Abstract
Objectives
To explain apparent differences among mammography screening services in Sweden using individual data on participation in screening and with breast cancer–specific survival as an outcome.
Methods
We analysed breast cancer survival data from the Swedish Cancer Register on breast cancer cases from nine Swedish counties diagnosed in women eligible for screening. Data were available on 38,278 breast cancers diagnosed and 4312 breast cancer deaths. Survival to death from breast cancer was estimated using the Kaplan–Meier estimate, for all cases in each county, and separately for cases of women participating and not participating in their last invitation to screening. Formal statistical comparisons of survival were made using proportional hazards regression.
Results
All counties showed a reduction in the hazard of breast cancer death with participation in screening, but the reductions for individual counties varied substantially, ranging from 51% (95% confidence interval 46–55%) to 81% (95% confidence interval 74–85%). Survival rates in nonparticipating women ranged from 53% (95% confidence interval 40–65%) to 74% (95% confidence interval 72–77%), while the corresponding survival in women participating in screening varied from 80% (95% confidence interval 77–84%) to 86% (95% confidence interval 83–88%), a considerably narrower range.
Conclusions
Differences among counties in the effect of screening on breast cancer outcomes were mainly due to variation in survival in women not participating in screening. Screening conferred similarly high survival rates in all counties. This indicates that the performance of screening services was similar across counties and that detection and treatment of breast cancer in early-stage reduces inequalities in breast cancer outcome.
Introduction
Cancer outcomes vary among and also within populations. Variation has been noted by geographic area, ethnic group, socioeconomic status and presence of comorbidities.1–4 These inequalities may be due to variable access to treatment, but populations with poorer outcomes tend to have cancers diagnosed at a later stage and have a lower coverage by cancer screening programmes. 1 , 3 ,5–7
The effect of cancer screening can also vary among and within population units in an individual nation. A study in Italy found that poorer breast cancer survival among women of lower educational status was rectified with the introduction of a breast screening programme. 8 We recently analysed data from nine Swedish counties and found that overall there was a significant 41% reduction in incidence of breast cancers proving fatal within 10 years in association with participation in mammography screening. However, the reduction varied among areas from 28% to 51%. 9 It is of interest to investigate the mechanism of an apparent difference in the effect of screening among these Swedish counties.
In any given year, the mortality from breast cancer is a function of incidence rates over time and the subsequent survival of individuals with breast cancer. Likewise, the incidence of subsequently fatal breast cancers is a combination of overall incidence of the disease in a given year and survival from it. The geographically varying effect of screening on the incidence of subsequently fatal breast cancers in the nine counties in any given year is unlikely to be influenced by variation in overall incidence, but may be due to geographic differences in survival among those participating in screening, among those not participating in screening, or both. In this paper, we examine differences in breast cancer survival among the nine counties in Sweden8 and compare how survival varies among counties in women participating or not participating in screening, defined as having attended or not attended the most recent scheduled screening appointment. The purpose of the survival analysis is not to evaluate screening per se, but to identify reasons for differences among counties with respect to the effects of screening on risk of breast cancer death.
Methods
We had data from the Swedish Cancer Register on invasive and in situ breast cancer cases diagnosed in women eligible for screening living in nine Swedish counties. The cause and date of death were collected from the Swedish Cause of Death Register of the Swedish National Board of Health and Welfare. Data were available for the following counties: Västmanland, Dalarna, Örebro, Gävleborg, Värmland, Västerbotten, Norrbotten, Västernorrland and Stockholm. Individual screening data on invitation and participation were provided by Sectra Medical Systems AB, Linköping, Sweden, which maintains the information system for breast cancer screening in these nine Swedish counties.
The breast cancer screening programme in Sweden offers two-view mammography, with the age range for screening determined at the county level. The recommended interscreening interval is 18 months at ages 40–54 and 24 months at ages 55–69. We abstracted data on breast cancer dates of diagnosis and death, and cause of death on women aged 40–69 for those counties which offer screening from age 40 (Dalarna, Västmanland, Gävleborg, Västernorrland and Norrbotten), and on women aged 50–69 for those counties which offered screening from age 50 (Stockholm, Värmland, Örebro and Västerbotten). Periods of observation are given in Table 1. All resident women in the appropriate age range for their counties receive regular personal invitations to screening. The data on invitation and attendance are recorded and stored in the information system for breast cancer screening. All healthcare units are mandated by law to report cancer cases to the Swedish Cancer Register. A linkage between the information on screening participation, cancer diagnosis, vital status and cause of death was determined using the Swedish Personal Identification Number (PIN), mandatory for all citizens.
