only 10% of women were <35 years of age when randomized in the TEXT–SOFT trials [27].
The additional benefit of OFS/OA is still a matter of debate. The 2005 EBCTG overview demonstrated that OA (by surgery/irradiation) or OFS with a GnRHa significantly reduce the risk of both recurrence and BC mortality in women <50 years with ER+ or ER-unknown early BC [29]. A larger EBCTG metaanalysis looked only at trials with known ER status and GnRHas as method of OFS [30]. OFS proved to be beneficial whether used alone (recurrence risk reduction of 28%; p = 0.08), in addition to tamoxifen or chemotherapy (recurrence risk reduction of 13%; p = 0.02), and as an alternative to chemotherapy. The effects were greater in women <40 years in whom chemotherapy is less likely to induce permanent amenorrhea. The recently published results of the SOFT trial showed that, after a median follow-up of 67 months, the addition of OFS/OA to tamoxifen did not result in a significant benefit in terms of DFS in the overall study population (86.6% in the tamoxifen–OFS group vs 84.7% in the tamoxifen group; HR: 0.83; 95% CI: 0.66–1.04; p = 0.10). However, for women at higher risk of recurrence (i.e., <40 years at diagnosis, node positive, with tumors >2 cm and grade 3) who received adjuvant chemotherapy and remained premenopausal within 8 months after its completion, the addition of OFS was associated with an absolute improvement of 4.5% in the 5-year BC-free interval, benefit which increased to 11.2% in women <35 years at diagnosis (11.5% of the entire population) [31]. The optimal duration of GnRHas is also unknown, although most studies have utilized 2–3 years of monthly injections with 5 years of tamoxifen. Data comparing the efficacy of monthly and trimonthly formulations of GnRHa in women with BC are lacking. The effects of monthly goserelin and trimonthly leuprolide on estradiol levels did not differ significantly in a study comparing 79 early BC patients receiving GnRHa for at least 6 months [31] but further research in needed before trimonthly administration can be recommended in women <40 years of age.
OFS is not always successfully achieved with GnRHa as demonstrated in patients with advanced BC (ABC) by a pooled analysis of 193 premenopausal women treated with goserelin from 29 European trials (5% of patients) [32] and in 32 patients treated with anastrozole in combination with goserelin (1/3 of patients) [33]. Menses cessation does not guarantee complete estrogen suppression, estradiol assays are not standardized and their accuracy and interpretation can be problematic in presence of very low levels of estradiol [34]. Despite these limitations, estradiol levels should be checked on a regular basis (at least every 6 months), always in the same laboratory and preferably in a central reference laboratory.
The reversible OFS with GnRHa can be particularly attractive to younger women, especially to those who did not complete childbearing before diagnosis. Prophylactic BSO is known to significantly reduce the risk of both breast and ovarian cancer in mutation carriers, who are often identified at the time of BC diagnosis. BSO may therefore be considered when discussing adjuvant ET in this subgroup of young women [35].
Chemotherapy-induced amenorrhea (CIA), even if transient, is associated with improved treatment outcome: in addition to direct cytotoxicity, adjuvant chemotherapy has an indirect endocrine effect in ER+ BC through the induction of OFS. A recent metaanalysis in 15,916 premenopausal patients from 46 randomized trials showed a significant DFS advantage in ER+ patients (HR: 0.61; 95% CI: 0.52–0.72; p < 0.00001) [36]. The three-drug combination regimens of cyclophosphamide/anthracycline/taxanes caused the highest rate of chemotherapy-induced amenorrhea. The limited evidence available on the addition of trastuzumab to anthracyclines and/or taxanes seems not to increase the rate of permanent amenorrhea [37].
Neoadjuvant endocrine therapy
Neoadjuvant ET should not be proposed to young women outside clinical trials [8].
In the Phase III, randomized, double-blind, multicenter STAGE study, 204 patients were treated with 24 months' neoadjuvant therapy with the GnRHa goserelin plus anastrozole or tamoxifen. Anastrozole plus goserelin showed a significantly better overall response rate (ORR) than tamoxifen plus goserelin (70.4 vs 50.5%; 95% CI: 6.5–33.3; p = 0.004) [38], both among patients who had a baseline Ki-67 index ≥20% and among those who had a baseline Ki-67 index <20% [39]. The use of letrozole and a GnRHa as primary therapy was investigated in 32 premenopausal women with ER+ BC. The ORR was 50% (95% CI: 32–68%) and no patient progressed during treatment. Response was significantly associated with younger age (p < 0.05) and a longer duration of treatment (p < 0.05) [40]. These results compare favorably to the ORR achieved with neoadjuvant ET in postmenopausal women [41] and neoadjuvant chemotherapy in Luminal B patients [42] and this evidence is reinforced by the recent results with AIs plus OFS in the adjuvant setting [27] but definitive data are awaited before neoadjuvant ET can be routinely proposed to young women.