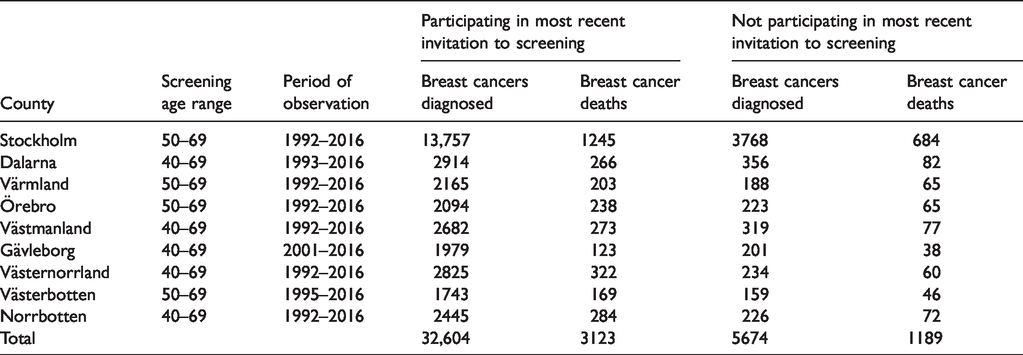
Counties, age ranges and periods of observation, breast cancers, and breast cancer deaths by screening participation.
From these data, we classified women as participating in screening or not, defined as having attended or not attended the most recent scheduled screening appointment based on data provided by Sectra Medical Systems AB. By implication, the cancers in those participating in screening were either screen-detected or interval cancers, and in those not participating were cancers diagnosed in women who had not attended their most recent screening appointment. Women who did not attend their most recent screening appointment may or may not have attended earlier screening rounds. This classification was made annually on the last day of each year.
Survival to death from breast cancer – defined as a report to the Causes of Death Register with breast cancer as primary or underlying cause of death – was estimated using the Kaplan–Meier estimate, for all cases in each county, and separately for cases having participated and not participated in the most recent scheduled screening appointment. Formal statistical comparisons of survival were made using proportional hazards regression. 10 To test whether the differences in survival among counties showed a different pattern between participating and non-participating women, we carried out a formal statistical test for interaction between county and participation status in the proportional hazards regression.
Results
In the period studied, there was an average screening eligible population of 599,875 women, 481,342 participants, and 118,533 non-participants. Table 1 lists the counties, with the periods of observation, age ranges offered screening, numbers of breast cancers (invasive and in situ) and numbers of breast cancer deaths in each county and period. There was a total of 38,278 breast cancers diagnosed and 4312 breast cancer deaths in the periods of observation.
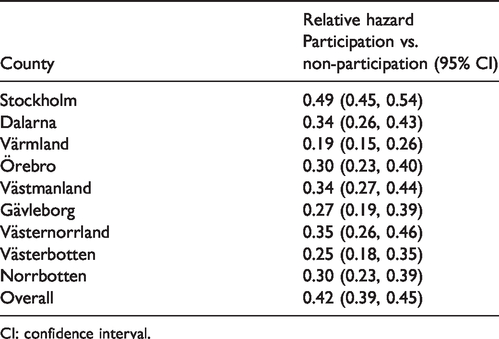
Table 2 shows the relative hazards for cancers diagnosed in those participating in screening compared to cancers in those not participating, for each county separately. Overall, there was a 58% reduction in the hazard of breast cancer death associated with participation in screening (hazards ratio (HR) = 0.42, 95% confidence interval (CI) 0.39–0.45). However, the reductions in hazard for individual counties varied substantially, ranging from a 51% (95% CI 46–55%) reduction in Stockholm to an 81% (95% CI 74–85%) reduction in Värmland.
Relative hazards of breast cancer death associated with participation in screening for each county separately, and all counties combined.
CI: confidence interval.
Figure 1 shows the survival to death from breast cancer for cancers diagnosed in women not participating in screening, for each county separately. There was a range of survival experience, with 20-year survival ranging from 53% (95% CI 40–65%) to 74% (95% CI 72–77%).