The activity of letrozole plus a GnRHa administered concurrently with preoperative chemotherapy (n = 119) was retrospectively compared with a nonrandomized unmatched control group of premenopausal women with locally advanced ER positive BC receiving preoperative chemotherapy, followed by tamoxifen and GnRHa after surgery (n = 95). The 5-year DFS was 78% vs 41% in the study and in the control group, respectively (adjusted HR: 0.46; 95% CI: 0.27–0.79; p = 0.0047) [43]. These retrospective data do not provide evidence-based indication to change clinical practice but suggest the role of chemoendocrine neoadjuvant therapy in this patients' population should be explored in randomized studies.
GnRHas have shown marked in vitro and in vivo antiproliferative activity in BC [44]. An International Breast Cancer Study Group (IBCSG) randomized Phase II trial (IBCSG 41–13 TREND) is evaluating the efficacy of the GnRHa degarelix versus triptorelin as neoadjuvant treatment in 50 premenopausal patients receiving letrozole.
Endocrine therapy in metastatic breast cancer
ET should be the preferred option for ER+ABC, unless there is evidence of endocrine resistance or need for rapid disease and/or symptom control [45,46].
In premenopausal women with ER+ABC, a variety of ETs have proven to be effective. A metaanalysis comparing GnRHa ± tamoxifen showed that the outcomes were significantly improved in patients who received the combination [47]. On the basis of these results, a GnRHa plus tamoxifen is currently recommended as the standard first-line therapy in ABC.
Based on the available evidence in postmenopausal women with recurrent BC [45], the role of AIs has also been studied in premenopausal women. In a prospective, single-arm, multicenter Phase II trial, 32 premenopausal patients were treated with goserelin and anastrozole achieving a clinic benefit rate of 71.9%, similar to that observed with AIs in postmenopausal women [48]. Other small Phase II studies confirm the efficacy of AIs as first- and second-line treatment in combination with a luteinizing hormone-releasing hormone (LH–RH) agonist [49–52]. The combination of goserelin and letrozole proved to be effective and well tolerated also in 35 very young (<35 years) women as first-line therapy with a clinic benefit rate of 65.7% [53].
In a Phase II parallel group study, at median follow-up of 27.4 months, there was no statistical difference in the median time to progression (TTP) between premenopausal patients receiving letrozole plus goserelin and postmenopausal patients treated with letrozole alone (9.5 months [95% CI: 6.4–12.1 months] vs 8.9 months [95% CI: 6.4–13.3 months]) [54].
Considering the demonstrated improved efficacy of AIs and OFS in the adjuvant setting [27], this combination can be reasonably proposed as first-line therapy in selected premenopausal women with ER+ABC but the exact role of this option requires further investigation in randomized trials.
Based on the documented efficacy of the selective estrogen receptor downregulator fulvestrant in postmenopausal patients [55], studies have been conducted in premenopausal patients as well [56,57]. Bartsch et al. demonstrated a clinical benefit rate of 58% with fulvestrant plus goserelin in 26 patients pretreated with tamoxifen and AIs in combination with goserelin: the median TTP was 6 months (95% CI: 2.4–9.6 months) and the OS 32 months (95% CI: 14.28–49.72 months [58].
In ER+ABC, most studies addressing the combination of ET and chemotherapy showed an increased ORR or an increased TTP but no improvement in OS with no age-related differences [59]. Trials examining concurrent versus sequential treatment with ET and chemotherapy need therefore to be conducted [60].
Side effects of endocrine therapy
Side effects of ET in young women mimic menopausal symptoms, including hot flashes, sweats, weight gain and sexual dysfunction which may negatively impact QoL [61]. Hot flashes are the most common side effect of tamoxifen. Nonhormonal and nonpharmacological therapies such as phytotherapy or acupuncture can be effective to reduce the intensity of symptoms as well as low-dose antidepressants [62]. The US FDA recently approved paroxetine for the treatment of moderate-to-severe vasomotor symptoms [63]: despite the known interaction between this class of drugs and the CYP2D6-mediated tamoxifen metabolism, there is currently no contraindications to prescribe antidepressants to control vasomotor symptoms [15]. Tamoxifen has both estrogen agonist and antagonist properties with different side effect profile depending on the target organ: there is little uterine cancer risk or excess risk of fatal pulmonary embolism before the age of 45 years [12]. To date, no evidence-based recommendations for routine screening (i.e., transvaginal ultrasound or endometrial biopsy) in women assuming tamoxifen are available. However, abnormal bleeding should be promptly investigated and expert opinion recommendations suggest annual gynecologic examinations [11]. As opposed to menopausal women, there is some evidence that tamoxifen may decrease bone mineral density (BMD) in premenopausal women, although the exact mechanism remains unclear. BMD has therefore to be regularly checked in premenopausal women receiving ET for BC [64].
Weight gain is often reported by women treated with tamoxifen ± OFS. Randomized trials have not reported an excess in weight gain in patients treated with tamoxifen as compared with those who received placebo [65]. In prevention trials, weight gain did not differ between anastrozole, tamoxifen and placebo [66,67]. Weight gain ≥10% after BC diagnosis was associated with a nonsignificant increased risk of death (HR: 1.15; 95% CI: 0.98–1.35) but not of BC-specific mortality (HR: 1.03; 95% CI: 0.84–1.26) in 12,915 patients with BC (diagnosed between 1990 and 2006) from four population-based prospective cohort studies examining the role of physical activity, BMI, dietary factors and interventions and QoL in BC prognosis [68]. Several mechanisms have been postulated through which weight gain may influence survival, including enhanced conversion in fat tissue of androgens to estrogens and associated high levels of fasting insulin which may bind to insulin receptors that are overexpressed on most human BC cells [69]. As a consequence, prevention of weight gain appears to be a sound public health goal for BC survivors [70].
Tamoxifen may adversely affect cognition [71], although few specific investigations on this side effect have been conducted and none in young women. In the ZIPP trial (six cycles of CMF ±2 years of goserelin, goserelin plus tamoxifen or tamoxifen), no effect of treatment on the patients' self-evaluation of memory and concentration was shown [72]. Cognitive function is being prospectively investigated in patients participating in the SOFT trial.
Tamoxifen may also increase plasma estradiol concentrations by interfering with the normal negative pituitary feedback mechanisms: endogenous sex hormone levels were not correlated with outcome in a high-risk postmenopausal population prevention trial [73] but further studies may be required to explore the potential impact on the breast of increased serum estrogen levels in premenopausal BC patients treated with tamoxifen.
Treatment with GnRHa is associated with menopausal side effects such as hot flushes and vaginal dryness [74] and with more severe sexual dysfunction than tamoxifen alone. Vaginal dryness can be mitigated by the addition of tamoxifen [72].
Side effects of AIs when combined with a GnRHa are consistent with the known safety profiles for each of the agents administered [24,27]. The recently reported SOFT–TEXT data show musculoskeletal symptoms, fractures, vaginal dryness, decreased libido and dyspareunia as specific AI side effects. Of note, depression was reported in 50% of patients, with grade 3–4 in 4% of the patients [27]. Bisphosphonates, although not yet approved for this indication, can prevent cancer therapy-induced bone loss and improve BMD in premenopausal BC patients [75,76] and should be promptly introduced at first signs of significant bone loss. Regular exercise has also a positive impact on bone mineralization and stimulation of osteogenesis [77].
Fertility issues, feasibility and safety of pregnancy after the disease should be addressed early after diagnosis and patients should ideally be referred to a fertility specialist before starting therapy [78,79]. In women <35 years, adjuvant chemotherapy is less likely to induce permanent amenorrhea [80]. The two largest randomized trials (PROMISE and POEMS) both demonstrated an increased resumption of ovarian function in women receiving a GnRHa during adjuvant chemotherapy and suggest this approach should be discussed to prevent premature ovarian failure [81,82]. Pregnancy following BC does not seem to negatively influence DFS or OS in ER+ premenopausal patients [83,84]. Within the Breast International Group–North American Breast Cancer Group collaboration the IBCSG has just launched a trial (POSITIVE–IBCSG 48–14 NCT02308085 [85]) (Figure 3) to assess patients' safety and pregnancy outcomes of interrupting ET after having received at least 18 months but no longer than 30 months, to attempt conception.

POSITIVE study design.
Patients should be informed of the possibility of getting pregnant while on tamoxifen, despite developing amenorrhea: the relatively high frequency of severe congenital abnormalities mandates a reliable nonhormonal contraception [86].
Younger age has been associated with lower rates of treatment adherence [87]. In a cohort of 288 French women diagnosed with BC <40 years, 29.7% (95% CI: 24.1–36.4%) had discontinued tamoxifen after 2 years; after 3 years the proportion increased to 39.5% (95% CI: 32.9–47.0%) [88]. In contrast, in the SOFT–TEXT trials, only 13.7% had stopped early all protocol assigned treatments (16.1% of the patients in the exemestane–OFS group and 11.2% of those in the tamoxifen–OFS group) and the overall QoL assessment did not favor either treatment [27]. These data suggest that motivated women, such those included in clinical trials, are willing to complete the full course of treatment and can be of particular interest when considering extended ET in young women and their impact on QoL.