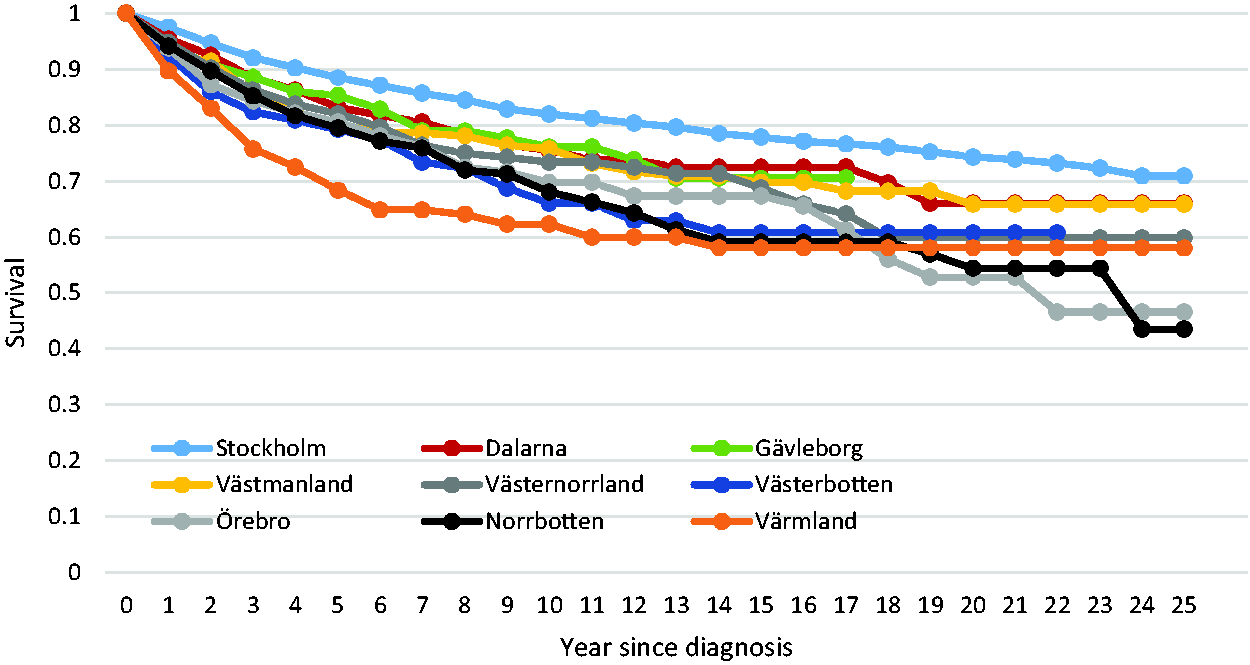
Survival of breast cancer patients not participating in mammography screening, by county.
The corresponding survival from cancers in women participating in screening is shown in Figure 2. Here, survival curves were very close together, with 20-year survival varying from 80% to 86%. Thus, in women participating in screening, breast cancer survival was higher, and more uniform than in women not participating. The difference between the survival patterns in Figures 1 and 2 was highly statistically significant (chi-squared for interaction = 69.50,
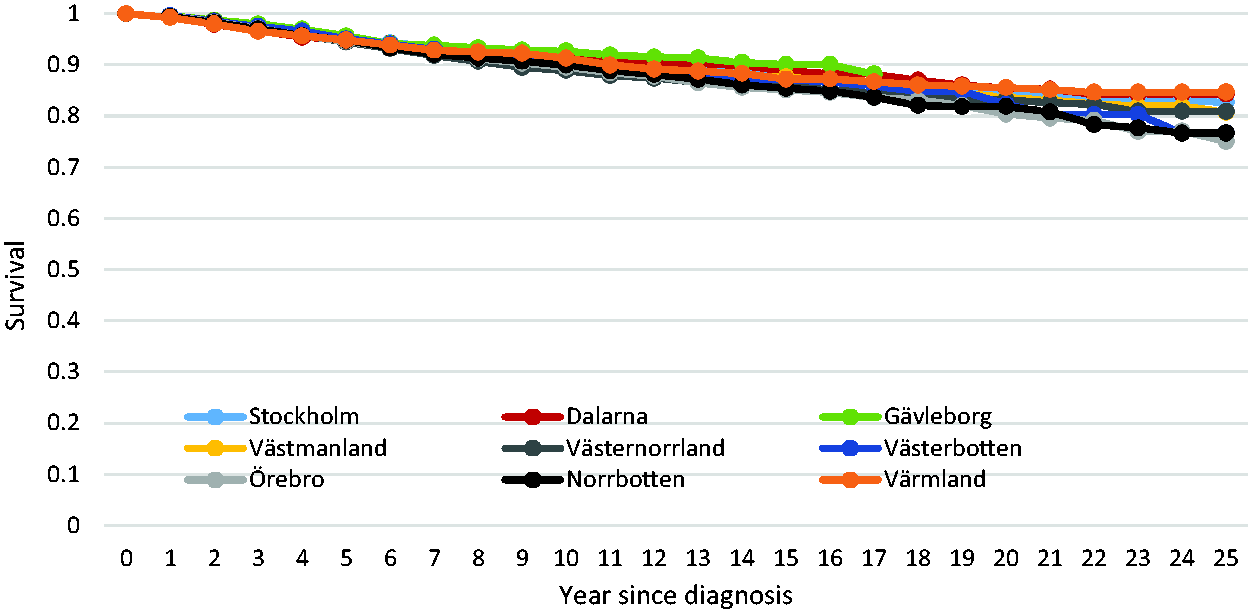
Survival of breast cancer patients participating in mammography screening, by county.
Discussion
In survival analysis of 38,278 breast cancers in nine Swedish counties, we found a 58% overall reduction in hazard of breast cancer death in women participating in their most recent screening examination prior to breast cancer diagnosis. We also found differences among counties in the effects of screening participation on breast cancer survival, ranging from a 51% reduction in hazard to an 81% reduction. However, the survival rates varied widely among counties only in those women not participating in screening. Breast cancer survival rates were much more uniform across counties in women participating in screening.
There are social inequalities in breast cancer survival in many countries, showing poorer survival in patients of lower socioeconomic status,1–4 and Sweden is no exception.11–13 Our results show substantial geographic differences in breast cancer survival, but these are largely confined to women not participating in mammography screening. Participation in screening is characterised by strikingly similar, high survival rates in all nine counties studied. In terms of screening reducing social inequalities in breast cancer outcome, similar results have been seen in other countries. A study in Italy suggested greater equality of outcomes with screening. 8 In the Gironde area of France, the five-year net survival ranged from 78.1% in the most deprived geographical quintile to 91.1% in the least deprived, for cancers in women not attending screening. 14 In screen-detected cancers, the five-year net survival was 91.9% in the most deprived quintile and 92.7% in the least deprived.
This work is not intended to formally evaluate the screening services in the counties studied. We are aware that survival rates alone are insufficient for this and that mortality results or absolute incidence of tumours proving fatal within a period of time are needed for evaluation. Our intention was to use the survival rates to identify the reason for variation in the results on tumours proving fatal within 10 years of diagnosis. We found that the main reason for the variation was the wide range of differences in survival of women
The strengths of this study include a substantial number of breast cancer cases and deaths with more than 20 years of follow-up and virtually no losses to follow-up due to the Swedish personal identification number. We had individual data on exposure and disease-specific death in contrast to ecological or semi-ecological studies of breast cancer screening. We used methods to identify exposure and outcome that are independent of the screening centres and the investigators. The use of death certificates to define breast cancer–specific death entails some misclassification, but is largely non-differential regarding screening status. 15
In conclusion, women participating in screening showed similarly high survival rates in all counties. These observations indicate that the differences in screening effects observed in our paper on 10-year fatal cancers were not due to variation in quality of the screening, but to variation among counties of hazards of breast cancer death for women not being screened. By implication, in settings where stage-specific state-of-the-art treatment is reliably delivered, participation in screening shows strong potential for reducing social and geographic inequalities in breast cancer outcomes.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the American Cancer Society through a gift from the Longaberger Company’s Horizon of Hope Campaign® (Project NHPDCSGBR-GBRLONG). Additional financial support was obtained from Bröstcancerföreningarnas Riksorganisation (BRO), Sweden.