Conclusion
The incidence of ER+ BC among younger women has increased in the past decade. Current available therapies in young women with ER+ early and ABC either modulate ER activity (tamoxifen) or decrease estrogen production (OFS/OA/AIs).
In early BC, recent data on the combination of AIs and OFS show that outcomes in young patients with ER+ disease, still considered at high risk due to age, are extraordinarily good, also in women who did not receive chemotherapy.
In advanced disease, ET has a major role in the long-term control of indolent disease and most of the time is associated with an acceptable toxicity profile.
Better tools to manage early menopause signs/symptoms (e.g., bone resorption, cognitive problems, fertility impairment and sexual disturbances) and careful monitoring of late toxicities (e.g., second cancers) need to be routinely implemented and specifically investigated.
The complex physical and psycho-social scenario of BC at young age mandates individualized and multidisciplinary approaches in order to maximize outcome while minimizing the impact of diagnosis and treatment in women with demanding personal, social and family commitments.
Future perspective
Further progress in the management of young women with BC cannot be achieved without a global commitment by healthcare professionals, researchers, patients, payers and regulators.
In early BC, several questions still need to be answered in order to maximize treatment individualization: the exact role and tailoring added value of OFS added to tamoxifen in the routine clinical management of young women with early BC, especially in those <35 years at diagnosis; the optimal duration of OFS; the optimal extended ET and duration in high-risk patients; and the value of sequential versus concomitant chemotherapy and OFS, particularly in those women for whom chemotherapy is indicated.
Executive summary
Five years of adjuvant tamoxifen is standard endocrine therapy (ET) for premenopausal women: the addition of OFS to tamoxifen should be considered in women who remain premenopausal after adjuvant chemotherapy, especially if <35 years at diagnosis.
The ATLAS and aTTom trial showed 10 years of tamoxifen reduce the risk for BC recurrence and mortality compared with 5-year treatment.
The NCIC-CTG MA.17/BIG 1–97 trial reported a significant advantage to extended adjuvant ET with 5 years of letrozole in premenopausal women with ER+ tumors at diagnosis who became definitively postmenopausal and who had received 5 years of tamoxifen.
Data from TEXT and SOFT showed that adjuvant exemestane plus OFS compared with adjuvant tamoxifen plus OFS improves disease-free survival, BC and distant recurrence-free interval.
Optimal duration of LH-RH agonists is unknown, although most studies have utilized 2–3 years of luteinizing hormone-releasing hormone agonists with 5 years of tamoxifen.
Chemotherapy-induced amenorrhea, even if transient, is associated with improved outcome.
Neoadjuvant ET should not be proposed to young women outside clinical trials.
Neoadjuvant ET with aromatase inhibitor (AI) and gonadotropin-releasing-hormone agonist showed to be effective and should be investigated in randomized trials.
ET is the preferred option for ER+ disease, unless there is evidence of endocrine resistance or need for rapid disease and/or symptom control.
OFS + tamoxifen is the standard first-line therapy in ABC.
OFS + AIs showed comparable activity to AIs in menopausal women and require further investigation in randomized trials.
Further studies are needed with fulvestrant + OFS.
Trials examining concurrent versus sequential treatment with ET and chemotherapy need to be conducted.
Side effects of tamoxifen and OFS/OA mimic menopausal symptoms, including hot flashes, sweats, weight gain, cognitive impairment and sexual dysfunction.
Side effects of AIs when combined with OFS include fractures, musculoskeletal symptoms, vaginal distress, decreased libido and dyspareunia.
Fertility preservation should be discussed early after diagnosis and patients should be referred to a fertility specialist before treatment start.
Pregnancy following BC does not seem to negatively influence disease outcome and should not be discouraged.
Neoadjuvant ET should be appropriately studied in young women with particular focus on translational end points of different endocrine pathways of proliferation in this patients' population.
In ABC, efforts should be made to extend to young women the efficacy data onselective estrogen receptor downregulators to widen their therapeutic armamentarium and to study endocrine resistance compounds (i.e., mTOR-PI3K-MEK-CDK inhibitors).
The number of long-term young survivors either cured after early BC or living with the disease, will increase in the next decades. Resources need to be invested to specifically address their needs (i.e., return to work, social coverage, family management) and to support their caregivers.
Financial & competing interests disclosure
The authors have no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending or royalties.
No writing assistance was utilized in the production of this manuscript.
